Abstract
Background and aims
Diverticulosis is a common disease of not completely defined pathogenesis. Motor abnormalities of the intestinal wall have been frequently described but very little is known about their mechanisms. We investigated in vitro the neural response of colonic longitudinal muscle strips from patients undergoing surgery for complicated diverticular disease (diverticulitis).
Methods
The neural contractile response to electrical field stimulation of longitudinal muscle strips from the colon of patients undergoing surgery for colonic cancer or diverticulitis was challenged by different receptor agonists and antagonists.
Results
Contractions of colonic strips from healthy controls and diverticulitis specimens were abolished by atropine. The β adrenergic agonist (−) isoprenaline and the tachykinin NK1 receptor antagonist SR140333 had similar potency in reducing the electrical twitch response in controls and diseased tissues, while the cannabinoid receptor agonist (+)WIN 55,212‐2 was 100 times more potent in inhibiting contractions in controls (IC50 42 nmol/l) than in diverticulitis strips. SR141716, a selective antagonist of the cannabinoid CB1 receptor, had no intrinsic activity in control preparations but potentiated the neural twitch in diseased tissues by up to 196% in a concentration dependent manner. SR141716 inhibited (+)WIN 55,212‐2 induced relaxation in control strips but had no efficacy on (+)WIN 55,212‐2 responses in strips from diverticular disease patients. Colonic levels of the endogenous ligand of cannabinoid and vanilloid TRPV1 receptors anandamide were more than twice those of control tissues (54 v 27 pmol/g tissue). The axonal conduction blocker tetrodotoxin had opposite effects in the two preparations, completely inhibiting the contractions of control strips but potentiating those in diverticular preparations, an effect selectively inhibited by SR140333.
Conclusions
Neural control of colon motility is profoundly altered in patients with diverticulitis. Their raised levels of anandamide, apparent desensitisation of the presynaptic neural cannabinoid CB1 receptor, and the SR141716 induced intrinsic response, suggest that endocannabinoids may be involved in the pathophysiology of complications of colonic diverticular disease.
Keywords: colon diverticulitis, colonic motility, cannabinoids, substance P, tetrodotoxin
Colon diverticula are small out‐pouchings from the colonic lumen caused by mucosal herniations through the wall. Diverticula are a frequent finding during colonoscopy and in elderly people, and are more frequent in Western countries (for reviews see Stollman and Raskin,1 Bassotti and colleagues,2 Kang and colleagues,3 Eastwood,4 and Colecchia and colleagues5). The aetiology and pathogenesis of diverticulosis are largely unknown but factors such as a low fibre diet as well as age related or genetically determined changes in collagen composition leading to weakening of the colonic wall seem to play a substantial role. Although most patients remain asymptomatic, with the increase in the aged population diverticulosis may become a clinical issue for a significant proportion (10–20%) who will develop symptoms or inflammatory and/or vascular complications resulting in diverticulitis and haemorrhage.1,5
Diverticular disease is often associated with motor abnormalities of the affected colonic segment2,6 but only very few studies have dealt with the role of the endogenous neurotransmitters and their receptors in the genesis and maintenance of motility alterations.7,8,9,10 Endocannabinoids and substance P are the most important neurotransmitters modulating colonic motility in animals and humans, mainly through an action on the cholinergic system.11,12,13,14 These mediators are involved in important physiological functions and/or pathological events in the intestinal wall, such as water exchange and inflammatory reactions.15,16,17,18,19
The endocannabinoid system is involved in several intestinal pathologies, including inflammation, paralytic ileus, and cholera toxin toxicity.20,21,22,23,24 β‐Adrenergic receptors are also present on enteric neurones where they apparently influence the release of neurotransmitters inducing muscular contractions and affecting colonic propulsion and water reabsorption.25,26,27
In order to investigate diverticulitis associated alterations in colonic neurotransmitter functions, we compared the in vitro contractile response to electrical field stimulation (EFS) of longitudinal smooth muscle strips from the colon of patients undergoing surgery for complicated diverticulosis and healthy strips obtained from the colon of patients with colon cancer. To assess the pharmacological reactivity of apparently healthy and disease specimens, we tested atropine, tetrodotoxin (TTX), the cannabinoid agonist (+)WIN 55,212‐2,28 the selective cannabinoid CB1 receptor antagonist SR141716,29 the β‐adrenergic agonist (−) isoprenaline, and the selective antagonist of the tachykinin NK1 receptor SR140333.30
Methods
The study was approved by the ethics committee of the “San Raffaele Hospital”, Milan, and patients gave their written consent.
Patients and tissue preparation
Specimens of human transverse or distal colon were obtained from patients undergoing surgery for non‐obstructive colonic cancer (eight males and six females; aged 55–65 years) or from patients with diverticular disease operated on for complications at the “San Raffaele Hospital” (seven males and six females; aged 64–73 years). Of these patients, 10 had chronic diverticulitis and underwent surgery for colic stenosis being a subocclusive or occlusive condition with persistent abdominal pain. Often these patients had developed perivisceritis after several episodes of diverticulitis. One patient had been operated on for persistent proctorrhagia due to diverticular bleeding. Two patients with acute diverticulitis had an emergency operation for diverticular perforation. Segments for the in vitro study were taken from macroscopically normal regions of colon cancer patients or, in patients with diverticulitis, from the apparently normal area closest to the diverticulum. In diverticulitis affected patients, nine segments were taken from the transverse and four from the distal colon. Patients with colon cancer and diverticulitis had the same standard anaesthesia and had not received radiotherapy or chemotherapy and were not being treated with steroids or opioids in the days preceding surgery.
Colon segments were collected in the operating theatre, washed in saline, and immediately placed in a cold (4°C) pre‐aerated (95% O2, 5% CO2) Krebs solution, as previously described,11 and transported to the Sanofi‐Aventis laboratories within approximately 30 minutes. Mucosa and submucosa were gently removed; the muscular region was cut along the longitudinal axis into strips approximately 3 mm wide (approximate total length of each preparation 1.5 cm). Smooth muscle strips were used immediately or stored overnight (16–18 hours) in cold (4°C) pre‐aerated Krebs solution. Under these conditions, strips remained fully sensitive to EFS and to the chemical agents under study.
