Abstract
Seeds of a Chinese traditional medicine plant, Cochinchina momordica were used in the present study for the improvement of influenza vaccine (H5N1) in chicken. Crude extraction from Cochinchina momordica seed (ECMS) was obtained by ethanol extraction method. In experiment No. 1, two weeks old chickens were immunized with influenza vaccine (H5N1) alone or combined with ECMS (5, 10, 20, 40 and 80 μg/dose). Serum IgG antibody levels (by ELISA) as well as effects on daily weight gain were measured on 0, 7, 14 and 28th day after immunization. Results revealed that all ECMS groups numerically increased the antibody levels while 10 and 20 μg/dose groups significantly (P<0.05) enhanced total IgG antibody on day 28, when compared with control. Average daily weight gain was also significantly higher in 20 μg/dose ECMS group. Adjuvant effect was also confirmed in experiment No. 2 when chickens were immunized with 20 μg/dose ECMS and antibody titer was measured through hemagglutination inhibition (HI). It is concluded that ECMS has potential to improve the immune responses and deserve further study as an adjuvant.
Keywords: Cochinchina momordica, ECMS, Adjuvant, Chicken, Avian influenza (H5N1), Bird flue
INTRODUCTION
Influenza is an important disease of a wide variety of warm-blooded animals and is characterized by respiratory syndromes. It is recognized as a zoonotic disease with the most commonly affected being man, pigs, horses and species of aquatic birds (McCauley et al., 1995). The ability to cause disease and the ability of the host to respond to influenza varies greatly by species (Rimmelzwaan and Osterhaus, 2001). Influenza viruses belong to the family Orthomyxoviridae and are divided into three types, A, B and C. A type viruses are responsible for major disease problem in birds, as well as in humans (Alexander and Brown, 2000; Rimmelzwaan and Osterhaus, 2001; Normile, 2005) and are further classified into subtypes on the basis of the antigenic properties. Infection of poultry with avian influenza (AI) viruses (bird flu) causes a wide range of clinical signs including asymptomatic infections, mild to severe respiratory disease, production losses, and rarely, severe disease with high morbidity and mortality. Avian influenza viruses are typically characterized as either being highly pathogenic avian influenza (HPAI) viruses or mildly pathogenic avian influenza (MPAI) viruses (Suarez and Schultz-Cherry, 2000). HPAI virus is highly lethal in poultry and can cause large outbreaks leading to substantial economic loss and can spread directly from poultry to human, constituting a possible “pandemic threat” to the human population (Alexander and Brown, 2000). Highly pathogenic virus strains cause ‘fowl plague’, a generalized disease with mortality rates close to 100% (Easterday et al., 1997; Alexander, 2000). HPAI has continued to cause serious losses to the poultry industry and due to the numerous antigenic subtypes and rapid evolution, make avian influenza virus a challenging pathogen (Suarez and Schultz-Cherry, 2000). The availability and use of effective vaccines can be a valuable tool in controlling outbreaks of avian influenza. Numerous vaccines against avian influenza have been developed and experimentally shown to be efficacious for the prevention of disease, although the number of HPAI outbreaks in commercial poultry has been increasing. Adjuvanted killed vaccines can provide a strong humoral response and have proven to be effective against preventing disease from MPAI and HPAI challenges (Suarez and Schultz-Cherry, 2000). In order to increase the efficacy of influenza vaccines much attention is being paid to the development of new adjuvants and antigen delivery system (Rimmelzwaan and Osterhaus, 2001). A large number of potential adjuvants have been developed (Edelman and Tacket, 1990) or under investigation in laboratory animal. An adjuvant can be used for increasing the immunogenicity of poor antigen, improving the efficacy of vaccine in new born and reducing the amount of antigen or the number of immunizations needed (McElrath, 1995).
In the present study, we investigated the humoral immune response of influenza vaccine (H5N1) combined with the crude extraction of Cochinchina momordica seed (ECMS) as an adjuvant. Cochinchina momordica is a Chinese medicine plant and traditionally used against many diseases in animals and man (Zheng et al., 1992; Gao, 2005). Saponins have been reported to have unique adjuvant effects. Two kinds of saponins have been separated from the seed of Cochinchina momordica plant (Iwamoto et al., 1985). The present study is designed to determine the immune response of ECMS and its effect on growth performance.
