Abstract
Aims
To estimate the expression of substance P in the haematopoietic cells of hypoplastic bone marrow and define its relationship with the course of bone marrow hypoplasia.
Methods
Bone marrow specimens were obtained from 42 children with bone marrow hypoplasia who were hospitalised in the Department of Pediatric Oncology, Hematology and Transplantology, Poznan University of Medical Sciences, Poznan, Poland, between 1996 and 2003. Substance P and Ki‐67 expression were evaluated using immunochemical and hybridocytochemical assays.
Results
The expression of substance P (as evidenced by both immunocytochemical and hybridisation techniques) was confirmed in the cytoplasm of B lymphocytes in 8 of 11 children who developed acute leukaemia in 45 (SD 12) days. The percentage of substance P‐positive cells ranged from 67.6 to 95.8 (mean of 81.5% cells with immunocytochemistry and 84.3% with in situ hybridisation). The risk of development of leukaemia secondary to bone marrow hypoplasia was found to be significant (p<0.001) in those children who expressed substance P in normal‐looking lymphocytes at the initial bone marrow evaluation.
Conclusions
The presence of substance P in B lymphocytes of hypoplastic bone marrow may predict its neoplastic transformation. A marked correlation between substance P‐positive bone marrow pattern and the expansion of tumour cells may prove the potential value of this oligopeptide in the pathogenesis of leukaemia.
Bone marrow hypoplasia seems to be a morphological equivalent of clinically diagnosed haematopoietic insufficiency.1 It is defined by the following laboratory values: haemoglobin <8.5 g/dl, mean corpuscular volume <88 fl, a white cell count <2 g/l with a neutrophil count <1 g/l, a platelet count <50 g/l and reticulocytes <0.1%.1,2 Bone marrow aspirate contains hypocellularity. Transient bone marrow hypoplasia is usually caused by infections (both viral and bacterial) or by different chemical (including iatrogenic) or physical factors.3,4 Inherited aplastic anaemia, or bone marrow hypoplasia before non‐haematopoietic tissue in bone marrow (ie, neuroblastoma metastases in bone marrow), is a much rarer phenomenon.
A considerable number of cases of bone marrow hypoplasia may be subjected to spontaneous remission and may not even be recognised. The first hospitalisation of a child with bone marrow hypoplasia is usually after two or more ineffective courses of antibiotic treatment (for some undiagnosed chronic infection), followed by symptoms of anaemia and thrombocytopenia. In such cases, bone marrow hypoplasia can be diagnosed as myelodysplastic syndrome, severe aplastic anaemia or neoplasia. The correct diagnosis usually takes several weeks, during which time the affected children are not treated but usually receive continuous monitoring, with laboratory evaluation of peripheral blood, bone marrow, chest radiographs and so on being carried out at least once a month.
The acute leukaemia of both lymphoblastic and non‐lymphoblastic types, which develops secondary to bone marrow hypoplasia, usually has a bad prognosis.1 This may, at least in part, result from the late introduction of a chemotherapy regimen. In cases of neoplastic transformation, the type of chemotherapy used will depend on a precise recognition of the neoplasm concerned. Such treatment, however, can probably be introduced much faster if, in the mean time, the expression of certain markers of early neoplastic transformation can be detected sooner. The difficulty at present is that the full list of possible markers is not universally accepted. On the other hand, some authors5,6,7 emphasised that several of the cytokines which regulate physiological haematopoiesis may themselves be possible risk factors of neoplastic transformation in bone marrow hypoplasia.
One of these cytokines is substance P, a neuromediator and oligopeptide consisting of 11 amino acids (H‐Arg‐Pro‐Lys‐Pro‐Gln‐Gln‐Phe‐Phe‐Gly‐Leu‐Met‐NH2), with a C‐terminal fragment typical of tachykinins.7,8 It is widely distributed in the central and peripheral nervous systems, being responsible for contraction of skeletal and smooth muscle, salivation, micturition, vasodilatation and sensory perception. Substance P and neurokinin A are products of the common preprotachykinin I gene and are also capable of modulating the functions of the immune and haematopoietic systems.8
Substance P stimulates proliferative activity of cells with surface receptors for substance P. It is generally accepted that substance P shows a preference for neurokinin‐1 receptor (NK‐1R) and has less affinity for NK‐2R and NK‐3R.9,10 The expression of NK‐1R has been reported in T and B lymphocytes, macrophages, haematopoietic progenitor (CD34) cells, endothelial cells and CD10 lymphoblasts. It is of particular significance that neoplastic lymphoblasts in acute lymphoblastic leukaemia (ALL) in children carry a threefold to fourfold excess of NK‐1R as compared with normal B and T lymphocytes.11
According to some authors,12,13 peptidergic nerve terminals in closest proximity to immunocompetent cells in the organs most exposed to exogenous antigens represent a morphological exponent of functional inter‐relations between the nervous and immune systems.
