Abstract
Aims
To study platelet volume indices (PVI) in the spectrum of ischaemic heart diseases.
Methods
A total of 210 cases were studied; 94 patients had unstable angina (UA) or acute myocardial infarction (AMI) diagnosed on the basis of history, characteristic electrocardiographic changes, and increased cardiac enzyme activities. Seventy patients had stable coronary artery disease (stable CAD) or were admitted for a coronary angiography or coronary artery bypass graft procedure. The third group comprised 30 age and sex matched healthy controls with no history of heart disease and a normal electrocardiogram.
Results
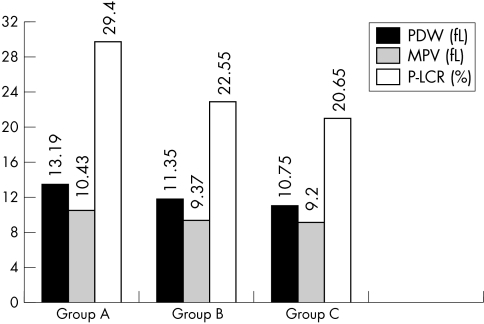
All PVI—mean platelet volume (MPV), platelet distribution width (PDW), and platelet large cell ratio (P‐LCR)—were significantly raised in patients with AMI and UA (mean MPV, 10.43 (SD, 1.03) fL; mean PDW, 13.19 (SD, 2.34) fL; mean P‐LCR, 29.4% (SD, 7.38%)) compared with those with stable CAD (mean MPV, 9.37 (SD, 0.99) fL; mean PDW, 11.35 (SD, 1.95) fL; mean P‐LCR, 22.55% (SD, 6.65%)) and the control group (mean MPV, 9.2 (SD, 0.91) fL; mean PDW, 10.75 (SD, l.42) fL; mean P‐LCR, 20.65% (SD, 6.14%)).
Conclusions
Larger platelets are haemostatically more active and are a risk factor for developing coronary thrombosis, leading to myocardial infarction. Patients with larger platelets can easily be identified during routine haematological analysis and could possibly benefit from preventive treatment. Thus, PVI are an important, simple, effortless, and cost effective tool that should be used and explored extensively, especially in countries such as India, for predicting the possibility of impending acute events.
Keywords: acute myocardial infarction, coronary artery disease, mean platelet volume, platelet distribution width, platelet volume indices
In the new millennium, coronary artery disease (CAD) is looming large as the new epidemic afflicting Indians. Both endogenous and exogenous risk factors such as smoking, diabetes mellitus, hypertension, hypercholesterolaemia, mental stress, and obesity—acting either singly or in combination—significantly increase the chances of developing coronary atherosclerosis. Platelets have been implicated in the pathogenesis of cardiovascular disorders, including atherosclerosis and its complications, such as acute myocardial infarction (AMI), unstable angina (UA), and sudden cardiac death. Platelet hyper‐reactivity and local platelet activation have been suggested to play a causal role in acute coronary events.1 Platelet size has been shown to reflect platelet activity. Large platelets are metabolically and enzymatically more active than small platelets and produce more thromboxane A2.2,3
“Our aim was to study platelet parameters in the spectrum of ischaemic artery disease and to attempt a clinicopathological correlation”
Automated cell counters have made the platelet count (PC) and the platelet volume indices (PVI)—mean platelet volume (MPV), platelet distribution width (PDW), and platelet large cell ratio (P‐LCR)—routinely available in most clinical laboratories. However, there is scope to make better use of the platelet parameters generated. The MPV can reflect changes in either the level of platelet stimulation or the rate of platelet production. Platelet activation is indirectly measured via MPV. Thus, our aim was to study platelet parameters in the spectrum of ischaemic artery disease and to attempt a clinicopathological correlation.
Materials and methods
A prospective hospital based study was carried out on 210 patients over a period of one year. Institutional ethical committee clearance was obtained. Informed consent was given.
Three groups were studied. Group A: patients admitted to the intensive care unit with UA and/or AMI (94 patients); group B: patients with stable CAD admitted for coronary angiography or a coronary artery bypass graft procedure for a previous ischaemic event (70 patients); and group C: age and sex matched normal healthy controls with a normal electrocardiogram (30 patients).
