Abstract
Background
Stress induced increase in colonic paracellular permeability results from epithelial cell cytoskeleton contraction and is responsible for stress induced hypersensitivity to colorectal distension (CRD). The probiotic Lactobacillus farciminis releases spontaneously nitric oxide (NO) in the colonic lumen in vivo and exerts anti‐inflammatory effects. This study aimed: (i) to evaluate the effects of L farciminis on stress induced hypersensitivity to CRD and increase in colonic paracellular permeability; and (ii) to ascertain whether these effects are NO mediated and related to changes in colonocyte myosin light chain phosphorylation (p‐MLC).
Methods
Female Wistar rats received either 1011 CFU/day of L farciminis or saline orally over 15 days before partial restraint stress (PRS) or sham‐PRS application. Visceral sensitivity to CRD and colonic paracellular permeability was assessed after PRS or sham‐PRS. Haemoglobin was used as an NO scavenger. Western blotting for MLC kinase, MLC, and p‐MLC were performed in colonic mucosa from L farciminis treated and control rats after PRS or sham‐PRS.
Results
PRS significantly increased the number of spike bursts for CRD pressures of 30–60 mm Hg as well as colonic paracellular permeability. L farciminis treatment prevented both effects, while haemoglobin reversed the protective effects of L farciminis. p‐MLC expression increased significantly from 15 to 45 minutes after PRS, and L farciminis treatment prevented this increase.
Conclusion
L farciminis treatment prevents stress induced hypersensitivity, increase in colonic paracellular permeability, and colonocyte MLC phosphorylation. This antinociceptive effect occurs via inhibition of contraction of colonic epithelial cell cytoskeleton and the subsequent tight junction opening, and may also involve direct or indirect effects of NO produced by this probiotic.
Keywords: probiotics, stress, colon sensitivity, epithelial cells, nitric oxide
Probiotics have been defined as living organisms in food and dietary supplements which improve the health of the host beyond inherent basic nutrition.1 Preventive and curative efficacy of probiotic treatments has been described in several gastrointestinal pathologies and in the past decade encouraging results have been obtained in inflammatory bowel disease using different strains.2,3 Similarly, treatment with several lactobacilli strains has been found to reduce the severity of experimental colitis in animals.4,5 Based on a recent approach using bacteria as a carrier of anti‐inflammatory compounds delivered into the colonic lumen,6 we have previously shown that Lactobacillus farciminis given orally for 15 days can release spontaneously nitric oxide (NO) into the colonic lumen and reduce the severity of trinitrobenzene sulphonic acid (TNBS) induced colitis in rats.7 Among the targets which have benefited from probiotics, studies have reported enhancement of intestinal barrier function8,9,10 by strengthening tight junctions between enterocytes11,12,13 and thereby preventing an increase in paracellular permeability and subsequent bacterial translocation. L farciminis shares these properties as it prevents bacterial translocation and the increase in colonic paracellular permeability in TNBS induced colitis in rats.5
On the other hand, irritable bowel syndrome is a gastrointestinal disease with unknown aetiology, frequently associated with psychological distress, and characterised by abdominal pain.14,15 An increase in intestinal permeability has recently been described in post‐dysenteric16 and other irritable bowel syndrome patients.17 Manifestations such as increased gut permeability and visceral hypersensitivity are similar to those initiated by various stressful stimuli in rats and mice.18,19,20,21 In rats, partial restraint stress induces colonic hypersensitivity to distension triggered by an increase in gut paracellular permeability.21 Moreover, this stress induced increase in gut permeability results from epithelial cell cytoskeleton contraction.19,21 In contrast, despite controversies in the literature concerning the role of NO in pain transmission and mucosal barrier protection, an antinociceptive action of NO in visceral or peritoneal pain22,23,24 as well as improvement in mucosal barrier function by NO donors25 have already been described.
Based on this background, the aim of our study was to evaluate: (i) whether L farciminis treatment prevents stress induced visceral hypersensitivity to colorectal distension (CRD) and increases colonic paracellular permeability; (ii) the involvement of NO in these effects; and (iii) the effect of L farciminis treatment on stress induced changes in colonocyte phosphorylated myosin light chain (p‐MLC) expression, reflecting contraction of the cytoskeleton.
