Abstract
Background and aim
Abdominal pain and discomfort are common symptoms in functional disorders and are attributed to visceral hypersensitivity. These symptoms fluctuate over time but the basis for this is unknown. Here we examine the impact of changes in gut flora and gut inflammatory cell activity on visceral sensitivity.
Methods
Visceral sensitivity to colorectal distension (CRD) was assessed at intervals in healthy mice for up to 12 weeks, and in mice before and after administration of dexamethasone or non‐absorbable antibiotics with or without supplementation with Lactobacillus paracasei (NCC2461). Tissue was obtained for measurement of myeloperoxidase activity (MPO), histology, microbiota analysis, and substance P (SP) immunolabelling.
Results
Visceral hypersensitivity developed over time in healthy mice maintained without sterile precautions. This was accompanied by a small increase in MPO activity. Dexamethasone treatment normalised MPO and CRD responses. Antibiotic treatment perturbed gut flora, increased MPO and SP immunoreactivity in the colon, and produced visceral hypersensitivity. Administration of Lactobacillus paracasei in spent culture medium normalised visceral sensitivity and SP immunolabelling, but not intestinal microbiota counts.
Conclusion
Perturbations in gut flora and in inflammatory cell activity alter sensory neurotransmitter content in the colon, and result in altered visceral perception. Changes in gut flora may be a basis for the variability of abdominal symptoms observed in functional gastrointestinal disorders and may be prevented by specific probiotic administration.
Keywords: probiotic therapy, visceral hypersensitivity, gut flora, inflammation, mice
Up to 20% of the general population is affected by irritable bowel syndrome (IBS) although most of these patients do not consult a physician. Abdominal pain and discomfort are hallmarks of IBS and are believed to reflect increased visceral sensitivity. Symptoms in IBS wax and wane over time in terms of intensity1 and character2 but mechanisms underlying these fluctuations are unclear. Factors such as stress, intestinal infection, drugs, and diet are known to exacerbate symptoms in IBS patients and may have indirect effects on visceral perception in the gut.
Several observations suggest a role for the gut flora in the expression of IBS. Gastrointestinal infection is known to alter gut flora3 and also to exacerbate or induce IBS.4 Antibiotics disrupt the gut flora and their use has also been linked to expression of functional gastrointestinal symptoms. Patients treated with antibiotics for non‐gastrointestinal complaints are three times more likely to report functional bowel symptoms.5 Another study investigating the risk factors for developing IBS after acute gastroenteritis showed that IBS symptoms develop more frequently in patients treated with antibiotics for their initial illness.6
Subtle changes in the mucosal immune system and low grade inflammation have been implicated in the pathophysiology of IBS.7,8,9 While the determinants of immune activation in IBS have not been identified, putative factors include diet and commensal bacteria. For example, intolerances to specific foods occur in IBS and may reflect an IgG mediated immune response to dietary antigen.10 Commensal bacteria are important determinants of immune activity in the gut11,12 and changes in gut bacteria have been implicated in symptom generation of IBS.13,14,15,16 Taken together, these observations support the hypothesis that perturbations in gut flora and changes in the degree of immunological activation in the gut influence expression of functional gastrointestinal symptoms.
Our laboratory has focused on the development of murine models of functional gastrointestinal disorders.17,18,19 During the course of these studies, we observed significant changes in visceral perception in control mice maintained in a specific pathogen free environment but without sterile animal handling procedures. This observation prompted us to consider whether fluctuations in the bacterial content of the gut, and subtle increases in its inflammatory cell activity, could be a basis for the changes in visceral perception observed in these mice.
The present study was designed to examine whether deliberate perturbation of the gut flora enhances visceral perception and whether this is mediated by changes in inflammatory cell activity in the gut. Our results show that antibiotic induced perturbation in the gut flora produces changes in inflammatory cell activity and sensory neuropeptide immunolabelling in the gut, and that this results in visceral hypersensitivity. These changes were prevented by administration of Lactobacillus paracasei suspended in spent culture medium.
Material and methods
Animal housing and handling
Female NIH Swiss mice were purchased from NCI (Bethesda, Massachusetts, USA) and Balb/c mice (6–8 weeks of age) from Harlan (Indianapolis, Indiana, USA). Mice were kept under specific pathogen free (SPF) conditions at McMaster University Central Animal Care Facility. Sentinel SPF mice were screened according to the tracking profile of Charles River's Diagnostic laboratories (www.criver.com).
In the first set of experiments, mice were exposed to unfiltered air during colorectal distension (CRD) recordings. Prior to use, intrarectal balloons were thoroughly cleaned but not sterilised. In the experiments using antibiotics, a “sterile protocol” in which mice were exposed to HEPA filtered air was applied. Mice were placed in restrainers inside level B hoods and kept in custom made sterile HEPA filtered cages during CRD recordings. Fistulas and cables were sterilised at the Central Sterilisation Unit at McMaster University. Intrarectal balloons and instruments were sterilised using CIDEX OPA, and rinsed in sterile distilled water prior to use.
All experiments were approved by the McMaster University Animal Care Committee and the Canadian Council on Animal Care.
Overall design
To confirm preliminary observations of the spontaneous development of visceral hypersensitivity in mice, we measured CRD in Balb/c mice at day 0, and weeks 4, 8, 10, and 12. To examine the strain specificity of time dependent changes in visceral sensitivity, we also studied a group of NIH Swiss mice at day 0 and at week 6. To investigate whether changes in visceral perception were accompanied by an increase in inflammatory cell activity, we measured myeloperoxidase (MPO) activity in colonic samples in additional Balb/c mice that were sacrificed at day 0 and at week 10. To establish an association between inflammatory cell activity and changes in visceral sensitivity, responses to CRD and MPO activity were measured after intraperitoneal administration of placebo (sterile saline 100 μl) or dexamethasone (0.5 mg/kg) at week 12.