Blood (2 ml) was collected in dipotassium EDTA tubes from all the patients on day 1 and also on day 7 for patients with acute coronary events by a clean puncture, avoiding bubbles and froth. The sample was run within two hours of venepuncture using the Sysmex K‐4500 automated cell counter (TOA Electronics, Koebe, Japan)
Quality control
Internal and external quality controls were strictly followed.
Statistical analysis
Statistical analysis of the data was carried out using the statistical package for social sciences. The Student's t test and the one way analysis of variance test were used for comparing the group means. The χ2 test was used and p values of < 0.05 were taken as significant.
Results
Of the 210 patients enrolled in our study, all 18 parameters were generated by the Sysmex analyser in 194 and were statistically analysed. Five samples were clotted and were excluded from the study. In the remaining 11 (nine in group A and two in group B) samples, only the PC value was obtained and PVI were flagged, indicating an abnormality sensed by the machine and the need for manual evaluation of a peripheral blood smear by an expert morphologist. Many giant sized platelets (more than 5 μm in diameter as judged on the blood smear) were noted in these patients.
The mean (SD) age of the patients was 53.9 (11.3) years with an equal sex predilection (male to female ratio, 1 : 1). This is in accordance with the Asian population at risk for an ischaemic heart disease, occurring one decade earlier than in developed countries,4 and equally prevalent among premenopausal women. In our present study, risk factors were evaluated and were evenly distributed. However, significantly more numbers of patients who were obese and had hypercholesterolaemia were present in groups A and B than in group C (table 1).
Table 1 Distribution of risk factors in all the patients.
| Groups | Group A (n = 94) | Group B (n = 70) | Group C (n = 30) | χ2 | p Value |
|---|---|---|---|---|---|
| Hypertension | 23 (24.4) | 20 (28.5) | 7 (23.3) | 0.46 | NS |
| Diabetes mellitus | 14 (14.8) | 10 (14.2) | 2 (6.6) | 1.4 | NS |
| Current smokers | 18 (19.1) | 15 (21.4) | 5 (16.6) | 0.32 | NS |
| Hypercholesterolaemia | 20 (21.2) | 17 (24.2) | 0 (0) | 8.6 | <0.05 |
| Hypertriglyceridaemia | 14 (14.8) | 7 (10) | 0 (0) | 1.47 | NS |
| BMI (obese) | 11 (11. 7) | 13 (18.5) | 0 (0) | 6.7 | <0.05 |
| Manual activity | 13 (13.8) | 10 (14.2) | 4 (13.3) | 0.02 | NS |
| Aspirin | 17 (18) | 20 (28.5) | 0 (0) | 1.96 | NS |
| Clopidogrel | 8 (8.5) | 14 (20) | 0 (0) | 3.62 | NS |
Figures in parenthesis indicate percentages.
BMI, body mass index; NS, not significant.
The white blood cell count was significantly raised in the AMI group compared with the CAD and control groups (table 2).
Table 2 Distribution of haematological (Sysmex) parameters in all the cases.
| Group A | Group B | Group C | F value | p Value | |
|---|---|---|---|---|---|
| Haemoglobin (g/l) | 121.7 (2.68) | 122.1 (2.59) | 126.6 (1.68) | 0.45 | NS |
| Haematocrit | 353.8 (7.38) | 354.8 (7.42) | 372.5 (4.09) | 0.87 | NS |
| WBC (×109/l) | 10.4 (4.37) | 9.52 (5.80) | 7.42 (1.49) | 4.68 | <0.05 |
| PC (×109/l) | 232.84 (88.8) | 240.79 (79.0) | 270.77 (75.2) | 3.39 | <0.05 |
| PDW (fL) | 13.19 (2.34) | 11.35 (1.95) | 10.75 (1.42) | 5.32 | <0.001 |
| MPV (fL) | 10.43 (1.03) | 9.37 (0.99) | 9.20 (0.91) | 6.63 | <0.001 |
| P‐LCR (%) | 29.4 (7.38) | 22.55 (6.65) | 20.65 (6.14) | 6.09 | <0.001 |
Values are mean (SD).
