Abstract
Background
β‐Glucosylceramide, a naturally occurring glycolipid, exerts modulatory effects on natural killer T (NKT) lymphocytes.
Aim
To determine whether β‐glucosylceramide can alter NKT function in opposite directions, colitis was induced by intracolonic installation of trinitrobenzenesulphonic acid, and hepatocellular carcinoma (HCC) was induced by transplantation of Hep3B cells.
Methods
The immunological effect of β‐glucosylceramide was assessed by analysis of intrahepatic and intrasplenic lymphocyte populations, serum cytokines and STAT protein expression.
Results
Administration of β‐glucosylceramide led to alleviation of colitis and to suppression of HCC, manifested by improved survival and decreased tumour volume. The beneficial effects were associated with an opposite immunological effect in the two models: the peripheral:intrahepatic CD4:CD8 lymphocyte ratio increased in the colitis model and decreased in the HCC group. The peripheral:intrahepatic NKT lymphocyte ratio decreased in β‐glucosylceramide‐treated mice solely in the HCC model. The effect of β‐glucosylceramide was associated with decreased STAT1 and 4 expression, and with overexpression of STAT6, along with decreased interferon γ levels in the colitis model, whereas an opposite effect was noted in the HCC model.
Conclusions
β‐glucosylceramide alleviates immunologically incongruous disorders and may be associated with “fine tuning” of immune responses, by changes in plasticity of NKT lymphocytes.
Natural killer T (NKT) cells are innate regulatory lymphocytes that express a conserved T cell receptor, and play an important part in diverse neoplastic, autoimmune and infectious processes.1,2,3 The versatile behaviour of NKT lymphocytes in various conditions, their plasticity, is manifested by a shift between enhanced and suppressed immunity.4,5,6 Combinations of these elements may result in primarily T helper cell (Th)1‐type pro‐inflammatory or Th2‐type anti‐inflammatory responses.7,8
In immune‐mediated disorders, reduced numbers and defective function of NKT lymphocytes were shown in NOD mice.9 In the context of immune‐mediated disease, oral administration of mouse‐derived colitis‐extracted proteins (CEPs) induced amelioration of experimental colitis that was mediated by NKT cells.10 By contrast, in non‐tolerised conditions, NKT cells mediated a pro‐inflammatory response, resulting in aggravation of colitis.11
NKT lymphocyte‐related anti‐tumour activity is associated with a Th1‐type immune response.12,13 NKT cells down regulate tumour immunosurveillance by interleukin (IL)13 production and by signalling via the IL4R‐STAT6 pathway,14 and suppress adaptive immune responses towards skin cancers in vivo.15 These lymphocytes promote rejection of tumours in experimental models of cancer immunotherapy.16,17,18 In a murine hepatocellular carcinoma (HCC) model, NKT lymphocytes were shown to have a role in oral immune regulation and in adoptive transfer of dendritic cells pulsed ex vivo with the same antigens19,20; both methods have led to effective suppression of HCC. NKT lymphocytes have also been implicated in the regulation of autoimmune processes in both mice and humans.21
The target lipid antigens for NKT regulatory lymphocytes have remained elusive. Several synthetic glycolipids, including αGalCer and many of its derivatives, activate NKT cells.22,23 Administration of αGalCer had a protective role in experimental autoimmune encephalomyelitis and collagen‐induced arthritis in mice.24,25 Disease amelioration was associated with a shift in the immune balance from a pathological Th1‐type response towards a protective Th2‐type response. Lysosomal glycosphingolipid, isoglobotrihexosylceramide, was suggested as a natural ligand for NKT cells.26 β‐Glucosylceramide, a naturally occurring glycolipid, is a metabolic intermediate in the pathways of complex glycosphingolipids. Patients with Gaucher's disease, in which accumulation of β‐glucosylceramide occurs owing to glucocerebrosidase deficiency, were shown to have a markedly raised peripheral blood NKT lymphocyte number.27 β‐Glucosylceramide can alter NKT lymphocyte number and function. CD1d‐bound β‐glucosylceramide inhibits NKT cell proliferation in vitro.28 In vivo, administration of β‐glucosylceramide attenuated immune‐mediated liver damage secondary to intravenous administration of concanavalin A to mice, associated with a 20% decrease in the intrahepatic NKT lymphocyte number.32
Despite extensive literature on the role of NKT lymphocytes in apparently incongruous experimental and clinical settings, no studies have directly dealt with the subject of NKT lymphocyte plasticity. We have selected two murine models, HCC and experimental colitis. In the experimental colitis model, β‐glucosylceramide‐mediated disease alleviation was related to attenuation of inflammation and Th2‐type directed immunity. In the HCC model, a beneficial clinical effect resulted from augmentation of inflammatory activity and a Th1‐type immune response.
Materials and methods
Preparation of glycolipids
β‐Glucosylceramide was purchased from Avanti Polar Lipids (Alabaster, Alabama, USA; catalogue number 131304), dissolved in ethanol and emulsified in phosphate‐buffered saline (PBS).
Effect of β‐glucosylceramide on immune‐mediated colitis
Animals
Normal inbred C57/Bl female mice, aged 2–4 months, were obtained from Harlan and maintained in the Animal Core of the Hadassah‐Hebrew University Medical School (Jerusalem, Israel). Mice were maintained on standard laboratory chow and kept in 12‐h light/dark cycles. All animal experiments were approved by the Institutional Committee for Care and Use of Laboratory Animals.
Induction of colitis
Trinitrobenzenesulphonic acid (TNBS) colitis was induced by rectal instillation of TNBS, 1 mg/mouse, dissolved in 100 μl of 50% ethanol as previously described.29
Experimental groups
Four groups of mice were studied (table 1, groups A–D, n = 10 mice per group). Colitis was induced by intracolonic installation of TNBS in groups A and B. Group A mice were treated by daily intraperitoneal administration of 1.5 μg β‐glucosylceramide for 10 days; group B mice received daily intraperitoneal injections of normal saline. Group C and D mice were similarly injected with β‐glucosylceramide (group C) or normal saline (group D), without induction of colitis.
Table 1 Experimental and control groups.
| Group | Mice | Animal model | GC treatment | Follow‐up (days) |
|---|---|---|---|---|
| A | C57/Bl | Colitis | + | 14 |
| B | C57/Bl | Colitis | – | 14 |
| C | C57/Bl | Naive | + | 14 |
| D | C57/Bl | Naive | – | 14 |
| E | Athymic C57/Bl | HCC | + | 54 |
| F | Athymic C57/Bl | HCC | – | 54 |
| G | Athymic C57/Bl | Naive | + | 54 |
| H | Athymic C57/Bl | Naive | – | 54 |
GC, β‐glucosylceramide; HCC, hepatocellular carcinoma.