To determine whether changes in gut flora result in altered visceral perception, NIH Swiss mice received by gavage a combination of non‐absorbable antibiotics or drinking water (placebo) for 10 days. Responses to CRD were measured before and at 10 and 30 days after starting the antibiotics. Samples of colonic tissue and lumen content were obtained at the different time points for MPO activity and for bacterial counts. We used a combination of antibiotics that had been well characterised in terms of dose, drug concentration, and tissue toxicity in the mouse.20,21 Bacitracin and neomycin were diluted in sterile deionised water and the pH of the solution was adjusted to 4.0 to prevent inactivation of bacitracin. Primaricin was added to the antibiotic solution to prevent yeast overgrowth. Mice received bacitracin 4 mg/ml, neomycin 4 mg/ml, and primaricin 0.2 g/ml of drinking water during the first five days. Antibiotic concentrations in drinking water were reduced to 2 mg/ml for bacitracin and neomycin and to 0.1 g/ml for primaricin during the last five days of treatment.
To examine whether probiotic therapy could protect against the antibiotic induced changes in visceral sensitivity, we administered the above combination of antibiotics plus 100 μl of 1010L paracasei NCC2461/ml in spent culture medium, or placebo, for 10 days by oral gavage. L paracasei was chosen based on results from previous experiments in a model of post‐infective IBS showing that this probiotic strain attenuated post‐infective hypercontractility in part by exerting an anti‐inflammatory effect. The beneficial effect was also observed with its spent culture medium (SCM) devoid of live bacteria.22 Responses to CRD were investigated before antibiotic therapy (day 0) and on day 10. Colonic content and tissue were obtained for histology (haematoxylin‐eosin stain), for substance P (SP) immunohistochemistry, as well as for measurement of total lactobacilli counts and for specific detection of L paracasei NCC 2461.
To test the effect of L paracasei on normal visceral perception in mice that did not receive antibiotics, additional mice (n = 6) were investigated at day 0 after a 10 day gavage with Man‐Rogosa‐Sharpe broth (MRS; Becton Dickinson, Sparks, USA) and eight weeks later after a 10 day gavage with MRS, followed by a 10 day gavage with L paracasei.
To discriminate between the effect of distensions or of handling of mice using an unsterile technique on MPO activity, additional groups of mice were sham distended. For this, mice were handled in level B hoods and placed in HEPA filtered restrainers during experiments. A sterile balloon was inserted intrarectally but was not distended. Groups of mice were sacrificed at day 0, week 10, and week 12 (n = 5/group).
To determine whether dexamethasone or the antibiotic combination exerted direct effects on visceral sensitivity, responses to CRD were measured in a separate group of mice that received either dexamethasone or the antibiotic combination directly into the colon via a double lumen catheter (n = 12). Local administration of saline or histamine was used as negative and positive controls, respectively. A washout period of three days was respected between experiments. Dexamethasone (0.5 mg/kg; 0.2 ml), neomycin (4 mg/ml) + bacitracin (4 mg/ml) + primaricin (0.2 mg/ml) (0.2 ml), histamine (10−6 mM; 0.2 ml), or saline (0.2 ml) were administered intrarectally after the third distension.
Pseudoaffective response to colorectal distension
Electromyographic (EMG) electrodes were surgically implanted under sterile conditions in the anterior abdominal wall muscle of mice and a chronic fistula was exteriorised. Mice were then allowed to recover for a period of at least seven days.
The response to CRD was assessed using a method described previously.19 CRD was performed in a stepwise fashion. Each 10 second distension was followed by a five minute resting period. Each level of distension (30 and 60 mm Hg) was repeated three times. EMG activity of the abdominal muscle was continuously recorded using customised software (Acquire 5.0; A Bayatti). The area under the curve was calculated for five seconds before and after the beginning of each distension period using customised software (GrafView 4.1; A Bayatti).
For experiments using the double lumen catheter, a single non‐painful level of distension (40 mm Hg) was chosen and CRD responses were recorded before and 15 minutes after drug administration. The level of distension was chosen based on previous studies demonstrating robust CRD responses to 40 mm Hg.19
L paracasei culture and spent culture medium
L paracasei was chosen based on previous results showing that this strain attenuated post‐infective muscle hypercontractility by attenuating the inflammatory response to infection. This effect was also observed with its SCM devoid of live bacteria.22L paracasei NCC2461 was obtained from the Nestle Culture Collection (Lausanne, Switzerland) and grown under anaerobic conditions in MRS. After 48 hours at 37°C, the number of bacteria was estimated by measuring optical density at 600 nm (1 OD600 = 108 bacteria/ml). Bacterial cells were pelleted by centrifugation for 15 minutes at 5000 g at 4°C, further resuspended at a concentration of 1010/ml in its SCM, and kept in frozen aliquots until use.
Intestinal microbiota counts
Analysis of a portion of the intestinal microbiota was performed before, on day 10 during antibiotic therapy, and on day 30 after discontinuation of antibiotics. Total lactobacilli counts were also performed before and on day 10 during antibiotic therapy in mice treated with placebo and supplemented with L paracasei. The presence of L paracasei was specifically investigated in the latter group. As no significant differences in intestinal microbiota were observed in control samples at different time points (days 0, 10, and 30), these samples were pooled in one single control group.
Colonic segments (2 cm) were obtained under sterile conditions. Contents were pooled with 1 ml of 0.9% NaCl‐10% glycerol used to wash the lumen. Tissues were ground in 2 ml of 0.9% NaCL‐10% glycerol using a polytron (Kinematica, Littau‐Lucerne, Switzerland). Samples were stored at −70°C until analysis. On plating on semi‐selective media,23 bacterial populations were estimated by counting colony forming units (CFU). Lactobacilli and Bacteroides were incubated anaerobically (AnaeroGen; Oxoid, Basingstoke, UK) at 37°C for 48 hours. Enterobacteria, enterococci, and yeast were incubated aerobically at 37°C for 24 hours. Bacterial counts were expressed in log10 CFU/g faeces or tissue. Presence of L paracasei NCC2461 was monitored by random amplification of polymorphism DNA (RAPD) fingerprint as described previously.22
Immunohistochemistry for substance P
SP is a neurotransmitter demonstrated in the terminals of primary afferent nerves and in enteric neurones of both animals and humans.24,25 To detect SP we used immunohistochemistry in colonic frozen sections from mice treated with antibiotics, antibiotics plus L paracasei, or placebo. Tissue was obtained before (day 0) and at day 10 during administration of antibiotics.
