Abstract
Background
Numerous studies examining the prevalence and natural history of dyspepsia in the general population have been conducted. However, few have reported the effect of quality of life on the development of dyspepsia. A 10‐year longitudinal follow‐up study examining the effect of quality of life on subsequent dyspepsia was performed.
Methods
Individuals originally enrolled in a population‐screening programme for Helicobacter pylori were contacted through a validated postal dyspepsia questionnaire. Baseline demographic data, quality of life at original study entry, and dyspepsia and irritable bowel syndrome (IBS) symptom data were already on file. Consent to examine primary‐care records was sought, and data regarding non‐steroidal anti‐inflammatory drugs (NSAID) and aspirin use were obtained from these.
Results
Of 8407 individuals originally involved, 3912 (46.5%) provided symptom data at baseline and 10‐year follow‐up. Of 2550 (65%) individuals asymptomatic at study entry, 717 (28%) developed new‐onset dyspepsia at 10 years, an incidence of 2.8% per year. After multivariate logistic regression, lower quality of life at study entry (OR 2.63; 99% CI 1.86 to 3.71), higher body mass index (OR per unit 1.05; 99% CI 1.02 to 1.08), presence of IBS at study entry (OR 3.1; 99% CI 1.51 to 6.37) and use of NSAIDs and/or aspirin (OR 1.32; 99% CI 0.99 to 1.75) were significant risk factors for new‐onset dyspepsia.
Conclusions
The incidence of new‐onset dyspepsia was almost 3% per year. Low quality of life at baseline exerted a strong effect on the likelihood of developing dyspepsia at 10 years.
There have been numerous cross‐sectional surveys and longitudinal studies examining the population prevalence of dyspepsia,1,2,3,4,5,6,7 which varies according to whether a broad definition of dyspepsia is used, in line with the 1988 working party report,8 or whether the more narrow classification, excluding those individuals with heartburn or acid reflux, espoused by the Rome consensus is taken.9 However, published data regarding the incidence of new‐onset dyspepsia are scarce.
This information is important, as are data on the effect of sociodemographic variables in determining the acquisition rates of dyspepsia. One topic that would be particularly interesting to study is the effect of quality of life on dyspepsia. A recent systematic review highlighted that there have been few studies of dyspepsia and health‐related quality of life conducted among samples of the general population.10 However, several studies report a reduced quality of life in patients with functional dyspepsia attending secondary care compared with non‐symptomatic individuals, healthy controls or the general population,11,12,13,14 but it is not clear from these whether dyspepsia symptoms cause a reduced quality of life or whether individuals with a poor quality of life develop dyspepsia symptoms. Cross‐sectional surveys suffer constraints imposed by their design, as they can only examine associations at one particular point in time, whereas longitudinal surveys can determine whether a particular factor pre‐dates the acquisition of dyspepsia.
This longitudinal study follows up a large group of individuals from the community over 10 years, and has been conducted in an attempt to deal with the lack of information regarding the incidence of new‐onset dyspepsia, as well as to assess how numerous social and demographic factors may influence this, particularly the effect of a poor quality of life at baseline on the development of subsequent dyspepsia.
Methods
Participants and setting
This study was a 10‐year follow‐up of individuals, now aged between 50 and 59 years, involved in the recruitment phase of a randomised controlled trial of population Helicobacter pylori screening and treatment conducted in Leeds and Bradford, both cities in West Yorkshire, Northern England, with a combined population of approximately 1 200 000. The study began in 1994,4 and individuals were randomly selected from the databases of 36 local primary‐care centres and invited to attend for screening for H pylori by the 13‐carbon‐labelled urea breath test.
Over 30 000 people were contacted, of whom 8407 attended and were eligible for inclusion in the trial. For the purposes of this study, an attempt was made to contact all participants, regardless of H pylori status, 10 years later. To trace those involved, the primary‐care centres that had initially recruited the individuals were visited, and their computer databases accessed, to obtain the current address for all subjects. People successfully located were sent a validated dyspepsia questionnaire by post, and informed written consent to re‐examine their primary‐care records was requested. Non‐responders were sent a second postal questionnaire. The initial questionnaires were sent out in August 2003, and the re‐examination of participants' primary‐care records began in January 2004. The relevant local research ethics committees in Leeds and Bradford approved the study in March 2003.
Data collection and synthesis
Baseline data
Baseline demographic data and lifestyle factors, including age, sex, marital status, ethnicity, consumption of tobacco, alcohol and coffee, social class (as defined by occupation), and H pylori status and treatment allocation (if positive), were collected at study entry, and were available for all individuals.
Dyspepsia data at baseline were collected using a validated 15‐item instrument, the Leeds Dyspepsia Questionnaire (LDQ), which is designed to be administered by a trained researcher.15 This obtains information on both frequency and severity of indigestion, heartburn, regurgitation, nausea, vomiting, belching, early satiety and dysphagia over the preceding 6 months. The most troublesome of these symptoms is also recorded. The presence or absence of dyspepsia at trial entry was classified using the validated scoring system derived from the LDQ, where subjects are defined as having dyspepsia if any of the first five symptom items are present at a frequency of once a month or more. These data were stored on file from the original study for all individuals.