MATERIALS AND METHODS
Preparation of ECMS
Dried Cochinchina momordica seeds were purchased from the market of Traditional Chinese Material Medica, Anhui Province, China and the seeds were appraised by Zhejiang Institute of Veterinary Drug Control. Seeds were extracted as described by Sheng et al.(2003). In brief, the seeds were homogenized using a blender, and then immersed in 50% ethanol for 24 h. The mixture was immersed in water bath and refluxed three times at 90 °C with each reflux taking 2~3 h. After filtration, ethanol was removed using a R502B rotary evaporator (Shenko Tech. Co. Ltd., Shanghai, China) and the condensed material was then dissolved in water. After addition of 101 detergent solution (5%), it was centrifuged at 3000 r/min for 20 min. Collected upper layer of liquid was rotated with the help of rotary evaporator and mixed with diethyl ether (anhydrous) at ratio of 1:2 for 2 h to dissolve the fat, colour pigments or some other unwanted substances. Diethyl ether was evaporated with the help of rotary evaporator. It was then immersed in n-butanol for 3 h and the upper layer of liquid was collected. It was repeated 3~4 times and rotated again for ethanol evaporation. Extracted fraction was dissolved in water (pH 6.5) and purified by passing through a chromatography column with resin D101A (Hai Guang Chemical Co. Ltd., Tianjin, China). A purified extract of Cochinchina momordica seeds (ECMS) was obtained by evaporating the liquid eluted from the column.
A water based solution of ECMS (1 mg/ml) was prepared and sterilized by passing through 0.22 μm filter and then analyzed for endotoxin level by a gel clot Limulus amebocyte lysate assay (Zhejiang A and C Biological, Zhejiang, China). The endotoxin level was less than 0.5 EU (endotoxin unit)/ml.
Vaccine
Inactivated avian influenza vaccine (H5N1 subtype, Re1 strain) was purchased from Harbin Veterinary Research Institute, China and combined with different concentration of ECMS to immunize the subject of interest.
Experiment No. 1: adjuvant effects and dose selection of ECMS
This experiment was conducted with different concentration of ECMS combined with vaccine, for adjuvant effects and selection of best dose of ECMS as well as its effects on growth.
1. Animals
Sixty, one-day-old Isa chickens (male), purchased from Hangzhou Layer Experimental Farm, were housed in wire cages in air-conditioned room at 37 °C and lighted for 24 h at the beginning of pretrial period. The temperature was gradually decreased to the room temperature and the light time to 12 h per day, which were kept constant in the following days. Chickens were fed with the commercial starter diet, purchased from the same company mentioned above.
2. Experimental design
At the age of two weeks, the chickens were randomly distributed into six groups of 10 individuals each. Serum samples were collected for the detection of maternal antibody level before immunization. All the birds were intramuscularly immunized with 0.2 ml of vaccine alone (control) or combined with 5, 10, 20, 40 or 80 µg/dose of ECMS. Blood samples were collected on days 0, 7, 14 and 28 and separated serum was stored at −20 °C until use. Live body weight of each bird was also measured at same time.
3. Measurement of serum IgG antibody
Serum samples were analyzed by an enzyme-linked immunosorbent assay (ELISA). ELISA kit was purchased from IDEXX Laboratories Inc., USA and assay was performed according to their instructions. Samples of test or standard sera were diluted in serum diluent (1:500) into test tubes. One hundred microlitres of diluted serum samples were added in duplicate to appropriate wells of AI antigen coated microtiter plates and incubated for 30 min at room temperature. After washing three times with distilled water, 100 µl of goat anti-chicken/turkey, horseradish peroxidase conjugate (IDEXX) was added to the wells. After 30 min incubation at room temperature, the plates were washed again and 100 µl of TMB-substrate solution was added to each well for 15 min at room temperature. The reaction was stopped by adding 100 µl of sulphuric acid and the optical density (OD) values at 630 nm were read with microplate spectrophotometer (Dialab, GMBH, Austria). The relative level of antibody in the unknown was determined by calculating the sample to positive ratio:
| Serum antibody level=OD value of tested sample/OD value of positive control. |
4. Effect on growth performance
Weight of each bird was measured on days 0, 7, 14 and 28 and effect on weight gain was studied. Average of live body weight, total weight gain and daily weight gain of different groups were compared.