Clinical assessment of colitis
Body weights were followed up daily throughout the study.
Macroscopic score of colitis
Colitis assessment was performed 14 days after induction of colitis using standard parameters. Four macroscopic parameters were determined—namely, the degree of colonic ulcerations, intestinal and peritoneal adhesions, wall thickness and degree of mucosal oedema. Each parameter was graded on a scale from 0 (completely normal) to 4 (most severe) by two experienced blinded examiners.
Grading of histological lesions
For histological evaluation of inflammation, distal colonic tissue (last 10 cm) was removed and fixed in 10% formaldehyde. Five paraffin‐wax‐embedded sections from each mouse were stained with haematoxylin and eosin using standard techniques. The degree of inflammation on microscopic cross sections of the colon was graded semiquantitatively from 0 to 4: 0, normal with no signs of inflammation; 1, very low level of leucocyte infiltration; 2, low level of leucocyte infiltration; 3, high level of infiltration with high vascular density and bowel wall thickening; and 4, transmural infiltrates with loss of goblet cells, high vascular density, wall thickening and disruption of normal bowel architecture. Grading was performed by two experienced blinded examiners.
Effect of β‐glucosylceramide on hepatocellular carcinoma
Cell cultures
The hepatitis B antigen‐expressing human hepatoma cell line, Hep‐3B, was grown in culture as monolayers, in a medium supplemented with non‐essential amino acids and 10% heat‐inactivated fetal bovine serum.
Mice
Female immunocompetent (heterozygotic) and athymic C57/Bl mice were purchased from Jackson Laboratories, USA. Animals were kept in laminar flow hoods in sterilised cages, and were given irradiated food and sterile acidified water.
Experimental groups
Four groups of recipient mice (10 per group) were studied (table 1, groups E–H). Athymic recipient mice in groups E and F were conditioned with sub‐lethal irradiation (600 cGy) and injected subcutaneously with 107 cells from the Hep3B human hepatoma cell line, as described previously.3 Three days after the injection of tumour cells, mice were transplanted with 1×107 splenocytes from naive donors for reconstitution of their immune system. Mice were treated by daily intraperitoneal injections of β‐glucosylceramide (1.5 μg in 100 μl PBS, group E) or PBS (100 μl, group F); group G and H mice were similarly injected with β‐glucosylceramide (group G) or normal saline (group H), without HCC transplantation.
Follow‐up of tumour growth
Mice were followed up for 8 weeks. Biweekly measurements of tumour volume and body weight were performed.
Effect of β‐glucosylceramide on the systemic immune response in colitis and HCC models
The immune modulatory effect of β‐glucoslyceramide was evaluated by monitoring the following parameters.
FACS analysis for determination of CD4+, CD8+ and NKT lymphocyte subpopulations
Lymphocytes were isolated from mice in all experimental groups (A–H). Splenic lymphocytes and NKT cells were isolated, and red blood cells removed. Intrahepatic lymphocytes were isolated at the end of the study. The inferior vena cava was cut above the diaphragm and the liver was flushed with 5 ml of cold PBS until it became pale. The connective tissue and the gall bladder were removed, and livers were placed in a 10‐ml dish in cold sterile PBS. Livers and spleens were crushed through a stainless steel mesh (size 60, Sigma Chemical, St Louis, Missouri, USA). The cell suspension was placed in a 50‐ml tube for 3 min, washed twice in cold PBS (1250 rpm for 10 min), and debris was removed. Cells were resuspended in PBS, the cell suspension was placed through a nylon mesh presoaked in PBS and unbound cells were collected. Cells were washed twice in 45 ml PBS (1250 rpm at room temperature). For isolation of lymphocytes from liver and spleen, 20 ml of histopague 1077 (Sigma Diagnostics, St Louis, Missouri, USA) was slowly placed underneath the cells suspended in 7 ml of PBS, in a 50‐ml tube. The tube was centrifuged at 1640 rpm for 15 min at room temperature. Cells at the interface were collected, diluted in a 50‐ml tube and washed twice with ice‐cold PBS (1250 rpm for 10 min). Approximately 1×106 cells/mouse liver were recovered. The viability by trypan blue staining was expected to be >95%. Immediately after lymphocyte isolation, triplicates of 2–5×104 cells/500 μl PBS were placed into Falcon 2052 tubes, incubated with 4 ml of 1% bovine serum albumin for 10 min and centrifuged at 1400 rpm for 5 min. Cells were resuspended in 10 μl FCS; for analysis of the different subsets of T lymphocytes, anti‐CD3 antibodies were combined with anti‐NK1.1, anti‐CD4 or anti‐CD8 antibodies (Pharmingen, USA). Analytical cell sorting was performed on 1×104 cells from each group with a fluorescence‐activated cell sorter (FACSTAR plus, Becton Dickinson, Oxnard, California, USA). Data were analysed with the Consort 30 two‐colour contour plot program (Becton Dickinson), and the CELLQuest 25 program.
Cytokine secretion
Supernatant and serum samples were collected from all mice in groups A–D on day 12 and from all mice in groups E–H on day 56. Levels of interferon (IFN)γ, IL4, IL10 and IL12 were measured by “sandwich” ELISA using commercial kits (Genzyme Diagnostics, Massachusetts, USA), according to the manufacturer's instructions.
STAT protein expression
Expression of the transcription factors STAT1, 3, 4, 5 and 6 in splenocytes was determined by western blot analysis of splenocytes harvested from mice in groups A–H. Splenocytes (10×106) were lysed in 100 μl of lysis solution (Sigma). Proteins (100 μg/lane) were resolved by electrophoresis on sodium dodecyl sulphate‐polyacrylamide (7.5%) gels and electroblotted to nitrocellulose membranes (Schleicher & Scuell, Germany). Probing with a polyclonal rabbit anti‐mouse antibody for the different STAT proteins (anti‐STAT1–6 antibodies, Santa Cruz Biotechnology) was followed by addition of horseradish peroxidase‐conjugated goat anti‐rabbit immunoglobulin G (Jackson Immuno Research, Pennsylvania, USA).