Materials and methods
Animals
Female Wistar rats (Janvier SA, Le Genest St Isle, France) weighing 200–225 g and housed individually in a temperature controlled room (21±1°C) were used. They were allowed free access to water and fed standard pellets (UAR pellets; Epinay, France). The local committee for animal use and care approved all experimental protocols described in this study.
Bacteria preparation
L farciminis (CIP 103136; Institut Pasteur Collection, Paris, France) was grown at 37°C in MRS broth (VWR International, Fontenay‐sous‐Bois, France). After 17 hours of incubation, cultures were harvested by centrifugation at 4500 g for 10 minutes. Strain extract was resuspended in 0.9% NaCl and conserved at −20°C. Bacterial suspension was prepared daily in order to administer orally 1011 CFU/day/rat.
Animal preparation
Under general anaesthesia induced by intraperitoneal administration of 0.6 mg/kg acepromazine (Calvimet; Vetoquinol, Lure, France) and 120 mg/kg ketamine (Imalgene 1000; Rhône‐Mérieux, Lyon, France), animals were equipped with a polyethylene catheter (OD 0.7 mm, ID 0.3 mm, length 60 cm) inserted into the proximal colon at 1 cm from the caecocolonic junction. Rats were also equipped with three groups of three NiCr wire electrodes (60 cm in length, 80 µm in diameter) implanted into the abdominal external oblique muscle, 2 cm superior to the inguinal ligament.26 Catheter and electrodes were exteriorised on the back of the neck and protected by a glass tube attached to the skin. Myoelectrical activity was recorded on an electromyograph (Mini VIII; Alvar, Paris, France) using a short time constant (0.03 seconds) to remove low frequency signals (<3 Hz) and with a paper speed of 3.6 cm/min.
Partial restraint stress
All stress sessions were performed at the same time of day (between 10am and 12 noon) to minimise any influence of circadian rhythms. Stress effects were studied using the wrap partial restraint stress (PRS) model which is a mild non‐ulcerogenic stressor.27 Thus animals were lightly anaesthetised with ethyl‐ether and their fore shoulders, upper forelimbs, and thoracic trunk were wrapped in a confining harness of paper tape to restrict, but not to prevent, body movements. Then, rats were placed in their home cage for two hours. Rats recovered from ethyl‐ether within 2–3 minutes and immediately moved around in their cage, although the restricted mobility of their forelimb prevented grooming behaviour. Sham‐PRS rats, considered as controls, were anaesthetised as above but were not wrapped and were allowed to move freely in their cages.
Colorectal distension
Rats were accustomed to being in polypropylene tunnels (diameter 7 cm; length 20 cm) for several days before CRD in order to minimise recording artefacts. A 4 cm long latex balloon fixed on a rigid catheter was used. CRD was performed by insertion of the balloon into the rectum 1 cm from the anus. Isobaric distensions of the colon were performed by connecting the balloon to a computerised barostat. Colonic pressure and balloon volume were continuously monitored on a potentiometric recorder (L6514; Linseis, Selb, Germany). Isobaric distensions were performed from 0 to 60 mm Hg, each distension step lasting five minutes. The first distension was performed at a pressure of 15 mm Hg and an increment of 15 mm Hg was added at each following step until a maximal pressure of 60 mmHg.
Colonic paracellular permeability
Colonic paracellular permeability was evaluated using 51Cr‐ethylenediaminetetraacetic acid (EDTA; Perkin Elmer Life Sciences, Paris, France) as a marker of paracellular permeation of tight junctions.2851Cr‐EDTA (0.7 µCi), diluted in 250 µl of saline, was injected through the intracolonic catheter 20 minutes after PRS or sham‐PRS. Then, animals were placed in metabolic cages, and faeces and urine were collected separately over 24 hours. Radioactivity in urine was measured on a gamma counter (Cobra II; Packard, Meriden, Connecticut, USA). Permeability to 51Cr‐EDTA was expressed as percentage of administered radioactivity recovered in urine.