Experiment No. 2: adjuvant effects of 20 µg/dose of ECMS
This experiment was conducted on the basis of results obtained from experiment No. 1.
1. Animals
One-day old Lohmann Brown layer chickens were raised at Yuhang Poultry Breeding Farm (Hangzhou, China) in hygienic condition.
2. Experimental design
Fifty chickens were randomly divided into two groups (25 each). At the age of two weeks, serum samples were collected for the detection of maternal antibody level before immunization. All the birds were intramuscularly immunized with 0.2 ml of vaccine alone (control) or combined with 20 µg/dose of ECMS. Blood samples were collected on days 0, 14 and 28. Serum was separated and stored at −20 °C until use.
3. Serum HI antibody assay
Antibody titer was measured by hemagglutination inhibition (HI) test (Swayne et al., 1998). The HI test was a standard beta test, using 4 hemagglutinating units of antigen in 96-well plates, where the test serum had been diluted twofold. HI endpoint titers were determined as the reciprocal of the highest serum dilution that produced complete inhibition of hemagglutinating activity.
Statistical analysis
SPSS software was used for data analysis. Data are expressed as the mean±SD. Duncan’s multiple range test was used to determine the difference among various groups. Difference between means was considered significant at P<0.05.
RESULTS
Serum IgG antibody response
Serum samples collected from chicken were analyzed for IgG antibody level to avian influenza virus (H5N1) using the ELISA test. The results are shown in Table 1. On day 7, total IgG antibody titer of all groups decreased and then increased when ECMS group (5, 10 and 20 µg) was a little higher than control. On day 14, titers were numerically higher in the birds receiving 20 and 80 µg ECMS than in control. Significantly higher value (P<0.05) was found in 10 or 20 µg ECMS groups on day 28, when compared with control.
Table 1.
Measurement of IgG antibody level by ELISA
| EMCS | 0 d | 7 d | 14 d | 28 d |
| 5 µg/dose | 0.492±0.05 | 0.413±0.04 | 0.743±0.38 | 1.791±0.85 |
| 10 µg/dose | 0.603±0.09 | 0.401±0.05 | 0.858±0.34 | 1.912±0.71* |
| 20 µg/dose | 0.552±0.13 | 0.397±0.04 | 0.971±0.39 | 2.070±0.39* |
| 40 µg/dose | 0.580±0.12 | 0.390±0.05 | 0.866±0.73 | 1.824±0.78 |
| 80 µg/dose |
0.542±0.08 |
0.386±0.04 |
0.970±0.52 |
1.846±0.70 |
| Control | 0.507±0.07 | 0.383±0.03 | 0.928±0.59 | 1.537±0.71 |
Significant (P<0.05)
Effect on growth performance
Changes in weight during experimental period are mentioned in Tables 2 and 3. Numerical increase of weight changes in 10 and 20 µg/dose ECMS groups was recorded on day 14, but was not significance when compared with control. Dynamic changes in weight were observed on day 28, as the value of 20 µg/dose ECMS group was significantly higher (P<0.05) than all groups including control. Average of total weight gain and daily weight gain of 20 µg/dose ECMS group were significantly higher (P<0.05) as compared to all tested groups.
Table 2.