As a primary antibody, we used a rabbit anti‐substance P (1:2000) (1:1000) antiserum (Chemicon International, Temecula, California, USA). Negative controls were performed by omitting the primary antibody or by blocking antibody/protein complex formation for SP (peptide concentration 10 mM). For the latter, SP blocking peptide (Sigma‐Aldrich, Oakville, Canada) was incubated with the primary antibody before its application on the slides. Vectastain Elite ABC kit was used for secondary antibody and reporter solutions (Vector, Burlingame, California, USA).
Tissue sections were analysed using light microscopy (DMLS, Leica, Germany), and quantification of immunostaining was performed on computer using public domain software (image J 1.32, http:rsb.info.nih.gov/ij/) selecting the area of the submucous plexus and muscularis externa, and positive staining was expressed as percentage of total tissue area.
Data presentation and statistical analysis
Parametric data are presented as means (SD) and non‐parametric data as box plots (box median; 25%, 75% percentiles; whiskers 5th and 95th percentiles). EMG responses to CRD are expressed as AUC or as percentage of change versus day 0 (day 0 = 100%) for each distension level. In experiments using the double lumen catheter, EMG responses to CRD are presented as percentage of change versus the first distension (1st distension = 100%).
Parametric data were analysed using the paired or unpaired t test, as appropriate. ANOVA was used for multiple comparisons. Paired comparisons of non‐parametric data were performed by the Mann‐Whitney U test. For multiple comparisons of non‐parametric data, the Friedman test followed by Wilcoxon‐Wilcox was used.
Results
Spontaneous changes in visceral hypersensitivity
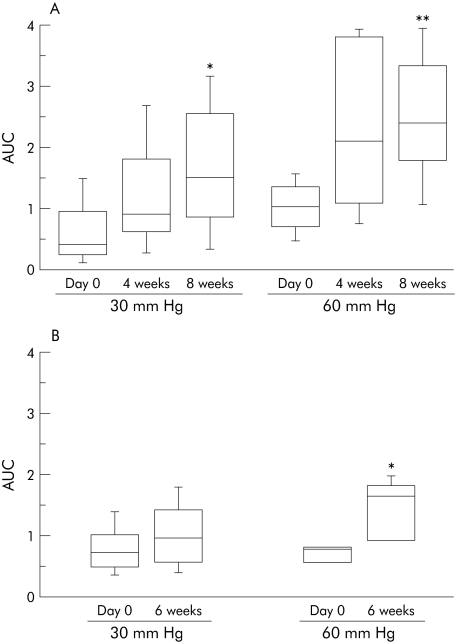
Visceral sensitivity increased with time at four and eight weeks compared with day 0 in Balb/c mice (fig 1A). A 270% and a 140% increase in CRD responses to 30 and 60 mm Hg, respectively, was observed at eight weeks compared with day 0. This observation was not strain specific as a 100% increase in CRD responses to 60 mm Hg was also observed in NIH Swiss mice at six weeks compared with day 0 (fig 1B).
Figure 1 Visceral sensitivity to colorectal distension to 30 and 60 mm Hg in (A) Balb/c mice (n = 8/group). At 30 mm Hg, *p = 0.02 versus day 0; at 60 mm Hg, **p = 0.0008 versus day 0. (B) NIH Swiss mice (n = 10/group). At 60 mm Hg, *p = 0.02 versus day 0. AUC, area under the curve for electromyographic responses to colorectal distension. Data are presented as box plots.
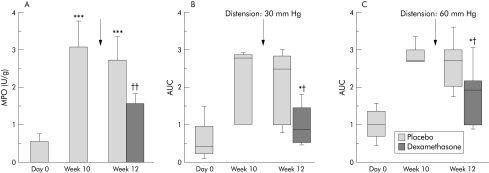
To determine whether the time dependent increase in visceral perception was accompanied by changes in inflammatory cell activity in the gut, we next measured MPO at week 10 in Balb/c mice. As shown in fig 2A, there was a mild elevation in MPO values at week 10 in the hyperalgesic mice.
Figure 2 (A) Myeloperoxidase (MPO) activity for Balb/c mice at day 0 (n = 5), week 10 (n = 5), and week 12 (placebo n = 5; dexamethasone n = 8). ***p<0.001 versus day 0, ††p = 0.01 versus week 12 placebo. Data are means (SD). (B) Visceral sensitivity to colorectal distension to 30 mmHg on day 0, week 10, and week 12 (n = 8). Dexamethasone (n = 8) or placebo (n = 5) was administered at week 11 (arrow). *p = 0.02 versus placebo; †p = 0.03 versus week 10. Data are presented as box plots. (C) Visceral sensitivity to colorectal distension to 60 mm Hg on day 0, week 10, and week 12. *p = 0.07 versus placebo; †p = 0.04 versus week 10. AUC, area under the curve for electromyographic responses to colorectal distension. Data are presented as box plots.
Effect of dexamethasone
To determine whether the increased inflammatory cell activity was associated with the emergence of visceral hypersensitivity, we measured responses to CRD before and after dexamethasone treatment. As shown in fig 2A, MPO activity increased at weeks 10 and 12 compared with day 0. In contrast with placebo, dexamethasone reduced MPO activity at week 12 compared with week 10. This was accompanied by a 70% and a 25% drop in CRD responses to 30 and 60 mm Hg, respectively, at 12 weeks (fig 2B).