Experimental conditions for the isolated organs
The muscle strips were mounted in a 20 ml organ bath containing warm (37°C) aerated (95% O2, 5% CO2) Krebs solution and stretched with 1 g. After washing, 20 μmol/l choline and 10 μmol/l indomethacin were added to the incubation medium, respectively, as a precursor of acetylcholine and to reduce spontaneous phasic contractions due to prostaglandin release. Isotonic contractions were evoked by EFS. Two platinum wire electrodes were placed on the top and bottom of the organ bath and EFS was elicited by a Power Lab stimulator (AD Instruments PTY Ltd, Castle Hill, Australia) coupled to a multiplexing pulse booster (Ugo Basile, Varese, Italy). Supramaximal stimulation (20 Hz; 2 ms pulse width; 10 second trains every two minutes; 150 mA) was set up to elicit maximal strip contractility then mA was reduced to a submaximal level of stimulation (20% reduction in maximal contractions); in order to better investigate the contractile effect of SR141716 and TTX, EFS was further reduced to 50% of its maximal stimulation in human as well as in guinea pig tissues. Contraction and relaxation were monitored by computer with a data recording and analysis system (Power Lab) linked to isotonic transducers (Ugo Basile, Varese, Italy) through preamplifiers (Octel Bridge Amp).
After stabilisation (approximately two hours), test substances were added to the incubation bath. Incubation time was one hour for SR141716, SR140333 and for the combination of β1 and β2 adrenergic antagonists ICI 118551 and CGP 20712, 30 minutes for atropine, and 15 minutes for TTX. For cumulative agonist concentration‐response curves, (+) WIN 55212‐2 and (−) isoprenaline were left in contact for approximately 15–20 minutes at each concentration. The response to 1 μmol/l atropine (100% inhibition of EFS induced contraction) was determined for each strip at the end of the experiment and used as reference for calculating the responses to the test substances. At least one preparation for each specimen was used as control (tissue incubated with the test substance vehicle only). To prevent oxidation, (−)isoprenaline was dissolved in 0.1% ascorbic acid in distilled water just before starting the experiments. All other substances, if not otherwise specified, were dissolved in distilled water.
Identification and quantification of anandamide (AEA) and 2‐arachidonylglycerol (2‐AG)
Biopsy samples were weighed, immersed in liquid nitrogen, and stored at −70°C until extraction of endocannabinoids. Tissues were extracted with chloroform/methanol (2:1, by volume), each containing 100 pmol/l of d8‐anandamide, d4‐palmitoylethanolamide, synthesised as described previously,31 and d5‐2‐AG (provided by Cayman Chemicals, Ann Arbor, Michigan, USA). Lipid extracts were purified by silica column chromatography, as described previously,31 and fractions containing AEA, palmitoylethanolamide, and 2‐AG were analysed by isotope dilution liquid chromatography‐atmospheric pressure chemical ionisation‐mass spectrometry in the selected monitoring mode, as described in detail elsewhere.32 Results were expressed as pmol or nmol/g of wet tissue. Since during tissue extraction/purification both d5‐ and native 2‐AG are partly transformed into the 1(3)‐isomers, and only a limited amount of arachidonic acid is present at the sn‐1(3) position of (phospho)glycerides, the amounts of 2‐AG reported here are for the combined mono‐arachidonyl‐glycerol peaks.
Semiquantitative reverse transcription‐polymerase chain reaction (RT‐PCR)
Total RNA was extracted using Trizol reagent according to the manufacturer's directions (GibcoBRL, Carlsbad, California, USA). After extraction, RNA was precipitated using ice cold isopropanol and resuspended in diethyl pyrocarbonate (Sigma, St Louis, Missouri, USA) treated water. The integrity of RNA was verified after separation by electrophoresis on a 0.8% agarose gel containing ethidium bromide. RNA was treated with RNAse‐free DNAse I (Ambion DNA‐free kit; Ambion, Woodeard, Austin, Texas, USA) according to the manufacturer's instructions, to digest contaminating genomic DNA. DNAse and divalent cations were removed.
We examined expression of mRNAs for GAPDH (glyceraldehyde‐3‐phosphate dehydrogenase) as the housekeeping gene and CB1 by RT‐PCR. Total RNA was reverse transcribed using oligo dT primers. DNA amplifications were carried out in PCR buffer (Q‐Biogen, Woodeard, Austin, Texas, USA) containing 3 μl cDNA, 25 mM dNTP, 50 mM MgCl2, 10 μM of each primer, and 0.5 U Taq polymerase (Q‐Biogen). The thermal reaction profile consisted of a denaturation step at 94°C for one minute, annealing at 60°C for one minute, and extension at 72°C for one minute with a final 20 minute extension at 72°C.
Thirty five PCR cycles were optimal, in the linear portion of the amplification curve. The reaction was run in a PE Gene Amp PCR System 9600 (Perkin Elmer, Wellesley, Massachusetts, USA). PCR products were electrophoresed on 2% agarose gel containing ethidium bromide for UV visualisation.
Specific oligonucleotides were synthesised on the basis of cloned cDNA sequences of GAPDH and CB1 common to the rat and mouse. For GAPDH, the primer sequences were 5′‐CCC TTC ATT GAC CTC AAC TAC ATG GT‐3′ (nt 208–233; sense) and 5′‐GAG GGC CAT CCA CAG TCT TCT G‐3′ (nt 655–677; antisense, accession No AH007340). The CB1 sense and antisense primers were 5′‐GAT GTC TTT GGG AAG ATG AAC AAG C‐3′ (nt 1095–1119) and 5′‐AGA CGT GTC TGT GGA CAC AGA CAT GG‐3′ (nt 1380–1405). The expected sizes of the amplicons were 470 bp for GAPDH and 309 bp for CB1. We used expression of the housekeeping gene GAPDH as internal standard. No PCR products were detected when the reverse transcriptase step was omitted.
Calculations and statistical analysis
The agonist concentration producing 50% maximal effect (IC50) was calculated using a four parameter logistic model according to Ratkowsky and Reedy,33 with adjustment by non‐linear regression using the Levenberg‐Marquard algorithm in RS/1 software. Differences between means and IC50 values were determined by analysis of variance (ANOVA) followed by Bonferroni's test. Values of p<0.05 were considered statistically significant. All data points or bars on graphics represent means (SEM).