Effect of β‐glucoslyceramide on NKT lymphocytes in vitro
In vitro preparation of NKT lymphocytes
In vitro experiments were performed on NKT cells harvested from wild‐type naive Balb/c mice. NKT lymphocytes were separated using anti‐CD3 and anti‐NK beads Magnetic Cell Sorting (MACS, Miltenyl Biotec, Germany). Anti‐CD11c beads were used for separation of dendritic cells. Quadruplicates of 104 NKT lymphocytes were prepared for each of the 12 combinations (table 2). Isolated NKT cells were pulsed ex vivo for 48 h with β‐glucosylceramide, CEP or HCC lysate (10 μg/ml), with or without dendritic cells (1×104 DC/1×106 NKT cells). Supernatants were analysed for IFNγ and IL4 levels (Genzyme Diagnostics). Experiments were performed in quadruplicate to account for biological variability.
Table 2 Cytokine secretion by natural killer T cells in vitro (pg/ml).
| IFNγ (pg/ml) | IL4 (pg/ml) | ||
|---|---|---|---|
| a | NKT | 12 | 11 |
| b | NKT+GC | 15 | 10 |
| b | NKT+CEP | 13 | 7 |
| d | NKT+HCC lysate | 15 | 14 |
| e | NKT+GC+CEP | 13 | 12 |
| f | NKT+GC+HCC lysate | 15 | 17 |
| a1 | NKT+DC | 42 | 38 |
| b1 | NKT+GC+DC | 23 | 18 |
| c1 | NKT+CEP+DC | 44 | 39 |
| d1 | NKT+HCC lysate+DC | 52 | 41 |
| e1 | NKT+GC+CEP+DC | 24 | 20 |
| f1 | NKT+GC+HCC lysate+DC | 19 | 18 |
DC, dendritic cells; GC, β‐glucosylceramide; HCC, hepatocellular carcinoma; IL, interleukin; INF, interferon; NKT, natural killer T lymphocytes.
Preparation of CEP
Colons were removed from mice with TNBS‐induced colitis, cut into small strips and mechanically homogenised. After filtration through a 40‐mm nylon cell strainer, intact cells were spun down and removed. Proteins were quantified by using a protein assay kit (Biorad, Munich, Germany).
Preparation of HCC lysate
To prepare HCC lysate, Hep3B cells were mechanically homogenised. After filtration through a 40‐mm nylon cell strainer, intact cells were spun down and removed. Proteins were quantified using a protein assay kit (Biorad, Munich, Germany).
Statistical analysis
Statistical analysis was performed using Student's t test.
Results
Administration of β‐glucosylceramide exerted a beneficial effect in the two incongruous murine models.
Effect of β‐glucosylceramide on macroscopic score of colitis
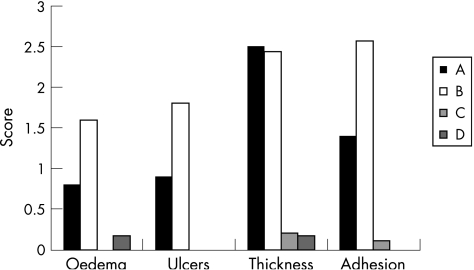
Administration of β‐glucosylceramide alleviated three of the four tested macroscopic parameters of colitis. The mucosal oedema score improved from 1.6 to 0.8, colonic ulceration score improved from 1.8 to 0.9 and adhesions score from 2.5 to 1.4 (for groups B v A, respectively, p<0.005). No significant effect was noted with respect to wall thickness (fig 1).
Figure 1 Effect of β‐glucosylceramide on macroscopic score of colitis: administration of β‐glucosylceramide alleviated three of the four tested macroscopic parameters of colitis (group A in the black bars).
Effect of β‐glucosylceramide on microscopic score of colitis
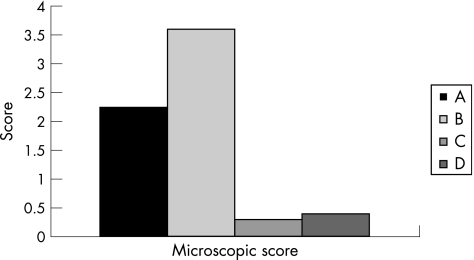
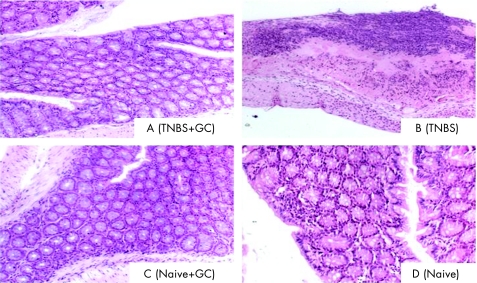
Five sections were evaluated from each mouse in each of the experimental and control groups. Administration of β‐glucosylceramide had a significant effect on the microscopic score of colitis (3.6 for group B v 2.25 for group A; fig 2; p<0.005). Figure 3 shows a representative figure from each group, manifesting significant amelioration of the inflammation and preservation of the normal mucosa, as compared with marked inflammatory response throughout all the layers of the mucosa and destruction of villi noted in untreated controls. β‐Glucosylceramide had no effect on the naive controls in group C, and no inflammatory response was noted in these mice.
Figure 2 Effect of β‐glucosylceramide on the microscopic score of colitis: significant improvement in all histological parameters was noted in β‐glucosylceramide treated animals in the black bars.
Figure 3 Effect of β‐glucosylceramide on bowel histology (×10): a representative figure from each group is shown; significant amelioration of the inflammation and preservation of the normal mucosa are seen in β‐glucosylceramide‐treated mice in group A, as compared with marked inflammatory response throughout all mucosa layers and destruction of villi in untreated controls.
Effect of β‐glucosylceramide on survival of tumour‐bearing mice
β‐Glucosylceramide had a beneficial effect on tumour‐bearing mice in the HCC experimental model. During the 8‐week follow‐up period, mortality decreased from 40% in control animals (group B) to 0% in β‐glucosylceramide‐treated animals (group A; fig 4).
Figure 4 Effect of β‐glucosylceramide on survival of tumour‐bearing mice: during the 8‐week follow‐up period, mortality decreased from 40% in control animals to 0% in β‐glucosylceramide‐treated animals (black bars).