However, substance P present in the bone marrow can be also of non‐neural origin. It is synthesised in the macrophages, eosinophils and neoplastically transformed lymphoblasts. It has been shown that substance P induces interleukin 1 (IL1) and stem cell factor in bone marrow stromal cells, with levels of IL1b (a major isoform of this cytokine) being twice those of IL1a. Both cytokines also perform a potent autoregulatory role in NK‐1R expression in bone marrow stromal cells.7
Therefore, our study aimed to estimate the expression of substance P in haematopoietic cells of hypoplastic bone marrow and define its relationship with the future course of bone marrow hypoplasia.
Patients and methods
Patients
Patients were children with bone marrow hypoplasia, hospitalised in the Department of Pediatric Oncology, Hematology and Transplantology (Poznan University of Medical Sciences, Poznan, Poland) between 1 January 1996 and 31 December 2003. In all, 485 children were referred to our department during that time. Bone marrow biopsies were performed on all of them and evidence of bone marrow hypoplasia was found in 42. Twenty children, all of whom presented with only one enlarged lymph node, served as the control group. Histopathological examination of the enlarged node indicated an inflammatory response only. Subsequent observation of these children in the outpatient clinic for hyperplastic diseases for around 12 months detected no clinical traits of a neoplastic disease. This research protocol was approved by the ethical commission of the university.
To participate in the investigation, children had to meet the following eligibility criteria: age 4–18 years, laboratory values: haemoglobin <8.5 g/dl, mean corpuscular volume <88 fl, white cell count <2 g/l with a neutrophil count <1 g/l, platelet count <50 g/l and reticulocytes < 0.1%, hypocellularity of bone marrow aspirate, with no neoplastic cells evaluated by microscopy and flow cytometry test; normal chest radiograph; no enlarged lymph nodes, spleen or liver; and sterile blood cultures (obtained at least three times) at the time of the first hospitalisation. All the children underwent reverse transcriptase‐polymerase chain reaction evaluation for hepatitis C virus, Epstein–Barr virus and cytomegalovirus. Table 1 presents the relevant data.
Table 1 Study group characteristics: the evolution of bone marrow hypoplasia during the 12‐month observation period.
| Study group | 12‐month observation (n) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Subgroup | No of patients | Age (year) | RT‐PCR viral infection markers (n) | Spontaneous remission | Severe aplastic anaemia | Neoplastic transformation (diagnosis) | No evolution | ||
| HCV | EBV | CMV | |||||||
| I | 17 | 10 (6.2) | 1 | — | — | 17 | — | — | — |
| II | 10 | 8 (2.5) | — | 1 | 1 | — | 10 | — | — |
| III | 11 | 10 (1.8) | — | 6 | — | — | — | 8 (ALL), 3 (ANLL) | — |
| IV | 4 | 14 (2.7) | 2 | — | — | — | — | — | 4 |
Age was expressed as the mean (SD).
ALL, acute lymphoblastic leukaemia; ANLL, acute non‐lymphoblastic leukaemia; CMV, cytomegalovirus; EBV, Epstein–Barr virus; HCV, hepatitis C virus; RT‐PCR, reverse transcriptase‐polymerase chain reaction. –, absence of the trait in all patients from a given group.
Bone marrow smear preparation
The samples of bone marrow were taken from the posterior superior iliac spine. Bone marrow smears prepared on Super Frost were fixed in 96% ethanol (30 min at room temperature) within 24 h of sampling and kept at −80°C until immunophenotyping and in situ hybridisation (ISH) were carried out.