Western blot analysis for myosin light chain kinase (MLCK), myosin light chain (MLC), and p‐MLC
After sacrifice, a 2 cm segment of proximal colon was removed, washed with saline, and the colonic mucosa was scraped off. Proteins were extracted in RIPA buffer and protein concentrations assessed using the BC Assay kit (Interchim, Montluçon, France). Equal amounts of each extract were subjected to 10% sodium dodecyl sulphate‐polyacrylamide gel electrophoresis and then electrotransferred onto PVDF membrane. Membranes were blocked overnight at 4°C with Tris buffered saline /bovine serum albumin 3%, before incubation with primary antibodies. Immunoblotting was performed using specific anti‐MLC, anti‐MLCK (Sigma, Saint Quentin Fallavier, France), and anti‐phospho‐MLC (Clinisciences, Montrouge, France) antibodies at the following dilutions: 1:500, 1:500, and 1:10 000, respectively. After washing, horseradish peroxidase conjugated secondary antibody was used (Upstate, Charlottesville, Virginia, USA). Bands were identified by ECL detection reagents (Amersham Biosciences Europe GmbH, Orsay, France). Band intensities were quantified by densitometry and expressed as mean area density using Quantify one 4.1.1 software.
Experimental protocol
In the first series of experiments, 10 groups of eight rats were used. Over 15 days, animals received orally either saline (groups 1, 2, 4, 5, 7, and 9) or 1011CFU/day L farciminis (groups 3, 6, 8, and 10). Animals were equipped with NiCr electrodes implanted in the abdominal striated muscles (groups 1, 2, 3, 7, and 8) or an intracolonic catheter (groups 4, 5, 6, 9, and 10).
Rats were subjected to PRS (groups 2, 3, 5, 6, 7, 8, 9, and 10) or sham‐PRS (groups 1 and 4). Stepwise CRD were performed in groups 1, 2, 3, 7, and 8, 20 minutes after PRS or sham‐PRS. Groups 4, 5, 6, 9, and 10 received 51Cr‐EDTA intracolonically at the end of PRS or sham‐PRS session, and colonic paracellular permeability was assessed by 24 hours post‐PRS urine collection. Groups 7–10 were continuously infused with haemoglobin, an NO scavenger (0.25 ml/h; 200 mg/kg/day) through the intracolonic catheter over 48 hours before PRS until the end of the CRD session (groups 7 and 8) or until the end of the urine collection period (groups 9 and 10).
In a second series of experiments, 12 groups of four rats were used for the kinetics of MLCK, MLC, and p‐MLC protein expression of stress exposure by western immunoblotting. Groups 1–6 and groups 7–12 were subjected to sham‐PRS and PRS, respectively. After 15, 30, 45, 60, 75, and 90 minutes of sham‐PRS or PRS application, the corresponding group was sacrificed and colonic segments were removed for western blot assays.
In the last series of experiments and according to the results obtained concerning the effect after 45 minutes of PRS application on MLC phosphorylation, four groups of four rats were used in order to evaluate the influence of L farciminis treatment on these stress induced changes. Saline (groups 1 and 2) or 1011 CFU/ml L farciminis were given orally over 15 days before sham‐PRS or PRS, respectively. All groups were sacrificed at 45 minutes after sham‐PRS or PRS application and colonic segments were removed for p‐MLC protein level determination by western blotting.
Statistical analysis
All values are expressed as mean (SEM). Statistical analysis of the number of spike bursts for each five minute period during CRD and colonic paracellular permeability were performed using one way analysis of variance. Differences were considered significant when post hoc test p<0.05.
Results
L farciminis and stress induced visceral hypersensitivity
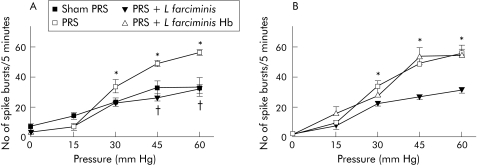
PRS significantly (p<0.05) increased the number of spike bursts for CRD pressures of 30–60 mm Hg compared with sham‐PRS (50 (5) v 29 (3) spike bursts/5 min at 45 mm Hg) (fig 1A). L farciminis treatment prevented the increase in the number of spike bursts induced by PRS, whatever the pressure of CRD (fig 1A). The beneficial effect of L farciminis treatment on stress induced visceral hyperalgesia was abolished when animals received haemoglobin infusion (54 (3) v 28 (2) spike bursts/5 min at 45 mm Hg) (fig 1B). Haemoglobin infusion had no effect per se on PRS induced increase in the number of spike bursts (data not shown).