At original study entry, participants were also asked to complete a copy of the Psychological and General Well‐Being Index (PGWBI),16 a validated scale consisting of 22 questions, with a maximum value of 132 and a minimum of 22, a higher score indicating a better quality of life. The PGWBI has been used in many surveys of gastrointestinal symptoms, and it has been confirmed that the scale possesses good sensitivity to detect symptoms attributable to gastrointestinal disease.17 However, no clinical threshold score exists for use with the PGWBI with which to categorise the quality of life. These baseline quality of life data were therefore split into three equal‐sized groups among all responders at 10 years according to the total PGWBI score: low (score 31–100), medium (101–112) and high (⩾113).
Finally, a questionnaire that used the Manning criteria to facilitate the diagnosis of irritable bowel syndrome (IBS) was completed at baseline.18 This enquired about the presence of six symptoms: abdominal pain eased by defaecation, increased stool frequency when abdominal pain commenced, loose stools when abdominal pain commenced, tenesmus, mucus per rectum and abdominal distension. Symptom frequency was rated on a three‐point Likert scale: never, occasionally, or more than once a week. Individuals were defined as having IBS if they provided a positive response to three or more of these six symptoms at a frequency greater than once a week.
Ten‐year data
When following up such a large number of individuals at 10 years, it would be logistically difficult to persuade those involved to attend an appointment to administer a questionnaire after such a long time interval. For this reason, a shortened, self‐administered version of the LDQ, the short‐form Leeds dyspepsia questionnaire (S‐FLDQ), was used to collect dyspepsia data at 10 years. The S‐FLDQ assesses both the frequency and severity of four symptoms over the preceding 2 months: indigestion, heartburn, regurgitation and nausea (which are rated on a five‐point Likert scale from 0 to 4, with a higher score indicating more frequent or severe symptoms), as well as asking the subject which is the most troublesome of these four symptoms. The S‐FLDQ has been previously validated in a primary and a secondary care setting, and is an acceptable, reliable and responsive instrument for measuring both the presence and the severity of dyspepsia.19 Dyspepsia symptom status at 10 years was dichotomised into symptomatic or asymptomatic using the summed symptom frequency score from the S‐FLDQ. A score of ⩾4 (of a possible total of 16) has been shown to predict dyspepsia as diagnosed by a clinician with good sensitivity and specificity.
In addition, participants were asked to give their current weight (in kilograms), and their height (in metres), to allow calculation of their body mass index (BMI) at 10 years. Data on the use of prescribed non‐steroidal anti‐inflammatory drugs (NSAIDs) and/or aspirin over the entire 10 years were also obtained, but only for those individuals who allowed access to their primary‐care records.
Definition of dyspepsia
Using data from both baseline and 10‐year dyspepsia questionnaires allowed the identification of individuals with new‐onset dyspepsia at the 10‐year follow‐up. There remain concerns over the most appropriate definition of dyspepsia to use in clinical practice. At present, the British Society of Gastroenterology (BSG) uses a broad definition in line with the 1988 working party report,8 whereas the American Gastroenterological Association prefers the Rome II definition.20 The BSG definition is probably more relevant in primary care, where there is considerable overlap of both individual symptoms and symptom subgroups,21 and neither have been shown to accurately predict endoscopic findings,22 whereas the Rome II definition is often used in studies of patients with functional dyspepsia recruited in secondary care to reduce heterogeneity among subjects. Both the LDQ and the S‐FLDQ measure dyspepsia using the broad definition espoused by the BSG but, as they capture the predominant symptom reported by the subject, they are also capable of providing a narrower definition of dyspepsia, in line with the Rome II criteria. As this was a community‐based study, our primary analysis used the broader definition of dyspepsia, but we also conducted a secondary analysis using the Rome II definition of dyspepsia, where those complaining of predominant heartburn or regurgitation were excluded.
Sample size
The original power calculation was based on the expected prevalence of H pylori and the difference in dyspepsia rates predicted in those receiving eradication therapy versus placebo.4 This is not the relevant outcome for this study. Retrospective sample size calculations were performed to determine the power of this study. Assuming there were 2500 subjects without dyspepsia (using a broad definition) at baseline, when quality of life was divided into three equal groups, 19% developed dyspepsia at 10 years in the highest quality of life group. This sample size would detect a 7‐percentage point difference in dyspepsia rates between quality of life groups at 80% power and 1% significance level.
Statistical analysis
To assess whether those who were successfully contacted were representative of the original study population, demographic data were compared between responders and those who were unable to be traced or who did not respond, using the χ2 test. The associations between demographic data, lifestyle factors, quality of life, IBS at baseline, BMI at 10 years, NSAID and/or aspirin use, and H pylori status and the new onset of dyspepsia at 10 years in those who were asymptomatic at original study entry were explored using univariate analysis. Independent risk factors were determined by performing multivariate logistic regression to control for all these demographic data and lifestyle factors. These analyses were performed using both the BSG and Rome II definitions of dyspepsia. Due to multiple comparisons, a two‐tailed p value of <0.01 was considered to be statistically significant for all analyses, and the results of univariate and multivariate analyses were expressed as odds ratios (ORs) with 99% confidence intervals (CIs). As the effect of baseline quality of life on the subsequent new onset of dyspepsia was the primary interest, the proportion of new‐onset dyspepsia in the individuals under study that was attributable to low quality of life was calculated from the population attributable risk percentage (PAR%). This was performed by dividing the individuals into two equal groups at baseline according to total PGWBI score, with a score of ⩽107 indicating lower quality of life, and a score of ⩾108 indicating higher quality of life. This variable was then put into the logistic regression model used above to obtain an adjusted OR, and the PAR% was calculated using the following formula (p = prevalence of lower quality of life at baseline in those asymptomatic at baseline, OR = adjusted OR after multivariate logistic regression):
PAR% = p(OR−1)/1+p(OR−1).