Effect of perturbation of the intestinal microbiota
To determine whether minor increases in MPO activity were associated with the distension procedure itself or to exposure of mice to unfiltered air, additional groups of mice were sham distended. We minimised the risk of bacterial contamination of these mice by adopting sterile techniques and avoiding exposure of these mice to unfiltered air. MPO activity in the colon was 0.2 (0.2), 0.3 (0.2), and 0.14 (0.1) on day 0, week 10, and week 12, respectively (p>0.05).
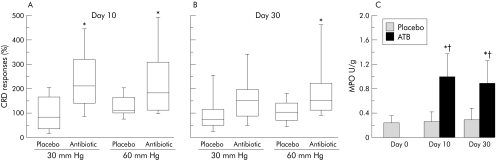
To perturb the gut flora, mice were gavaged with a combination of non‐absorbable antibiotics. Controls received phosphate buffered saline (placebo). MPO activity in placebo treated mice was 0.1 (0.01) at day 0, 0.5 (0.01) at day 10, and 0.3 (0.01) at day 30 (both p>0.05 versus day 0). In addition, no significant increase in CRD responses was observed in placebo treated mice at days 10 and 30 compared with day 0 (fig 3A, 3B; table 1).
Figure 3 (A, B) Percentage of colorectal distension (CRD) responses to 30 mm Hg versus day 0 in placebo and antibiotic treated mice. On day 10, antibiotic treated mice had increased CRD responses to 30 mm Hg (*p = 0.02) and 60 mm Hg (*p = 0.04). On day 30, antibiotic treated mice had increased CRD responses to 60 mm Hg (*p = 0.04) (n = 15 mice/group). Data are presented as box plots. (C) Myeloperoxidase (MPO) activity in placebo and antibiotic treated mice at days 0 (before antibiotics, n = 5), day 10 (n = 5/group), and day 30 (n = 15/group). Day 10: *p = 0.042 versus day 0 and †p = 0.049 versus placebo. Day 30: *p = 0.042 versus day 0 and †p = 0.05 versus placebo. Data are means (SD).
Table 1 Median (interquartile range) area under the curve values in placebo and antibiotic treated mice.
| Placebo | Antibiotic | |||||
|---|---|---|---|---|---|---|
| 0 mm Hg | 30 mm Hg | 60 mm Hg | 0 mm Hg | 30 mm Hg | 60 mm Hg | |
| Day 0 | 0.2 | 1.2 | 1.7 | 0.2 | 0.7 | 1.3 |
| (0.2–0.3) | (0.6–1.3) | (1.0–2.0) | (0.2–0.6) | (0.5–1.4) | (1.0–2.0) | |
| Day 10 | 0.2 | 1.1 | 2.2 | 0.4 | 1.6** | 2.8*† |
| (0.1–0.3) | (0.5–1.4) | (1.0–2.4) | (0.1–0.6) | (0.6–2.0) | (2.0–3.1) | |
| Day 30 | 0.3 | 0.7 | 1.6 | 0.4 | 1.0 | 2.1** |
| (0.2–0.4) | (0.5–1.5) | (1.4–2.3) | (0.3–0.4) | (0.5–1.5) | (1.4–2.4) | |
*p<0.05 and **p = 0.08 versus day 0; †p<0.05 versus placebo.
As shown in fig 3A and 3B, a 10 day course of antibiotics was associated with the emergence of visceral hypersenstitivity to CRD. This was associated with a mild but significant increase in MPO activity on day 10 and 30 after antibiotic therapy (fig 3C).
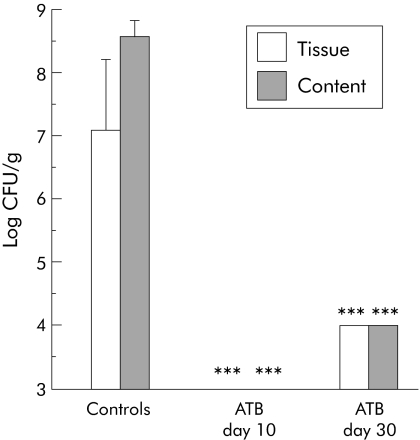
To evaluate the impact of the antibiotic combination used in this study on selected gut microbiota, Bacteroides, enterobacteria, enterococci, and lactobacilli counts were performed. Ten days after administration of antibiotics, we were not able to culture lactobacilli from colonic content or tissue (<3.0 log CFU/g). As shown in fig 4, on day 30, lactobacilli populations were still markedly reduced in mice previously treated with antibiotics compared with placebo treated controls.
Figure 4 Total lactobacilli populations from colonic content and tissue on days 10 and 30 in antibiotic treated mice (ATB, n = 5 per group) and in controls. All placebo controls (day 0, day 10, and day 30) were pooled into one single group (n = 15). ***p<0.0001 versus controls. Data are means (SD). CFU, colony forming units.
Table 2 shows the effect of antibiotics on enterobacteria, Bacteroides, and enterococci populations. Bacteroides and enterococci decreased significantly on day 10 during antibiotic therapy. On day 30, increases in enterobacteria and Bacteroides were observed with respect to control values. However, enterococci counts remained lower than control values on day 30.
Table 2 Markers of microbiota changes in control and antibiotic treated (ATB) mice.
| Enterobacteria | Bacteroides | Enterococci | ||||
|---|---|---|---|---|---|---|
| Content | Tissue | Content | Tissue | Content | Tissue | |
| Control | 4.1 (0.3) | 4.0 (0.0) | 6.9 (1.1) | 5.0 (1.0) | 5.3 (1.2) | 4.5 (0.7) |
| Day 10 ATB | 4.0 (0.0) | 4.0 (0.0) | 4.0 (0.0)* | 4.0 (0.0) | 4.0 (0.0)* | 4.0 (0.0) |
| Day 30 ATB | 5.3 (1.1)* | 4.8 (1.2)* | 7.0 (2.4) | 6.1 (2.0)* | 4.08 (0.0)* | 4.0 (0.0) |
*p<0.05 versus control.
