Abstract
Objective
To evaluate the clinical utility of a targeted screening approach for the detection of genetic haemochromatosis.
Methods
Screening by measuring fasting serum transferrin saturation (TS) and gene testing was carried out in patients in whom a raised serum alanine amino transferase (ALT) activity and raised random serum TS had been found on routine blood testing.
Results
During the 29 month study period, 32 patients homozygous for the C282Y genotype were detected from a catchment population of 330 000 by screening blood samples referred initially for routine laboratory liver function tests. By comparison, during the same period of time and within the same population, only seven patients were found by clinical suspicion alone. The patients in the study, after treatment by venesection, have shown both clinical and biochemical improvement.
Conclusions
The study shows that from a population of patients in whom a routine liver function profile had been requested, it is possible to detect subjects homozygous for the C282Y HFE genotype who have clinical or biochemical markers of iron overload.
Keywords: screening, haemochromatosis, ALT, iron overload
Genetic haemochromatosis is one of the commonest inherited metabolic disorders among white populations of Northern European descent. The disease is predominantly caused by a mutation of the HFE gene, with approximately 1 in 200 people being homozygous for the C282Y mutation and thus at increased risk of developing iron overload.1 When the diagnosis is made early, iron can be removed by repeated venesection and life expectancy is improved. Once cirrhosis has developed, it is not reversible by treatment and though patients can live for extended periods they have a markedly increased risk of developing hepatic cancer.2,3
The identification of mutations in the HFE gene provides a specific genetic test for the disorder.4 However, as the HFE genotype shows incomplete penetrance5,6,7 and as other genetic and environmental factors are known to affect the development of the disease,8 whole population screening for HFE gene mutations is unlikely to be an effective strategy for disease prevention. Screening potentially high risk groups—for example, diabetics and patients with liver disease—has similarly been proven to be largely ineffective,9 although a recent study by Poullis et al suggests that targeted screening of liver clinic patients may be both effective and cost‐efficient.10 Most authorities therefore suggest that there should be an initial screen for the phenotype and follow up genetic testing.11,12 Serum transferrin saturation (TS) is increased in most subjects homozygous for the HFE C282Y mutation7,13 and it is the recommended phenotypic screening test in the United Kingdom.14 Following an initial pilot study,15 we have introduced a screening programme for genetic haemochromatosis (using raised alanine aminotransferase (ALT) and TS) in a targeted population of patients in whom raised ALT activity had been detected in a blood sample sent for routine biochemistry. In this paper, we present the results of this screening programme—a combined phenotype/genotype approach.
Methods
Wrightington Wigan & Leigh Trust serves a mainly white population of 330 000. Over the 29 month study period, blood samples received for routine liver function tests were screened for HFE mutations using the protocol shown in fig 1. During this period, 210 000 blood samples for routine liver function tests were received from approximately 100 000 patients (40% inpatients, 49% outpatients, and 11% general practitioner patients). The male to female distribution of the subjects was 1:1.2. Serum liver function tests, including ALT, were measured on a Roche Modular analyser using standard methodology. In those samples with an ALT >50 U/l (reference range 5 to 45 U/l), serum iron and transferrin were also assayed on the same instrument. The transferrin saturation was calculated as [serum iron (μmol/l)/transferrin (g/l)]×4.
Figure 1 Study screening protocol used to detect C282Y homozygotes in patients having a routine serum liver function test undertaken. Numbers in italics refer to the numbers of subjects identified at each stage of the screening protocol over the 29 month study period.
Serum ferritin concentration was measured on a Roche Elecsys 2010 analyser. HFE mutations were detected by polymerase chain reaction (PCR)–heteroduplex analysis.16,17 Spearman's rank correlation and linear regression analysis were used to compare the biochemical variables and to assess the statistical significance of the biochemical response to treatment.
Patients with fasting TS >55% were invited to attend an outpatient clinic to see one of the authors (MB), when a detailed history and clinical examination were carried out, permission sought to perform the gene test, and venesection arranged as necessary.
Results
Over 29 months of the study, from samples received by the laboratory for routine liver function tests 8770 patients were identified as having a raised serum ALT (>50 U/l) and were further analysed for TS. Of these samples, 376 (4.3%) were found to have a TS above the cut off point (55%) and a request for a repeat fasting sample was made to the referring clinician. Approximately half the clinicians responded, no difference in response rate being noted between those from primary or secondary care. Of the 194 fasting samples obtained, 70 had a fasting TS above 55%. In those patients with a fasting TS greater than 55%, an invitation was sent to attend the haematology clinic for genetic testing and assessment of iron status. Following counselling and consent, genetic testing was carried out on 66 of these patients (fig 1).