All statistical analyses were performed using StatsDirect statistical software V.2.2.5, and SPSS for Windows V.11.5.
Results
Demographic data of involved individuals
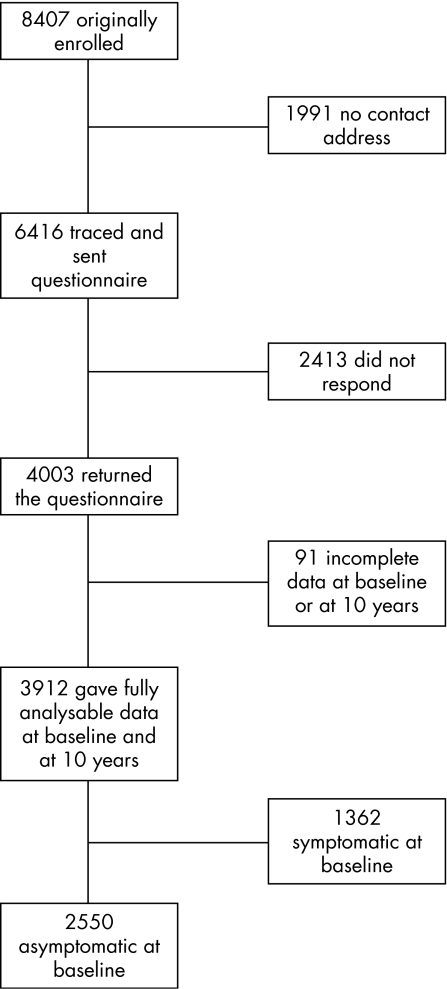
Of the 8407 individuals originally enrolled, 1991 (24%) could not be traced to a current address. Questionnaires were sent to the remaining 6416 (76%), and 4003 (48%) responded (fig 1). Of those who responded, 3912 (46.5%) provided complete symptom data both at baseline and at the 10‐year follow‐up. Consent to access primary‐care records was provided by 3266 individuals (39%) of the original sample. Females, non‐smokers, alcohol users, those of higher social class and those who were asymptomatic at baseline were significantly more likely to be successfully followed up (p<0.001 for all analyses; table 1), and there was a trend for those with a higher quality of life at baseline being more likely to be successfully followed up (p = 0.012).
Figure 1 Flowchart of study participants.
Table 1 Demographics and baseline characteristics of individuals who were successfully contacted compared with those who were not contacted or did not respond.
| Responded (n = 4003) | Not contacted or no response (n = 4404) | p Value | |
|---|---|---|---|
| Sex | |||
| Male | 1756 (44%) | 2180 (49.5%) | <0.001 |
| Female | 2247 (56%) | 2224 (50.5%) | |
| Lifestyle | |||
| Smoker | 879 (22%) | 1324 (30%) | <0.001 |
| Alcohol user | 3642 (91%) | 3876 (88%) | <0.001 |
| Social class | |||
| I and II | 1531 (38%) | 1502 (34%) | <0.001 |
| III | 1697 (42%) | 1916 (43.5%) | |
| IV and V | 409 (10%) | 490 (11%) | |
| Baseline quality of life | |||
| High | 1237 (31%) | 1247 (28%) | 0.012 |
| Medium | 1343 (33.5%) | 1417 (32%) | |
| Low | 1269 (32%) | 1558 (35%) | |
| Dyspepsia at baseline | 1375 (34%) | 1792 (41%) | <0.001 |
| IBS at baseline | 221 (5.5%) | 246 (6%) | 0.90 |
| Mean (SD) age (years) | 55.4 (2.8) | 55.2 (2.9) | 0.30 |
IBS, irritable bowel syndrome.
Ten‐year natural history of dyspepsia in the study population using the BSG definition
Of the 3912 individuals who provided complete symptom data at baseline and the 10 years, 1362 (35%; 95% CI 33.5% to 36.5%) had dyspepsia at baseline using the BSG definition, and 2550 were asymptomatic. Of those individuals who were symptomatic at baseline, 891 (65%; 95% CI 62.5% to 67.5%) had persistent dyspepsia at 10 years. Among the 2550 individuals asymptomatic at trial entry, 717 (28%; 95% CI 26% to 30%) had developed new‐onset dyspepsia at 10 years, giving an incidence of new‐onset dyspepsia in this population of 2.8% (95% CI 2.6% to 3.0%) per year. Predominant symptom was reported in 606 of these individuals; 205 complained of indigestion‐predominant dyspepsia (30%; 95% CI 27% to 33%), 297 heartburn/regurgitation‐predominant dyspepsia (43%; 95% CI 39% to 47%) and 104 nausea‐predominant dyspepsia (15%; 95% CI 12% to 18%), whereas 83 were unable to specify their predominant symptom. In total, 1608 of the 3912 individuals who provided data at both baseline and the 10‐year follow‐up reported dyspepsia at 10 years, giving a prevalence of dyspepsia of 41% (95% CI 39.5% to 42.5%) in all individuals at 10 years.