Effect of probiotics
To investigate the effect of L paracasei administration of visceral perception in untreated mice, visceral sensitivity was measured in six mice after a 10 day gavage with MRS, and eight weeks later after a 10 day gavage with MRS followed by a 10 day gavage with L paracasei. After eight weeks, median AUC (interquartile range) in mice treated with MRS was 1.1 (0.7–1.8) and 1.7 (0.7–2.7) for 30 and 60 mm Hg, and not different from that at day 0 (1.1 (0.6–2.0); 1.5 (0.6–1.8)) (table 3). L paracasei administration in the absence of antibiotic treatment did not affect visceral perception significantly (1.2 (0.7–2.1) and 1.4 (0.7–2.5) for 30 and 60 mm Hg; both p>0.05 versus MRS).
Table 3 Median (interquartile range) area under the curve values in placebo, antibiotic alone, and antibiotic plus Lactobacillus paracasei (L pa) in spent culture medium (SCM) treated mice at day 0 and day 10 after antibiotic administration.
| Placebo | Antibiotic | Antibiotic+L pa/SCM | ||||
|---|---|---|---|---|---|---|
| 30 mm Hg | 60 mm Hg | 30 mm Hg | 60 mm Hg | 30 mm Hg | 60 mm Hg | |
| Day 0 | 0.7 | 1.5 | 0.6 | 1.5 | 0.8 | 1.7 |
| (0.5–1.1) | (0.7–1.7) | (0.4–0.8) | (1.0–1.6) | (0.4–1.1) | (0.7–2.0) | |
| Day 10 | 0.7 | 1.7 | 1.5* | 2.6* | 1.2 | 2.3 |
| (0.2–1.5) | (1.0–2.5) | (0.9–2.1) | (1.6–3.3) | (0.5–1.6) | (1.0–3.1) | |
*p<0.05 versus day 0.
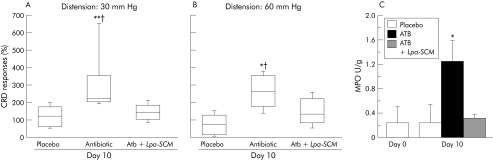
To test whether probiotics or their products protect against antibiotic induced increases in visceral sensitivity, we gavaged L paracasei resuspended in its culture medium from day 1 until day 10 concomitantly with antibiotics. Responses to CRD were measured before and on day 10 during antibiotics. As shown in fig 5A and 5B, the increase in visceral sensitivity on day 10 during antibiotic therapy was attenuated in mice supplemented with L paracasei in SCM and was not different than that at day 0. In parallel with this, administration of L paracasei in SCM decreased MPO activity in colonic samples compared with antibiotic alone treated mice (fig 5C).
Figure 5 (A, B) Percentage of colorectal distension (CRD) responses to 30 and 60 mm Hg versus day 0 in placebo, antibiotic (ATB), and antibiotic + Lactobacillus paracasei resuspended in spent culture medium (ATB+L pa‐SCM) treated mice. On day 10, antibiotic treated mice had increased CRD responses compared with placebo and mice supplemented with L paracasei resuspended in culture medium (n = 15/group). At 30 mm Hg, **p = 0.008 versus placebo and †p = 0.03 versus ATB+L pa‐SCM; at 60 mmHg, *p = 0.04 versus placebo and †p = 0.07 versus ATB+L pa‐SCM. Data are presented as box plots. (C) Myeloperoxidase (MPO) activity at days 0 (before antibiotics) and 10 in placebo, antibiotic treated, and antibiotic + Lpa in SCM treated mice. MPO in ATB treated mice at day 10 was higher versus day 0 (*p = 0.04), versus day 10 in placebo (*p = 0.049), and versus day 10 in ATB+ Lpa‐SCM treated mice (*p = 0.01). Data are means (SD).
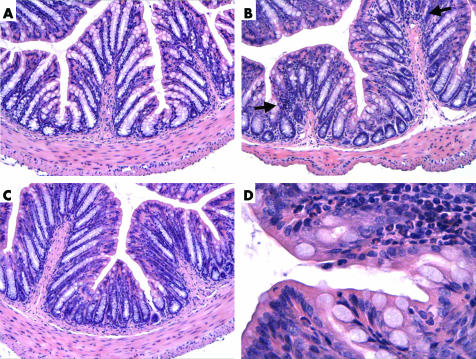
Histological examination of haematoxylin‐eosin stained slides from mice treated with placebo, antibiotics, or antibiotics supplemented with L paracasei in SCM revealed normal colonic structure at day 10. Few isolated foci of polymorphonuclear cells were observed at high power view in antibiotic treated mice but not in placebo or antibiotic treated mice supplemented with L paracasei in SCM (fig 6).
Figure 6 Haematoxylin‐eosin stained slides of mouse colon on day 10. (A) Placebo. (B) Antibiotic treated mouse. (C) Antibiotic supplemented with Lactobacillus paracasei resuspended in culture medium treated mouse. (D) High power view of (B) showing a polymorphonuclear focus in one of the villi. Arrows, polymorphonuclear cells.
Despite improvement in visceral hypersensitivity and histology, total lactobacilli populations and L paracasei NCC 2461 were undetectable (content and tissue <3.0 log 10 CFU/g) in L paracasei/SCM supplemented mice during antibiotic therapy.
Substance P immunoreactivity
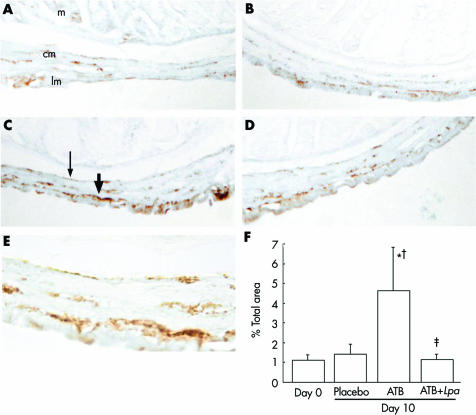
There was increased immunostaining for SP in the area of the submucous and myenteric plexus of mice treated with antibiotics compared with day 0 and with placebo controls (fig 7A, B). Quantification of staining in 5–6 slides per group confirmed these results (fig 7, bottom panel). Administration of L paracasei in SCM to antibiotic treated mice markedly reduced SP immunostaining.