The pA2 for antagonists, as defined by Arunlakshana and Schild,34 was obtained from linear regression of the mean of the log (DR‐1) against the negative log of the antagonist concentration. The negative logarithm of the dissociation constant (pKB) was calculated using the Cheng‐Prusoff equation.35 Computer analysis was carried out as described by Tallarida and Murray.36
Chemicals
SR141716 (rimonabant), 5‐(4‐chlorophenyl)‐4‐methyl‐N‐(1‐piperidinyl)‐1H‐pyrazole‐3‐carboxa‐mide and SR140333, 1‐(2‐(3‐(3,4‐dichlorophenyl)‐1‐[(3‐isopropoxyphenyl)acetyl]‐piperidin‐3‐yl}ethyl)‐4‐phenyl‐1‐azoniabicyclo2‐octane hydrochloride, were synthesised at Sanofi‐Aventis Research (Montpellier, France). The following chemicals were purchased from Sigma‐Aldrich Corp. (St Louis, Missouri, USA): TTX, indomethacin, atropine sulphate, (+)WIN 55,212‐2, (−) isoprenaline, ICI 118551, and CGP 20712. AEA, 2‐AG, and palmitoylethanolamide were purchased from Tocris (Northpoint, UK)
Results
EFS of colonic strips from longitudinal muscle (intertaenial segments) evoked similar twitch‐like regular contractions in tissues from both controls (healthy specimens from the colon of cancer patients) and patients with diverticular disease. However, approximately 30% of diverticular colonic strips had to be discarded because they were completely unresponsive; the remaining 70% generally required more time for twitch stabilisation but their contractile response (after stabilisation) was similar to controls.
The evoked contractions in both control and diverticular preparations were virtually abolished (95–98%) by addition of 1 µmol/l atropine.
The actions of the various receptor agonists and antagonists expected to inhibit intestinal smooth muscle neural contractions were compared in colonic strips from controls and diverticulitis patients (figs 1–5).
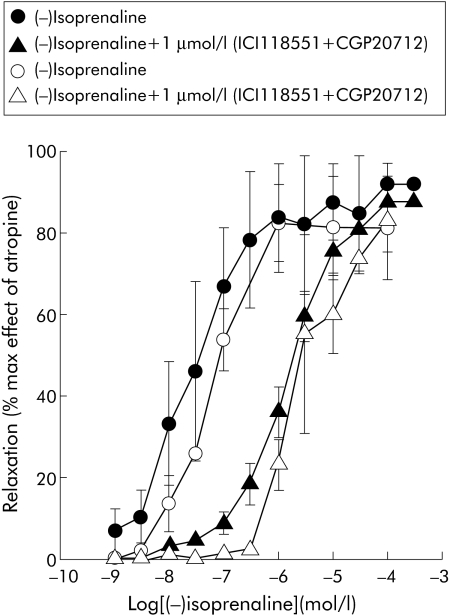
Figure 1 Inhibition by cumulative concentrations of (−)isoprenaline of electrical field stimulation induced contractions of longitudinal colonic muscle strips from controls (filled symbols) and patients with diverticulitis (open symbols). The inhibitory response was recorded in the same strip with or without selective β1 and β2 adrenergic antagonists (CGP20712 + ICI118551). Antagonists were added to the bath one hour before (−)isoprenaline challenge. Results are means (SEM) of 5–12 preparations from different patients. The response is expressed as a percentage of the maximal effect induced by 1 μmol/l atropine.
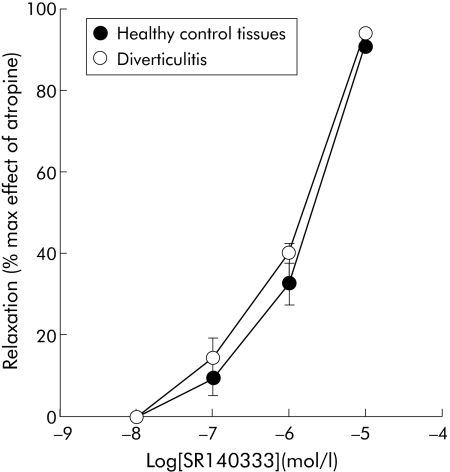
Figure 2 Inhibition by the tachykinin NK1 receptor antagonist SR140333 of electrical field stimulation induced contractions of longitudinal colonic muscle strips from diverticulitis and control patients. Results are mean (SEM) of 3–5 preparations from different patients. The response is expressed as a percentage of the maximal effect induced by 1 μmol/l atropine.
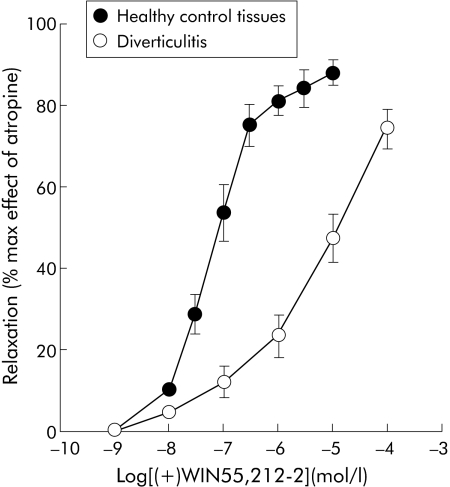
Figure 3 Inhibition by the cannabinoid agonist (+)WIN 55212‐2 of electrical field stimulation induced contractions of longitudinal colonic muscle strips from diverticulitis and control patients. Results are mean (SEM) of 3–5 preparations from different patients. The response is expressed as a percentage of the maximal effect induced by 1 μmol/l atropine.
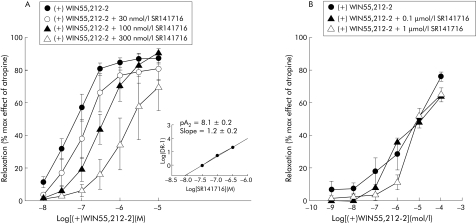
Figure 4 Logarithm concentration‐response curves of (+)WIN 55212‐2 in the absence or presence of increasing concentrations of SR141716 on electrical field stimulation induced contractions of longitudinal colonic muscle strips from controls (A) and patients with diverticulitis (B). The small panel in (A) shows the Schild plot from the curves for control colons. Results are mean (SEM) of 5–8 preparations from different patients. The response is expressed as a percentage of the maximal effect induced by 1 μmol/l atropine.
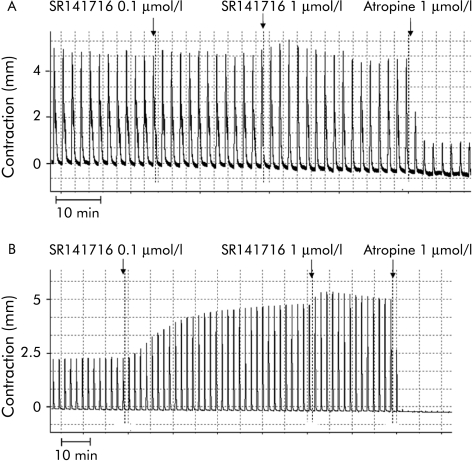
Figure 5 Representative tracings of the intrinsic effect of the cannabinoid antagonist SR141716 on electrical field stimulation induced contractions of longitudinal colonic muscle strips from controls (A) and patients with diverticulitis (B).