Univariate analysis in the 717 individuals who developed new‐onset dyspepsia at 10 years showed that female sex, higher BMI, lower social class, lower quality of life at baseline, presence of IBS at baseline, and use of NSAIDs and/or aspirin over the preceding 10 years were all risk factors for the new onset of dyspepsia. In addition, there was a trend towards smokers being more likely to develop dyspepsia compared with non‐smokers (table 2).
Table 2 Univariate and multivariate analyses for new‐onset dyspepsia at 10 years using the British Society of Gastroenterology definition.
| Variable | With dyspepsia, n (%) | Unadjusted OR | 99% CI | p Value | Adjusted OR | 99% CI | p Value |
|---|---|---|---|---|---|---|---|
| Age (per year) | 0.98 | 0.95 to 1.02 | 0.31 | 0.99 | 0.95 to 1.04 | 0.73 | |
| BMI at 10 years (per unit) | 1.05 | 1.03 to 1.08 | <0.001 | 1.05 | 1.02 to 1.08 | <0.001 | |
| Sex | |||||||
| Male | 273/1099 (25) | 1.0 | 1.0 | ||||
| Female | 444/1451 (31) | 1.33 | 1.05 to 1.69 | 0.001 | 1.14 | 0.85 to 1.53 | 0.25 |
| Marital status | |||||||
| Single | 35/152 (23) | 1.0 | 1.0 | ||||
| Married | 678/2376 (28.5) | 1.33 | 0.80 to 2.32 | 0.16 | 1.51 | 0.78 to 2.93 | 0.11 |
| Smoking status | |||||||
| Non‐smoker | 551/2026 (27) | 1.0 | 1.0 | ||||
| Smoker | 164/510 (32) | 1.27 | 0.95 to 1.68 | 0.03 | 1.25 | 0.89 to 1.75 | 0.10 |
| Alcohol use | |||||||
| No | 59/188 (31) | 1.0 | 1.0 | ||||
| Yes | 657/2356 (28) | 0.85 | 0.55 to 1.32 | 0.31 | 1.01 | 0.59 to 1.72 | 0.98 |
| Coffee drinker | |||||||
| No | 84/297 (28) | 1.0 | 1.0 | ||||
| Yes | 632/2243 (28) | 0.99 | 0.70 to 1.44 | >0.99 | 0.96 | 0.63 to 1.48 | 0.82 |
| Ethnicity | |||||||
| Caucasian | 702/2490 (28) | 1.0 | 1.0 | ||||
| Non‐Caucasian | 12/42 (29) | 1.02 | 0.37 to 2.51 | >0.99 | 0.75 | 0.21 to 2.63 | 0.55 |
| Social class | |||||||
| I and II | 252/1053 (24) | 1.0 | 1.0 | ||||
| III | 325/1072 (30) | 1.38 | 1.07 to 1.79 | 0.001 | 1.29 | 0.96 to 1.74 | 0.028 |
| IV and V | 62/228 (27) | 1.19 | 0.76 to 1.83 | 0.31 | 0.99 | 0.58 to 1.69 | 0.97 |
| Quality of life | |||||||
| High | 191/984 (19) | 1.0 | 1.0 | ||||
| Medium | 234/829 (28) | 1.63 | 1.22 to 2.19 | <0.001 | 1.56 | 1.11 to 2.17 | 0.001 |
| Low | 272/661 (41) | 2.90 | 2.16 to 3.91 | <0.001 | 2.63 | 1.86 to 3.71 | <0.001 |
| IBS at baseline | |||||||
| No | 665/2459 (27) | 1.0 | 1.0 | ||||
| Yes | 45/75 (60) | 4.05 | 2.18 to 7.51 | <0.001 | 3.10 | 1.51 to 6.37 | <0.001 |
| NSAID/aspirin use in last 10 years | |||||||
| No | 228/945 (24) | 1.0 | 1.0 | ||||
| Yes | 377/1,136 (33) | 1.56 | 1.21 to 2.03 | <0.001 | 1.32 | 0.99 to 1.75 | 0.01 |
| H pylori status | |||||||
| Negative | 541/1925 (28) | 1.0 | N/A* | ||||
| Positive | 176/625 (28) | 1.0 | 0.76 to 1.31 | >0.99 | N/A | N/A | N/A |
| H pylori status and treatment arm | |||||||
| Negative | 541/1,925 (28) | 1.0 | 1.0 | ||||
| Positive, eradication therapy | 88/316 (28) | 0.99 | 0.69 to 1.40 | 0.95 | 0.84 | 0.55 to 1.28 | 0.28 |
| Positive, placebo | 88/309 (28.5) | 1.02 | 0.71 to 1.45 | 0.93 | 0.99 | 0.65 to 1.52 | 0.95 |
BMI, body mass index; IBS, irritable bowel syndrome; N/A, not applicable; NSAID, non‐steroidal anti‐inflammatory drug.
*H pylori status was not used in the logistic regression model, as H pylori status and treatment arm variable was used.
After multivariate logistic regression, higher BMI, lower quality of life at study entry, presence of IBS at study entry, and use of NSAIDs and/or aspirin remained significant risk factors for the new onset of dyspepsia. The adjusted OR for new‐onset dyspepsia at 10 years in the lower quality of life group was 1.94 (99% CI 1.47 to 2.56) after logistic regression, suggesting that being in the lower half of quality of life at baseline had a 30% PAR for new‐onset dyspepsia.