Changes in weight at different intervals
| ECMS | 0 d | 7 d | 14 d | 28 d |
| 5 µg/dose | 88.4±7.50 | 138.7±12.50 | 182.9±14.60 | 262.6±32.84 |
| 10 µg/dose | 91.3±6.65 | 144.2±12.92 | 192.3±16.98 | 275.7±31.17 |
| 20 µg/dose | 86.6±7.89 | 145.4±9.23 | 196.7±15.00 | 302.1±38.69* |
| 40 µg/dose | 84.8±4.02 | 130.7±6.18 | 176.7±17.86 | 245.3±34.85 |
| 80 µg/dose |
85.7±4.37 |
130.7±9.97 |
176.8±14.86 |
245.0±29.34 |
| Control | 86.6±8.03 | 144.0±20.63 | 188.8±30.95 | 267.8±33.26 |
Significant (P<0.05)
Table 3.
Average growth performance
| ECMS | Average initial weight | Average final weight | Average total weight gain | Average daily weight gain |
| 5 µg/dose | 88.4±7.50 | 262.6±32.84 | 174.2±31.46 | 6.22±1.12 |
| 10 µg/dose | 91.3±6.65 | 275.7±31.17 | 184.4±27.32 | 6.59±0.98 |
| 20 µg/dose | 86.6±7.89 | 302.1±38.69 | 215.5±38.65* | 7.70±1.38* |
| 40 µg/dose | 84.8±4.02 | 245.3±34.85 | 160.5±34.55 | 5.73±1.23 |
| 80 µg/dose |
85.7±4.37 |
245.0±29.34 |
159.3±29.12 |
5.69±1.04 |
| Control | 86.6±8.03 | 267.8±33.26 | 181.2±30.63 | 6.47±1.09 |
Significant (P<0.05)
Changes in serum antibody titer
Changes in serum antibody titer are given in Table 4. On days 14 and 28 after vaccination, the antibody titers of 20 µg/dose ECMS group were significantly higher (P<0.05) than control.
Table 4.
Variation of HI antibody titer (log2)
Significant (P<0.05)
DISCUSSION
Technologies are now in place to design and construct new influenza virus vaccines that have the potential to be cheaper and more cross-protective than current vaccine preparations, while at the same time being equally safe (Palese, 2006). Numerous approaches to develop more effective influenza vaccine are under investigation. Adjuvants are reported to broaden the immune response which could enhance immunity to influenza (Halman, 1993). Technologic breakthroughs should result in effective adjuvants for use with killed and live vaccines, increasing the number of available doses (Palese, 2006). Many adjuvants are now under investigation. Liposome-like preparations containing cholesterol and viral particles (immune-stimulating complexes) (Sambhara et al., 2001) or synthetic adjuvants, such as synthetic lipid A, muramyl peptide derivatives, and cationic molecules (Fritz et al., 2004) have been reported as successful adjuvants. Saponins based adjuvants have the unique ability to enhance the immunity (Oda et al., 2000). Saponins produced from the Ginseng, bark of Quillaja saponaria, Astragalus, Achyranthes bidentata, Acacia victoriae, soybean, etc. (Yang et al., 2005; Hu et al., 2001; Wang et al., 2001; Francis et al., 2002; Haridas et al., 2004; Rivera et al., 2005; Kong et al., 2004; Li et al., 2003; 2005; Chen et al., 2005) have been reported as active adjuvant to enhance the immune responses. Iwamoto et al.(1985) isolated two kinds of saponins from presently investigated seeds of Momordica cochinchinensis. It is produced in large area of South Asian countries and is very cheap. The seeds have been traditionally used for the treatment of inflammatory swelling, scrofula, tinea, diarrhoea as well as suppurative skin infections such as sore, carbuncles, furuncles and boils in humans and animals (Zheng et al., 1992; Gao, 2005).
Currently, the hemagglutination inhibition (HI) assay is the preferred serological assay to detect antibodies to avian influenza virus (Huang et al., 1990), while ELISA is also commonly used method (Gan, 2004).