Effect of β‐glucosylceramide on percentage of tumour development and tumour volume
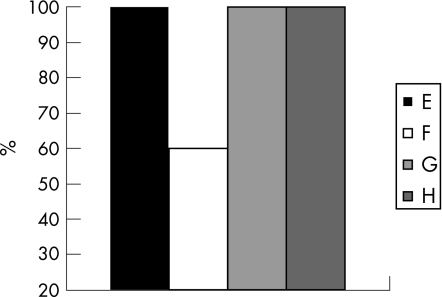
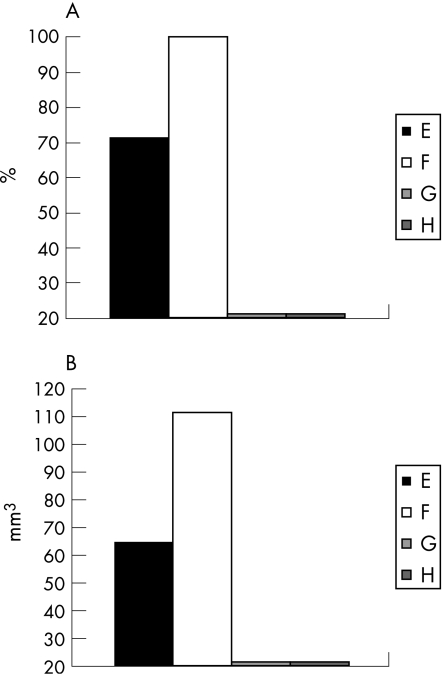
As compared with 100% of control animals that developed tumours, only 70% of β‐glucosylceramide‐treated animals developed noticeable tumours (fig 5A; p<0.005). Tumour volumes, measured in animals that developed them, decreased from 110 mm3 in group B to 72 mm3 in group A (fig 5B; p<0.005).
Figure 5 Effect of β‐glucosylceramide on percentage of tumour development (A) and tumour volume (B). As compared with 100% of control animals who developed tumours, only 70% of β‐glucosylceramide‐treated animals developed noticeable tumours (A). Tumour volumes decreased from 110 mm3 in group B to 72 mm3 in group A (B).
Effect of β‐glucosylceramide on CD4 and CD8 lymphocyte distribution
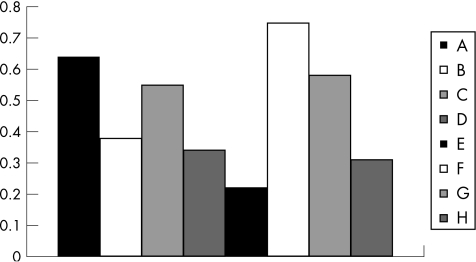
The beneficial effects of β‐glucosylceramide in the two models were associated with opposite effects on intrahepatic and peripheral lymphocyte distribution (fig 6). CD4:CD8 lymphocyte ratios were calculated for each of the eight experimental and control groups, for both splenic and intrahepatic lymphocytes. A general ratio between each of the splenic‐to‐intrahepatic CD4:CD8 ratios was calculated (fig 6). In the experimental colitis model, β‐glucosylceramide‐mediated alleviation of colitis was associated with increased ratio (0.64 for group A v 0.38 for group B; p<0.005), suggesting an increased intrahepatic CD8 lymphocyte trapping. By contrast, the anti‐tumour effect of β‐glucosylceramide in the HCC model was associated with a decreased ratio (0.22 for group E v 0.75 for group F; p<0.005), suggesting a decreased intrahepatic CD8 lymphocyte trapping. In naive animals (groups C and G), administration of β‐glucosylceramide led to an increase in the ratio (0.55 for group C v 0.34 for group D, and 0.58 for group G v 0.31 for group H).
Figure 6 Effect of β‐glucosylceramide on CD4 and CD8 lymphocyte distribution: the beneficial effects of β‐glucosylceramide in the two models were associated with opposite effects on intrahepatic and peripheral lymphocyte distribution. In the experimental colitis model, β‐glucosylceramide‐mediated alleviation of colitis was associated with an increased splenic‐to‐intrahepatic CD4:CD8 ratio (groups A and B), suggesting an increased intrahepatic CD8 lymphocyte trapping. By contrast, the anti‐tumour effect of β‐glucosylceramide in the HCC model was associated with a decreased ratio (groups E and F), suggesting a decreased intrahepatic CD8 lymphocyte trapping. In naive animals (groups C and G), administration of β‐glucosylceramide led to an increase in the ratio (groups C v D, and G v H).
Effect of β‐glucosylceramide on NKT lymphocyte distribution
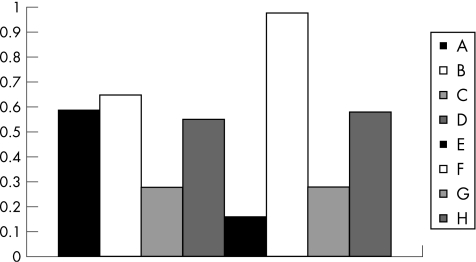
The anti‐inflammatory effect of β‐glucosylceramide in the experimental colitis model was associated with a mild increase of the intrahepatic NKT cell population. The peripheral‐to‐intrahepatic NKT lymphocyte ratio decreased from 0.65 for group B to 0.59 for group A (fig 7; p<0.005). By contrast, the anti‐tumour effect of β‐glucosylceramide in the HCC model was associated with a marked effect on NKT lymphocyte distribution. The peripheral‐to‐intrahepatic NKT ratio decreased significantly from 0.98 for group F to 0.16 for group E, suggesting a significant increase in intrahepatic NKT lymphocytes (fig 7; p<0.005). In both naive controls, administration of β‐glucosylceramide was associated with a decreased ratio, suggesting a relative increase in intrahepatic NKT cell population (0.27 for group C v 0.55 for group D, and 0.28 for group G v 0.58 for group H; fig 7; p<0.005 for both).
Figure 7 Effect of β‐glucosylceramide on NKT lymphocyte distribution: the peripheral‐to‐intrahepatic NKT lymphocyte ratio mildly decreased in group A mice. By contrast, the anti‐tumour effect of β‐glucosylceramide in the HCC model was associated with a marked effect on NKT lymphocyte distribution. The peripheral‐to‐intrahepatic NKT ratio significantly decreased in group E, suggesting a significant increase in intrahepatic NKT lymphocytes. In both naive controls, administration of β‐glucosylceramide was associated with a decreased ratio, suggesting a relative increase in intrahepatic NKT cells (groups C and D, and G and H).
