Abstract
Aim
To investigate the ocular surface inflammatory response to chronic topical treatments in patients with glaucoma by measuring the cytokine level in tears using multiplex bead analysis.
Methods
Tear samples were collected from 21 patients with glaucoma and 12 healthy volunteers. Tears were analysed for the presence of 17 cytokines: interleukin (IL)1β, IL2, IL4, IL5, IL6, IL7, IL8, IL10, IL12, IL13, IL17, granulocyte‐colony stimulating factor, granulocyte‐macrophage stimulating factor, interferon (INF)γ, monocyte chemotactic protein (MCP)1, macrophage inflammatory protein 1β and tumour necrosis factor (TNF)α. The cytokines in each sample of tears were measured using multiplex bead analysis with microspheres as solid support for immunoassays.
Results
In the tears of treated patients, proinflammatory cytokines (IL1β, IL6, IL12, TNFα) were significantly increased compared with controls. T helper (Th)1 (INFγ, IL2) and Th2 (IL5, IL10, IL4) type cytokines were also significantly higher (p<0.05); however, the most marked increase was observed with Th1 cytokines. The expression of chemokine IL8 and MCP1 was also increased in the treated group.
Conclusion
This study shows that pro‐inflammatory cytokine secretion by conjunctival cells is increased in response to topical treatments for glaucoma. The characterisation of cytokines in tears was previously limited by the small volume attainable, a limitation that has been overcome by multiplex analysis.
Topical intraocular pressure‐lowering drugs have been shown to induce ocular surface changes in patients treated for glaucoma or ocular hypertension.1 Clinically, local disturbances such as ocular stinging or burning, decrease in tear break‐up time and superficial punctuate keratitis have been reported.2 Ocular surface inflammation is involved in all of these clinical disorders3 and could be a risk factor for failure of glaucoma surgery.4 It has been reported that the preservative used by manufacturers is mainly responsible for the toxic effects of repeated instillations. Benzalkonium chloride (BAC), the most frequently used preservative, has been shown to be toxic on conjunctival cells.5,6 Cytokines have a key role in the immunological and inflammatory response, as they can regulate activation, differentiation and proliferation of immunocompetent cells in the conjunctiva. Raised levels of inflammatory cytokines have been reported in tears of various ocular diseases such as allergies,7 ocular rosacea8 and dry eye.9 Measuring several cytokines in tears can identify an inflammatory profile of the ocular surface in response to topical treatment for gloucoma. However, to date measurement of cytokines in tears has been limited because of the small amount of tears available per sample. This has now been overcome with the use of cytometric multiplex bead analysis, which can determine many cytokines in a single tear sample.10 The measurement of cytokines within one sample with multiplex bead assays11 is of particular interest, as it can simultaneously identify several inflammatory cytokines within a small volume (<10 μl) and further assess the type of inflammatory response according to the T helper (Th)1 and Th2 cytokines detected.
To investigate the ocular surface inflammatory response to treatments for chronic glaucoma, we assessed the cytokine level in tears of patients with glaucoma with multiplex bead analysis.
Methods
Patients and sample collection
Samples were obtained from 21 treated patients with glaucoma after their fully informed consent, and from 12 controls with no history of ocular surface disorders or contact lens wear. The protocol of this prospective, observational case–control study was approved by the local ethics committee of the Burgundy region, located in Dijon, France. Patients with diabetes and allergies were excluded. All patients had been treated for >6 months with preserved topical intraocular pressure‐lowering drugs, and they received ⩾1 instillation per day. None of them had undergone ocular surgery. Tear samples were collected without topical anaesthesia, non‐traumatically, using a capillary tube to obtain 2 μl of tears from the inferior meniscus. Tears were expelled from the capillary tube in a 1‐ml tube and diluted in 48 μl of phosphate‐buffered saline (total volume 50 μl). After dilution, they were stored at −80°C. Tears were analysed for 17 cytokines with multiplex bead analysis, using microspheres as the solid support for immunoassays.