Ten‐year natural history of dyspepsia in the study population using the Rome II definition
When the Rome II definition of dyspepsia was used, 795 (20%; 95% CI 19% to 21%) individuals had dyspepsia at baseline, and 3117 were asymptomatic. Of those symptomatic at baseline, 323 (41%; 95% CI 39% to 42%) continued to experience dyspepsia at the 10‐year follow‐up, whereas 569 (18%; 95% CI 17.5% to 19%) of those who were asymptomatic at original study entry reported Rome II dyspepsia at 10 years.
After univariate analysis in those individuals who did not have Rome II dyspepsia at baseline, lower quality of life at baseline, presence of IBS at baseline, and use of NSAIDs and/or aspirin over the preceding 10 years were again found to be significantly associated with the new onset of symptoms at 10 years, while there were also trends towards those of lower social class and higher BMI developing new‐onset dyspepsia (table 3).
Table 3 Univariate and multivariate analyses for new‐onset dyspepsia at 10 years using the Rome II definition.
| Variable | With dyspepsia, n (%) | Unadjusted OR | 99% CI | p Value | Adjusted OR | 99% CI | p Value |
|---|---|---|---|---|---|---|---|
| Age (per year) | 0.98 | 0.94 to 1.03 | 0.32 | 0.99 | 0.94 to 1.04 | 0.60 | |
| BMI at 10 years (per unit) | 1.02 | 1.00 to 1.05 | 0.02 | 1.02 | 0.99 to 1.05 | 0.12 | |
| Sex | |||||||
| Male | 232/1360 (17) | 1.0 | 1.0 | ||||
| Female | 337/1757 (19) | 1.15 | 0.91 to 1.47 | 0.13 | 1.06 | 0.78 to 1.44 | 0.63 |
| Marital status | |||||||
| Single | 32/175 (18) | 1.0 | 1.0 | ||||
| Married | 534/2916 (18) | 1.0 | 0.60 to 1.68 | 0.99 | 1.04 | 0.55 to 1.97 | 0.88 |
| Smoking status | |||||||
| Non‐smoker | 437/2454 (18) | 1.0 | 1.0 | ||||
| Smoker | 129/647 (20) | 1.15 | 0.86 to 1.53 | 0.21 | 1.11 | 0.78 to 1.57 | 0.46 |
| Alcohol use | |||||||
| No | 49/240 (20) | 1.0 | 1.0 | ||||
| Yes | 519/2870 (18) | 0.86 | 0.56 to 1.32 | 0.37 | 0.89 | 0.52 to 1.51 | 0.57 |
| Coffee drinker | |||||||
| No | 75/386 (19) | 1.0 | 1.0 | ||||
| Yes | 492/2719 (18) | 0.92 | 0.64 to 1.31 | 0.53 | 1.0 | 0.64 to 1.54 | 0.98 |
| Ethnicity | |||||||
| Caucasian | 553/3037 (18) | 1.0 | 1.0 | ||||
| Non‐Caucasian | 11/57 (19) | 1.33 | 0.58 to 3.02 | 0.38 | 1.30 | 0.44 to 3.84 | 0.53 |
| Social class | |||||||
| I and II | 193/1226 (16) | 1.0 | 1.0 | ||||
| III | 257/1333 (19) | 1.28 | 0.98 to 1.67 | 0.02 | 1.20 | 0.88 to 1.65 | 0.14 |
| IV and V | 50/297 (17) | 1.08 | 0.69 to 1.70 | 0.65 | 0.93 | 0.54 to 1.62 | 0.74 |
| Quality of life | |||||||
| High | 132/1107 (12) | 1.0 | 1.0 | ||||
| Medium | 177/1023 (17) | 1.55 | 1.12 to 2.13 | <0.001 | 1.44 | 0.99 to 2.09 | 0.01 |
| Low | 241/892 (27) | 2.73 | 2.01 to 3.72 | <0.001 | 2.44 | 1.70 to 3.51 | <0.001 |
| IBS at baseline | |||||||
| No | 516/2976 (17) | 1.0 | 1.0 | ||||
| Yes | 50/123 (41) | 3.27 | 2.00 to 5.33 | <0.001 | 2.83 | 1.60 to 5.01 | <0.001 |
| NSAID/aspirin use in last 10 years | |||||||
| No | 162/1057 (15) | 1.0 | 1.0 | ||||
| Yes | 305/1482 (21) | 1.43 | 1.09 to 1.89 | 0.001 | 1.25 | 0.92 to 1.69 | 0.06 |
| H pylori status | |||||||
| Negative | 424/2301 (18) | 1.0 | N/A* | ||||
| Positive | 145/816 (18) | 0.96 | 0.73 to 1.26 | 0.68 | N/A | N/A | N/A |
| H pylori status and treatment arm | |||||||
| Negative | 424/2301 (18) | 1.0 | 1.0 | ||||
| Positive, eradication therapy | 80/414 (19) | 1.06 | 0.75 to 1.50 | 0.67 | 0.94 | 0.61 to 1.43 | 0.68 |
| Positive, placebo | 65/402 (16) | 0.85 | 0.59 to 1.24 | 0.28 | 0.88 | 0.56 to 1.37 | 0.46 |
BMI, body mass index; IBS, irritable bowel syndrome; N/A, not applicable; NSAID, non‐steroidal anti‐inflammatory drug.
*H pylori status not used in the logistic regression model, as H pylori status and treatment arm variable was used.