Antibodies and immunocytochemistry
To show the presence of substance P in bone marrow cells, an indirect immunocytochemical procedure was carried out using rabbit polyclonal antibodies against human substance P (Serotec, PEP A40, Oxford, UK) and the streptavidin–biotin complex method with the use of horseradish peroxidase modified by application of biotinylated tyramine (Dako Catalysed Signal Amplification System, Peroxidase, K 1500, Dako, Copenhagen, Denmark).14 Heat‐induced antigen demasking pretreatment was also carried out (Target retrieval solution, Dako S 1699).15 The endogenous activity of peroxidase was blocked by 10‐min pre‐incubation in 10% hydrogen peroxide. The smears were then incubated with anti‐substance P antibodies diluted 1:2000 for 12 h at 4°C. Incubation with a secondary antibody (biotinylated goat anti‐rabbit, Dako E 0432, diluted 1:300) was carried out at room temperature for 60 min followed by incubation with diaminobenzidine (Dako S 3000).
Ki‐67 antigen detection included an immunocytochemical procedure with mouse monoclonal antibodies directed against human Ki‐67 (Dako, M 7187). The smears were incubated with anti‐Ki‐67 antibodies, diluted 1:100 for 12 h at 4°C. Incubation with the secondary antibody (biotinylated goat anti‐mouse, Dako E 0433, diluted 1:300) was carried out at room temperature for 60 min.
After incubation with antibodies against human leucocyte antigen‐DR, CD3, CD10, CD13, CD14, CD15, CD20, CD33, CD34, CD41, CD61 and glycophorin A were used in white cell (including blasts) immunophenotyping.16
In situ hybridisation
For detection of mRNA encoding the amino acid sequence of human substance P, a 5′‐biotinylated probe of the nucleotide sequence 5′‐TCT GGG TTC GGA GTC GTC AAG AAA CCT AAT TAC 3′, was used.17,18,19 The probe was synthesised by the DNA‐Gdansk (Gdansk, Poland) and was complementary to the nucleotide sequence of human substance P (locus = 7q21–q22, gene = “TAC1”, product = “substance P”, bp = 68–100; http://www.ncbi.nlm.nih.gov/entrez/viewer.fcgi?val = M68907). The smears were incubated with the probe (concentration: 200 ng/1 ml) for 18 h at 37°C in a hybridisation chamber. This was followed by incubation with a streptavidin–biotin complex (Dako K 1500, diluted 1:100) at room temperature for 60 min and incubation with diaminobenzidine for 5 min.
Controls and microscopical analysis
Immunocytochemistry (ICC) and hybridocytochemistry complied with the principles of positive and negative controls.20 Both analyses were carried out blind on coded samples. In control incubations, primary antibodies or oligoprobes were replaced by rabbit or mouse serum (positive control) or phosphate‐buffered saline (negative control). The results of the immunocytochemical reactions and of the ISH were examined under a light microscope (Eclipse 600, Nikon), at magnifications of 1:200–1:400. On the basis of the results of the haematological staining, which were analysed through the use of Microimage (Olympus) morphometric software, the content of reaction‐positive cells was determined by comparing the number of cells with a positive reaction for substance P, Ki‐67 or mRNA substance P with the total number of blasts. A percentage of substance P‐positive cells <5% was deemed to reflect technical errors and was classified as a negative result.
Statistical analysis
As the group studied was relatively small, statistical analysis was based on McNemar analysis and Fisher's exact test. The first was used to verify the difference in percentage of substance P‐positive cells noted by the immunocytochemical and in ISH techniques. The relationship between substance P‐positive cases and an unfavourable course of bone marrow hypoplasia was determined using the Fisher's exact test. Significance was defined at p<0.05.
Results
12‐month monitoring
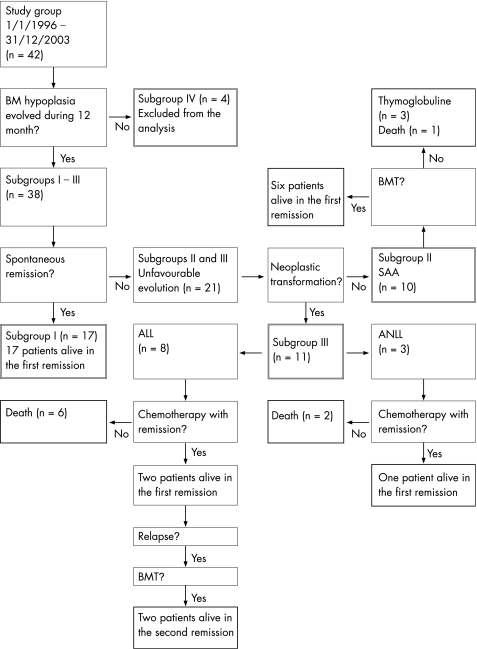
Every child from the study group underwent a 12‐month period of observation. During that time, laboratory values of peripheral blood were evaluated at least once a week; bone marrow, depending on the clinical course, was aspirated every 30–60 days; and chest radiographs and abdomen ultrasonography were carried out every 3 months. Reverse transcriptase‐polymerase chain reaction markers of viral infection (see Patients and methods) were estimated only once. Of the patients, 17 children underwent a spontaneous remission (subgroup I, median time of observation 93 (standard deviation (SD) 23) days) and 10 children developed severe aplastic anaemia (subgroup II, median time 212 (162) days). Eleven children were diagnosed with acute leukaemia (subgroup III): eight had ALL with a median time of 44 (12) days and three had acute non‐lymphoblastic leukaemia (ANLL) with a median time of 48 (13) days. In the remaining 4 children (subgroup IV), bone marrow hypoplasia did not develop during the 12‐month period of observation, and these children were excluded from further analysis (table 1).