Multiplex analyses of cytokines in tears with the Bio‐Plex system
A standard capture sandwich assay was used to determine the levels of different cytokines in tears. Each captured antibody was coupled to a different bead set (Bio‐Rad Laboratories, Hercules, California, USA). The system uses a liquid suspension array of 17 sets of 5.5‐μm beads (Bio‐Plex Human Cytokine 17‐plex panel) internally dyed with different ratios of two spectrally distinct fluorochromes to assign a unique spectral address. Each set of beads was combined with a monoclonal antibody raised against interleukin (IL)1β, IL2, IL4, IL5, IL6, IL7, IL8, IL10, IL12 (p70), IL13, IL17, granulocyte‐colony stimulating factor, granulocyte‐macrophage colony stimulating factor, interferon (IFN)γ, MCP1 (monocyte chemotactic and activating factor), macrophage inflammatory protein (MIP)1β or tumour necrosis factor (TNF)α. Beads were incubated first (30 min to 2 h, at room temperature) with diluted standards (serial dilutions from 1.95 to 32 000 pg/ml) or tears, and then with biotinylated detector antibodies (30 min, at room temperature). They were washed twice in phosphate‐buffered saline, and incubated for 30 min at room temperature with phycoerythrin‐conjugated streptavidin. Cytokine levels were measured on a Bio‐Plex system (Bio‐Rad Laboratories). Each measurement was taken in duplicate. Standard curves were generated by using the reference cytokine concentrations supplied by the manufacturer. Raw data (mean fluorescent intensity) were analysed by Bio‐Plex Manager Software (Bio‐Rad Laboratories) to obtain concentration values. The lower limit of detection was <10 pg/ml (based on detectable signal >2SD above background).
Statistical analysis
Data were stored and analysed using the Bio‐Plex Manager software. Statistical comparisons were performed using the non‐parametric Mann–Whitney U test. The statistical significance was set at p<0.05.
Results
Population
Table 1 displays the patient characteristics. The treatments were: latanoprost (n = 7), carteolol (n = 3), fixed combination of latanoprost with timolol (n = 3), latanoprost and timolol (n = 3), fixed combination of timolol with dorzolamide and brimonidine (n = 2), fixed combination of latanoprost with timolol and brimonidine (n = 2), and bimatoprost and pilocarpine (n = 1).
Table 1 Patient characteristics.
| Control n = 12 | Glaucoma‐treated n = 21 | |
|---|---|---|
| Sex ratio (male:female) | 1.4 | 1.3 |
| Age, mean (SD) | 69.4 (9.6) | 66.6 (15.2) |
| Duration of treatment (years), mean (SD) | 8.6 (5.4) |
Mean cytokine concentration in tears
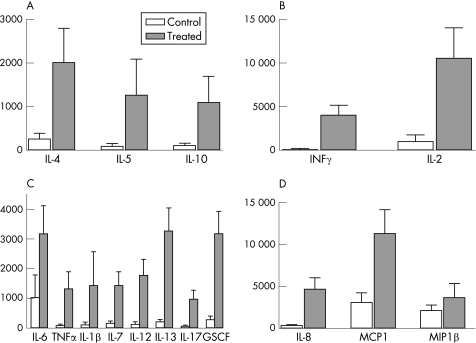
There was a significant increase in the secretion of all cytokines measured in the tears of treated patients compared with controls, except for MIP1β (p = 0.79; fig 1). The mean level of cytokines was <500 pg/ml for all cytokines in the control group, except for the two chemokines MIP1β (2100 (610) pg/ml) and MCP1 (3059 (1176) pg/ml), whereas in the treated group the mean level of all cytokines was >1000 pg/ml.
Figure 1 Mean (SEM) concentrations (pg/ml) of 17 cytokines in patients with preserved glaucoma treatment and in controls. (A) IL10, IL4 and IL5 cytokines were significantly increased in the treated patients. (B) The concentration of cytokines involved in the T helper 1(Th)1 pathway (interferon (IFN)γ, interleukin (IL)2) was significantly increased (**p<0.01) in the group of patients with glaucoma compared with controls. (C) For the major pro‐inflammatory cytokines (IL6, IL1β, IL12 and TNFα), the level was significantly (*p<0.05) increased. (D) The chemokine concentration of MCP1 and IL8 was significantly increased in the treated group (*p<0.05; **p<0.01; ***p<0.001), but MIP1β was not increased.