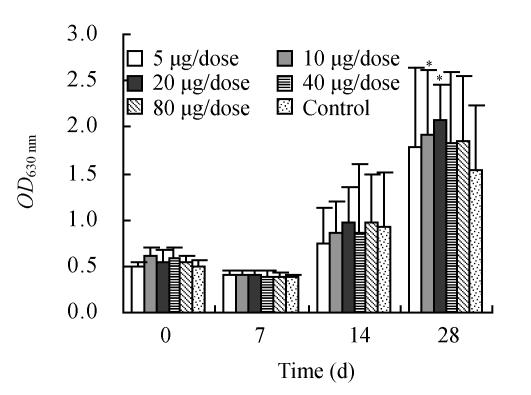
The present results show that incorporation of influenza vaccine (H5N1) into ECMS enhanced the serum IgG response (Fig.1). From the results presented here it is clear that immunization with formulations of influenza vaccine containing 10 and 20 µg/dose ECMS yields significantly higher IgG antibody levels when compared with control on 28th day. Comparing the IgG antibody level among all groups, 20 µg/dose ECMS had the best effect.
Fig. 1.
Changes in IgG antibody level
* indicates significant values (P<0.05)
The changes of antibody titer reflected the state of the humoral immunity in the animal. Ascertained results evidenced that antibody titer of ECMS group was significantly higher than control (Fig.2) which suggested that ECMS could strengthen the humoral immunity. The humoral immunity is one of the major ways to resist infectious diseases.
Fig. 2.
Variation of HI antibody titer
* indicates significant values (P<0.05)
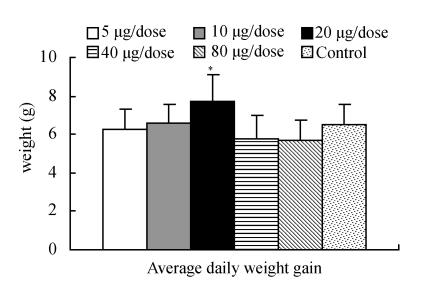
The effect on growth was also recoded during the study and no adverse effect was observed. Comparison of weight gain on days 0, 7, 14 and 28, average daily weight gain, and average of total weight gain in all groups are depicted in Figs.3, 4 and 5 respectively. Results revealed that average daily weight gain of 10 and 20 µg/dose ECMS group was better than all groups including control during the whole the period of study. The value of 20 µg/dose ECMS group was numerically higher on days 7 and 14 but significant increase in weight gain was observed on day 28 when compared with control. Total weight gain of 20 µg/dose ECMS group was also significantly higher than control.
Fig. 3.
Comparison of weight gain of all groups on days 0, 7, 14 or 28
* indicates significant value (P<0.05)
Fig. 4.
Comparison of average daily weight gain
* indicates significant value (P<0.05)
Fig. 5.
Comparison of average total weight gain
* indicates significant value (P<0.05)
CONCLUSION
The present experiment was conducted in controlled condition and no other antigen was introduced into the chicken except influenza vaccine (H5N1).
It is concluded that ECMS has adjuvant properties to enhance the antibody level of total IgG when used in combination with influenza vaccine (H5N1) and has no adverse effects on growth performance. Therefore, ECMS deserves further study as a vaccine adjuvant.
Acknowledgments
Higher Education Commission of Pakistan is appreciated for financial support of a Ph.D. scholarship to Rajput Zahid Iqbal (first author).