Effect of β‐glucosylceramide on serum cytokines: IFNγ and IL10
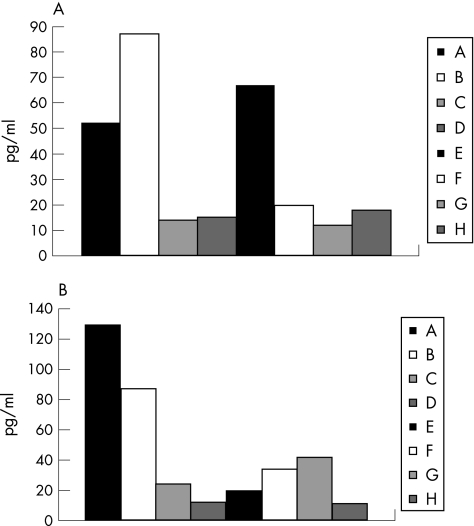
In the experimental colitis model, disease alleviation and attenuation of inflammation was associated with Th2‐type directed immunity. Serum levels of IFNγ decreased in β‐glucosylceramide‐treated animals in the colitis model from 87 pg/ml in group B to 52 pg/ml in group A (fig 8A; p<0.005). By contrast, in the HCC model, the β‐glucosylceramide‐mediated beneficial clinical effect was associated with augmentation of inflammatory activity and a Th1‐type immune response, manifested by a significant increase in IFNγ serum levels from 20 pg/ml in group F to 67 pg/ml in group E (fig 8A; p<0.005). An opposite effect was noted for IL10 serum levels. β‐Glucosylceramide‐mediated alleviation of colitis was associated with increased IL10 levels (130 pg/ml in group A v 87 pg/ml in group B; fig 8B; p<0.005). In the HCC model, a non‐significant decrease was noted (20 pg/ml in group E v 37 pg/ml in group F; fig 8B).
Figure 8 Effect of β‐glucosylceramide on serum cytokines: interferon (IFN)γ (A) and interleukin (IL)10 (B). Serum levels of IFNγ decreased in β‐glucosylceramide‐treated animals in the colitis model (group A). By contrast, in the HCC model, the β‐glucosylceramide‐mediated beneficial clinical effect was associated with augmentation of inflammatory activity manifested by a significant increase in IFNγ serum levels (group E). An opposite effect was noted for IL10 serum levels. β‐Glucosylceramide‐mediated alleviation of the colitis was associated with increased IL10 levels (group A). In the HCC model, a non‐significant decrease was noted in group E.
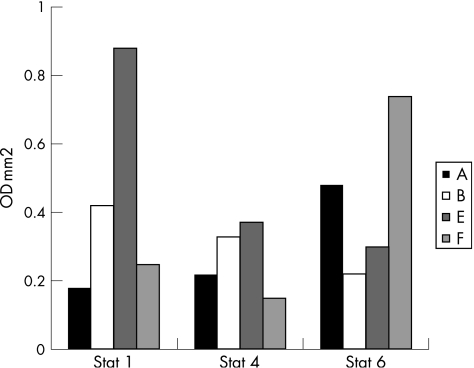
Effect of β‐glucosylceramide on STAT protein expression
β‐Glucosylceramide treatment in experimental colitis was associated with decreased STAT1 and STAT4 expression and increased STAT6 expression in group A versus untreated mice in group B (0.18 v 0.42, 0.22 v 0.33 and 0.48 v 0.33, optical density×mm2, for STAT1, 4 and 6, for groups A and B, respectively; fig 9). An opposite effect on STAT protein expression was noted in the HCC model. β‐Glucosylceramide‐treated animals in group E manifested increased STAT1 and 4 expression and a relative decrease in STAT6 expression (0.88 v 0.25, 0.37 v 0.5 and 0.3 v 0.74, optical density×mm2, for STAT1, 4 and 6, for groups E and F, respectively; fig 9). These results correspond to overexpression of IL4, and decreased expression of IFN and IL12 in the experimental colitis model, and to overexpression of IFN and IL12, with decreased IL4 expression in the HCC model. No significant effect of β‐glucosylceramide was noted in naive animals (groups C and G).
Figure 9 Effect of β‐glucosylceramide on STAT protein expression: β‐glucosylceramide treatment in experimental colitis was associated with decreased STAT1 and 4 expression and increased STAT6 expression in group A versus untreated mice in group B. An opposite effect on STAT protein expression was noted in the hepatocellular carcinoma (HCC) model. β‐Glucosylceramide‐treated animals in group E manifested increased STAT1 and 4 expression and a relative decrease in STAT6 expression.
Effect of β‐glucosylceramide on in vitro cytokine production by NKT cells
To determine the ex vivo effect of β‐glucosylceramide, NKT lymphocytes were exposed to β‐glucosylceramide in the presence or absence of dendritic cells and disease target proteins. The effect of β‐glucosylceramide on IL4 and IFNγ secretion in vitro was enhanced in the presence of dendritic cells, but no significant differences were noted when target antigens, colitis‐extracted proteins or HCC lysate were added (table 2).
Discussion
Administration of β‐glucosylceramide alleviated immune‐mediated experimental colitis, improving the macroscopic and microscopic scores, and suppressed HCC tumour growth, decreasing tumour volume and improving survival. The beneficial effects of β‐glucosylceramide were associated with opposite immunological effects in the two models. In the Th1‐mediated colitis model, β‐glucosylceramide led to an increased peripheral/intrahepatic CD4:CD8 lymphocyte ratio, and to a Th2 immune shift. By contrast, the anti‐tumour effect of β‐glucosylceramide was associated with a decreased peripheral to intrahepatic CD4:CD8 ratio and a Th1 immune shift.
The inherent plasticity of NKT cells is an important requirement for their function as regulatory lymphocytes.29 NKT cells are able to produce both Th1‐type and Th2‐type cytokines simultaneously after stimulation in vivo, an unusual characteristic that at face value seems paradoxical.30
The data shown in this study further support the notion that in keeping with this unique cytokine pattern, NKT cells “can go both ways”, as their activation polarised the immune response in a Th1 direction, or in a Th2 response. In the experimental colitis model, β‐glucosylceramide‐mediated disease alleviation and attenuation of inflammation were associated with Th2‐type immunity. β‐Glucosylceramide treatment led to decreased STAT1 and 4 expression and increased STAT6 expression, which corresponded to decreased IFNγ and increased IL10 serum levels. An opposite effect was noted in the HCC model. β‐Glucosylceramide‐mediated tumour suppression was associated with augmentation of inflammatory activity and a Th1‐type immune response, manifested by a significant increase in STAT1 and 4 expression, and increased IFNγ serum levels. STAT1 and 4 play an important part in the regulation of IFNγ and IL12 cytokine production at a transcriptional level.31 STAT6 is involved in signal transduction of the anti‐inflammatory cytokine IL4, and NKT cells have been shown to down regulate tumour immunosurveillance by signalling through the IL4R–STAT6 pathway. The decrease in STAT6 expression and consequently in IL4 levels in the HCC model, may have induced the beneficial immunosurveillance and tumour inhibition.