Pro‐inflammatory cytokines (IL1β, IL6, IL12 and TNFα) were increased (p = 0.02, 0.02, 0.03 and 0.03 respectively) in treated patients using preserved eye drops (fig 1C). The level of these cytokines was low in controls (<150 pg/ml) for IL1β, IL12 and TNFα, whereas the mean level was >1000 pg/ml for all these cytokines in treated patients, with a maximum value of 3062 (953) pg/ml for IL6.
The chemokine expression of IL8 (356 (93) pg/ml) and MCP1 (3059 (1059) pg/ml) in controls was decreased compared with patients with glaucoma (4650 1383 pg/ml; p = 0.001 and 11 250 (2879) pg/ml; p = 0.039, respectively), whereas MIP1β did not vary (fig 1C).
Cytokine inflammatory profile in tears
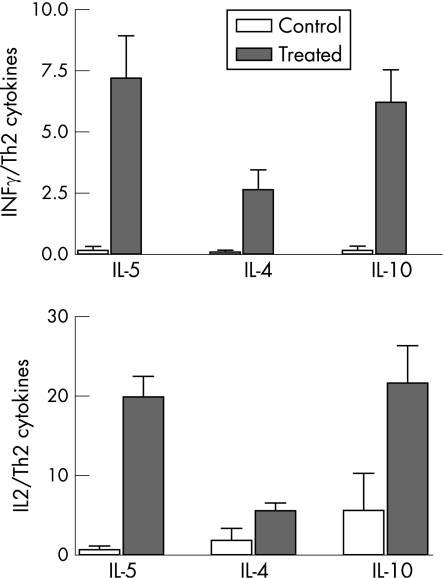
The Th1 type cytokines (IFNγ, IL2) and the Th2 type (IL4, IL5, IL10) were all found at significantly higher concentrations (p<0.05) in the treated group (fig 1A,B). The Th1/Th2 cytokine ratio was higher in patients with glaucoma than in controls (p<0.05) (fig 2). The balance of the cytokine concentration suggested a predominantly Th1 inflammatory response.
Figure 2 Th1 (IL2, INFγ)/Th2 cytokine (IL5, IL4, IL10) ratio in tears of treated and non‐treated patients. There was a significant increase (*p<0.05; **p<0.01) in the Th1/Th2 cytokine ratio in glaucoma‐treated patients compared with controls. The increase in the secretion of Th1 cytokines in the treated group was greater than the increase in Th2 cytokines.
Comparison of cytokine level in tears of patients in relation to the number of instillations per day
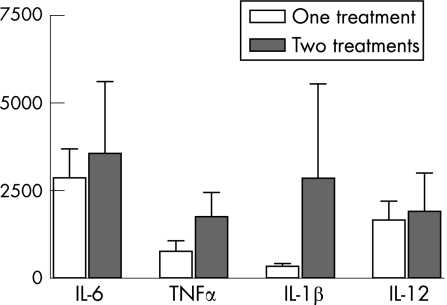
In the treated group, 13 patients received one treatment with preserved eye drops, whereas eight patients had at least two different preserved topical treatments with more than two instillations per day. The mean concentration of cytokines between these two groups did not differ significantly, although a tendency to higher expression in multitreated patients was found with TNFα and IL1β (fig 3).
Figure 3 Concentration of cytokines (pg/ml) in tears according to the number of instillations per day. The increase in the main pro‐inflammatory cytokine concentration in tears of patients who used more than one topical treatment was not statistically significant.
Discussion
Topical treatments for glaucoma have been shown to induce ocular surface disorders subsequent to the activation of inflammatory mechanisms.12 Conjunctival cells are able to produce cytokines, which are secreted in tears and have a key role in the initiation and development of the immunoinflammatory response. In this study, we used a recently developed flow cytometry method to measure cytokine level in tears, which provides many advantages over the traditional ELISA method, with the same or better results for characterisation of cytokines.13,14 The multiplex bead analysis makes it possible to measure several cytokines in a single sample to establish a profile of the inflammatory reaction. The results confirmed that inflammatory cytokines are overexpressed in tears of patients with glaucoma treated with preserved intraocular pressure‐lowering drugs compared with controls. The results obtained in our control group are in agreement with the concentrations observed in tears of healthy patients measured in another study by an alternative multiplex bead analysis system.15 The increase in IL6 and IL8 in patients with glaucoma receiving topical treatments has already been observed by flow cytometry in conjunctival epithelial cells obtained with impression cytology, and was associated with an increase of human leucocyte antigen (HLA)‐DR expression.16 In this study, we did not use impression cytology to harvest the material as our multiplex bead analysis kit had a better yield with tears than with collected cells. It would have been advantageous to perform a simultaneous assessment of HLA‐DR expression on our samples; however, our kit did not allow this measurement.