The β‐adrenergic agonist (−)isoprenaline had comparable potency and efficacy in reducing the twitches in colonic strips from controls and diverticulitis patients: IC50 (95% confidence limits) were 25 nmol/l (19–48) and 63 nmol/l (45–81), respectively. Combined addition of the β1 and β2 receptor selective antagonists CGP 20712 and ICI 118551 produced a similar parallel shift to the right of the log concentration‐response curve to (−)isoprenaline in control and diverticular preparations: IC50 1.5 (0.9–4.5) µmol/l and 3.9 (1.0–7.9) µmol/L, respectively (fig 1). Similarly, the selective NK1 receptor antagonist SR140333 inhibited electrically evoked contractions with equal potency in control and diverticular preparations in a concentration dependent manner: IC50 0.3 µmol/l (0.1–4) and 0.2 µmol/l (0.4–5.2), respectively (fig 2). In the absence of EFS stimulation, a submaximal concentration of carbachol (0.1 µmol/l) contracted the healthy and diseased preparations with a similar effect (data not shown).
To assess the involvement of the cannabinoid system in colonic contraction, we compared the effects of the cannabinoid agonist (+)WIN 55,212‐2 and the selective CB1 receptor antagonist SR141716 in control and diverticular colon strips. The non‐selective cannabinoid receptor agonist (+)WIN 55,212‐2 was approximately 100 times more potent in inhibiting contractions in the colon from controls than diverticular disease patients: IC50 42 (38–94) nmol/l and 6300 nmol/l (4100–22000), respectively (p<0.01) (fig 3).
Figure 4 shows SR141716 antagonism of the response to (+)WIN 55,212‐2. In control colon strips, the curve representing relaxation induced by cumulative concentrations of (+)WIN 55,212‐2 was shifted to the right by increasing concentrations of SR141716 (fig 4A). Inhibition of (+)WIN 55,212‐2 by SR141716 appears to be competitive at the CB1 receptors, as shown by the Schild plot reported in the small panel of fig 4A: the slope was not significantly different from 1 and pA2 was 8.1 (0.2), corresponding to the affinity of SR141716 for the CB1 receptor. Unlike control colon strips, in those from patients with diverticular disease inhibition of contractions induced by high concentrations of (+)WIN 55,212‐2 was not antagonised by SR141716, even at 1 µmol/l (fig 4B).
The intrinsic activity of the cannabinoid CB1 receptor antagonist SR141716 was also studied in control and diverticular colon strips (fig 5A, B). SR141716 (0.1–1 µmol/l) had no effect in controls but markedly increased twitch contractions in diverticular colon (42% and 196%).
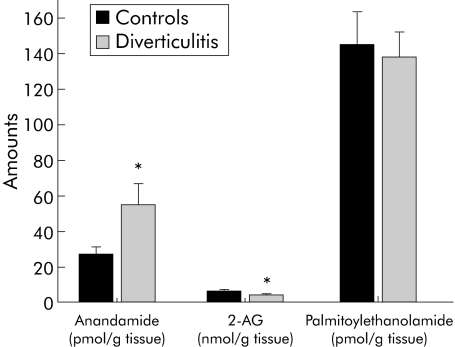
The two endocannabinoids, AEA and 2‐AG, were identified and quantified in colon segments from control and diverticulitis patients by high pressure liquid chromatography coupled to an isotope dilution gas chromatography‐mass spectrometry method. Tissue levels (fig 6) of AEA, an endogenous ligand of cannabinoid and vanilloid TRPV1 receptors, were twice (54.5 (12.0) and 27.0 (4.0) pmol/g tissue; p<0.05) those in control colon whereas 2‐AG levels were slightly lower in diverticulitis than in control segments (4.0 (0.5) v 6.1 (0.9) nmol/g tissue; p<0.05). No change was observed in the AEA congener palmitoylethanolamide.
Figure 6 Anandamide (AEA), 2‐arachydonoylglycerol (2‐AG), and palmitoylethanolamide levels in the colon wall of diverticulitis patients and controls patients. Results are shown as the means (SEM) of 6–10 separate measurements. *p<0.05 by ANOVA followed by Bonferroni's test.
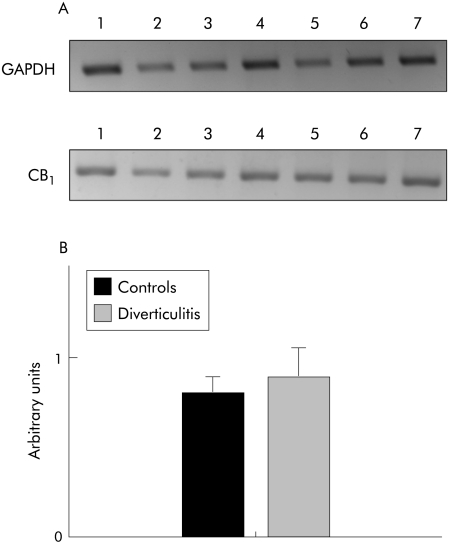
Expression levels of cannabinoid CB1 receptors, measured by RT‐PCR, were similar in diverticular and control colonic segments (fig 7).
Figure 7 Reverse transcription‐polymerase chain reaction (RT‐PCR) expression of cannabinoid CB1 receptors in healthy control or diverticulitis longitudinal colon tissues. (A) Representative agarose gel of amplicons from RT‐PCR of RNAs from healthy (lanes 1–4) and diverticulitis (lanes 5–7) colon tissues. (B) Densitometric scanning of the bands shown in (A), background subtracted and normalised to the corresponding bands of the housekeeping gene (GAPDH, glyceraldehyde‐3‐phosphate dehydrogenase).