Outcome
The 17 children who underwent spontaneous remission are still alive. The median time of observation is 37 (10.2) months. These patients are regularly evaluated for clinical traits of aplastic anaemia or neoplastic disease.
Of the 10 patients who developed severe aplastic anaemia (haemoglobin <7.0 g/dl, mean corpuscular volume <85 fl, white cell count <1.0 g/l with a neutrophil count <0.5 g/l, platelet count <20 g/l), 6 underwent allogenic bone marrow transplantation and they are alive at the first remission. Three children in this group were given thymoglobulin (they are also alive at the first remission). The one remaining child died 50 days after first hospitalisation due to a cytomegalovirus infection. The median time of observation of the children of subgroup II who survived is 32 (12.5) months.
The children of subgroup III had the worst outcome. Six of the eight patients with ALL died, with no clinical and haematological signs of remission (median time of observation after the development of ALL was 45 (12) days). In the remaining two children ALL remission was diagnosed, but they later relapsed (median time of observation 284 (68) days) and underwent allogenic bone marrow transplantation. They are still alive in the second remission, but the period of observation is not long (12 (4.5) months). Of the three patients, two with ANLL died, with no clinical and haematological signs of remission (median time of observation after ANLL was recognised as 34 (16) days). The remaining child with ANLL is in remission and is still alive (time of observation 24.5 months).
Overall, 9 of the 42 patients (21.4%) investigated in this study died between 1 January 1996 and 31 December 2003, 27 (64.3%) are alive at the first remission and 2 (4.8%) at the second remission. Four patients (9.5%) were excluded from the analysis. The relevant data are presented in fig 1.
Figure 1 Diagrammatic representation of sample outcome. ALL, acute lymphoblastic leukaemia; ANLL, acute non‐lymphoblastic leukaemia; BM, bone marrow; BMT, bone marrow transplantation; SAA, severe aplastic anaemia.
White cell immunophenotyping
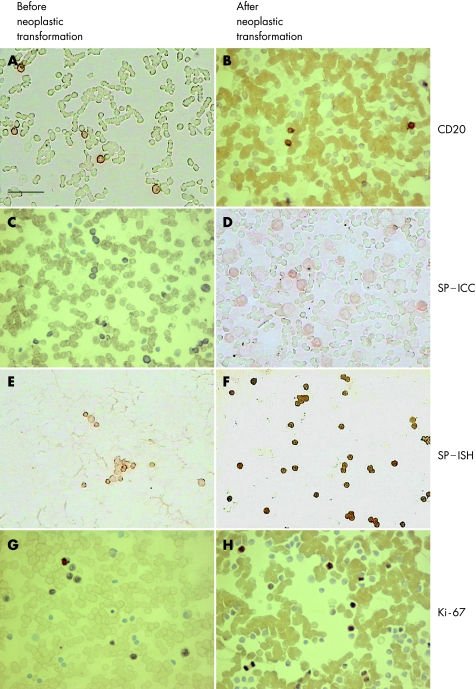
In all the children of the study group the population of nucleated cells in the hypoplastic bone marrow was mainly composed of lymphocytes (mean of 96.4%). The percentage of B lymphocytes (CD20) was >95% (mean of 95.1%), whereas T lymphocytes (CD3) constituted <4% of all lymphocytes. Other individual nucleated cells were first represented by neutrophils (fig 2A). After neoplastic transformation B lymphocytes constituted <5% of all nucleated cells (fig 2B), which consisted mostly CD10‐positive blasts in cases of ALL, or CD13/CD14‐positive cells in cases of ANLL (data not shown).