There were 32 C282Y HFE homozygotes among the patients screened as described above. Serum ferritin concentrations, ALT activity, and fasting TS at diagnosis are given in table 1, compared with values in the normal/heterozygous patients,. There were no significant differences in any of these variables between subjects homozygous for the C282Y mutation and the other genotypes apart from serum ferritin in male subjects, where the homozygotes showed a significantly higher value (p = 0.005). Serum ferritin levels at presentation ranged from 321 to 5419 μg/l (median 1362) in the male homozygotes, and from 451 to 1629 μg/l (median 949) in female homozygotes. There was no significant correlation between serum ALT and serum ferritin at diagnosis in the C282Y homozygotes or in the other genotypes group.
Table 1 Blood indices at diagnosis in patients having fasting transferrin saturation greater than 55% and referred for genetic testing.
| Measurement | Genotype | No of patients | Mean (SD) | Median | Significance‡ | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ferritin (μg/l) | C282Y homozygotes | M 27 | 17 93 (266) | 1362 | ||||||
| F 5 | 984 (453) | 949 | ||||||||
| Other genotypes | M 26* | 904 (844) | 645 | p = 0.005 | ||||||
| F 8* | 1414 (1229) | 946 | NS | |||||||
| ALT (U/l) | C282Y homozygotes | 32 | 91 (73) | 69 | ||||||
| Other genotypes | 36 | 142 (254) | 75 | NS | ||||||
| Fasting TS (%) | C282Y homozygotes | 32 | 78 (11) | 78 | ||||||
| Other genotypes | 31† | 64 (16) | 66 | NS |
*Serum ferritin values were not available on 2 of the 36 non‐C282Y genotypes.
†Fasting samples were not provided on 5 of the non‐C282Y patients. However, repeat transferrin saturation on non‐fasting samples from these patients were all >55% and the results therefore included in the analysis.
‡Versus respective homozygotes.
ALT, alanine aminotransferase; F, female; M, male; TS, transferrin saturation.
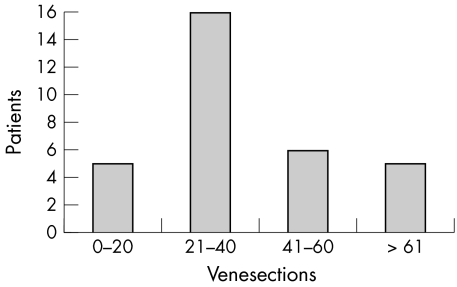
During the study period 24 men and two women underwent venesection (fig 2). The number of venesections to reduce serum ferritin to less than 50 μg/l was 15 to 72 units (median 35) in men.
Figure 2 Venesections carried out to date on the 32 patients homozygous for the C282Y mutation. For the five patients requiring fewer than 20 venesections, serum ferritin concentrations were less than 500 μg/l at presentation.
There was a strong relation between the serum ferritin concentration at diagnosis and the number of venesections required to reduce the serum ferritin to <50 μg/l (r = 0.8, p<0.0001). This confirms that the presenting serum ferritin concentration provides a good indicator of the degree of iron overload. There was also a significant correlation between the serum ferritin value at diagnosis and the number of venesections to reduce the serum ALT to <50 U/l, suggesting that the liver cell damage at this stage of the disease is reversibly related to the degree of iron overload (r = 0.65, p = 0.0064).
Serum ALT values were reduced in all patients undergoing venesection, the majority having values within the reference range at the time of writing (fig 3).
Figure 3 Reduction in serum alanine aminotransferase (ALT) following venesection in patients homozygous for the C282Y mutation.
At presentation before the diagnosis of genetic haemochromatosis was made, the 32 C282Y homozygotes (27 men and five women) showed symptoms and signs similar to published studies2,18—that is, fatigue (30%), arthralgia (30%), and diabetes mellitus (10%). However, in our study more patients appeared to be asymptomatic (52%), possibly suggesting recognition of the condition at an earlier stage of the disease process. Of those patients undergoing venesection, 20 of 24 have reported a marked improvement in clinical wellbeing.
Of the 32 homozygotes, five either declined venesection (1), or were lost to follow up (4).