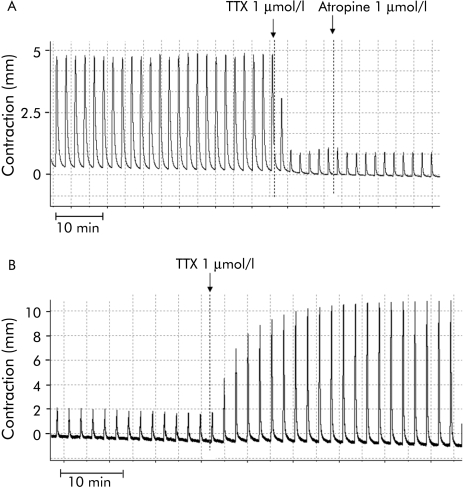
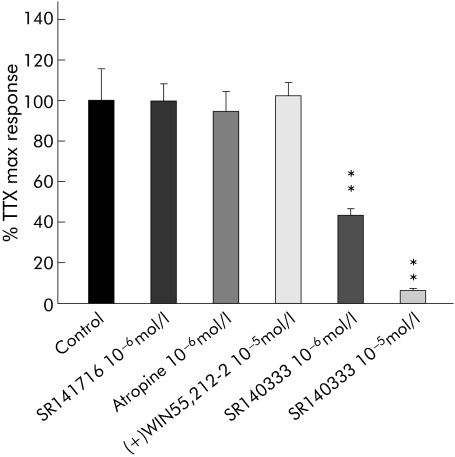
Figure 8 compares the action of the Na+ channel blocker TTX at 1 µmol/l on electrically evoked contractions in normal and diverticular colon strips. TTX had opposite actions in the two preparations, almost completely inhibiting contractions of normal colonic strips but increasing the generally lower EFS evoked contractions in diverticular colon up to a level comparable to, or even higher (198%) than, controls. This increase in contractility induced by TTX in diverticular segments was not amplified by SR141716 1 µmol/l and was not antagonised by 1 µmol/l atropine or 10 µmol/l (+)WIN 55,212‐2, but was inhibited by the tachykinin NK1 receptor antagonist SR140333 in a concentration dependent manner (fig 9).
Figure 8 Representative effect of tetrodotoxin (TTX) on electrical field stimulation induced contractions of colonic longitudinal muscle strips from a control (A) and a patient with diverticulitis (B). TTX was added to the bath at a concentration of 1 μmol/l.
Figure 9 Effects of atropine, (+)WIN 55212‐2, and SR140333 on potentiation of electrical field stimulation induced contractions of longitudinal muscle by 10−6 M tetrodotoxin (TTX) on the colon from patients with diverticulitis. Columns represent the means (SEM) of 5–8 patients. **p<0.01 versus control (ANOVA followed by Bonferroni's test).
Discussion
Motor abnormalities of the intestinal wall in patients with diverticular disease have been frequently described but very little is known about their mechanisms or their role in disease progression.6,7,8,9,10 In our study, we compared the in vitro contractile response to EFS of longitudinal smooth muscle strips from colonic segments close to diverticula and anatomically healthy segments from patients with non‐obstructive colon cancer (controls). Selection of longitudinal muscle was based on previous evidence of lower responsiveness of circular compared with longitudinal muscle to EFS and cannabinoids.12 Diverticular strips were generally less responsive and took longer to stabilise; however, after stabilisation, colon strips from control and diverticular disease patients responded with similar neural twitch‐like contractions to electrical stimulation. These evoked contractions were virtually abolished by atropine, confirming our previous results in the healthy human colon demonstrating the neuronal cholinergic nature of these contractions.12 It is noteworthy that Golder et al showed evidence of cholinergic denervation and hypersensitivity to exogenous acetylcholine in circular but not longitudinal colonic smooth muscle preparations from patients with diverticular disease.37 Consistently in this study, our smooth muscle longitudinal colonic preparations from healthy and diverticular disease patients responded in the same way to carbachol, isoprenaline, and the tachykinin NK1 receptor antagonist SR141333 as well as to EFS. The main unexpected result of the present study was the striking difference between healthy control and diverticular colon strips in response to compounds acting on neural conduction (TTX effect) and cannabinoid CB1 receptors. In the healthy colon, the cannabinoid agonist (+)WIN 55,212‐2 inhibited electrically evoked contractions and this effect was competitively antagonised by SR141716 with a pA2 of 8.1, a value corresponding to its affinity for the CB1 receptor29 and already reported for other in vitro intestinal preparations.38 In healthy colon strips, SR141716 had no intrinsic effects, being unable to potentiate or inhibit electrical twitches. Similar results have already been obtained for circular and longitudinal muscles from human healthy colon segments.12 Unlike the healthy colon strips, those from diverticular disease patients were markedly less sensitive to the inhibitory action of (+)WIN 55,212‐2. (+)WIN 55,212‐2 inhibited electrically evoked contractions only at high concentrations by a CB1 independent mechanism (absence of inhibition by SR141716). In these strips, in contrast with controls, SR141716 had marked intrinsic action (up to 196%). It increased twitch contractions at the same concentrations that blocked cannabinoid CB1 receptors, suggesting that diverticular strips were tolerant to the inhibitory action of endogenous cannabinoids. It is noteworthy that in a previous study we showed that long term preincubation of human intestinal preparations with 10 µmol/l of (+)WIN 55,212‐2 completely abolished the inhibitory effect of (+) WIN 55,212‐2 on EFS contractions. In these tolerant preparations, preincubated with (+)WIN 55,212‐2, twitch responses were markedly enhanced by 1 µmol/l SR141716.39,40 Moreover, although (+)WIN 55,212‐2 is a non‐selective cannabinoid CB1/CB2 agonist, involvement of CB2 receptors in the relaxing action of (+)WIN 55,212‐2 appears unlikely as CB2 receptors are mainly located on immunomodulatory and mucosal cells and do not appear to modulate contractions by acting either on neurones or smooth muscle.12,27
These marked differences in responses to the exogenous cannabinoid agonist (+)WIN 55,212‐2 and the CB1 receptor antagonist SR141716 could be explained by a profound functional alteration of the endogenous cannabinoid system in the wall of colonic segments close to diverticula. A possible interpretation is that in diverticular, but not the healthy colon, there is a strong tonic inhibitory drive sustained by endocannabinoids which reduce the release of contracting neurotransmitters from enteric nerve terminals, through an action at the presynaptic cannabinoid CB1 receptors.
The reason for this substantial activation of the endogenous cannabinoid system is not known. During acute and chronic intestinal inflammation, endogenous cannabinoids may be released with inflammatory mediators, and the cannabinoid CB1 receptors overactivated.20,22,23,24 Chronic inflammation of the colon is often associated with diverticular disease.1,5 In fact, the patients in our study undergoing surgery for complications of diverticular disease showed high serum values for C reactive protein before surgery (from 4 to 10‐fold normal values) and lymphocyte infiltration in most mucosal samples (data not reported), attesting to the presence of an active inflammatory status. Although it is not yet clear whether hyperactivity of the endocannabinoid system counteracts inflammation or is involved in its persistence,24 it would appear that motility is inhibited by endogenously released cannabinoids.11,20,24
It may be argued that the difference in the effects of the cannabinoid system on electrically induced contractions depends on a non‐specific generalised neuronal response or muscle contractility impairment in colon segments with diverticula. However, this is unlikely as the inhibitory responses to the tachykinin NK1 receptor antagonist SR140333 or the β‐adrenergic agonist (−)isoprenaline, either with or without β1 and β2 receptor antagonists, were not modified and were similar in control and diverticular colon strips.