NKT cells are responsible for the promotion or maintenance of immunological tolerance without intentional exogenous stimulation.33 Their plasticity is manifested by the fact that in some cases NKT cells promote autoimmunity, even in the absence of α‐GalCer stimulation.34 NKT lymphocytes are reduced in NOD mice, and adoptive transfer of these cells reduced disease progression.35,36 αGalCer was effective in preventing experimental autoimmune encephalomyelitis by shifting the balance from a Th1 to a Th2 response to central nervous system antigens.37,38 CD1d deficiency has been associated with exacerbated lupus in lupus‐prone MRL lpr/lpr mice.39,40 Compared with these disease‐preventing influences, NKT cells exert an opposite effect, and promote autoimmunity in (NZB/NZW) F1 mice,41,34 and in the OVA airway hypersensitivity model.42,43 In atherosclerosis‐prone apoE–/– mice crossed with CD1d–/– mice, lesion sizes in the arteries were decreased compared with controls.44 In the experimental colitis model, NKT lymphocytes play a dual part: in the presence of peripheral tolerance, they were accountable for keeping a high CD4+IL4+:CD4+IFNγ+ ratio and disease alleviation. However, in non‐tolerised conditions, they induced a pro‐inflammatory shift.7
In tumour models, NKT cells act to up regulate the immune response, a type of regulation not observed with CD25+CD4+ T regulatory cells, which are uniformly suppressive.34 NKT lymphocytes were necessary for IL12‐mediated tumour treatment in mice.9 αGalCer promotes NKT cell‐dependent rejection of a broad range of experimental tumour lines,9,14 and protects against spontaneous, carcinogen‐induced primary tumour formation.45,46 Compounding the paradox of the differential immune activity mediated by NKT cells, in other mouse models, they play a suppressive part. Experimental tumours –15–12RM and 4T1 were rejected in CD1d–/– mice, but grew in wild‐type mice.47,48 Similarly, NKT cells can operate in either a stimulating or suppressive fashion in the setting of infectious disorders. Clearance of herpes simplex virus type 1 is reduced in mice deficient in NKT cells.49 Salmonella infection leads to an altered environment, where NKT cells are stimulated by inflammatory cytokines induced in response to infection.50 By contrast, NKT cells negatively regulate the response to lymphocytic choriomeningitis virus.51
Our data further support the paradoxical function of NKT cells in immunoregulation.52 The beneficial effect of β‐glucosylceramide was associated with a significant decrease in the peripheral‐to‐intrahepatic NKT lymphocyte distribution in the HCC model, but not in the colitis model. A similar opposing effect was noted on CD4 and CD8 lymphocyte distribution. Activation of CD8+ T cells in the periphery is often accompanied by lymphocyte trafficking to the liver, where T cell trapping and Fas‐mediated apoptosis occur.53 The splenic‐to‐intrahepatic CD4:CD8 ratio was increased in the experimental colitis model, suggesting an increased intrahepatic CD8 lymphocyte trapping. By contrast, the anti‐tumour effect of β‐glucosylceramide was associated with a decreased ratio, suggesting an increased peripheral CD8+‐mediated anti‐tumour effect.
NKT plasticity can be maintained as an intrinsic property, endowing these cells with immunoregulatory properties.34 In this study, no correlation was shown between the in vivo effects of β‐glucosylceramide and its in vitro effect on naive NKT cells. These findings suggest that the plasticity of these cells is dependent on the immune environment. The diverse role of NKT cells in different settings may reflect the existence of several subpopulations of cells rather than true plasticity of the same cell type.54 NKT cells include at least two subsets, distinguishable as CD4+ or CD4–, which may be functionally distinct.55,56 Conversely, cells with similar phenotypes may exert a site‐dependent effect. NKT cells from the liver, thymus and spleen exert different abilities to mediate rejection of the sarcoma cell line.57 NKT cells might produce pro‐inflammatory or anti‐inflammatory cytokines, depending on the type of signals they receive. IL12 or anti‐NK1.1 cross linking has been shown to preferentially induce IFN production, whereas IL7 promotes IL4 production.58 The OCH analogue of αGalCer preferentially induces Th2 responses, whereas the C glycoside preferentially induces Th1 cytokines.34,59 The type of APC may skew NKT activation. When αGalCer was loaded on to dendritic cells, mice developed a stronger response with sustained IFNγ production.60
In this study, the same ligand, β‐glucosylceramide, was able to activate NKT cells to induce both Th1 and Th2 immune responses. β‐Glucosylceramide may activate or suppress different subtypes of NKT cells activated in the two models. Alternatively, β‐glucosylceramide may directly activate or inactivate different receptors on the same cell. β‐Glucosylceramide may have opposing effects on the same cells that are being activated differently in various immune environments, so that its effect is dependent on the type of NKT cell polarisation (eg, towards a Th1 or Th2 immune response). Another possible mechanism relates to displacement of a natural activating ligand from the CD1d molecule. Finally, β‐glucosylceramide may alter the function of NKT or effector T cells, via a direct effect on dendritic cells.30
In summary, NKT cells represent highly potent immunoregulatory cells, with a conserved specificity that makes them attractive targets for immunotherapy of autoimmune diseases and malignancy. The identification and characterisation of the various molecules or signals that lead to their activation is an important goal to allow a more precise manipulation of their activity. The results of our study, in which β‐glucosylceramide ameliorated immunologically incongruous disorders suggests that β‐glucosylceramide can serve as a possible “fine tuner” of immune responses via changes in NKT lymphocyte plasticity, making it a potential novel form of immunotherapy.
Abbreviations
CEP - colitis‐extracted protein
HCC - hepatocellular carcinoma
IFN - interferon
NKT lymphocyte - natural killer T lymphocyte
PBS - phosphate‐buffered saline
TNBS - trinitrobenzenesulphonic acid
Footnotes
Funding: This work was supported in part by the Roaman‐Epstein Liver Research Foundation (to YI).
Competing interests: None.