The measurement of the secreted part of these cytokines suggests that intracellular cytokines are excreted in tears after being produced in cells. BAC is probably the main factors responsible for this inflammation found on conjunctival cells. This has already been observed in an in vitro study on conjunctival cells17 where BAC was identified, even at a low level, as a promoter of cellular death. This is supported by the comparison of inflammatory markers in patients treated with preservative‐containing, and preservative‐free eye drops.18
Two major species of cytokines that are smaller than the others and show powerful attractive chemotactic properties on T cells and neutrophil polynuclear cells were considerably increased in the treated group: IL8 and MCP1. The level of IL8 in the control group was a mean of <1000 pg/ml, which is comparable with the results obtained by Nakamura et al on pooled samples of normal tears of 270 controls measured by ELISA.19
The conjunctival profile of interleukin expression showed that the Th1 pathway is activated to a greater extent than is Th2 in patients with glaucoma, although cytokines of both systems are overproduced. In fact, the Th1/Th2 ratio is higher in the treated group compared with controls. This observation suggests that cellular mediation is mainly involved in patients with glaucoma treated with topical treatments, whereas in tears of allergic patients a Th2 profile was observed instead.7 However, in our series, an increase in cytokines involved in the Th2 immune response was also observed. These results suggest that conjunctival immune response induced by topical treatments for glaucoma could be a mixed mechanism involving both systems, even if the Th1 pathway seems to be more preponderant. The involvement of both Th1 and Th2 systems was also suggested by the study of CCR4 and CCR5 expression on conjunctival cells in patients with a long‐term use of topical treatments.20
We could expect that the number of instillations per day would increase the BAC proinflammatory effects on the ocular surface. However, the cytokine concentration in tears was not significantly correlated with the number of daily instillations in our small series. Further studies will be required for better tackling of this issue, as a clear dose dependence was found with BAC toxicity, leading to expectations of higher inflammatory profiles in multitreated eyes, as was found with other cellular markers in impression cytology specimens.21
In this study, we have shown the feasibility of using multiplex bead analysis in analysing the tears of patients with glaucoma to determine the conjunctival inflammatory reaction more precisely. However, our series is limited and it should be advantageous to use this technique in patients receiving preservative‐free eye drops. The characterisation of the cytokine profile with multiplex analysis in tears seems to be a promising method to discriminate between Th1 and Th2 immune response and could help to screen less toxic/proinflammatory drugs and choose appropriate anti‐inflammatory treatment in these patients when needed.
Abbreviations
BAC - benzalkonium chloride
HLA - human leucocyte antigen
IFN - interferon
MCP - monocyte chemotactic protein
MIP - macrophage inflammatory protein
Footnotes
Competing interests: None declared.