Over the same period of time as the screening study described here, the laboratory had received 68 samples for gene tests from all clinicians in the hospital, from cases in whom genetic haemochromatosis was suspected on clinical grounds alone. Seven C282Y homozygotes were detected by these traditional clinical methods. Thirty six samples were requested as part of family studies and five homozygotes were detected. Thus the diagnostic yield on samples referred for genetic testing from the screening study was 32 of 66 (48%), compared with 7 of 68 (10%) from traditional clinical methods, and 5 of 36 (14%) from tests done for family studies (table 2).
Table 2 Results of gene testing in the screening study compared to routine clinical diagnosis.
| Screening study | Clinical suspicion | Family follow up | ||||
|---|---|---|---|---|---|---|
| Total gene tests | 66 | 68 | 36 | |||
| C282Y homozygotes | 32 | 7 | 5 | |||
| C282Y heterozygotes | 5 | 13 | 16 | |||
| Compound heterozygotes (C282Y/H63D) | 5 | 6 | 1 | |||
| H63D homozygotes | 3 | 3 | 1 | |||
| H63D heterozygotes | 8 | 15 | 5 | |||
| Normal | 13 | 24 | 8 | |||
| Diagnostic yield of C282Y homozygote detection | 48% | 10% | 14% |
Discussion
In this study we set out to evaluate the clinical utility of a targeted screening approach for the detection of genetic haemochromatosis. Non‐fasting TS, used as the initial phenotypic screen, is the biochemical marker of iron overload that offers the best combination of sensitivity and specificity over serum iron or ferritin.19 We acknowledge that the use of transferrin saturation, as a screen, in acutely ill hospital inpatients may be problematic because of the acute phase response which lowers serum iron and transferrin saturation,20 but this does not affect its use in ambulatory patients.
Expert opinion varies over the best threshold to define a raised TS, and values range from 45% to 65%.21 In our study we have adopted a non‐fasting TS threshold of 55% to offer the best compromise of sensitivity and specificity. To further improve specificity, we have followed up the non‐fasting TS in those patients with a value above the cut off with a fasting TS value.14 The selection of 55% as a cut off for the fasting TS does not seem to have excluded C282Y homozygotes, as of 32 patients identified as being homozygous for C282Y, 30 had a fasting transferrin saturation greater than 65%. This has resulted in a diagnostic yield of 48% (32 patients homozygous for C282Y found in the 66 patients who had a raised fasting TS) compared with 27% in our previous study,15 in which non‐fasting TS alone was used.
Take home message
Targeted screening by clinical biochemistry laboratories through routine liver function tests and random transferrin saturation estimation is an effective strategy to detect homozygotes for the C282Y mutation of the HFE gene, who may well benefit from treatment.
Our previous pilot study15 showed that it was possible to detect homozygotes with iron overload and then to offer them treatment by venesection. This study confirms the validity of this approach over a longer time frame. It demonstrates that ALT measurement and subsequent TS estimation is an effective method of screening for genetic haemochromatosis.
In the United Kingdom about 1 in 200 of the population of northern European origin are homozygous for HFE C282Y.22 Thus the 100 000 subjects tested should include over 500 C282Y homozygotes. The yield of only 32 homozygotes shows the low clinical penetrance of the condition, as fewer than 5% of the expected number of C282Y homozygotes had both a raised ALT activity (indicating possible liver disease) and a raised transferrin saturation (indicating iron accumulation). This confirms the survey of iron overload in south Wales,6 in which it was calculated that the lifetime cumulative incidence of a potential homozygote receiving treatment for iron overload in hospital was about 5.1%. Despite the low penetrance, the simplicity of testing within a routine clinical laboratory enables clinicians to identify and treat asymptomatic patients during the early stages of iron overload. Over the same period of time, the yield of homozygotes detected by traditional clinical methods was less effective, with only seven patients being identified by these means.
There are no controlled studies showing that venesection benefits individuals with haemochromatosis detected in the early stages of iron accumulation. However, if they remain untreated, some will progress to liver damage. As iron removal is simple and safe it was considered justified to reduce iron stores, but long term follow up of these patients will be undertaken to determine whether arthritis, diabetes mellitus, and other clinical sequelae of haemochromatosis have been prevented.
In conclusion, our study shows that from a population of patients in whom a routine liver function profile had been requested, it is possible to detect those homozygous for the C282Y HFE genotype who display clinical and biochemical markers of iron overload. The patients so identified may be at a presymptomatic stage of the disease process, and with treatment to remove the excess iron, may not develop irreversible organ damage.