Figure 2 (A) Immunohistochemical expression of CD20 in the hypoplastic bone marrow of a 7‐year‐old child assigned to subgroup III. Individual cells present at the time of neoplastic transformation are mostly B lymphocytes. (B) A bone marrow specimen of a 7‐year‐old child who developed acute lymphoblastic leukaemia. Mature CD20‐positive B lymphocytes form a minority of all blasts. (C) Substance P (SP) immunoreactivity in normal‐looking B lymphocytes before neoplastic transformation of a 5‐year‐old child. Its expression is present in the cytoplasm of most nucleated cells. (D) The same 5‐year‐old child (as in C), who developed acute non‐lymphoblastic leukaemia. Weak substance P expression in all blasts. (E) A hypoplastic bone marrow of a 9‐year‐old child assigned to subgroup III. Expression of substance P mRNA in individual groups of nucleated cells. (F) Expression of SP mRNA in the same 9‐year‐old child (as in E) after neoplastic transformation (common acute lymphoblastic leukaemia). SP transcript immunoreactivity in all nucleated cells. (G) Expression of Ki‐67 antigen at the moment of neoplastic transformation. Strong nuclear immunocytochemical reaction in about half of the normal‐looking B‐lymphocytes (7‐year‐old child assigned to subgroup III). (H) Immunocytochemical expression of Ki‐67 in the bone marrow specimen of a 7‐year‐old child who developed acute leukaemia. Ki‐67‐positive cells constitute one third of all nucleated cells. Scale bar = 50 µm. ICC, immunocytochemistry; ISH, in situ hybridisation.
ICC and ISH
Before and at the time of neoplastic transformation the expression of substance P (in both the immunocytochemical and hybridisation techniques) was confirmed exclusively in the cytoplasm of B lymphocytes in 8 of the 11 children assigned to subgroup III (fig 2C,E). The number of substance P‐positive cells ranged from 67.6% to 95.8% (mean of 81.5% cells for ICC and 84.3% with ISH). This positive expression was also found in the subsequent bone marrow examination and the range of positive cells did not differ markedly from that in the initial findings. During the 12‐month observation period, seven substance P‐positive patients developed ALL and one developed ANLL. The mean percentages of substance P‐positive cells at this stage of the diseases were 82.7 (ICC, fig 2D) and 88.9 (ISH, fig 2F), respectively. No differences were observed among the densities from substance P staining in individual patients.
The expression of Ki‐67 antigen in the bone marrow haematopoietic cells was not evaluated at the first hospitalisation. However, it was detected at the time of (fig 2G), and after (fig 2H), neoplastic transformation, when the percentage of Ki‐67‐positive cells (blasts) ranged from 39.6% to 49.8% (mean of 42.6% blast cells) of all nucleated cells.
Substance P was absent from the cytoplasm and Ki‐67 from the nucleus of the normal haematopoietic cells of the 20 children in the control group (data not shown). Table 2 presents the information in detail.
Table 2 Immunocytochemical and hybridocytochemical expression of substance P and Ki‐67 antigen in the children of the study group during a 12‐month observation period (shown as the mean percentage of immunopositive cells).
| Subgroup | Initial BM evaluation | BM evaluation after the neoplastic transformation | ||||
|---|---|---|---|---|---|---|
| ICC | ISH | ICC | ISH | |||
| SP (%) | Ki‐67 (%) | SP (%) | Ki‐67 (%) | |||
| I | <5 | <5 | <5 | — | — | — |
| II | <5 | <5 | <5 | — | — | — |
| III | 81.5 | <5 | 84.3 | 82.7 | 42.6 | 88.9 |
| IV | <5 | <5 | <5 | — | — | — |
BM, bone marrow; ICC, immunocytochemistry; ISH, in situ hybridisation; SP, substance P. –, absence of the trait in all patients from a given group.
Statistical analysis
McNemar analysis showed no significant difference between the results obtained by the ICC and ISH techniques (p values ranged from 0.25 to 1.00).
When all the patients were considered (regardless of leukaemia type), the risk of developing leukaemia secondary to bone marrow hypoplasia was found to be significantly greater (p<0.001, Fisher's exact test) in those children who showed expression of substance P in normal‐looking lymphocytes at the initial bone marrow evaluation—that is, before neoplastic transformation. However, in subgroup III the risk of death resulting from progression of the leukaemia was not significantly greater in the children who were substance P‐positive at the initial bone marrow examination (ALL‐related death: p = 0.248; ANLL‐related death: p = 1.0; death regardless of leukaemia type: p = 0.153). On the other hand, statistical analysis of those patients in whom no treatment failure was noted during the observation period showed that the probability of remission of bone marrow hypoplasia was significantly greater in those children who were initially substance P‐negative (p<0.001).