References
- 1.Baudouin C. Side effects of antiglaucomatous drugs on the ocular surface. Curr Opin Ophthalmol 1996780–86. [DOI] [PubMed] [Google Scholar]
- 2.Broadway D, Grierson I, Hitchings R. Adverse effects of topical antiglaucomatous medications on the conjunctiva. Br J Ophthalmol 199377590–596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Broadway D, Grierson I, O'Brien C.et al Adverse effects of topical antiglaucoma medication. I. The conjunctival cell profile. Arch Ophthalmol 19941121437–1445. [DOI] [PubMed] [Google Scholar]
- 4.Baudouin C. Mechanisms of failure in glaucoma filtering surgery: a consequence of antiglaucomatous drugs? Int J Clin Pharmacol Res 19961629–41. [PubMed] [Google Scholar]
- 5.Guenoun J M, Baudouin C, Rat P.et al In vitro study of inflammatory potential and toxicity profile of latanoprost, travoprost, and bimatoprost in conjunctiva‐derived epithelial cells. Invest Ophthalmol Vis Sci 2005462444–2450. [DOI] [PubMed] [Google Scholar]
- 6.Mietz H, Niesen U, Krieglstein G K. The effect of preservatives and antiglaucomatous medication on the histopathology of the conjunctiva. Graefe's Arch Clin Exp Ophthalmol 1994232561–565. [DOI] [PubMed] [Google Scholar]
- 7.Cook E B, Stahl J L, Lowe L.et al Simultaneous measurement of six cytokines in a single sample of human tears using microparticle‐based flow cytometry: allergics vs. non‐allergics. J Immunol Methods 2001254109–118. [DOI] [PubMed] [Google Scholar]
- 8.Barton K, Monroy D C, Nava A.et al Inflammatory cytokines in the tears of patients with ocular rosacea. Ophthalmology 19971041868–1874. [DOI] [PubMed] [Google Scholar]
- 9.Pflugfelder S C, Jones D, Ji Z.et al Altered cytokine balance in the tear fluid and conjunctiva of patients with Sjogren's syndrome keratoconjunctivitis sicca. Curr Eye Res 199919201–211. [DOI] [PubMed] [Google Scholar]
- 10.Wilson M R, Wotherspoon J S. A new microsphere‐based immunofluorescence assay using flow cytometry. J Immunol Methods 1988107225–230. [DOI] [PubMed] [Google Scholar]
- 11.Vignali D A. Multiplexed particle‐based flow cytometric assays. J Immunol Methods 2000243243–255. [DOI] [PubMed] [Google Scholar]
- 12.Noecker R J, Herrygers L A, Anwaruddin R. Corneal and conjunctival changes caused by commonly used glaucoma medications. Cornea 200423490–496. [DOI] [PubMed] [Google Scholar]
- 13.Khan S S, Smith M S, Reda D.et al Multiplex bead array assays for detection of soluble cytokines: comparisons of sensitivity and quantitative values among kits from multiple manufacturers. Cytometry B Clin Cytometry 20046135–39. [DOI] [PubMed] [Google Scholar]
- 14.DuPont N C, Wang K, Wadhwa P D.et al Validation and comparison of luminex multiplex cytokine analysis kits with ELISA: determinations of a panel of nine cytokines in clinical sample culture supernatants. J Reprod Immunol 200566175–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Uchino E, Sonoda S, Kinukawa N.et al Alteration pattern of tear cytokines during the course of a day: diurnal rhythm analyzed by multicytokine assay. Cytokine 20063336–40. [DOI] [PubMed] [Google Scholar]
- 16.Baudouin C, Hamard P, Liang H.et al Conjunctival epithelial cell expression of interleukins and inflammatory markers in glaucoma patients treated over the long term. Ophthalmology 20041112186–2192. [DOI] [PubMed] [Google Scholar]
- 17.De Saint‐Jean M, Brignole F, Bringuier A F.et al Effects of benzalkonium chloride on growth and survival of Chang conjunctival cells. Invest Ophthalmol Vis Sci 199940619–630. [PubMed] [Google Scholar]
- 18.Pisella P J, Fillacier K, Elena P P.et al Comparison of the effects of preserved and unpreserved formulations of timolol on the ocular surface of albino rabbits. Ophthalmic Res 2000323–8. [DOI] [PubMed] [Google Scholar]
- 19.Nakamura Y, Sotozono C, Kinoshita S. Inflammatory cytokines in normal human tears. Curr Eye Res 199817673–676. [PubMed] [Google Scholar]
- 20.Baudouin C, Liang H, Bremond‐Gignac D.et al CCR 4 and CCR 5 expression in conjunctival specimens as differential markers of T(H)1/T(H)2 in ocular surface disorders. J Allergy Clin Immunol 2005116614–619. [DOI] [PubMed] [Google Scholar]
- 21.Pisella P J, Debbasch C, Hamard P.et al Conjunctival proinflammatory and proapoptotic effects of latanoprost and preserved and unpreserved timolol: an ex vivo and in vitro study. Invest Ophthalmol Vis Sci 2004451360–1368. [DOI] [PubMed] [Google Scholar]