Figure 1 (A) Effect of Lactobacillus farciminis (1011 CFU/day) treatment on stress induced hypersensitivity in rats. *p<0.05, significantly different from sham‐PRS; †p<0.05, significantly different from partial restraint stress (PRS). (B) Reversion by haemoglobin (200 mg/kg/day) of the effect of L farciminis treatment (1011 CFU/day) on hyperalgesia induced by PRS. *p<0.05 significantly different from PRS+L farciminis.
L farciminis and stress induced increase in colonic paracellular permeability
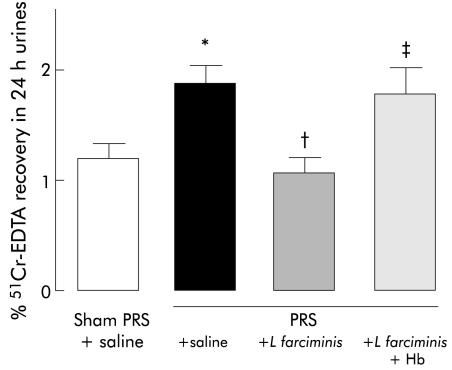
PRS significantly (p<0.05) increased colonic paracellular permeability (2.07 (0.24) v 1.20 (0.20)%) compared with sham‐PRS (fig 2). L farciminis treatment prevented the increase in colonic paracellular permeability induced by PRS (0.95 (0.09) v 2.07 (0.24)%) (fig 2). Haemoglobin infusion reversed the effect of L farciminis on the increase in colonic paracellular permeability induced by PRS (1.78 (0.18) v 2.07 (0.24) %) (fig 2). Haemoglobin infusion did not affect the increase in colonic paracellular permeability induced by PRS (data not shown).
Figure 2 Effect of Lactobacillus farciminis (1011 CFU/ml/day) treatment on the increase in colonic paracellular permeability induced by partial restraint stress (PRS) and its reversion by haemoglobin (Hb 200 mg/kg/day). *p<0.05, significantly different from sham‐PRS; †p<0.05 significantly different from PRS; ‡p<0.05 significantly different from PRS+L farciminis. EDTA, ethylenediaminetetraacetic acid.
Time dependent changes in MLC and p‐MLC protein levels after stress exposure
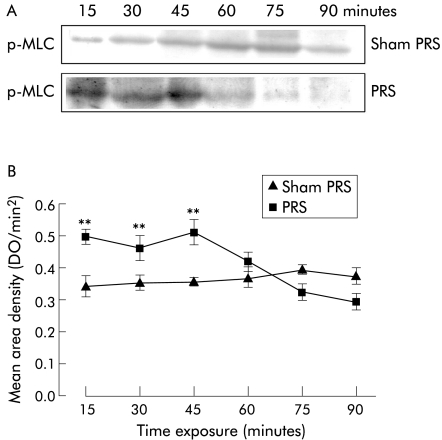
MLCK protein levels were not affected by exposure to stress (table 1). Conversely, significantly (p<0.05) higher levels of p‐MLC were observed from 15 to 45 minutes after the beginning of stress application, with a maximal effect at 45 minutes (fig 3). Then, p‐MLC levels decreased and returned to basal values at 75 minutes after the beginning of stress application (fig 3). MLC protein levels were similar to those observed in sham stressed rats during the early stages of stress application but they increased significantly (p<0.05) after 60 minutes from the beginning of the stress session (table 1).
Table 1 Kinetics of myosin light chain kinase (MLCK) and myosin light chain (MLC) protein level determination after sham‐partial restraint stress (PRS) and PRS exposure*.
| Times of sham‐PRS or PRS exposure (minutes) | |||||||
|---|---|---|---|---|---|---|---|
| 15 | 30 | 45 | 60 | 75 | 90 | ||
| MLCK | Sham‐PRS | 0.49 (0.01) | 0.47 (0.02) | 0.45 (0.03) | 0.48 (0.02) | 0.50 (0.01) | 0.47 (0.02) |
| PRS | 0.48 (0.01) | 0.52 (0.02) | 0.54 (0.03) | 0.54 (0.03) | 0.48 (0.01) | 0.49 (0.01) | |
| MLC | Sham‐PRS | 0.49 (0.01) | 0.47 (0.02) | 0.45 (0.03) | 0.48 (0.02) | 0.50 (0.01) | 0.48 (0.02) |
| PRS | 0.44 (0.05) | 0.53 (0.01) | 0.56 (0.03) | 0.83 (0.03)† | 0.82 (0.03)† | 0.68 (0.04)† | |
Data are represented as mean (SEM) area density.