No marked relationship was found between initially detected viral infection and the outcome of bone marrow hypoplasia.
Discussion
The search for causes and aids to the diagnosis of bone marrow hypoplasia includes taking a thorough history, a detailed physical examination and several laboratory tests, including microscopic analysis of bone marrow, sampled by needle biopsy or during surgery, determination of infectious diseases and, occasionally, genetic studies of patients and their families. The application of ICC techniques in the diagnosis of bone marrow hypoplasia has been tested already.21 However, attempts to use this technique have been restricted to the differentiation of myelodysplastic syndromes from various grades of bone marrow aplasia.21,22,23,24 Among other antigens, Ki‐67 antigen and proliferating cell nuclear antigen are useful in such a procedure.24 In patients with bone marrow aplasia the markers could not be seen in nucleated cells, whereas in patients with myelodysplastic syndromes the percentage of immunopositive cells ranged from 20% to 60%.24 These studies, however, have not attempted to determine the future trend of such bone marrow hypoplastic lesions.
Our selection of substance P as an indicator of the potential evolving trend of bone marrow hypoplasia was prompted by detection of its role and the role of other neuropeptides in the physiological control of haematopoiesis. Owing to the peptidergic nerve endings in bone marrow, substance P easily comes into contact with haematopoietic and stromal cells of the bone marrow.25 Previous investigations have proved that substance P is released in bone marrow by macrophages,26 eosinophils27,28 and vascular endothelial cells.29,30,31 Most bone marrow cells—that is, haematopoietic cells,32 the cells forming bone marrow stroma,33 and T and B lymphocytes present in bone marrow34—are equipped with the substance P‐specific NK‐1R receptor. Substance P, tested in short‐term human bone marrow cultures in methylcellulose, is capable of supporting haematopoiesis in vitro.32 This indicates that at a concentration of 10−8–10−11 mol/l it can substitute for IL3 and for granulocyte–monocyte colony stimulating factor, the presence of which is indispensable for growth of the colonies. However, substance P cannot substitute for erythropoietin, although, given in parallel, it stimulates its effects. The specificity of such a stimulatory function of substance P was confirmed when it was given with blockers of the known subtypes of substance P receptor. Substance P given with the blocker for the NK‐1R subtype of the receptor yielded results similar to those in negative controls. On the other hand, substance P, when given with a blocker of the NK‐2R receptor, exerted no effect on the activity of substance P.7,32 Substance P was also found to act indirectly on haematopoietic cells through the stromal cells, stimulating them to produce cytokines. In substance P‐supplemented cultures, IL1‐specific, IL3‐specific, IL6‐specific and granulocyte–monocyte colony stimulating factor‐specific antibodies induced only partial growth inhibition, indicating that substance P acts by inducing synthesis of the cytokines.7,32,35 Substance P can also induce synthesis of IL1 and of stem cell factor in cells of the bone marrow sublayer.7,32,35
Some investigators have suggested that substance P released from nerve endings not only augments IL1 synthesis but also increases the density of IL1 receptors.7 In turn, IL1, released by the sublayer cells, augments the density of NK‐1R receptors on the surface of haematopoietic and sublayer cells.
We have previously reported that substance P is also secreted by blast cells in ALL.18,19 A possible connection between the substance P‐positive blast pattern and leukaemia relapse may prove that substance P is associated with the proliferation of the tumour cells.
Our studies of the bone marrow of healthy people (who formed the control group) showed that the immunocytochemical expression of peripherally located substance P (most probably coupled to NK‐1R) on nucleated cells of the bone marrow was ⩽5%. These results prompted us to carry out analogous determinations in the material sampled from patients with clinical symptoms of bone marrow hypoplasia. In some of them the proportion of substance P‐positive cells among all nucleated cells was 67.6–95.8% (mean of 81.5% cells for ICC and 84.3% with ISH), with an absence of neoplastic cells. Subsequent observation of these patients showed that they developed a proliferative disease of the bone marrow. Throughout the entire time preceding the neoplastic transformation, an increased number of substance P‐positive cells was noted even if the proportion of Ki‐67‐positive cells was similar to control values. Neoplastic transformation was manifested by the presence of poorly differentiated cells with a positive reaction for Ki‐67 (39.6–49.8%) in bone marrow. In line with the above, we suggest that the presence of substance P in normal‐looking B lymphocytes of hypoplastic bone marrow becomes the second (after nerve endings) source of this peptide, which may stimulate other neoplastically transformed cells for uncontrolled proliferation. Substance P possibly accelerates the already initiated development of leukaemia. The origin of leukaemia, however, seems to be independent of substance P expression.