When we measured tissue levels of the two most widely studied endocannabinoids AEA and 2‐AG, we found that even with wide individual variability, levels of AEA were at least twice as high in diverticular than in normal colons, while 2‐AG was reduced by 34% in the former tissue. On the other hand, no change was observed in the AEA congener palmitoylethanolamide, which has non‐cannabinoid receptor mediated inhibitory activity on small intestine motility.41
Although the relative importance of the different endocannabinoids in colon motility is unknown, it has been suggested that AEA, in spite of its much lower tissue levels, is more important than 2‐AG because of its higher affinity for the CB1 receptors and the specific system of cellular uptake and enzymatic metabolism that regulates its concentration at the site of action.20,21,22,42 Levels of endogenous 2‐AG in control colons are theoretically high enough to fully stimulate cannabinoid CB1 receptors tonically. As expression of cannabinoid CB1 receptors is similar in diverticulitis and control healthy colon, endogenous 2‐AG apparently does not exert any role in the inhibition of EFS responses. Indeed, we did not observe an enhanced response in the presence of the cannabinoid CB1 antagonist SR141716. We therefore suggest that continuous increased release of AEA or reduction in its breakdown during chronic intestinal inflammation in the diverticular colon leads to sustained activation of CB1 receptors on enteric cholinergic and substance P nerve terminals, inhibiting the release of these mediators. Both the lack of CB1 mediated response to the exogenous CB1 agonist (+)WIN 55,212‐2 and potentiation of contraction by the CB1 antagonist SR141716 are consistent with the hypothesis that in diverticular colon cannabinoid CB1 receptors are already fully activated by endogenously released cannabinoids and probably tolerant to the agonist action.
A difference in the functional regulation of longitudinal muscle contractility in diverticular and control colon is also suggested by the effect of TTX, a blocker of Na+ channels and axonal conduction. Like atropine, TTX completely abolished the effects of EFS in healthy tissues, indicating the neuronal nature of this response and confirming our previous results.12 In contrast with the healthy colon, in colon strips with diverticulitis we observed an unexpected sustained potentiation of contractions after addition of TTX. These TTX potentiated contractions were not affected by SR141716, atropine, or (+)WIN 55,212‐2, but were inhibited by the tachykinin NK1 receptor antagonist SR140333 in a concentration dependent manner. Although electrically induced release of substance P from non‐neuronal sources is theoretically possible, a more likely explanation of this intriguing response entails the presence in the diverticular colon of neuronal terminals actively releasing substance P and insensitive to the action of TTX. A TTX resistant component in the electrically evoked contraction of colon strips from diverticular disease patients was also described by Maselli et al although these authors could not demonstrate the potentiation of response observed in our study.7 The measure of isometric instead of isotonic contractions and the use, in their study, of different strips (circular instead of longitudinal) may possibly account for the different response. In any case it is relevant that some extrinsic and intrinsic primary afferent neurones regulating intestinal reflexes that control motility, blood flow, and fluid movements express a particular subtype of Na+ channel resistant to the blocking action of TTX.43,44,45,46 The function of these afferent neurones, like that of nociceptive terminals for pain transmission, is increased in inflamed tissues.43,44 On stimulation, they may release substance P which can act as a local transmitter,43,47 contracting smooth muscles by an action both on tachykinin NK1 and NK2 receptors.48,49 In the diverticular colon, TTX, by blocking transmission of neurones releasing inhibitory transmitters, including endocannabinoids, might unmask the action of a TTX resistant neuronal network inducing muscle contraction through substance P release. Another possibility is that when cannabinoid releasing neurones are blocked, the already high AEA levels act directly on the vanilloid VR1 receptor whose stimulation causes substance P release in a TTX insensitive manner. Indeed, AEA activated VR1 receptors are presumably a causative factor in toxin A induced small intestine inflammation50 and might also be involved in inflammatory conditions of the human colon.51 Finally, it may be argued that a direct non‐neuronal stimulation of diverticular smooth muscle strips by EFS could account for the increased twitch contractions after TTX. However, this hypothesis is not supported by the study of Snape and colleagues52 who found similar in vitro sensitivity of colonic smooth muscles from diverticular and cancer patients to different contractile stimuli, including electrical, as well as to the very low degree EFS used in the present study.
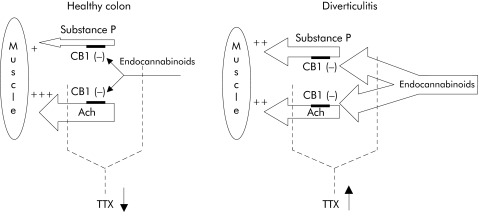
Taken together, the results of this study indicate that the diverticulitis affected colon has a profound alteration in its neuronal control of motility, with substantial involvement of the endocannabinoid system. As shown in fig 10, in diverticular colon, unlike the healthy one, nerve terminals releasing the muscle contracting transmitters acetylcholine and substance P are under the strong inhibitory control of the endocannabinoid AEA, released by a network of functionally activated neurones. In the diseased tissue, this could explain the generally lower contractility in response to EFS, the increased tissue levels of AEA and, most importantly, the potentiating action of SR141716, which promptly relieves the tonic inhibitory control of the endocannabinoids on the presynaptic CB1 receptors located on acetylcholine and/or substance P terminals. When TTX is added to healthy colon strips it almost completely abolishes EFS evoked contractions by blocking the largely dominant cholinergic tone. However, in diverticular colon, where it again blocks neuronal transmission of cholinergic terminals, it also unmasks the action of TTX resistant nerve terminals which directly contract the smooth musculature by releasing substance P. These substance P releasing neurones are functionally more active in inflamed than healthy tissue, and are very likely to express VR1A receptors whose activation by AEA leads to substance P release.
Figure 10 Suggested functional model for regulation of longitudinal colonic smooth muscle contraction in healthy controls and in the colon of diverticulitis patients (see discussion). Ach, acetylcholine; TTX, tetrodotoxin; CB1, cannabinoid receptor subtype 1.
Our experimental approach has the inherent limitations of any in vitro system. For instance, neural contractions of colon smooth muscles are artificially induced by EFS and we measured only contractions of longitudinal muscle. The absence in vitro of any blood supply to the tissue may have a considerable influence on both the release and removal of transmitters from the site of action. In view of these limitations, much care is needed in extrapolating these results to the clinical setting. Further studies are essential to see how these in vitro findings relate to altered patterns of colon motility in patients with diverticular disease.