References
- 1.Godfrey D I, MacDonald H R, Kronenberg M.et al NKT cells: what's in a name? Nat Rev Immunol 20044231–237. [DOI] [PubMed] [Google Scholar]
- 2.Godfrey D I, Hammond K J, Poulton L D.et al NKT cells: facts, functions and fallacies. Immunol Today 200021573–583. [DOI] [PubMed] [Google Scholar]
- 3.Moody D B, Zajonc D M, Wilson I A. Anatomy of CD1‐lipid antigen complexes. Nat Rev Immunol 20055387–399. [DOI] [PubMed] [Google Scholar]
- 4.Kronenberg M, Gapin L. The unconventional lifestyle of NKT cells. Nat Rev Immunol 20022557–568. [DOI] [PubMed] [Google Scholar]
- 5.Hammond K J L, Pellicci D G, Poulton L D.et al NKT cells are phenotypically and functionally diverse. Eur J Immunol 1999293768–3781. [DOI] [PubMed] [Google Scholar]
- 6.Kronenberg M, Hammond K J L. Natural killer T cells: natural or unnatural regulators of autoimmunity? Curr Opinion Immunol 200315683–689. [DOI] [PubMed] [Google Scholar]
- 7.Trop S, Ilan Y. NK1.1+ T cell: a two‐faced lymphocyte in immune modulation of the IL4/IFN‐g paradigm. J Clin Immunol 200222270–280. [DOI] [PubMed] [Google Scholar]
- 8.Hammond K J, Godfrey D I. NKT cells: potential targets for autoimmune disease therapy? Tissue Antigens 200259353–363. [DOI] [PubMed] [Google Scholar]
- 9.Berzins S P, Kyparissoudis K, Pellicci D G.et al Systemic NKT cell deficiency in NOD mice is not detected in peripheral blood: implications for human studies. Immunol Cell Biol 200482247–252. [DOI] [PubMed] [Google Scholar]
- 10.Trop S, Samsonov D, Gotsman I.et al Liver associated lymphocytes expressing NK1.1 are essential for oral tolerance induction in a murine model. Hepatology 199929746–755. [DOI] [PubMed] [Google Scholar]
- 11.Margalit M, Ilan Y. Induction of immune tolerance: a role for natural killer T lymphocytes? Liver Int 200525501–504. [DOI] [PubMed] [Google Scholar]
- 12.Smyth M J, Crowe N Y, Hayakawa Y.et al NKT cells—conductors of tumor immunity? Curr Opin Immunol 200214165–171. [DOI] [PubMed] [Google Scholar]
- 13.Ostrand‐Rosenberg S, Sinha P, Clements V.et al Signal transducer and activator of transcription 6 (Stat6) and CD1: inhibitors of immunosurveillance against primary tumors and metastatic disease. Cancer Immunol Immunother 20045386–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Terabe M, Matsui S, Noben‐Trauth N.et al NKT cell‐mediated repression of tumor immunosurveillance by IL‐13 and the IL‐4R‐STAT6 pathway. Nat Immunol 20001515–520. [DOI] [PubMed] [Google Scholar]
- 15.Moodycliffe A M, Nghiem D, Clydesdale G.et al Immune suppression and skin cancer development: regulation by NKT cells. Nat Immunol 20001521–525. [DOI] [PubMed] [Google Scholar]
- 16.Kawano T, Cui J, Koezuka Y.et al Natural killer‐like non‐specific tumor cell lysis mediated by specific ligand‐activated Valpha14 NKT cells. Proc Natl Acad Sci USA 955690–5698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kawano T, Nakayama T, Kamada N.et al Antitumor cytotoxicity mediated by ligand‐activated human Valpha24 NKT cells. Cancer Res 1999595102–5112. [PubMed] [Google Scholar]
- 18.Cui J, Shin T, Kawano T.et al Requirement for Valpha14 NKT cells in IL‐12 mediated rejection of tumors. Science2781623–1625. [DOI] [PubMed] [Google Scholar]
- 19.Shibolet O, Alper R, Zolotarov L.et al Suppression of hepatocellular carcinoma via oral immune regulation towards tumor associated antigens is associated with increased NKT and CD8+ lymphocytes. Oncology 200466323–330. [DOI] [PubMed] [Google Scholar]
- 20.Shibolet O, Alper R, Zolotarov L.et al NKT and CD8 lymphocytes mediate suppression of hepatocellular carcinoma growth via tumor antigen‐pulsed dendritic cells. Int J Cancer 2003106236–242. [DOI] [PubMed] [Google Scholar]
- 21.Van Kaer L. Natural killer T cells as targets for immunotherapy of autoimmune diseases. Immunol Cell Biol 200482315–322. [DOI] [PubMed] [Google Scholar]
- 22.Stanic A K, De Silva A D, Park J J.et al Defective presentation of the CD1d‐restricted natural Va14Ja18 NKT lymphocyte antigen caused by β‐D‐glucosylceramide synthase deficiency. Proc Natl Acad Sci USA 20031001849–1854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Parekh V V, Singh A K, Wilson M T.et al Quantitative and qualitative differences in the in vivo response of NKT cells to distinct alpha‐ and beta‐anomeric glycolipids. J Immunol 200415173, 3693–173, 3706. [DOI] [PubMed] [Google Scholar]
- 24.Furlan R, Bergami A, Cantarella D.et al Activation of invariant NKT cells by alphaGalCer administration protects mice from MOG35‐55‐induced EAE: critical roles for administration route and IFN‐gamma. Eur J Immunol 2003331830–1838. [DOI] [PubMed] [Google Scholar]
- 25.Chiba A, Oki S, Miyamoto K.et al Suppression of collagen‐induced arthritis by natural killer T cell activation with OCH, a sphingosine‐truncated analog of alpha‐galactosylceramide. Arthritis Rheum 200450305–313. [DOI] [PubMed] [Google Scholar]
- 26.Zhou D, Mattner J, Cantu C I I I.et al Lysosoma glycosphingolipid recognition by NKT cells. Science 20043061786–1789. [DOI] [PubMed] [Google Scholar]
- 27.Margalit M, Zolotarov L, Hemed N.et al Glucocerebroside induces altered anti hepatitis C immunity in patients with Gaucher's disease. Hepatology 200338490A [Google Scholar]
- 28.Margalit M, Abu Ghazala S, Alper R.et al Glucocerebroside treatment ameliorates conA hepatitis by inhibition of NKT lymphocytes. Am J Physiol 2005289G917–G925. [DOI] [PubMed] [Google Scholar]
- 30.Shlomai A, Trop A, Gotsman I.et al Immunemodulation of experimental colitis: the role of NK1.1+ liver lymphocytes and surrogate antigen‐bystander effect. J Pathol 2001195498–507. [DOI] [PubMed] [Google Scholar]
- 31.Miyake S, Yamamura T. Therapeutic potential of glycolipid ligands for natural killer (NK) T cells in the suppression of autoimmune diseases. Curr Drug Targets Immune Endocr Metabol Disord 20055315–322. [DOI] [PubMed] [Google Scholar]
- 32.Yu K O, Porcelli S A. The diverse functions of CD1d‐restricted NKT cells and their potential for immunotherapy. Immunol Lett 200510042–55. [DOI] [PubMed] [Google Scholar]