The expression of substance P in normal‐looking bone marrow lymphocytes before neoplastic transformation could not predict the death of the patient, which is certainly related to the small number of patients in subgroup III and the different chemotherapy protocols used in the treatment of ALL and ANNL.
Bone marrow hypoplasia, followed by neoplastic transformation, often remains asymptomatic. Only a few patients receive specialist care before the symptoms of bone marrow proliferative disease become evident. In these few patients, the diagnosis of bone marrow hypoplasia leads to subsequent check‐ups, during the course of which immunocytochemical analysis of cellular inducers of differentiation on the surface of bone marrow cells may provide a simple screening test pointing to the potential trend of evolution of the lesion.
Conclusions
Substance P expressed in the B lymphocytes of hypoplastic bone marrow may predict its leukaemic transformation.
The significant correlation between a substance P‐positive bone marrow pattern and expansion of tumour cells in the bone marrow may prove the potential role of this oligopeptide in the pathogenesis of leukaemia.
The results reported here, although promising, require confirmation by the investigation of a larger group of patients.
Acknowledgements
We thank Professor Geoffrey Shaw for his invaluable help in English text editing.
Abbreviations
ALL - acute lymphoblastic leukaemia
ANLL - acute non‐lymphoblastic leukaemia
ICC - immunocytochemistry
IL1 - interleukin 1
ISH - in situ hybridisation
NK‐1R - nerokinin‐1 receptor
Footnotes
Competing interests: None.
References
- 1.Stockman J A. Aplastic anemia. In: Nelson textbook of pediatrics (14/e by Behrman). Warszawa: PWN, 1996, 1426–8 (in Polish)
- 2.Alter B P, Potter N U. Classification and aetiology of the aplastic anaemias. Clin Haematol 19787431–439. [PubMed] [Google Scholar]
- 3.Williams D M, Lynch R E, Cartwright G E. Drug‐induced aplastic anemia. Semin Hematol 197310195–201. [PubMed] [Google Scholar]
- 4.Baranski B, Armstrong G, Truman J T.et al Epstein‐Barr virus in the bone marrow of patients with aplastic anemia. Ann Intern Med 1988109695–704. [DOI] [PubMed] [Google Scholar]
- 5.Maggio J E. Tachykinins. Ann Rev Neurosci 19901113–26. [DOI] [PubMed] [Google Scholar]
- 6.Pernow B. Substance P. Pharmacol Rev 19833585–98. [PubMed] [Google Scholar]
- 7.Rameshwar P, Ganea D, Gascon P. Substance P mediates production of stem cell factor and Il‐1 in bone marrow stroma: potential autoregulatory role for these cytokines in SP receptor expression and induction. Blood 19958698–113. [PubMed] [Google Scholar]
- 8.Aronin N. Substance P and the tachykins. In: Endocrinology and metabolism. Philadelphia: JB Lippincott Company, 19901309–1322.