MPV, mean platelet volume; PC, platelet count; PDW, platelet distribution width; P‐LCR, platelet large cell ratio; WBC, white blood cell count.
No significant difference in PC was seen between groups A and B (t value, −0.59; p > 0.05) or between groups B and C (t value, −1.76; p > 0.05), although the lowest values were seen in group A, followed by group B, and group C. However, a significant difference was seen when the mean PC was compared between the AMI and control groups (t value, 2.10; p < 0.05).
PVI—PDW, MPV, and P‐LCR—were significantly raised in group A compared with the stable CAD and control groups (fig 1).
Figure 1 Platelet volume indices in all patients (groups A, B, and C). MPV, mean platelet volume; PDW, platelet distribution width; P‐LCR, platelet large cell ratio.
PVI could be measured on day 7 in only 78 of the 94 patients in group A because six patients died before day 7 and nine patients were discharged. The PC values on day 1 (mean, 236.7 (SD, 93.7) and day 7 (mean, 237.2 (SD, 93.2) in group A patients were not significantly different. Similarly, PVI values on day 1 and day 7 were not significantly different. Of the 94 patients, six succumbed to the disease within a week of admission. Mortality among female patients was higher: five women died whereas only one man died. There were no significant differences in the PC and PVI values between the six patients who died and the survivors in group A.
The PC and PVI were independently correlated with: (1) type of infarct/ischaemia on electrocardiogram—that is, subendocardial type (70 patients) versus transmural type (24 patients); (2) the site of the infarct (anterior wall, lateral wall, posterior wall, others, and unknown); (3) the severity of the infarct as subtyped using Killips classification—types I–IV; (4) the time before starting treatment (within 24 hours, one to three days, four to seven days); (5) the number of ischaemic episodes; and (6) the number of coronaries blocked. No correlations were seen between these parameters. There were no significant differences in PVI and PC values between patients with UA and AMI (table 3).
Table 3 Comparison of PVI values in patients with AMI and UA in group A.
| Type | Total no. | PC (×109/l) | PDW (fL) | MPV (fL) | P‐LCR (%) |
|---|---|---|---|---|---|
| AMI | 77 | 235.3 (92.6) | 13.l (2.1) | 10.4 (1.0) | 29.30 (7.4) |
| UA | 17 | 221.4 (70.4) | 13.7 (3.1) | 10.5 (1.0) | 29.8 (7.2) |
| t Value | 0.58 | 1.06 | 0.24 | 0.27 | |
| p Value | NS | NS | NS | NS |
AMI, acute myocardial infarction; MPV, mean platelet volume; NS, not significant; PC, platelet count; PDW, platelet distribution width; P‐LCR, platelet large cell ratio; PVI, platelet volume indices; UA, unstable angina.
Risk factors such as diabetes mellitus, hypertension, body mass index, physical activity, smoking, hypercholesterolaemia, and hypertriglyceridaemia were not a confounding factor in the determination of PVI and PC in acute coronary events, and neither were the readings affected by age or sex. No correlation was seen between the PVI and haemoglobin value, haematocrit value, or total leucocyte count. No significant effect was noted of antiplatelet drugs, aspirin, or clopidogrel on the PC and PVI results.
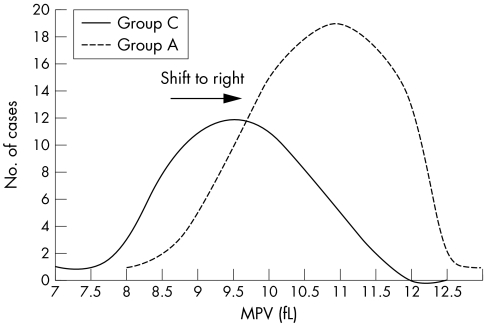
The log normality of the platelet distribution curve was preserved in acute coronary events and showed a shift of the whole curve to the right compared with the controls (fig 2). Univariate analysis of the MPV quartiles indicated a value of more than 9.6 fL to be a significant risk factor for developing a myocardial infarction (MI).
