Abstract
Sickle cell trait (SCT) has a UK urban population rate estimated at 3.2%. The condition may remain unrecognised, but sudden death is a recognised feature. The sudden death of a 51‐year‐old man with SCT who presented with hyperosmolar non‐ketotic acidosis (HONK) as his first presentation of diabetes is reported here. After admission to hospital, a rapid deterioration was seen in his condition, with loss of cardiac output, leading to death. He was found to have had acute‐chest syndrome and hepatosplenic infarction on postmortem examination. The pathophysiology of sudden death in patients with SCT and the role of HONK in the patient's demise is discussed here.
The number of people in the UK with homozygous sickle cell disease (SCD) was expected to exceed 10 000 by 2000.1 In urban areas, the heterozygous rate of sickle cell trait (SCT) has been estimated as 3.2%.2 In many of these patients the trait may remain unrecognised, owing to the fact that carriers are invariably asymptomatic.
Sudden death in SCD has been recognised for decades, with deaths attributable predominantly to acute chest crises, splenic sequestration and bacterial infection. Increasing numbers of reports have highlighted the potential for patients with SCT to die suddenly and unexpectedly. A review of the risk of sudden death in SCT has shown a 28–40‐fold increased risk in the incidence of sudden unexplained death during exertion among soldiers with SCT, when compared with their haemoglobin AA counterparts.3 Variations in the reported circumstances of sudden unexpected deaths in SCT include death after police pursuit,4 death during pregnancy5 and death in athletes with SCT.6 The potential combination of deoxygenation, dehydration, acidosis and increased temperature is a common feature of the above situations. The above factors are also stresses that are present in hyperosmolar non‐ketotic acidosis (HONK).
Case history
The patient was a 51‐year‐old Afro‐Caribbean man who had been diagnosed with SCT several years previously. He had a medical history of high blood pressure and high cholesterol. In the days before he was admitted to hospital, he had felt unwell and was thirsty and drinking excessive fluids. On the day of admission, he collapsed at home and in casualty was found to have a blood glucose concentration of 54 mmol/l, and glycosuria but no ketonuria. He was newly diagnosed as having diabetes with HONK. The pH of his blood on admission was 7.2, with a base excess of −10.6 mmol/l. Other tests showed a mildly raised white cell count and normal clotting. He had renal failure (Na 160 mmol/l, K 3.1 mmol/l, urea 22 mmol/l, creatinine 331 μmol/l). His creatinine kinase value was 383 U/l, which rose to >11 000 U/l perimortem. He was also noted to have an amylase value of 1181 U/l. Over the next several hours, his blood glucose and acid–base balance responded to standard medical management, but he continued to have right upper quadrant abdominal pain. No acute cause was found and he was managed conservatively. The following morning he was reviewed and found to be hypotensive, with a falling urine output. Soon after this, while being reviewed, he had a sudden cardiac arrest from which he could not be resuscitated.
Postmortem findings
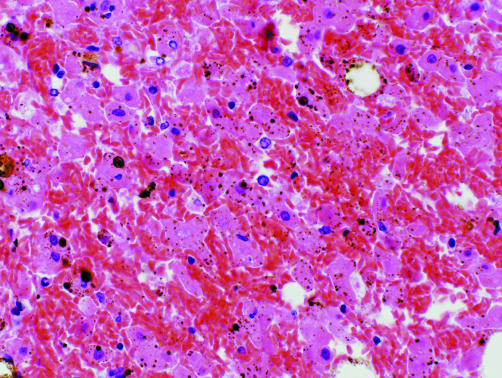
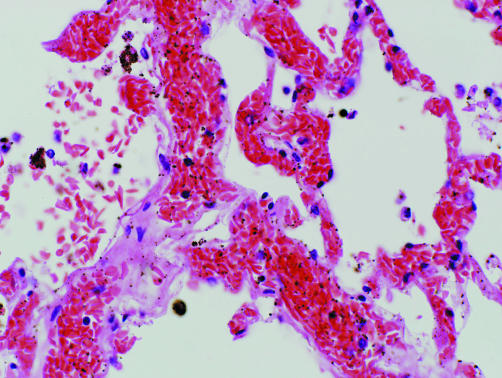
A postmortem examination showed that the lungs were heavy and oedematous, the right lung weighing 805 g and the left 745 g. The heart was normal and the coronary arteries were patent. Congestion and sickling was seen in the small and medium‐sized vessels of the lungs (fig 1). The spleen showed focal infarction and sickling. The liver showed zone 3 haemorrhage and infarction associated with sickled red blood cells in the sinusoids (fig 2). Samples of quadriceps muscle showed rhabdomyolysis, with no myositis or arteritis. The pancreas showed mild focal pancreatitis with duct obstruction by inspissated secretions. The histology of the remaining organs was unremarkable.
Figure 1 Liver at autopsy. Sickled red blood cells are seen in sinusoids and necrotic hepatocytes. High power, stained with haematoxylin and eosin.