- 9.Regoli D, Nguyen Q T, Jukic D. Neurokinin receptor subtypes characterized by biological assays. Life Sci 1994452035–2041. [DOI] [PubMed] [Google Scholar]
- 10.Gerard N P, Bao L, Ping H X.et al Molecular aspects of the tachykinin receptors. Regul Pept 19934321–25. [DOI] [PubMed] [Google Scholar]
- 11.Sirinek L, O'Dorisio M. Modulation of immune function by intestinal neuropeptides. Acta Oncol 199130509–518. [DOI] [PubMed] [Google Scholar]
- 12.Felten D, Felten S, Carlson S. Noradrenergic and peptidergic innervation of lymphoid tissue. J Immunol 1985135(Suppl)7559–7655. [PubMed] [Google Scholar]
- 13.Savino W, Dardenne M. Immuno‐neuroendocrine interactions. Immunol Today 199516318–329. [DOI] [PubMed] [Google Scholar]
- 14.Hsu S, Raine L, Fanger H. Use of avidin‐biotin peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 198129577–580. [DOI] [PubMed] [Google Scholar]
- 15.Shi S R, Cote R J, Taylor C R. Antigen retrieval immunocytochemistry: past, present and future. J Histochem Cytochem 199745327–343. [DOI] [PubMed] [Google Scholar]
- 16.Crist W M, Grosse C E, Pullen J. Immunologic markers in childhood acute lymphoblastic leukemia. Semin Oncol 198512105–112. [PubMed] [Google Scholar]
- 17.Coghlan J, Aldred P, Haralambidis J.et al Hybridisation histochemistry. Anal Biochem 19851491–28. [DOI] [PubMed] [Google Scholar]
- 18.Nowicki M, Miskowiak B, Ostalska‐Nowicka D. Detection of substance P and its mRNA in human blast cells in childhood lymphoblastic leukemia using immunocytochemistry and in situ hybridization. Folia Histochem Cytobiol 20034133–36. [PubMed] [Google Scholar]
- 19.Nowicki M, Miskowiak B. Substance P – a potent risk factor in childhood lymphoblastic leukemia. Leukemia 2003171096–1099. [DOI] [PubMed] [Google Scholar]
- 20.Elias J, Gown A, Nakamura R. Special report: quality control in immunohistochemistry. Am J Clin Pathol 198992836–843. [DOI] [PubMed] [Google Scholar]
- 21.Sadahira Y, Wada H, Manabe T.et al Immunohistochemical assessment of human bone marrow macrophages in hematologic disorders. Pathol Int 199949626–632. [DOI] [PubMed] [Google Scholar]
- 22.Kitagawa M, Kamiyama R, Kasuga T. Expression of the proliferating cell nuclear antigen in bone marrow cells from patients with myelodysplastic syndromes and aplastic anemia. Hum Pathol 199324359–363. [DOI] [PubMed] [Google Scholar]
- 23.Juneja H S, Gardner F H. Functionally abnormal marrow stromal cells in aplastic anemia. Exp Hematol 198513194–199. [PubMed] [Google Scholar]
- 24.Orazi A, Albitar M, Heerema N A.et al Hypoplastic myelodysplastic syndromes can be distinguished from acquired aplastic anemia by CD34 and PCNA immunostaining of bone marrow biopsy specimens. Am J Clin Pathol 1997107268–274. [DOI] [PubMed] [Google Scholar]
- 25.Felten S Y, Felten D L, Bellinger D L.et al Noradrenergic and peptidergic innervation of lymphoid organs. Neuroimmunoendocrinology 19922567–77. [PubMed] [Google Scholar]
- 26.Pascual D W, Bost K L. Substance P production by P388D1 macrophages: a possible autocrine function for this neuropeptide. Immunology 19907152–77. [PMC free article] [PubMed] [Google Scholar]
- 27.Weinstock J V, Blum A, Walder J.et al Eosinophils from granulomas in murine Schistosomatosis mansoni produce substance P. J Immunol 1988141961–966. [PubMed] [Google Scholar]
- 28.Weinstock J V, Blum A. Tachykinin production by granuloma eosinophils in murine Schistosomatosis mansoni. J Immunol 19891423256–3561. [PubMed] [Google Scholar]
- 29.Linnik D M, Moskowitz A M. Identification of immunoreactive substance P in human and other mammalian endothelial cells. Peptides 198910957–963. [DOI] [PubMed] [Google Scholar]
- 30.Ralevic V. Substance P is released from the endothelium of normal and capsaicin‐treated rat hind‐limb vasculature, in vivo, by increased flow. Circul Res 1990661178–1185. [DOI] [PubMed] [Google Scholar]
- 31.Walsh D A, Wharton J, Blake D R.et al Neural and endothelial regulatory peptides, their possible involvement in inflammation. Int J Tissue Reac 199214101–112. [PubMed] [Google Scholar]
- 32.Rameshwar P, Ganea D, Gascon P. In vitro effect of substance P on hematopoiesis. Blood 199381391–399. [PubMed] [Google Scholar]
- 33.Ziche M, Morbidelli L, Pacini M.et al NK‐1 receptors mediate the proliferative response of human fibroblasts to tachykinins. Br J Pharmacol 199010011–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Payan D G, Brewster D B, Missirian‐Bastian A.et al Substance P recognition by a subset of human T‐lymphocytes. J Clin Invest 1984741532–1537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rameshwar P, Gascon P. Induction of negative hematopoietic receptors by NK‐A in bone marrow stroma. Blood 19968898–107. [PubMed] [Google Scholar]