Multivariate logistic regression confirmed that lower quality of life and presence of IBS at baseline were independently associated with the new onset of Rome II dyspepsia at 10 years.
Discussion
This longitudinal study has examined the incidence and natural history of dyspepsia, as well as factors that may influence the new development of dyspepsia, in a large number of individuals over the longest period of follow‐up reported, to our knowledge, to date (10 years). It has shown that the prevalence of dyspepsia remains relatively stable over time, between 35% and 40%, which is of a magnitude similar to those of previous surveys conducted in the UK, using a broad definition of dyspepsia to include any symptom referable to the upper gastrointestinal tract.2,3,5 In addition, almost two thirds of those who had dyspepsia at study entry remained symptomatic at the 10‐year follow‐up, whereas almost a third of individuals who were asymptomatic at baseline reported new‐onset dyspepsia at 10 years. This is consistent with previous reports in the literature of the natural history of dyspepsia over a shorter time period.1,6
The estimate of an incidence of new‐onset dyspepsia of almost 3% per year is one of the novel findings of this study. A previous study reported the incidence to be approximately 1% over 3 months,6 which is of a magnitude similar to that noted in this population, but there are little other published data regarding this. The reason for the small increase in the prevalence of dyspepsia between baseline and the 10‐year follow‐up is not clear. The possible explanations include the fact that a lower proportion of the original participants with dyspepsia at baseline were successfully traced and contacted, and that their data were therefore not examined in this follow‐up study, a potential volunteer bias wherein those who returned questionnaires at 10 years were more likely to be experiencing symptoms at that point in time, and the fact that the individuals under study are now 10 years older. However, it should be remembered that the incidence of new‐onset dyspepsia in this study is based on the assessment of individuals at only two points in time, 10 years apart, and will therefore not include those who may have experienced one or more episodes of dyspepsia in the intervening years.
A further novel finding concerns the effect of quality of life on risk of subsequent development of dyspepsia in previously asymptomatic individuals. The negative effect of dyspepsia on quality of life is well recognised,11 although some of this association is thought to be due to other psychological factors,12 and indeed one large study has suggested that psychiatric disorders and recent major life events are more common in those who report the presence of dyspeptic symptoms.23 It has always been assumed that symptoms of dyspepsia, and other chronic gastrointestinal conditions, give rise to psychological distress, rather than the reverse. A recent post hoc analysis of pooled data from randomised controlled trials of different treatments for gastro‐oesophageal reflux disease showed that low well being at trial entry, again assessed using the PGWBI, predicted a poor response to treatment in terms of relief of symptoms.24 However, there have been no studies published, to our knowledge, that have examined the effect of an initially poor quality of life on the likelihood of subsequent development of dyspepsia. It would seem, from this study, that individuals who have a lower quality of life are at a significantly increased risk of new‐onset dyspepsia, and that this risk increases with worsening quality of life. This was an important predictor of developing new‐onset dyspepsia at 10 years in our model, and suggests that the poor quality of life seen in subjects with dyspepsia is due, at least in part, to those with a poor quality of life being prone to develop upper gastrointestinal symptoms. Indeed, assuming that the relationship is causal, our data suggest that dyspepsia could be reduced by 30% in this community sample if the PGWBI score improved in the half of the population with the worst quality of life to a similar level as those in the top half.
The reasons for the association between quality of life and dyspepsia are unclear, although it is possible that individuals with other chronic symptom complexes were more likely to report a low quality of life at baseline, and that they are more likely to report multiple symptoms, or subsequently develop new symptoms. As discussed earlier, the PGWBI is not designed to categorise an individual's quality of life, merely to quantify it, and it is therefore difficult to define the difference between individuals in the three equal‐sized groups we created. However, a previous Swedish study showed a mean PGWBI score in over 2000 randomly selected individuals from the general population of 102.9,25 suggesting that the range of values we chose to create the tertiles are plausible.
The influence of BMI on dyspepsia has been described previously, with an increase in prevalence of symptoms in those who are either overweight or obese.26 The new onset of symptoms of dyspepsia in those with pre‐existing IBS has also been reported elsewhere, and there is thought to be substantial overlap between the two conditions.6,27 These surveys did not evaluate the effect of NSAIDs on dyspepsia, and this is another novel finding of the current study, with NSAID prescription predicting the development of dyspepsia. It should however be pointed out that the temporal relationship between prescription of an NSAID and development of dyspepsia cannot be uncovered by the design of a study such as this, and it is conceivable that dyspepsia may have developed before NSAID use. In addition, association does not necessarily mean causation. For example, those with dyspepsia may have experienced relief of their symptoms with food, which may explain the increased risk of symptoms in those with higher BMI.
The most appropriate definition of dyspepsia is still a subject of great debate. The BSG currently advocates the use of a broad definition, including any symptom referable to the upper gastrointestinal tract, in line with the 1988 working party report.8 Others prefer the Rome criteria, which have recently been refined further.28 This study, which originally recruited subjects over 10 years ago, is not designed to examine the utility of these latest modifications. However, we were able to alter the definition of dyspepsia used in the study, and assess the effect on our observed results. The fact that this did not diminish the effect of low quality of life at baseline on the development of new‐onset dyspepsia suggests that these findings are robust, and that this is an important risk factor, probably relevant to all common upper gastrointestinal disorders in the community.