Figure 2 Normal mean platelet volume curve in patients in groups A and C. MPV, mean platelet volume.
Discussion
We found that PVI (MPV, PDW, and P‐LCR) are raised in patients who have suffered an acute coronary event compared with controls and those with stable CAD. This is in agreement with the results of similar studies by other workers (table 4).
Table 4 Comparison of MPV in AMI and controls in different studies.
| Publication | AMI | Controls | |||
|---|---|---|---|---|---|
| N | MPV (fL) | N | MPV (fL) | p Value | |
| O'Brien et al (1973)5 | 23 | 8.10 | 36 | 7.01 | <0.001 |
| Cameron et al (1983)6 | 100 | 9.07 (SE, 0.08) | 200 | 8.32 (SE, 0.07) | <0.001 |
| Martin et al (1983)7 | 15 | 7.3 | 22 | 6.32 | 0.05 |
| Martin et al (1991)8 | 126 | 10.09 (1.43) | 1590 | 9.72 (1.12) | <0.001 |
| Smyth et al (1993)9 | 24 | 8.54 (0.6) | 23 | 8.1 (0.69) | 0.04 |
| Pizulli et al (1998)10 | 108 | 9.4 (1.23) | 97 | 8.2 (0.95) | <0.001 |
| Present study | 94 | 10.43 (1.03) | 30 | 9.20 (0.91) | <0.001 |
Values are mean (SD) unless otherwise stated.
AMI, acute myocardial infarction; MPV, mean platelet volume.
Generalised platelet activation occurs before an acute coronary event. The increase in platelet consumption at the site of the coronary atherosclerotic plaque causes larger platelets to be released from the bone marrow. The fact that the increase persists even after discharge from hospital supports the view that platelet volume is chronically larger in the infarct group.6,8 This suggests that PVI, particularly MPV, are indicators of the degree of damage already done and that these markers maintain their strength and predictive value for a long time. However, some workers found no correlation between the time span between MI and measurement of MPV.10 The same group also suggested that MPV does not change during the acute phase reaction, but is determined by other factors. There are studies indicating that platelet size is determined at the level of the progenitor cell. Megakaryocyte ploidy is influenced by interleukin 3 and interleukin 6, which leads to the production of larger platelets that are more reactive.11,12,13 Using logistic regression analysis of MPV quartiles, we found that a value of > 9.6 fL was a significant risk for developing MI in patients with CAD, whereas a figure of > 11.6 fL was determined by Endler et al.14
“Our data suggest that the increased mean platelet volume contributes to the prethrombotic state in acute ischaemic syndromes and that larger platelets may play a specific role in infarction”
The preservation of log normality of the platelet volume, consistent with the study by Martin et al,7 indicates that the increase in volume is not the result of consumption or production of a platelet subpopulation, because the whole distribution curve was shifted to the right.
Take home messages
Platelet volume indices (PVI) are a useful means of identifying larger platelets, which are haemostatically more active and are a risk factor for developing coronary thrombosis, leading to myocardial infarction
Patients with larger platelets can easily be identified during routine haematological analysis and could possibly benefit from preventive treatment
Therefore, PVI are an important, simple, effortless, and cost effective tool that should be used more extensively to predict impending acute events
We found no association between the type and site of infarct and MPV, as has been reported by others.6,8 PC and PVI were not associated with mortality, morbidity, or the severity of MI in our present study, whereas Martin et al found that MPV was significantly higher in those patients who died of MI, compared with survivors.8 Some studies have found higher MPV values in patients with UA than those with MI.15 However, we found no such difference between patients with UA and AMI, as also reported by Senaran et al.16
The role of PDW specifically in patients with CAD and acute coronary events is yet to be explored. Similarly, the P‐LCR parameter is generated by only a few machines, with the Sysmex analyser being one of them. It is not often quoted in the literature, probably because it is a relatively new PVI parameter.