*By western blot band density.
†p<0.05 versus sham‐PRS.
Figure 3 Western blot for evaluation of phosphorylated myosin light chain (p‐MLC) protein levels on colonic mucosa after partial restraint stress (PRS) application for different time periods. (A) Western immunoblotting for p‐MLC. (B) Relative mean (SEM) area density. Significant increases in p‐MLC protein levels were observed from 15 to 45 minutes after the beginning of stress application. **p<0.01 significantly different from sham‐PRS.
L farciminis treatment effect on changes in p‐MLC protein levels induced by stress
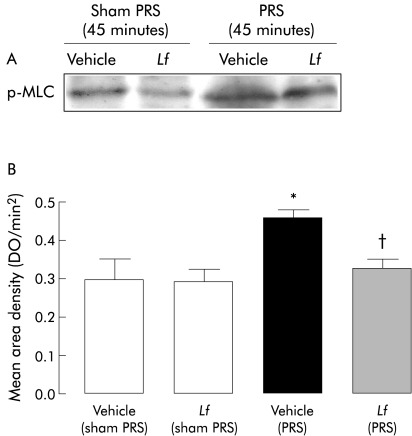
In sham treated animals, L farciminis did not change p‐MLC levels. Conversely, after 45 minutes of stress exposure, L farciminis treatment significantly (p<0.05) decreased p‐MLC protein levels (fig 4).
Figure 4 Western blot for phosphorylated myosin light chain (p‐MLC) protein levels on colonic mucosa after Lactobacillus farciminis treatment. (A) Western immunoblotting for p‐MLC. (B) Relative mean (SEM) area density. L farciminis treatment reduced phosphorylation of MLC induced by 45 minutes of stress exposure. Significantly different (p<0.05) from *sham‐PRS and †PRS, respectively.
Discussion
The present study shows that oral L farciminis treatment prevents stress induced visceral hypersensitivity to colorectal distension and the increase in colonic paracellular permeability. Delivery of exogenous NO by this strain into the colonic lumen is involved in these protective effects as they are both reversed by intracolonic infusion of haemoglobin, an NO scavenger. Moreover, L farciminis treatment suppresses stress induced colonocyte MLC phosphorylation, reflecting their cytoskeleton contraction.
In animals, acute restraint stress enhances abdominal responses to rectal distension18,21,29 and we have recently established that this hypersensitivity to distension results from increased gut permeability.21 In a previous study, we showed that L farciminis, given orally for 15 days, releases spontaneously NO into the colonic lumen.7 We can hypothesise that NO released luminally by L farciminis may act on colonic permeability. A protective effect of NO on mucosal barrier permeability has been described. Indeed, alterations in mucosal barrier permeability after ischaemic injury are also reduced in rats pretreated with molsidomine, an NO donor.25 Furthermore, it has been shown that administration of NO synthase inhibitors elicits large increases in the mucosal permeability of the feline intestine, independent of alterations in intestinal blood flow or adhesion of leucocytes to vascular endothelium.30 This effect of NO synthase inhibitors is reversed by simultaneous administration of NO donor or L‐arginine, underlying a modulatory role of NO produced in the gut mucosa on paracellular permeability.30,31
Increased colonic paracellular permeability resulting from stress depends on epithelial cell cytoskeleton contraction through MLCK activation, inducing MLC phosphorylation, as a specific MLCK inhibitor (ML‐7) prevents the stress induced increase in permeability in mice19 and rats.21 In this study, we showed that acute stress induced MLC phosphorylation at early stages of stress application, and L farciminis treatment suppressed this effect. Interestingly, in a recent study, NO was found to be involved in the modulation of intestinal permeability by preventing MLC phosphorylation in vitro.32 In contrast, the role of NO in pain transmission is controversial. NO has been proposed as being involved in inflammatory hyperalgesia33 but it is also considered a mediator of opiate analgesia.34 Only a few studies have investigated the role of NO on visceral or peritoneal pain models and they indicate an antinociceptive action of NO.22,23,24 Taken together, these data suggest that the antinociceptive effect of L farciminis on stress induced hyperalgesia is probably due to a primary regulatory action of NO on colonic permeability rather than a direct action of NO on nociceptive fibres.