Footnotes
Project (No. 2004C32047) supported by the Department of Science and Technology of Zhejiang Province, China
References
- 1.Alexander DJ. A review of avian influenza in different bird species. Vet Microbiol. 2000;74(1-2):3–13. doi: 10.1016/S0378-1135(00)00160-7. [DOI] [PubMed] [Google Scholar]
- 2.Alexander DJ, Brown IH. Recent zoonoses caused by influenza A viruses. Rev Sci Tech. 2000;19(1):197–225. doi: 10.20506/rst.19.1.1220. [DOI] [PubMed] [Google Scholar]
- 3.Chen XM, Xu YJ, Tian GY. Physical-chemical properties and structure elucidation of abPS isolated from the root of Achyranthes bidentata . Acta Pharm Sin. 2005;40(1):32–35. [PubMed] [Google Scholar]
- 4.Easterday BC, Hinshaw VS, Halvorson DA. Influenza. In: Calnek BW, Barnes HJ, Beard CW, et al., editors. Diseases of Poultry. Ames: Iowa State University Press; 1997. pp. 583–605. [Google Scholar]
- 5.Edelman R, Tacket CO. Adjuvants. Int Rev Immunol. 1990;7(1):51–66. doi: 10.3109/08830189009061764. [DOI] [PubMed] [Google Scholar]
- 6.Francis G, Kerem Z, Makkar HPS, Becker K. The biological action of saponins in animal system: a review. British J Nutr. 2002;88(6):587–605. doi: 10.1079/BJN2002725. [DOI] [PubMed] [Google Scholar]
- 7.Fritz JH, Brunner S, Birnstiel ML, Buschle M, Gabain A, Mattner F, Zauner W. The artificial antimicrobial peptide KLKLLLLLKLK induces predominantly a Th2-type immune response to co-injected antigens. Vaccine. 2004;22(25-26):3274–3284. doi: 10.1016/j.vaccine.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 8.Gan MH. Avian Influenza. 2nd Ed. Beijing, China: Chinese Agricultural Press; 2004. pp. 112–117. [Google Scholar]
- 9.Gao XM. Mu Bie Zi (Semen momordicae). Chinese Materia Medicia. Beijing, China: China Traditional Chinese Materia Medica Press; 2005. pp. 601–602. [Google Scholar]
- 10.Halman C. New Approaches to Vaccine Delivery. In: Hannoun C, Kendal AP, Klenk HD, et al., editors. Options for the Control of Influenza. Volume II. Amsterdam: Excerpta Medica; 1993. pp. 389–391. [Google Scholar]
- 11.Haridas V, Hanausek M, Nishimura G, Soehnge H, Gaikwad A, Narog M, Spears E, Zoltaszek R, Walaszek Z, Gutterman JU. Triterpenoid electrophiles (avicins) activate the innate stress response by redox regulation of a gene battery. J Clin Invest. 2004;113(1):65–73. doi: 10.1172/JCI200418699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hu S, Concha C, Johannisson A, Meglia G, Waller KP. Effect of subcutaneous injection of ginseng on cows with sub-clinical Staphylococcus aureus mastitis. J Vet Med B. 2001;48(7):519–528. doi: 10.1046/j.1439-0450.2001.00470.x. [DOI] [PubMed] [Google Scholar]
- 13.Huang ZX, Huang T, Liu CB. Medical Virology Base and Fundamental Techniques and Methods. Beijing, China: Science Press; 1990. pp. 150–164. [Google Scholar]
- 14.Iwamoto M, Okabe H, Yamauchi T, Tanaka M, Rokutani Y, Hara S, Mihashi K, Higuchi R. Studies on the constituents of Momordica cochinchinensis Spreng I. Isolation and characterization on the seed saponins, momordica saponins I and II. Chem Pharm Bull. 1985;33(2):464–478. [Google Scholar]
- 15.Kong X, Hu Y, Rui R, Wang D, Li X. Effects of Chinese herbal medicinal ingredients on peripheral lymphocyte proliferation and serum antibody titer after vaccination in chicken. Int Immunopharmacol. 2004;4(7):975–982. doi: 10.1016/j.intimp.2004.03.008. [DOI] [PubMed] [Google Scholar]
- 16.Li CC, Hu XG, Zhang WX, Xie LW, Zhang HY, Dong L, Cai XH, Wu RX, Zhang ZX, He QS. Eosinophils apoptosis, fas mRNA and bcl-2 mRNA expressions in asthma model of young rat and effects of Achyranthes bidentata polysaccharides. Chin J Pediatr. 2003;41(9):657–660. [PubMed] [Google Scholar]