Figure 7 Substance P (SP) immunostaining. (A) Day 0. (B) Day 10 after placebo therapy. (C) Day 10 after antibiotics alone. (D) Day 10 after antibiotics + Lactobacillus paracasei. (E) High power view of (C). m, mucosa; cm, circular muscle; lm, longitudinal muscle. Thick arrow indicates SP staining in the myenteric plexus, thin arrow indicates SP staining in the submucuous plexus. (E) Bar graph depicts quantification for SP staining (area selected: submucous and myenteric plexus, muscularis externa) in 5–6 mice per group. *p = 0.03 versus day 0, †p = 0.049 versus day 10 placebo, ‡p = 0.04 versus antibiotic alone. Data are means (SD).
Direct effects of dexamethasone or antibiotics on visceral sensitivity
Using a double lumen catheter, neither direct administration of dexamethasone, or of the antibiotic combination, caused changes in CRD responses. Histamine administration increased CRD responses when compared to before administration (p = 0.03) (table 4).
Table 4 Direct effects of dexamethasone or antibiotics on visceral sensitivity.
| Before intrarectal administration | After intrarectal administration | |
|---|---|---|
| Saline | 102% (80–145) | 97% (70–110) |
| Histamine | 100% (99–110) | 152% (130–250)* |
| Dexamethasone | 100% (79–108) | 95% (67–99) |
| Antibiotic combination | 100% (88–115) | 100% (60–115) |
*p = 0.03 versus before intrarectal administration. Mice (n = 12/group) were distended three times before and three times after administration of active substances.
Data are median (IQR) of three distensions expressed as percentage of change versus the first distension (1st distension = 100%).
Discussion
The aim of this study was to determine if changes in the commensal bacterial content and activity of resident inflammatory cells in the intestine of healthy mice alter visceral sensitivity. It was not our purpose to define the role of specific flora in modulating sensory function as this would involve performing comparative studies on germ free and colonised mice in a gnotobiotic facility.
Our results show that mice maintained under normal, but non‐sterile, conditions may exhibit a time dependent increase in visceral sensitivity which is accompanied by an increase in the activity of inflammatory cells in the intestine. These findings suggest that a small increment in inflammatory activity that is well below that seen in our models of intestinal inflammation17,26 and which is not accompanied by signs of ill health is sufficient to induce hyperalgesia. This is supported by demonstration that the time dependent increase in visceral sensitivity and myeloperoxidase activity could be reversed by dexamethasone. Dexamethasone administration has been reported to prevent hypersensitivity in dental nerves and to reduce sensory neuropeptides.27,28 Increased degradation of tachykinins released from sensory nerves by glucocorticoids may contribute to this effect.29 However, we found that instillation of dexamethasone directly into the colon of naïve mice without hypersensitivity failed to alter responses to CRD. We conclude that the antinociceptive effect of dexamethasone observed in mice with spontaneous visceral hyperalgesia was due to a primary suppressive effect on inflammatory cell activity. Taken together, these results suggest that the degree of immune activation in the gut modulates visceral perception, and is in keeping with other recent findings that the integrity of the mucosal immune system is necessary for normal visceral perception.30
As a time dependent shift in visceral sensitivity and increases in MPO activity were not observed in mice housed under similar conditions but handled using sterile techniques, we speculate that enhanced MPO and sensory state reflected low grade bacterial contamination. Support for the concept that shifts in resident intestinal bacteria may alter visceral sensitivity also comes from our demonstration that antibiotic administration to healthy mice produced visceral hyperalgesia. The antibiotics caused a decrease in Bacteroides and enterococci and eliminated lactobacilli from the gut. Commensal bacteria are involved in maintenance of normal mucosal immune responses and inflammatory activity in the gut.11,12 We believe that antibiotic treatment altered the bacterial content of the gut, eliminating bacteria that possess counterinflammatory properties and thus favouring a proinflammatory milieu and a hyperalgesic state.
There are other explanations for the antibiotic induced changes in this study that merit discussion. Neomycin has been reported to produce increased infiltrates of macrophages in the lamina propria and cause histological changes in small bowel mucosa.31 Although we cannot rule out direct induction of low grade inflammation, a direct toxic effect is unlikely as at the dose used in this study this antibiotic combination has previously been determined to be safe and no toxicity has been reported in rodents or humans.20,21 Moreover, while direct intrarectal administration of histamine significantly increased CRD responses, no increases in CRD responses were observed after intrarectal perfusion with the antibiotic combination. We also reason that any direct toxic effects would have also been present in antibiotic treated mice receiving L paracasei in SCM. These results suggest that an immediate toxic effect of the antibiotic combination leading to visceral hypersensitivity is unlikely. We have not tested the effect of repeated intrarectal administration of the antibiotic combination. It is possible that such a protocol would result in intestinal microbiota changes and altered visceral sensitivity.