Acknowledgements
We are grateful to the Wigan & Leigh Health Authority for funding
Abbreviations
ALT - alanine aminotransferase
TS - transferrin saturation
References
- 1.Worwood M. Inherited iron loading: genetic testing in diagnosis and management. Blood Rev 20051969–88. [DOI] [PubMed] [Google Scholar]
- 2.Niederau C, Fischer R, Purschel A.et al Long‐term survival in patients with hereditary hemochromatosis. Gastroenterology 19961101107–1119. [DOI] [PubMed] [Google Scholar]
- 3.Adams P C, Speechley M, Kertesz A E. Long‐term survival analysis in hereditary hemochromatosis. Gastroenterology 1991101368–372. [DOI] [PubMed] [Google Scholar]
- 4.Feder J N, Gnirke A, Thomas W.et al A novel MHC class I‐like gene is mutated in patients with hereditary haemochromatosis. Nat Genet 199613399–408. [DOI] [PubMed] [Google Scholar]
- 5.Asberg A, Hveem K, Thorstensen K.et al Screening for hemochromatosis: high prevalence and low morbidity in an unselected population of 65,238 persons. Scand J Gastroenterol 2001361108–1115. [DOI] [PubMed] [Google Scholar]
- 6.McCune C A, Al Jader L N, May A.et al Hereditary haemochromatosis: only 1% of adult HFE C282Y homozygotes in South Wales have a clinical diagnosis of iron overload. Hum Genet 2002111538–543. [DOI] [PubMed] [Google Scholar]
- 7.Beutler E, Felitti V J, Koziol J A.et al Penetrance of 845G→A (C282Y) HFE hereditary haemochromatosis mutation in the USA. Lancet 2002359211–218. [DOI] [PubMed] [Google Scholar]
- 8.Bomford A. Genetics of haemochromatosis. Lancet 20023601673–1681. [DOI] [PubMed] [Google Scholar]
- 9.Worwood M. HFE mutations as risk factors in disease. Best Pract Res Clin Haematol 200215295–314. [PubMed] [Google Scholar]
- 10.Poullis A, Moodie S J, Ang L.et al Routine transferrin saturation measurement in liver clinic patients increases detection of hereditary haemochromatosis. Ann Clin Biochem 200340521–527. [DOI] [PubMed] [Google Scholar]
- 11.Burke W, Reyes M, Imperatore G. Hereditary haemochromatosis: a realistic approach to prevention of iron overload disease in the population. Best Pract Res Clin Haematol 200215315–328. [PubMed] [Google Scholar]
- 12.Anon EASL International Consensus Conference on Haemochromatosis. J Hepatol 200033485–504. [DOI] [PubMed] [Google Scholar]
- 13.Adams P C, Reboussin D M, Barton J C.et al Hemochromatosis and iron‐overload screening in a racially diverse population. N Engl J Med 20053521769–1778. [DOI] [PubMed] [Google Scholar]
- 14.Dooley J, Worwood M. Guidelines on diagnosis and therapy: genetic haemochromatosis. British Committee for Standards in Haematology. 2000. Abingdon, Oxford, Darwin Medical Communications Ltd ( http://www.bcshguidelines.com/ )
- 15.Bhavnani M, Lloyd D, Bhattacharyya A.et al Screening for genetic haemochromatosis in blood samples with raised alanine aminotransferase. Gut 200046707–710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jackson H A, Bowen D J, Worwood M. Rapid genetic screening for haemochromatosis using heteroduplex technology. Br J Haematol 199798856–859. [DOI] [PubMed] [Google Scholar]
- 17.Worwood M, Jackson H A, Feeney G P.et al A single tube heteroduplex PCR for the common HFE genotypes. Blood 199994405a [Google Scholar]
- 18.Adams P C, Deugnier Y, Moirand R.et al The relationship between iron overload, clinical symptoms, and age in 410 patients with genetic hemochromatosis. Hepatology 199725162–166. [DOI] [PubMed] [Google Scholar]
- 19.Edwards C Q, Kushner J P. Screening for hemochromatosis. N Engl J Med 19933281616–1620. [DOI] [PubMed] [Google Scholar]
- 20.Distante S. Phenotypic expression of the HFE gene mutation (C282Y) among the hospitalised population. Gut 200047575–579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Looker A C, Johnson C L. Prevalence of elevated serum transferrin saturation in adults in the United States. Ann Intern Med 1998129940–945. [DOI] [PubMed] [Google Scholar]
- 22.Jackson H A, Carter K, Darke C.et al HFE mutations, iron deficiency and overload in 10 500 blood donors. Br J Haematol 2001114474–484. [DOI] [PubMed] [Google Scholar]