- 17.Li JX, Hareyama T, Tezuka Y, Zhang Y, Miyahara T, Kadota S. Five new oleanolic acid glycosides from Achyranthes bidentata with inhibitory activity on osteoclast formation. Planta Med. 2005;71(7):673–679. doi: 10.1055/s-2005-871275. [DOI] [PubMed] [Google Scholar]
- 18.McCauley JW, Pullen LA, Forsyth M, Penn CR, Thomas GP. 4-Guanidino-NeuAc2en fails to protect chickens from infection with highly pathogenic avian influenza virus. Antivir Res. 1995;27(1-2):179–186. doi: 10.1016/0166-3542(95)00005-7. [DOI] [PubMed] [Google Scholar]
- 19.McElrath MJ. Selection of potent immunological adjuvants for vaccine construction. Semin Cancer Biol. 1995;6(6):375–385. doi: 10.1016/1044-579X(95)90007-1. [DOI] [PubMed] [Google Scholar]
- 20.Normile D. North Korea collaborates to fight bird flu. Science. 2005;308(5719):175. doi: 10.1126/science.308.5719.175a. [DOI] [PubMed] [Google Scholar]
- 21.Oda K, Matsuda H, Murakami T, Katayama S, Ohgitani T, Yoshikawa M. Adjuvant and haemolytic activities of 47 saponins derived from medicinal and food plants. Biol Chem. 2000;381(1):67–74. doi: 10.1515/BC.2000.009. [DOI] [PubMed] [Google Scholar]
- 22.Palese P. Making better influenza virus vaccines? Emerg Infect Dis. 2006;12(1):61–65. doi: 10.3201/eid1201.051043. (Available from: http://www.cdc.gov/eid) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rimmelzwaan GF, Osterhaus AD. Influenza vaccine: new developments. Curr Opin Pharmacol. 2001;1(5):491–496. doi: 10.1016/S1471-4892(01)00085-6. [DOI] [PubMed] [Google Scholar]
- 24.Rivera E, Ekholm PF, Inganas M, Paulie S, Gronvik KO. The Rb1 fraction of ginseng elicits a balanced Th1 and Th2 immune response. Vaccine. 2005;23(46-47):5411–5419. doi: 10.1016/j.vaccine.2005.04.007. [DOI] [PubMed] [Google Scholar]
- 25.Sambhara S, Kurichh A, Miranda R, Tumpey T, Rowe T, Renshaw M, Arpino R, Tamane A, Kandil A, James O, et al. Heterosubtypic immunity against human influenza A viruses, including recently emerged avian H5 and H9 viruses, induced by FLU-ISCOM vaccine in mice requires both cytotoxic T-lymphocyte and macrophage function. Cell Immunol. 2001;211(2):143–153. doi: 10.1006/cimm.2001.1835. [DOI] [PubMed] [Google Scholar]
- 26.Sheng YX, Jun WL, Zhou WJ. Natural Medicine Chemistry. Triterpenoid Saponins (Chapter 7), Extraction and Separation. Beijing, China: People Hygiene Publisher; 2003. p. 294. (in Chinese) [Google Scholar]
- 27.Suarez DL, Schultz-Cherry S. Immunology of avian influenza virus: a review. Dev Comp Immunol. 2000;24(2-3):269–283. doi: 10.1016/S0145-305X(99)00078-6. [DOI] [PubMed] [Google Scholar]
- 28.Swayne DE, Senne DA, Beard CW. Avian Influenza. In: Swayne DE, editor. A Laboratory Manual for the Isolation and Identification of Avian Pathogens. Kennett Square, Pa: American Association of Avian Pathologists; 1998. pp. 150–155. [Google Scholar]
- 29.Wang M, Guilbert LJ, Ling L, Li J, Wu Y, Xu S, Pang P, Shan JJ. Immunomodulating activity of CTV-E002, a proprietary extract from North American ginseng (Panax equinquefolium) J Pharm Pharmacol. 2001;53(11):1515–1523. doi: 10.1211/0022357011777882. [DOI] [PubMed] [Google Scholar]
- 30.Yang ZG, Sun HX, Fang WH. Haemolytic activities and adjuvant effect of Astragalus membranaceus saponins (AMS) on the immune responses to ovalbumin in mice. Vaccine. 2005;23(44):5196–5203. doi: 10.1016/j.vaccine.2005.06.016. [DOI] [PubMed] [Google Scholar]
- 31.Zheng S, Li GE, Yan SM. Purification and characterisation of momordin. Acta Biochim Biophys Sin. 1992;24:311–316. (in Chinese) [Google Scholar]