Administration of L paracasei in SCM prevented the antibiotic induced increase in visceral sensitivity and inflammatory activity but did not restore normal counts of lactobacillus in the gut. This is in accordance with our recent study showing that supplementation with live L paracasei improved gut dysfunction without restoring counts of lactobacilli in the intestine.23 Moreover, in that study we showed the ability of the SCM medium to attenuate inflammation induced changes in muscle contractility following primary infection by the nematode Trichinella spiralis.23 Taken together with our previous results, and because L paracasei was undetectable on day 10 after antibiotic therapy, we suggest that the beneficial effect after antibiotic therapy may be mediated by a soluble product of L paracasei present in the SCM in which the bacteria were resuspended. Alternatively, bacterial fragments released as a consequence of antibiotic therapy may be involved, as probiotic DNA has been shown to attenuate inflammation in models of experimental colitis.32
In the present study, we showed that the antibiotic induced change in visceral sensitivity was accompanied by increased SP immunoreactivity, which was localised primarily in the myenteric and submucous plexus. It is likely that the change in sensory neurotransmitter content was secondary to the increase in inflammatory activity, as previous studies have shown increases in enteric SP following exposure of the myenteric plexus to interleukin 1β33 and in experimental colitis.34 However, we cannot exclude the possibility that bacteria directly influence neurotransmitter content. Previous studies have suggested that pathogenic microbes can upregulate SP in infected tissue.35,36 Direct communication between commensals and the enteric nervous system was suggested in the study of Hooper et al in which expression of genes encoding enteric neural transmission differed in germ free and colonised mice.37 In addition, Kamm et al showed changes in the localisation patterns of neuronal markers in myenteric neurones of the pig jejunum of Saccharomyces boulardi treated animals.38 While an oligopeptidase produced by L paracasei has been shown to hydrolyse the Pro‐Gln, Gln‐Phe, and Phe‐Gly bonds of SP,39 we consider this an unlikely explanation for its prevention of antibiotic induced increases in SP and visceral sensitivity as this action would not account for the decrease in MPO activity.
There are several implications of our results. Firstly, the hygiene status of mice maintained in conventional facilities may influence responsiveness to colorectal distension, and this may be important in disease models, which are not associated with overt inflammation. Secondly, the notion that subtle changes in inflammatory activity in the gut alter visceral perception may have bearing on the finding that IBS symptoms are less prevalent in those patients receiving oral corticosteroids for other indications,40 and on the association between antibiotic usage and expression of IBS.5,6,41 Thirdly, these findings provide a rationale for the use of selected probiotics in the management of IBS.
Acknowledgements
The authors thank G Bergonzelli for helpful discussions and G Reuteler for technical assistance with the bacterial cultures.
Abbreviations
IBS - irritable bowel syndrome
SPF - specific pathogen free
CRD - colorectal distension
MPO - myeloperoxidase
SCM - spent culture medium
SP - substance P
MRS - Man‐Rogosa‐Sharpe broth
EMG - electromyographic
CFU - colony forming units
Footnotes
We acknowledge the Canadian Institutes of Health Research (grant to SMC) and the Canadian Association of Gastroenterology and Astra Zeneca, Canada for scholarship support (EFV).
Conflict of interest: None declared.
Conflict of interest: None declared.
References
- 1.Agreus L, Svardsudd K, Talley N J.et al Natural history of gastroesophageal reflux disease and functional abdominal disorders: a population‐based study. Am J Gastroenterol 2001962905–2911. [DOI] [PubMed] [Google Scholar]
- 2.Locke GR I I I. Natural history of irritable bowel syndrome and durability of the diagnosis. Rev Gastroenterol Disord 2003S1212–17. [PubMed] [Google Scholar]
- 3.Guarner F, Malagelada J R. Gut flora in health and disease. Lancet 2003361512–519. [DOI] [PubMed] [Google Scholar]
- 4.Spiller R C. Postinfectious irritable bowel syndrome. Gastroenterology 20031241662–1671. [DOI] [PubMed] [Google Scholar]
- 5.Alun‐Jones V, Wilson A J, Hunter J O.et al The aetiological role of antibiotic prophylaxis with hysterectomy in irritable bowel syndrome. J Obstet Gynaecol 19845S22–S23. [Google Scholar]
- 6.Gwee K A, Graham J C, McKendrick M W.et al Psychometric scores and persistence of irritable bowel after infectious diarrhea. Lancet 1996347150–153. [DOI] [PubMed] [Google Scholar]
- 7.Rodriguez L A, Ruigomez A. Increased risk of irritable bowel syndrome after bacterial gastroenteritis: cohort study. BMJ 1999318565–566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Collins S M, Piche T, Rampal P. The putative role of inflammation in irritable bowel syndrome. Gut 200149743–745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chadwick V S, Chen W, Shu D.et al Activation of the mucosal immune system in irritable bowel syndrome. Gastroenterology 20021221778–1783. [DOI] [PubMed] [Google Scholar]
- 10.Atkinson W, Sheldon T A, Shaath N.et al Food elimination based on IgG antibodies in irritable bowel syndrome: a randomized controlled trial. Gut 2004531459–1464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cebra J J, Periwal S B, Lee G.et al Development and maintenance of the gut‐associated lymphoid tissue (GALT): the roles of enteric bacteria and viruses. Dev Immunol 1998613–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Macpherson A J, Harris N L. Interactions between commensal intestinal bacteria and the immune system. Nat Rev Immunol 20044478–485. [DOI] [PubMed] [Google Scholar]
- 13.King T S, Elia M, Hunter J O. Abnormal colonic fermentation in irritable bowel syndrome. Lancet 19983521187–1189. [DOI] [PubMed] [Google Scholar]