Figure 2 Lung at autopsy. Pulmonary venule and alveolar capillaries are distended with sickled red blood cells. Medium power, stained with haematoxylin and eosin.
Discussion
This case adds a novel scenario to the literature on sudden death in SCT. A recently published case reported a patient with SCT, who presented with non‐ketotic diabetes.7 The condition was controlled, and the patient was discharged normoglycaemic. He died 4 days later from “sudden idiopathic death”. Unfortunately, no autopsy data were presented. Hence, it is unclear if a pathogenic association was present between the SCT, the diabetes and the ultimate death of the patient. At the core of the present case are the fundamental pathophysiological mechanisms that lead to death and link reports of sudden death in SCT.
When deoxygenated, haemoglobin S undergoes polymerisation into needle‐like fibres in the red blood cells (RBCs), resulting in distortion. This leads to increased transit times through splenic sinusoids and peripheral vascular beds and increases the time spent by RBCs in a relatively deoxygenated environment. In hyperosmolar states associated with diabetes, RBCs have impaired blood flow properties,8 which would compound the problems for already sickled RBCs, as in this case.
Take‐home messages
Although usually asymptomatic, sickle cell trait is associated with sudden death under certain physiological extremes.
Dehydration, hypoxia and acidosis, alone or in combination, are frequently implicated in such deaths.
Undiagnosed diabetes is still responsible for a small number of fatalities a year.
Sickle cell trait may be a significant contributory factor in deaths that appear, at first sight, to have an alternative cause.
In patients with SCD and SCT, the nature of the stresses is similar—that is, dehydration and deoxygenation, but the magnitude of the insult needs to be greatly increased in patients with SCT. The factors that determine the critical point at which haemoglobin S RBCs will sickle are poorly understood and many variables play a part in it, including the presence of synchronous haemoglobinopathies, levels of fetal haemoglobin, which has a protective role,9 and the mean corpuscular haemoglobin concentration. Dehydration is a key feature in many of the reported cases of sudden death in SCT, as is hypoxia, and both were stresses in this case. One reason for dehydration is age‐related hyposthenuria, which is attributable to silent papillary necrosis.10 This lesion was not identified in our case. Despite this, the presence of rhabdomyolysis would have had a negative effect on renal function. The sudden nature of the final cardiac arrest was due to the acute chest syndrome, a well‐recognised fatal event in SCD.
In conclusion, given the frequency of both SCT and diabetes in the Afro‐Caribbean population, it is important for clinicians treating these patients to appreciate that the conditions may often coexist and patients with SCT may have a precipitous clinical decline. Pathologists investigating sudden death in SCT, and considering possible precipitating factors, should bear in mind the potential of undiagnosed diabetes.
Abbreviations
HONK - hyperosmolar non‐ketotic acidosis
RBC - red blood cell
SCD - sickle cell disease
SCT - sickle cell trait
Footnotes
Competing interests: None declared.
References
- 1.Davies S, Cronin E, Gill M.et al Executive summary. Screening for sickle cell disease and thalassaemia: a systematic review with supplementary research. Health Technol Assess 20004 [PubMed] [Google Scholar]
- 2.Brozovic M, Anionwu E. Sickle cell disease in Britain. J Clin Pathol 1984371321–1326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kark J A, Posey D M, Schumacher H R.et al Sickle‐cell trait as a risk factor for sudden death in physical training. N Engl J Med 1987317781–787. [DOI] [PubMed] [Google Scholar]
- 4.Thogmartin J R. Sudden death in police pursuit. J Forensic Sci 1998431228–1231. [PubMed] [Google Scholar]
- 5.Pastorek J G, Seiler B. Maternal death associated with sickle cell trait. Am J Obstet Gynaecol 1985151295–297. [DOI] [PubMed] [Google Scholar]
- 6.Holmes P S, Kerle K K, Seto C K. Sickle cell trait and sudden death in athletes. Am Fam Physician 1998572763–2770. [PubMed] [Google Scholar]
- 7.Schutt M, Meier M. Sudden death in sickle cell trait: could coexistent diabetes play a role? Med Hypotheses 200564217. [DOI] [PubMed] [Google Scholar]
- 8.Evan‐Wong L A, Davidson R J, Stowers J M. Alterations in erythrocytes in hyperosmolar diabetic decompensation: a pathophysiological basis for impaired blood flow and for an improved design of fluid therapy. Diabetologia 198528739–742. [DOI] [PubMed] [Google Scholar]
- 9.Harkness D R. Sickle cell trait revisited. Am J Med 19898730–34N. [PubMed] [Google Scholar]
- 10.Keitel H G, Thompson D, Itano H A. Hyposthenuria in sickle cell anaemia; a reversible renal defect. J Clin Invest 195635998–1007. [DOI] [PMC free article] [PubMed] [Google Scholar]