The hypothesis of NO involvement in the protective effects of L farciminis on both colonic paracellular permeability and visceral sensitivity is based on reversal of these effects by intracolonic infusion of haemoglobin. NO is known to form dinitrosyl complexes with iron and to bind easily and strongly with proteins that contain the haem group, such as haemoglobin.35 Consequently, haemoglobin has been used as an appropriate NO scavenger in several experimental studies.36,37 However, we cannot exclude the fact that haemoglobin infusion in the rat colon may impact on the colonic microecology in terms of specific bacterial metabolic activity and/or population composition changes, suggesting that observations obtained in animals undergoing haemoglobin infusion may result from a direct action of haemoglobin on colonic microbiota. Haemoglobin or other haemoproteins reaching the colonic lumen are metabolised by bacteria to a range of haem derived porphyrins lacking iron.38 In a previous work we have shown that L farciminis significantly increased colonic NO concentrations and haemoglobin infusion reduced these concentrations to basal levels, similar to saline treated rats, suggesting that there are no significant changes concerning the ability of the microflora to degrade haemoglobin as this haemoprotein remains able to scavenge NO. However, further investigations are needed to confirm the involvement of NO by using alternative NO scavengers, and also to exclude the possibility that the effect of haemoglobin is due to another mechanism, such as alteration of the microflora.
The protective effect of L farciminis treatment on mucosal barrier integrity is in agreement with previous data showing that in vitro live probiotic strains interact with intestinal epithelial cells to protect them from the deleterious effects of enteroinvasive Escherichia coli by blocking the fall in transepithelial resistance and acting on tight junction proteins.12 Similarly, supernatant of Lactobacillus thermophilus culture prevents aspirin induced inhibition of ZO‐1 expression.13 We therefore speculate that blockade of tight junction opening by L farciminis during stress may in turn prevent excessive uptake of luminal microbial antigens and bacterial products able to activate the submucosal immune system, which may sensitise terminals or sensory nerves through release of mediators. According to the literature, the most attractive potential use of probiotics seems to be colonisation of the gut and subsequent improvement of the intestinal microflora balance and fight against pathogens. We have previously shown that L farciminis given orally for two weeks can adhere to the mucosa of the proximal colon and modulate the immunity of the inflamed colon in rats.5 Thus we cannot exclude the fact that the ability to colonise and interact with the microflora could be involved in the beneficial effects of L farciminis treatment observed in this study. Furthermore, a recent study by Verdu et al showed that perturbations in the gut flora and inflammatory cell activity enhanced visceral sensitivity and this effect was prevented by Lactobacillus paracasei administration.39 Also, a preliminary report from our group showed that disturbances of the commensal flora resulting from oral antibiotic administration in mice decreased the effects of acute stress induced changes on colonic permeability, underlying the role of the flora in the responsiveness of epithelial cells.40
In conclusion, in this study we showed that probiotic treatment prevented the acute stress induced hypersensitivity to distension and the increase in colonic paracellular permeability. It is possible that NO released by L farciminis may affect directly or indirectly colonic paracellular permeability and subsequent colonic hypersensitivity but further investigations are needed to confirm this action. Finally, L farciminis exert a primary action on the colonic epithelial barrier and the effect at this level results from inhibition of MLC phosphorylation and cytoskeleton contraction. All of these data suggest that L farciminis may be of interest in the treatment of visceral hyperalgesia, particularly irritable bowel syndrome.
Abbreviations
CRD - colorectal distension
EDTA - ethylenediaminetetraacetic acid
MLC - myosin light chain
MLCK - myosin light chain kinase
p‐MLC - phosphorylated myosin light chain
NO - nitric oxide
PRS - partial restraint stress
TNBS - trinitrobenzene sulphonic acid
Footnotes
Conflict of interest: None declared.