- 14.Madden J A, Hunter J O. A review of the role of the gut microflora in irritable bowel syndrome and the effects of probiotics. Br J Nutr 200288S67–S72. [DOI] [PubMed] [Google Scholar]
- 15.Nobaek S, Johansson M L, Molin G.et al Alteration of intestinal microflora is associated with reduction in abdominal bloating and pain in patients with irritable bowel syndrome. Am J Gastroenterol 2000951231–1238. [DOI] [PubMed] [Google Scholar]
- 16.Pimentel M, Chow E J, Lin H C. Eradication of small intestinal bacterial overgrowth reduces symptoms of irritable bowel syndrome. Am J Gastroenterol 2000953503–3506. [DOI] [PubMed] [Google Scholar]
- 17.Barbara G, Vallance B A, Collins S M. Persistent intestinal neuromuscular dysfunction after acute nematode infection in mice. Gastroenterology 19971131224–1232. [DOI] [PubMed] [Google Scholar]
- 18.Barbara G, De Giorgio R, Deng Y.et al Role of immunologic factors and cyclooxygenase 2 in persistent postinfective enteric muscle dysfunction in mice. Gastroenterology 20011201729–1736. [DOI] [PubMed] [Google Scholar]
- 19.Bercik P, Wang L, Verdú E F.et al Visceral hyperalgesia and intestinal dysmotlity in a mouse model of post infective gut dysfunction. Gastroenterology 2004127179–187. [DOI] [PubMed] [Google Scholar]
- 20.Van Der Waaij D, Sturm C A. Antibiotic decontamination of the digestive tract of mice. Technical procedures. Lab Anim Care 1968181–10. [PubMed] [Google Scholar]
- 21.Van Der Waaij D, Berghuis‐De Vries J M, Korthals Altes C.et al Oral dose and faecal concentrations of antibiotics during antibiotic decontamination in mice and in a patient. J Hyg Camb 197473197–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Verdú E F, Bercik P, Bergonzelli G.et al Lactobacillus paracasei normalizes muscle hypercontractility in a murine model of post‐infective gut dysfunction. Gastroenterology 2004127826–837. [DOI] [PubMed] [Google Scholar]
- 23.Guigoz Y, Rochat F, Perruisseau‐Carrier G.et al Effects of oligosaccharide on the faecal flora and non‐specific immune system in elderly people. Nutr Res 2213–25. [Google Scholar]
- 24.Kamm K, Hoppes S, Breves G.et al Effect of the probiotic yeast Saccaromyces boulardii on the neurochemistry of myenteric neurons in pig jejunum. Neurogastroenterol Motil 20041653–60. [DOI] [PubMed] [Google Scholar]
- 25.Schneider J, Jehle E C, Starlinger M J.et al Neurotransmitter coding of enteric neurons in the submucous plexus is changed in non‐inlfamed rectum of patients with Crohn's disease. Neurogastroenterol Motil 200113255–264. [DOI] [PubMed] [Google Scholar]
- 26.Verdu E F, Deng Y, Bercik P.et al Modulatory effects of estrogen in two murine models of experimental colitis. Am J Physiol Gastrointest Liver Physiol 2002283G27–G36. [DOI] [PubMed] [Google Scholar]
- 27.Barron R P, Benoliel R, Zeltser R.et al Effect of dexamethasone and dypirone on lingual and inferior alveolar nerve hypersensitivity following third molar extractions: preliminary report. J Orofac Pain 20041862–68. [PubMed] [Google Scholar]
- 28.Hong D, Byers M R, Oswald R J. Dexamethasone treatment reduces sensory neuropeptides and nerve sprouting reactions in injured teeth. Pain 199355171–181. [DOI] [PubMed] [Google Scholar]
- 29.Piedimonte G, McDonald D M, Nadel J A. Endopeptidase and kininase II mediate glucocorticoid inhibition of neurogenic inflammation in the rat trachea. J Clin Invest 19918840–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Verma‐Gandhu M, Bercik P, Blennerhassett P.et al Immunodeficiency and visceral hyperalgesia: A putative mechanisms for abdominal pain in AIDS patients. Gastroenterology 2004126A161 [Google Scholar]
- 31.Dobins III W O, Herrero B A, Mansbach C M. Morphologic alterations associated with neomycin induced malabsorption. Am J Med Sci 196825563–77. [DOI] [PubMed] [Google Scholar]
- 32.Rachmilewitz D, Katakura K, Karmeli F.et al Toll‐like receptor 9 signaling mediates the anti‐inflammatory effects of probiotics in murine experimental colitis. Gastroenterology 2004126520–528. [DOI] [PubMed] [Google Scholar]
- 33.Hurst S M, Stanisz A M, Sharkey K A.et al Interleukin 1 beta‐induced increase in substance P in rat myenteric plexus. Gastroenterology 19931051754–1760. [DOI] [PubMed] [Google Scholar]
- 34.Miampamba M, Sharkey K A. Distribution of calcitonin gene‐related peptide, somatostatin, substance P and vasoactive intestinal polypeptide in experimental colitis in rats. Neurogastroenterol Motil 199810315–329. [DOI] [PubMed] [Google Scholar]
- 35.Gonkowski S, Kaminska B, Bossowska A.et al The influence of experimental Bacteroides fragilis infection on substance P and somatostatin‐immunoreactive neural elements in the porcine ascending colon ‐ a preliminary report. Folia Morphol (Warsz) 200362455–457. [PubMed] [Google Scholar]
- 36.Piedimonte G, Rodriguez M M, King K A.et al Respiratory syncytial virus upregulates expression of the substance P receptor in rat lungs. Am J Physiol 1999277L831–L840. [DOI] [PubMed] [Google Scholar]
- 37.Hooper L V, Wong M H, Thelin A.et al Molecular analysis of commensal host‐microbial relationships in the intestine. Science 2001291881–884. [DOI] [PubMed] [Google Scholar]
- 38.Kamm K, Hoppe S, Breves G.et al Effects of the probiotic yeast Saccharomyces boulardii on the neurochemistry of myenteric neurones in pig jejunum. Neurogastroenterol Motil 20041653–60. [DOI] [PubMed] [Google Scholar]
- 39.Tobiassen R O, Sorhaug T, Stepaniak L. Characterization of an intracellular oligopeptidase from Lactobacillus paracasei. Appl Environ Microbiol 1997631284–1287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Huerta C, Garcia Rodriguez L A, Wallander M A.et al Users of oral steroids are at a reduced risk of developing irritable bowel syndrome. Pharmacoepidemiol Drug Saf 200312583–588. [DOI] [PubMed] [Google Scholar]
- 41.Maxwell P R, Rink E, Kumar D.et al Antibiotics increase functional abdominal symptoms. Am J Gastroenterol 200297104–108. [DOI] [PubMed] [Google Scholar]