However, we have shown for the first time that there is altered neuronal control of motility involving the endocannabinoid system in diverticulitis and suggest that a similar dysfunction could also occur in other intestinal disorders with a common inflammatory basis such as some subtypes of irritable bowel disease or even ulcerative colitis and Crohn's disease. It is noteworthy that SR141716 has been reported to display a potent anti‐inflammatory activity involving inhibition of tumour necrosis factor α production.53 Agents acting on the endocannabinoid system could eventually find therapeutic application in colonic inflammatory and motility disorders.
Acknowledgements
We thank Mr Mauro Vezzola and Mr Gabriele De Nardis for expert technical assistance.
Abbreviations
AEA - anandamide or arachidonoyl‐ethanolamide
2‐AG - 2‐arachidonyl‐glycerol
CB1 - cannabinoid receptor subtype 1
EFS - electrical field stimulation
GAPDH - glyceraldehyde‐3‐phosphate dehydrogenase
NK1 - tachykinin receptor subtype 1
RT‐PCR - reverse transcription‐polymerase chain reaction
TTX - tetrodotoxin
VR1 (also known as TRPV1) - vanilloid receptor subtype 1
Footnotes
Conflict of interest: None declared.
A preliminary report of this work was presented at Digestive Diseases Week, May 2004, New Orleans, LA, USA (Gastroenterology 2004;126(Suppl 2): S1447, A219).
References
- 1.Stollman N, Raskin J B. Diverticular disease of the colon. Lancet 2004363631–639. [DOI] [PubMed] [Google Scholar]
- 2.Bassotti G, Chistolini F, Morelli A. Pathophysiological aspects of diverticular disease of colon and role of large bowel motility. World J Gastroenterol 200392140–2142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kang J Y, Melville D, Maxwell J D. Epidemiology and management of diverticular disease of the colon. Drugs Aging 200421211–228. [DOI] [PubMed] [Google Scholar]
- 4.Eastwood M. Colonic diverticula. Proc Nutr Soc 20036231–36. [DOI] [PubMed] [Google Scholar]
- 5.Colecchia A, Sandri L, Capodicasa S.et al Diverticular disease of the colon: New perspectives in symptom development and treatment. World J Gastroenterol 200391385–1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bassotti G, Battaglia E, Spinozzi F.et al Twenty‐four hour recordings of colonic motility in patients with diverticular disease: evidence for abnormal motility and propulsive activity. Dis Colon Rectum 2001441814–1820. [DOI] [PubMed] [Google Scholar]
- 7.Maselli M A, Piepoli A L, Guerra V.et al Colonic smooth muscle responses in patients with diverticular disease of the colon: effect of the NK2 receptor antagonist SR 48968. Dig Liver Dis 200436348–354. [DOI] [PubMed] [Google Scholar]
- 8.Liu L, Shang F, Markus I.et al Roles of substance P receptors in human colon circular muscle: alterations in diverticular disease. J Pharmacol Exp Ther 2002302627–635. [DOI] [PubMed] [Google Scholar]
- 9.Tomita R, Fujisaki S, Tanjoh K.et al Role of nitric oxide in the left‐sided colon of patients with diverticular disease. Hepatogastroenterology 200047692–696. [PubMed] [Google Scholar]
- 10.Huizinga J D, Waterfall W E, Stern H S. Abnormal response to cholinergic stimulation in the circular muscle layer of the human colon in diverticular disease. Scand J Gastroenterol 199934683–688. [DOI] [PubMed] [Google Scholar]
- 11.Pinto L, Capasso R, Di Carlo G.et al Endocannabinoids and the gut. Prostaglandins Leukot Essent Fatty Acids 200266333–341. [DOI] [PubMed] [Google Scholar]
- 12.Manara L, Croci T, Guagnini F.et al Functional assessment of neuronal cannabinoid receptors in the muscular layers of human ileum and colon. Dig Liver Dis 200234262–269. [DOI] [PubMed] [Google Scholar]
- 13.Scheurer U, Drack E, Halter F. Substance P activates rat colonic motility via excitatory and inhibitory neural pathways and direct action on muscles. J Pharmacol Exp Ther 19942717–13. [PubMed] [Google Scholar]
- 14.Rettenbacher M, Reubi J C. Localization and characterization of neuropeptide receptors in human colon. Naunyn‐Schmiedeberg Arch Pharmacol 2001364291–304. [DOI] [PubMed] [Google Scholar]
- 15.Pinto L, Izzo A A, Cascio M G.et al Endocannabinoids as physiological regulators of colonic propulsion in mice. Gastroenterology 2002123227–234. [DOI] [PubMed] [Google Scholar]
- 16.Venkova K, Greenwood‐Van Meerveld B. Effects of TAK‐637 on NK1 receptor‐mediated mechanisms regulating colonic secretion. Toxicol Appl Pharmacol 200496215–222. [DOI] [PubMed] [Google Scholar]
- 17.Tsukamoto M, Sarna S K, Condon R E. A novel motility effect of tachykinins in normal and inflamed colon. Am J Physiol (Gastrointest Liver Physiol) 1997272G1607–G1614. [DOI] [PubMed] [Google Scholar]
- 18.Keränen U, Kiviluoto T, Järvinen H.et al Changes in substance P‐immunoreactive innervation of human colon associated with ulcerative colitis. Dig Dis Sci 1995402250–2258. [DOI] [PubMed] [Google Scholar]
- 19.Riegler M, Castagliuolo I, So P T C.et al Effects of substance P on human colonic mucosa in vitro. Am J Physiol (Gastrointest Liver Physiol) 1999276G1473–G1483. [DOI] [PubMed] [Google Scholar]
- 20.Izzo A A, Fezza F, Capasso R.et al Cannabinoid CB1‐receptor mediated regulation of gastrointestinal motility in mice in a model of intestinal inflammation. Br J Pharmacol 2001134563–570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mascolo N, Izzo A A, Ligresti A.et al The endocannabinoid system and the molecular basis of paralytic ileus in mice. FASEB J 2002161973–1975. [DOI] [PubMed] [Google Scholar]
- 22.Izzo A A, Capasso F, Costagliola A.et al An endogenous cannabinoid tone attenuates cholera toxin‐induced fluid accumulation in mice. Gastroenterology 2003125765–774. [DOI] [PubMed] [Google Scholar]
- 23.Massa F, Marsicano G, Hermann H.et al The endogenous cannabinoid system protects against colonic inflammation. J Clin Invest 20041131202–1209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kunos G, Pacher P. Cannabinoids cool the intestine. Nat Med 200410678–679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ek B A, Bjellin L A, Lundgren B T.et al Beta‐adrenergic control of motility in the rat colon. I. Evidence for functional separation of the beta 1‐ and beta 2‐adrenoceptor‐mediated inhibition of colon activity. Gastroenterology 198690400–407. [DOI] [PubMed] [Google Scholar]