References
- 1.Fuller R. Probiotics in man and animals. J Appl Bacteriol 198966365–378. [PubMed] [Google Scholar]
- 2.Rembacken B J, Snelling A M, Hawkey P M.et al Non‐pathogenic Escherichia coli versus mesalazine for the treatment of ulcerative colitis: a randomised trial. Lancet 1999354635–639. [DOI] [PubMed] [Google Scholar]
- 3.Gionchetti P, Rizzello F, Venturi A.et al Probiotics in infective diarrhoea and inflammatory bowel diseases. J Gastroenterol Hepatol 200015489–493. [DOI] [PubMed] [Google Scholar]
- 4.Schultz M, Veltkamp C, Dieleman L A.et al Lactobacillus plantarum 299V in the treatment and prevention of spontaneous colitis in interleukin‐10‐deficient mice. Inflamm Bowel Dis 2002871–80. [DOI] [PubMed] [Google Scholar]
- 5.Lamine F, Eutamene H, Fioramonti J.et al Colonic responses to Lactobacillus farciminis treatment in trinitrobenzene sulphonic acid‐induced colitis in rats. Scand J Gastroenterol 2004391250–1258. [DOI] [PubMed] [Google Scholar]
- 6.Steidler L, Hans W, Schotte L.et al Treatment of murine colitis by Lactococcus lactis secreting interleukin‐10. Science 20002891352–1355. [DOI] [PubMed] [Google Scholar]
- 7.Lamine F, Fioramonti J, Bueno L.et al Nitric oxide released by Lactobacillus farciminis improves TNBS‐induced colitis in rats. Scand J Gastroenterol 20043937–45. [DOI] [PubMed] [Google Scholar]
- 8.Isolauri E, Majamaa H, Arvola T.et al Lactobacillus casei strain GG reverses increased intestinal permeability induced by cow milk in suckling rats. Gastroenterology 19931051643–1650. [DOI] [PubMed] [Google Scholar]
- 9.Salminen S, Isolauri E, Salminen E. Clinical uses of probiotics for stabilizing the gut mucosal barrier: successful strains and future challenges. Antonie Van Leeuwenhoek 199670347–358. [DOI] [PubMed] [Google Scholar]
- 10.Madsen K L. Inflammatory bowel disease: lessons from the IL‐10 gene‐deficient mouse. Clin Invest Med 200124250–257. [PubMed] [Google Scholar]
- 11.Lu L, Walker W A. Pathologic and physiologic interactions of bacteria with the gastrointestinal epithelium. Am J Clin Nutr 2001731124–30S. [DOI] [PubMed] [Google Scholar]
- 12.Resta‐Lenert S, Barrett K E. Live probiotics protect intestinal epithelial cells from the effects of infection with enteroinvasive Escherichia coli (EIEC). Gut 200352988–997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Montalto M, Maggiano N, Ricci R.et al Lactobacillus acidophilus protects tight junctions from aspirin damage in HT‐29 cells. Digestion 200469225–228. [DOI] [PubMed] [Google Scholar]
- 14.Whitehead W E, Bosmajian L, Zonderman A B.et al Symptoms of psychologic distress associated with irritable bowel syndrome. Comparison of community and medical clinic samples. Gastroenterology 198895709–714. [DOI] [PubMed] [Google Scholar]
- 15.Drossman D A, Creed F H, Olden K W.et al Psychosocial aspects of the functional gastrointestinal disorders. Gut 199945(suppl 2)II25–II30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Spiller R C, Jenkins D, Thornley J P.et al Increased rectal mucosal enteroendocrine cells, T lymphocytes, and increased gut permeability following acute Campylobacter enteritis and in post‐dysenteric irritable bowel syndrome. Gut 200047804–811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Marshall J K, Thabane M, Garg A X.et al Intestinal permeability in patients with irritable bowel syndrome after a waterborne outbreak of acute gastroenteritis in Walkerton, Ontario. Aliment Pharmacol Ther 2004201317–1322. [DOI] [PubMed] [Google Scholar]
- 18.Gue M, Del Rio‐Lacheze C, Eutamene H.et al Stress‐induced visceral hypersensitivity to rectal distension in rats: role of CRF and mast cells. Neurogastroenterol Motil 19979271–279. [DOI] [PubMed] [Google Scholar]
- 19.Ferrier L, Mazelin L, Cenac N.et al Stress‐induced disruption of colonic epithelial barrier: role of interferon‐gamma and myosin light chain kinase in mice. Gastroenterology 2003125795–804. [DOI] [PubMed] [Google Scholar]