This study has some limitations imposed by its design. The high attrition rate, with only 48% of those originally involved being successfully traced and responding to the questionnaire, is almost inevitable when following up a large number of individuals randomly selected from the general population after such a long period of time. Those who did respond were not entirely representative of the original sample, with an over‐representation of females, non‐smokers, alcohol users and those of higher social class, although the absolute difference in proportions between responders and non‐responders is modest. In addition, the fact that those with dyspepsia at baseline were less likely to be successfully followed up may have led to an underestimation of the true prevalence of dyspepsia, although, as has been discussed, the proportion of individuals who reported symptoms in this study is in line with previous reports from Western populations. The restricted age range of these individuals, between 50 and 59 years, means that the results may not be generalisable to any age group in the general population. Furthermore, we only have BMI data at 10 years and do not have information on change in BMI over time. One final criticism that could be levelled against this study is the use of different dyspepsia questionnaires at baseline and at the 10‐year follow‐up. However, both of these had been previously validated, and the use of a predefined cut‐off score to diagnose dyspepsia had been shown to correlate well with a doctor's diagnosis of the disorder. In addition, the S‐FLDQ is derived from the LDQ, and uses the symptom items with the greatest discriminant validity from that questionnaire, so this is unlikely to have affected the observed results. It is possible to use the validated scoring system from the S‐FLDQ to attempt to classify each individual's symptom status at baseline, rather than the scoring system derived from the LDQ. When this was done, only three individuals who were symptomatic at baseline according to the LDQ were misclassified as asymptomatic, and no individuals asymptomatic at baseline according to the LDQ were misclassified as symptomatic, showing that both questionnaires have a very similar ability to detect dyspepsia.
Despite these limitations, this study also has several strengths. The follow‐up period of 10 years is longer than any other study previously published. In addition, almost 4000 individuals provided data both at baseline and at 10 years, which is larger than any other longitudinal survey over a comparable time period. This is the first study to examine the influence of numerous social and demographic factors on the new onset of dyspepsia simultaneously, within such a large dataset. In addition, as most of these data were collected at baseline, this allowed us to assess their effect on the subsequent development of dyspepsia; therefore, this study is not susceptible to the inherent biases of case–control studies. Finally, the fact that this study was conducted in the general population means that the participants are more likely to be generalisable to individuals of a similar age encountered by primary‐care physicians, as referral bias is eliminated.
In summary, the incidence of new‐onset dyspepsia in this large sample of the community was almost 3% per year. Low quality of life at baseline exerted a strong effect on the likelihood of developing dyspepsia at 10 years. This has implications for treatment trials of patients with dyspepsia as these data predict that curing dyspepsia symptoms will not necessarily improve all patients' quality of life as dramatically as might be suggested by cross‐sectional surveys.
Supplementary Material
Acknowledgements
We are thank the original participating general practices for their help in facilitating follow‐up of involved individuals and, where applicable, allowing access to primary‐care records—Beeston Hill Health Centre, Bridge Street Surgery, Burley Park Medical Centre, Burton Croft Surgery, Carlton Gardens Surgery, Chapeloak Practice, Crossland Surgery, Fountains Medical Centre, Garforth Medical Centre, The Grange Medical Centre, High Field Surgery, Kippax Health Centre, Leigh View Medical Practice, Lingwell Croft Surgery, Manor Park Surgery, Marsh Street Surgery, Meanwood Health Centre, New Wortley Health Centre, Park Edge Medical Practice, Robin Lane Medical Centre, Silver Lane Surgery, St Martin's Practice, The Street Lane Practice, Tinshill Lane Surgery, West Lodge Surgery, The Whitfield Practice, The Windmill Health Centre, Windsor House Surgery, Woodhouse Medical Centre, Woodsley Health Centre, and Yeadon Health Centre in Leeds; and Cullingworth Medical Centre, Leylands Medical Centre, Parkhurst Medical Practice, Ridge Medical Practice, and Westcliffe Medical Centre in Bradford.
Abbreviations
BMI - body mass index
BSG - British Society of Gastroenterology
IBS - irritable bowel syndrome
LDQ - Leeds Dyspepsia Questionnaire
NSAID - non‐steroidal anti‐inflammatory drug
PAR% - population attributable risk percentage
PGWBI - Psychological and General Well‐Being Index
S‐FLDQ - Short‐Form Leeds Dyspepsia Questionnaire
Footnotes
Competing interests: declared (the declaration can be viewed on the Gut website at http://www gut.bmj.com/supplmental).