- 26.Roberts S J, Papaioannou M, Evans B A.et al Functional and molecular evidence for β1‐, β2‐ and β3‐adrenoceptors in human colon. Br J Pharmacol 19971201527–1535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Manara L, Croci T, Aureggi G.et al Functional assessment of β adrenoceptor subtypes in human colonic circular and longitudinal (Taenia coli) smooth muscle. Gut 200047337–342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Compton D R, Gold L H, Ward S J.et al Aminoalkylindole analogs: cannabimimetic activity of a class of compounds structurally different from Δ9‐tetrahydrocannabinol.J Pharmacol Exp Ther 19922631118–1126. [PubMed] [Google Scholar]
- 29.Rinaldi‐Carmona M, Barth F, Héaulme M.et al Biochemical and pharmacological characterization of SR 141716A, the first potent and selective brain cannabinoid receptor antagonist. Life Sci 1995561941–1947. [DOI] [PubMed] [Google Scholar]
- 30.Oury‐Donat F, Lefevre I A, Thurneyssen O.et al SR 140333, a novel, selective, and potent non‐peptide antagonist of the NK1 tachykinin receptor: characterization on the U373MG cell line. J Neurochem 1994621399–1407. [DOI] [PubMed] [Google Scholar]
- 31.Bisogno T, Berrendero F, Ambrosino G.et al Brain regional distribution of endocannabinoids: implications for their biosynthesis and biological function. Biochem Biophys Res Commun 1999256377–380. [DOI] [PubMed] [Google Scholar]
- 32.Marsicano G, Wotjak C T, Azad S C.et al The endogenous cannabinoid system controls extinction of aversive memories. Nature 2002418530–534. [DOI] [PubMed] [Google Scholar]
- 33.Ratkowsky D A, Reedy T J. Choosing near‐linear parameters in the four‐parameter logistic model for radioligand and related assays. Biometrics 198642575–582. [PubMed] [Google Scholar]
- 34.Arunlakshana O, Schild H O. Some quantitative uses of drug antagonists. Br J Pharmacol Chemother 19591448–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kenakin T P.Competitive antagonism. Pharmacologic analysis of drug‐receptor interaction , 3rd edn. Philadelphia‐New York: Lippincott‐Raven, 1997
- 36.Tallarida R J, Murray R B.Manual of pharmacological calculations with computer programs, 2nd edn. New York: Springer Verlag 1987
- 37.Golder M, Burleigh D E, Belai A.et al Smooth muscle cholinergic denervation hypersensitivity in diverticular disease. Lancet 20033611945–1951. [DOI] [PubMed] [Google Scholar]
- 38.Izzo A A, Mascolo N, Borrelli F.et al Excitatory transmission to the circular muscle of the guinea‐pig ileum: evidence for the involvement of cannabinoid CB1 receptors. Br J Pharmacol 19981241363–1368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Guagnini F, Croci T, Aureggi G.et al Tolerance to(+)WIN55,212‐2 inhibitory effect and withdrawal by the cannabinoid CB1 receptor antagonist SR141716 in isolated strips of human small intestine. International Cannabinoid Research Society 2000 Symposium on the Cannabinoids. Burlington, Vermont 200082
- 40.Pertwee R. Cannabinoids and the gastrointestinal tract. Gut 200148859–867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Capasso R, Izzo A A, Fezza F.et al Inhibitory effect of palmitoylethanolamide on gastrointestinal motility in mice. Br J Pharmacol 2001134945–950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hornby P J, Prouty S M. Involvement of cannabinoid receptors in gut motility and visceral perception. Br J Pharmacol 2004141335–345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Clerc N, Gola M, Vogalis F.et al Controlling the excitability of IPANs: a possible route to therapeutics. Curr Opin Pharmacol 20022657–664. [DOI] [PubMed] [Google Scholar]
- 44.Beyak M J, Ramji N, Krol K M.et al Two TTX‐resistant Na+ currents in mouse colonic dorsal root ganglia neurons and their role in colitis‐induced hyperexcitability. Am J Physiol (Gastrointest Liver Physiol) 2004287G845–G855. [DOI] [PubMed] [Google Scholar]
- 45.Su X, Wachtel R E, Gebhart G F. Capsaicin sensitivity and voltage‐gated sodium currents in colon sensory neurons from rat dorsal root ganglia. Am J Physiol (Gastrointest Liver Physiol) 1999277G1180–G1188. [DOI] [PubMed] [Google Scholar]
- 46.Wu Z Z, Pan H L. Tetrodotoxin‐sensitive and ‐resistant Na+ channel currents in subsets of small sensory neurons of rats. Brain Res 20041029251–258. [DOI] [PubMed] [Google Scholar]
- 47.Clerc N, Furness J B. Intrinsic primary afferent neurones of the digestive tract. Neurogastroenterol Motil 200416(Suppl 1)24–27. [DOI] [PubMed] [Google Scholar]
- 48.Croci T, Aureggi G, Manara L.et al In vitro characterization of tachykinin NK2‐receptors modulating motor responses of human colonic muscle strips. Br J Pharmacol 19981241321–1327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Holzer P, Holzer‐Petsche U. Tachykinins in the gut. Part I. Expression, release and motor function. Pharmacol Ther 199773173–177. [DOI] [PubMed] [Google Scholar]
- 50.McVey D C, Schmid P C, Schmid H H O.et al Endocannabinoids induce ileitis in rats via the capsaicin receptor (VR1). J Pharmacol Exp Ther 2003304713–722. [DOI] [PubMed] [Google Scholar]
- 51.Geppetti P, Trevisani M. Activation and sensitisation of the vanilloid receptor: role in gastrointestinal inflammation and function. Br J Pharmacol 20041411313–1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Snape W J, Jr, Williams R, Hyman P E. Defect in colonic smooth muscle contraction in patients with ulcerative colitis. Am J Physiol 1991261G987–G991. [DOI] [PubMed] [Google Scholar]
- 53.Croci T, Landi M, Galzin A M.et al Role of cannabinoid CB1 receptors and tumor necrosis factor‐alpha in the gut and systemic anti‐inflammatory activity of SR141716. Br J Pharmacol 2003140115–122. [DOI] [PMC free article] [PubMed] [Google Scholar]