- 20.Barreau F, Cartier C, Ferrier L.et al Nerve growth factor mediates alterations of colonic sensitivity and mucosal barrier induced by neonatal stress in rats. Gastroenterology 2004127524–534. [DOI] [PubMed] [Google Scholar]
- 21.Ait‐Belgnaoui A, Bradesi S, Fioramonti J.et al Acute stress‐induced hypersensitivity to colonic distension depends upon increase in paracellular permeability: role of myosin light chain kinase. Pain 2005113141–147. [DOI] [PubMed] [Google Scholar]
- 22.Fidecka S, Lalewicz S. Studies on the antinociceptive effects of sodium nitroprusside and molsidomine in mice. Pol J Pharmacol 199749395–400. [PubMed] [Google Scholar]
- 23.Smith S D, Wheeler M A, Foster H E., Jret al Improvement in interstitial cystitis symptom scores during treatment with oral L‐arginine. J Urol 1997158703–708. [DOI] [PubMed] [Google Scholar]
- 24.Rouzade M L, Anton P, Fioramonti J.et al Reduction of the nociceptive response to gastric distension by nitrate ingestion in rats. Aliment Pharmacol Ther 1999131235–1241. [DOI] [PubMed] [Google Scholar]
- 25.Schleiffer R, Raul F. Prophylactic administration of L‐arginine improves the intestinal barrier function after mesenteric ischaemia. Gut 199639194–198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Morteau O, Hachet T, Caussette M.et al Experimental colitis alters visceromotor response to colorectal distension in awake rats. Dig Dis Sci 1994391239–1248. [DOI] [PubMed] [Google Scholar]
- 27.Williams C L, Peterson J M, Villar R G.et al Corticotropin‐releasing factor directly mediates colonic responses to stress. Am J Physiol 1987253G582–G586. [DOI] [PubMed] [Google Scholar]
- 28.Bjarnason I, MacPherson A, Hollander D. Intestinal permeability: an overview. Gastroenterology 19951081566–1581. [DOI] [PubMed] [Google Scholar]
- 29.Bradesi S, Eutamene H, Garcia‐Villar R.et al Acute and chronic stress differently affect visceral sensitivity to rectal distension in female rats. Neurogastroenterol Motil 20021475–82. [DOI] [PubMed] [Google Scholar]
- 30.Kubes P. Nitric oxide modulates epithelial permeability in the feline small intestine. Am J Physiol 1992262G1138–G1142. [DOI] [PubMed] [Google Scholar]
- 31.Kanwar S, Wallace J L, Befus D.et al Nitric oxide synthesis inhibition increases epithelial permeability via mast cells. Am J Physiol 1994266G222–G229. [DOI] [PubMed] [Google Scholar]
- 32.Rivero‐Vilches F J, de Frutos S, Saura M.et al Differential relaxing responses to particulate or soluble guanylyl cyclase activation on endothelial cells: a mechanism dependent on PKG‐I alpha activation by NO/cGMP. Am J Physiol Cell Physiol 2003285C891–C898. [DOI] [PubMed] [Google Scholar]
- 33.Aley K O, McCarter G, Levine J D. Nitric oxide signaling in pain and nociceptor sensitization in the rat. J Neurosci 1998187008–7014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nozaki‐Taguchi N, Yamamoto T. Involvement of nitric oxide in peripheral antinociception mediated by kappa‐ and delta‐opioid receptors. Anesth Analg 199887388–393. [DOI] [PubMed] [Google Scholar]
- 35.Ortega Mateo A, Amava Aleixandre de Artinona Nitric oxide reactivity and mechanisms involved in its biological effects. Pharmacol Res 200042421–427. [DOI] [PubMed] [Google Scholar]
- 36.Larauche M, Anton P M, Garcia‐Villar R.et al Protective effect of dietary nitrate on experimental gastritis in rats. Br J Nutr 200389777–786. [DOI] [PubMed] [Google Scholar]
- 37.Fricker S P. Nitric oxide scavengers as a therapeutic approach to nitric oxide mediated disease. Expert Opin Investig Drugs 199981209–1222. [DOI] [PubMed] [Google Scholar]
- 38.Young G P, St John D J, Rose I S.et al Haem in the gut. Part II. Faecal excretion of haem and haem‐derived porphyrins and their detection. J Gastroenterol Hepatol 19905194–203. [DOI] [PubMed] [Google Scholar]
- 39.Verdu E L, Bercik P, Verma‐Gandhu M.et al Specific probiotic therapy attenuates antibiotic induced visceral hypersensitivity in mice. Gut 200655182–190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Demaude J, Ferrier L, Fioramonti J.et al Alteration of colonic paracellular permeability after stress: role of bacterial flora. Gastroenterology 2005128A86 [Google Scholar]