References
- 1.Weir R D, Backett E M. Studies of the epidemiology of peptic ulcer in a rural community: prevalence and natural history of dyspepsia and peptic ulcer. Gut 1968975–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jones R, Lydeard S. Prevalence of symptoms of dyspepsia in the community. BMJ 198929830–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jones R H, Lydeard S E, Hobbs F D R.et al Dyspepsia in England and Scotland. Gut 199031401–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Moayyedi P, Forman D, Braunholtz D.et al The proportion of upper gastrointestinal symptoms in the community associated with Helicobacter pylori, lifestyle factors, and nonsteroidal anti‐inflammatory drugs. Am J Gastroenterol 2000951448–1455. [DOI] [PubMed] [Google Scholar]
- 5.Penston J G, Pounder R E. A survey of dyspepsia in Great Britain. Aliment Pharmacol Ther 19961083–89. [DOI] [PubMed] [Google Scholar]
- 6.Agreus L, Svardsudd K, Nyren O.et al Irritable bowel syndrome and dyspepsia in the general population: overlap and lack of stability over time. Gastroenterology 1995109671–680. [DOI] [PubMed] [Google Scholar]
- 7.Talley N J, Zinsmeister A R, Schleck C D.et al Dyspepsia and dyspepsia subgroups: a population‐based study. Gastroenterology 19921021259–1268. [PubMed] [Google Scholar]
- 8.Colin‐Jones D G, Bloom B, Bodemar G.et al Management of dyspepsia: report of a working party. Lancet 1988331576–579. [PubMed] [Google Scholar]
- 9.Talley N J, Colin‐Jones D G, Koch K L.et al Functional dyspepsia: a classification with guidelines for diagnosis and management. Gastroenterol Int 19914145–160. [Google Scholar]
- 10.El‐Serag H B, Talley N J. Systematic review: health‐related quality of life in functional dyspepsia. Aliment Pharmacol Ther 200318387–393. [DOI] [PubMed] [Google Scholar]
- 11.Enck P, Dubois D, Marquis P. Quality of life in patients with upper gastrointestinal symptoms: results from the domestic/international gastroenterology surveillance study (DIGEST). Scand J Gastroenterol 199923148–54. [DOI] [PubMed] [Google Scholar]
- 12.Halder S L S, Locke G R, Talley N J.et al Impact of functional gastrointestinal disorders on health‐related quality of life: a population‐based case‐control study. Aliment Pharmacol Ther 200419233–242. [DOI] [PubMed] [Google Scholar]
- 13.Mones J, Adan A, Segu J L.et al Quality of life in functional dyspepsia. Dig Dis Sci 20024720–26. [DOI] [PubMed] [Google Scholar]
- 14.Talley N J, Weaver A L, Zinsmeister A R. Impact of functional dyspepsia on quality of life. Dig Dis Sci 199540584–589. [DOI] [PubMed] [Google Scholar]
- 15.Moayyedi P, Duffett S, Braunholtz D.et al The Leeds dyspepsia questionnaire: a valid tool for measuring the presence and severity of dyspepsia. Aliment Pharmacol Ther 1998121257–1262. [DOI] [PubMed] [Google Scholar]
- 16.Dupuy H J. The psychological general well‐being (PGWB) index. In: Wenger NK, Mattson ME, Fruberg CF, eds. Assessment of quality of life in clinical trials of cardiovascular therapies. New York: Le Jacq Publishing, 1984170–183.
- 17.Glise H, Hallerback B, Johansson B. Quality of life assessments in the evaluation of gastroesophageal reflux and peptic ulcer disease before, during and after treatment. Scand J Gastroenterol 1995208131–135. [DOI] [PubMed] [Google Scholar]
- 18.Manning A P, Thompson W G, Heaton K W.et al Towards positive diagnosis of the irritable bowel. BMJ 1978277653–654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fraser A, Qume M, Ford A C.et al Validation of the short‐form Leeds dyspepsia questionnaire (S‐FLDQ) in a community setting. Gastroenterology 2003124(Suppl 1)A224 [Google Scholar]
- 20.Talley N J, Stanghellini V, Heading R C.et al Functional gastroduodenal disorders. Gut 199945(Suppl 2)37–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tougas G, Chen M S, Hwang P.et al Prevalence and impact of upper gastrointestinal symptoms in the Canadian population: findings from the DIGEST study. Am J Gastroenterol 1999942845–2854. [DOI] [PubMed] [Google Scholar]
- 22.Thomson A B R, Barkun A N, Armstrong D.et al The prevalence of clinically significant endoscopic findings in primary care patients with uninvestigated dyspepsia: The Canadian Adult Dyspepsia Empiric Treatment—Prompt Endoscopy (CADET‐PE) study. Aliment Pharmacol Ther 2003171481–1491. [DOI] [PubMed] [Google Scholar]
- 23.Stanghellini V. Relationship between upper gastrointestinal symptoms and lifestyle, psychosocial factors and comorbidity in the general population: results from the domestic/international gastroenterology surveillance study (DIGEST). Scand J Gastroenterol 199934(Suppl 231)29–37. [PubMed] [Google Scholar]
- 24.Wiklund I, Carlsson R, Carlsson J.et al Psychological factors as a predictor of treatment response in patients with heartburn: a pooled analysis of clinical trials. Scand J Gastroenterol 200641288–293. [DOI] [PubMed] [Google Scholar]
- 25.Dimenas E, Carlsson G, Glise H.et al Relevance of norm values as part of the documentation of quality of life instruments for use in upper gastrointestinal disease. Scand J Gastroenterol 199631(Suppl 221)8–13. [DOI] [PubMed] [Google Scholar]
- 26.Stanghellini V. Three month prevalence rates of gastrointestinal symptoms and the influence of demographic factors: results from the domestic/international gastroenterology surveillance study. Scand J Gastroenterol 199934(Suppl 231)20–28. [DOI] [PubMed] [Google Scholar]
- 27.Agreus L, Svardsudd K, Talley N J.et al Natural history of gastroesophageal reflux disease and functional abdominal disorders. Am J Gastroenterol 2001962905–2914. [DOI] [PubMed] [Google Scholar]
- 28.Tack J, Talley N J, Camilleri M.et al Functional gastroduodenal disorders. Gastroenterology 20061301466–1479. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.