- 29.Rengarajan J, Szabo S J, Glimcher L H. Transcriptional regulation of Th1/Th2 polarization. Immunol Today 200021479–483. [DOI] [PubMed] [Google Scholar]
- 33.Berzins S P, Smyth M J, Godfrey D I. Working with NKT cells—pitfalls and practicalities. Curr Opin Immunol 200517448–454. [DOI] [PubMed] [Google Scholar]
- 34.Van Kaer L, Joyce S. Innate immunity: NKT cells in the spotlight. Curr Biol 200515R429–R431. [DOI] [PubMed] [Google Scholar]
- 35.Naumov Y N, Bahjot K S, Gausling R.et al Activation of CD1d‐restricted T cells protects NOD mice from developing diabetes by regulating dendritic cell subsets. Proc Natl Acad Sci USA 20019813838–13843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sharif S, Arreaza G A, Zucker P.et al Activation of natural killer T cells by alpha‐galactosylceramide treatment prevents the onset and recurrence of autoimmune type 1 diabetes. Nat Med 200171057–1062. [DOI] [PubMed] [Google Scholar]
- 37.Furlan R, Bergami A, Cantarella D.et al Activation of invariant NKT cells by alphaGalCer administration protects mice from MOG35‐55‐induced EAE: critical roles for administration route and IFN‐gamma. Eur J Immunol 2003331830–1838. [DOI] [PubMed] [Google Scholar]
- 38.Miyamoto K, Miyake S, Yamamura T. A synthetic glycolipid prevents autoimmune encephalomyelitis by inducing TH2 bias of natural killer T cells. Nature 2001413531–534. [DOI] [PubMed] [Google Scholar]
- 39.Yang J Q, Singh A K, Wilson M T.et al Immunoregulatory role of CD1d in the hydrocarbon oil‐induced model of lupus nephritis. J Immunol 20031712142–2153. [DOI] [PubMed] [Google Scholar]
- 40.Yang J Q, Saxena V, Xu H.et al Repeated alpha‐galactosylceramide administration results in expansion of NK T cells and alleviates inflammatory dermatitis in MRL‐lpr/lpr mice. J Immunol 20031714439–4446. [DOI] [PubMed] [Google Scholar]
- 41.Zeng D, Liu Y, Sidobre S.et al Activation of natural killer T cells in NZB/W mice induces Th1‐type immune responses exacerbating lupus. J Clin Invest 20031121211–1222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lisbonne M, Diem S, de Castro Keller A.et al Cutting edge: invariant Valpha14 NKT cells are required for allergen‐induced airway inflammation and hyperreactivity in an experimental asthma model. J Immunol 20031711637–1641. [DOI] [PubMed] [Google Scholar]
- 43.Campos R A, Szczepanik M, Itakura M.et al Cutaneous immunization rapidly activates liver invariant Valpha14 NKT cells stimulating B‐1 B cells to initiate T cell recruitment for elicitation of contact sensitivity. J Exp Med 20031981785–1796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tupin E, Nicoletti A, Elhage R.et al CD1d‐dependent activation of NKT cells aggravates atherosclerosis. J Exp Med 2004199417–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hayakawa Y, Rovero S, Forni G.et al Alpha‐galactosylceramide (KRN7000) suppression of chemical‐ and oncogene‐dependent carcinogenesis. Proc Natl Acad Sci USA 20031009464–9469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Schmieg J, Yang G, Franck R W.et al Superior protection against malaria and melanoma metastases by a C‐glycoside analogue of the natural killer T cell ligand alpha‐galactosylceramide. J Exp Med 20031981631–1641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Smyth M J, Thia K Y, Street S E.et al Differential tumor surveillance by natural killer (NK) and NKT cells. J Exp Med 2000191661–668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Terabe M, Matsui S, Park J M.et al Transforming growth factor‐beta production and myeloid cells are an effector mechanism through which CD1d‐restricted T cells block cytotoxic T lymphocyte‐mediated tumor immunosurveillance: abrogation prevents tumor recurrence. J Exp Med 20031981741–1752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Grubor‐Bauk B, Simmons A, Mayrhofer G.et al Impaired clearance of herpes simplex virus type 1 from mice lacking CD1d or NKT cells expressing the semivariant V alpha 14‐J alpha 281 TCR. J Immunol 20031701430–1434. [DOI] [PubMed] [Google Scholar]
- 50.Brigl M, Bry L, Kent S C.et al Mechanism of CD1d‐restricted natural killer T cell activation during microbial infection. Nat Immunol 200341230–1237. [DOI] [PubMed] [Google Scholar]
- 51.Roberts T J, Lin Y, Spence P M.et al CD1d1‐dependent control of the magnitude of an acute antiviral immune response. J Immunol 20041723454–3461. [DOI] [PubMed] [Google Scholar]
- 52.Exley M A, He Q, Cheng O.et al Cutting edge: compartmentalization of Th1‐like noninvariant CD1d‐reactive T cells in hepatitis C virus‐infected liver. J Immunol 20021681519–1523. [DOI] [PubMed] [Google Scholar]
- 53.Park S, Murray D, John B.et al Biology and significance of T‐cell apoptosis in the liver. Immunol Cell Biol 20028074–83. [DOI] [PubMed] [Google Scholar]
- 54.van der Vliet H J, Molling J W, von Blomberg B M.et al The immunoregulatory role of CD1d‐restricted natural killer T cells in disease. Clin Immunol 20041128–23. [DOI] [PubMed] [Google Scholar]
- 55.Prussin C, Foster B. TCR V‐alpha‐24 and V‐beta‐11 coexpression defines a human NK1 T cell analog containing a unique Th0 subpopulation. J Immunol 19971595862–5870. [PubMed] [Google Scholar]
- 56.Eberl G, Lees R, Smiley S T.et al Tissue‐specific segregation of CD1d‐dependent and CD1d‐independent NK T cells. J Immunol 19991626410–6419. [PubMed] [Google Scholar]
- 57.Crowe N Y, Coquet J M, Berzins S P.et al Differential antitumor immunity mediated by NKT cell subsets in vivo. J Exp Med 20052021279–1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Godfrey D I, Hammond K J, Poulton L D.et al NKT cells: facts, functions and fallacies. Immunol Today 200021573–583. [DOI] [PubMed] [Google Scholar]
- 59.Stetson D B, Mohr M, Reinhardt R L.et al Constitutive cytokine mRNAs mark natural killer (NK) and NK T cells poised for rapid effector function. J Exp Med 20031981069–1076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fujii S, Shimizu K, Kronenberg M.et al Prolonged IFN‐gamma‐producing NKT response induced with alpha‐galactosylceramide‐loaded DCs. Nat Immunol 20023867–874. [DOI] [PubMed] [Google Scholar]