Our data suggest that the increased MPV contributes to the prethrombotic state in acute ischaemic syndromes and that larger platelets may play a specific role in infarction. Because larger platelets are haemostatically more active, the presence of larger platelets is probably a risk factor for developing coronary thrombosis and MI. Patients with larger platelets can easily be identified during routine haematological analysis because PVI are generated as a byproduct of automated blood counts. Thus, in conclusion, PVI provide an important, simple, effortless, and cost effective tool, which can be useful in predicting an impending acute coronary event.
Abbreviations
AMI - acute myocardial infarction
CAD - coronary artery disease
MI - myocardial infarction
MPV - mean platelet volume
PC - platelet count
PDW - platelet distribution width
P‐LCR - platelet large cell ratio
PVI - platelet volume indices
UA - unstable angina
References
- 1.Trip M D, Cats V K, van Capelle F J L.et al Platelet hyperreactivity and prognosis in survivors of myocardial infarction. New Engl J Med 19903221549–1554. [DOI] [PubMed] [Google Scholar]
- 2.Corash L, Tan H, Grolnick H R. Heterogeneity of human whole blood platelet subpopulations. I. Relationship between buoyant density, cell volume and ultrastructure. Blood 19774971–87. [PubMed] [Google Scholar]
- 3.Thompson C B, Eaton K A, Princiotta S M.et al Size‐dependent platelet subpopulation: relationship of platelet volume to ultrastructure, enzymatic activity and function. Br J Haematol 198250509–520. [DOI] [PubMed] [Google Scholar]
- 4.Grover A, Bahl A, Vijayvergiya R.et al Changing trends of cardiovascular disease in India: implications for prevention based on 2001 census. Indian J Cardiol 2003659–63. [Google Scholar]
- 5.O'Brien J R, Etherington M, Jamieron S.et al Stressed template bleeding‐time and other platelet function tests in myocardial infarction. Lancet 197311778694–696. [DOI] [PubMed] [Google Scholar]
- 6.Cameron E A, Phillips R, Ibbatson R M.et al Platelet size in myocardial infarction. BMJ 1983287449–451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Martin J F, Plumb J, Kilbey R S.et al Changes in volume and density of platelets in myocardial infarction. BMJ 1983287456–459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Martin J E, Bath P M W, Burr M L. Influence of platelet size on outcome after myocardial infarction. Lancet 19913381403–1411. [DOI] [PubMed] [Google Scholar]
- 9.Smyth D W, Martin J F, Michalis L.et al Influence of platelet size before coronary angioplasty on subsequent restenosis. Eur J Clin Invest 199323361–367. [DOI] [PubMed] [Google Scholar]
- 10.Pizzulli L, Yang A, Martin J F.et al Changes in platelet size and count in UA compared to stable angina or non‐cardiac chest pain. Eur Heart J 19981980–84. [DOI] [PubMed] [Google Scholar]
- 11.Tschoepe D, Esser J, Schwippert B.et al Large platelets circulate in an activated state in diabetes mellitus. Semin Thromb Hemost 199117433–439. [DOI] [PubMed] [Google Scholar]
- 12.Burstein S A, Downs T, Friese P.et al Thrombocytopoiesis in normal and sublethally irradiated dogs: response to human interleukin‐6. Blood 199280420–428. [PubMed] [Google Scholar]
- 13.Debili N, Masse J M, Katz A.et al Effects of recombinant hematopoietic growth factors interleukin‐3, interleukin‐6, stem cell factor and leukemia inhibitory factor on the megakaryocytic differentiation of CD34+ cells. Blood 19938284–95. [PubMed] [Google Scholar]
- 14.Endler G, Klimesch A, Plassmann H S.et al Mean platelet volume is an independent risk factor for myocardial infarction but not for coronary artery disease. Br J Haematol 2002117399–404. [DOI] [PubMed] [Google Scholar]
- 15.Mathur A, Rokinson M S, Cotton J.et al Platelet reactivity in acute coronary syndromes: evidence for differences in platelet behaviour between unstable angina and myocardial infarction. Thromb Haemost 200185989–994. [PubMed] [Google Scholar]
- 16.Senaran H, Heri M, Altinbas A.et al Thrombopoietin and mean platelet volume in coronary artery disease. Clin Cardiol 200124405–408. [DOI] [PMC free article] [PubMed] [Google Scholar]