Abstract
Objective
To measure coronary flow reserve (CFR), an index of microvascular function, in Anderson‐Fabry disease (AFD) at baseline and after enzyme replacement therapy (ERT).
Methods and results
Mean (SD) myocardial blood flow (MBF) at rest and during hyperaemia (adenosine 140 μg/kg/min) was measured in 10 male, non‐smoking patients (53.8 (10.9) years, cholesterol 5.5 (1.3) mmol/l) and in 24 age matched male, non‐smoking controls (52.0 (7.6) years, cholesterol 4.5 (0.6) mmol/l) by positron emission tomography (PET). Resting and hyperaemic MBF and CFR (hyperaemic/resting MBF) were reduced in patients compared with controls (0.99 (0.17) v 1.17 (0.25) ml/g/min, p < 0.05; 1.37 (0.32) v 3.44 (0.78) ml/g/min, p < 0.0001; and 1.41 (0.39) v 3.03 (0.85), p < 0.0001, respectively). This coronary microvascular dysfunction was independent of cholesterol concentrations. PET was repeated in five patients after 10.1 (2.3) months of ERT; resting and hyperaemic MBF and CFR were unchanged after ERT (0.99 (0.16) v 0.99 (0.16) ml/g/min; 1.56 (0.29) v 1.71 (0.3) ml/g/min; and 1.6 (0.37) v 1.74 (0.28), respectively; all not significant).
Conclusions
The results of the present study show that patients with AFD have very abnormal coronary microvascular function. These preliminary data suggest that ERT has no effect on coronary microvascular dysfunction. Further work is necessary to determine whether treatment at an earlier stage in the course of the disease may improve coronary microvascular function in patients with AFD.
Keywords: cardiomyopathy, coronary circulation, myocardial blood flow, myocardial ischaemia, cardiac imaging
Anderson‐Fabry disease (AFD) is a multisystem disorder caused by an X‐linked deficiency of lysosomal α galactosidase A that results in renal, cardiac, and cerebrovascular disease and premature death.1 Patients with AFD have angina despite angiographically normal coronary arteries. Recent studies have also shown progressively deteriorating left ventricular (LV) systolic function and myocardial scarring in patients with AFD cardiomyopathy.2,3 We hypothesised that these clinical abnormalities may be caused by coronary microvascular dysfunction.
The objectives of this study were to measure coronary flow reserve (CFR), an index of coronary microvascular function, in a consecutive cohort of patients with AFD by using positron emission tomography (PET) and to determine the effect of enzyme replacement therapy (ERT) on microvascular abnormalities.
METHODS
Patient and controls
Ten non‐smoking male patients with AFD (mean (SD) age 53.8 (10.9) years, range 43–82 years; cholesterol 5.5 (1.3) mmol/l) referred to the Heart Hospital, London, UK were studied. The diagnosis of AFD was based on the identification of an α galactosidase A gene mutation and a low plasma α galactosidase A (mean (SD) 0.45 (0.35) nmol/h/ml, range 0.04–0.99 nmol/h/ml). Nine patients were identified through screening of patients presenting with unexplained LV hypertrophy; six of these patients have been reported on previously.4 One patient was referred with an established diagnosis of AFD (patient 6) and one was identified through family screening (patient 10). At the time of diagnosis, none of these patients was receiving ERT.
All patients underwent ECG and two dimensional Doppler echocardiography by previously described methods.5 LV mass was calculated by M mode echocardiography with the Devereux formula and indexed to body surface area.6 Nine patients had symptoms or signs suggestive of myocardial ischaemia and underwent coronary angiography to exclude coronary artery disease.
Twenty four healthy non‐smoking age matched men (52.0 (7.6) years, cholesterol 4.5 (0.6) mmol/l) with normal ECG and no evidence of cardiac disease served as controls for the myocardial blood flow (MBF) and CFR data.
Enzyme replacement therapy
Five patients (table 1) received Fabrazyme (Genzyme Corporation, Cambridge, Massachusetts, USA) at a dose of 1 or 2 mg/kg every two weeks as part of a separate randomised study. Follow up PET was obtained 17.1 (1.9) months after the baseline scan while patients were being treated. Plasma globotriaosylceramide (neutral glycosphingolipid) concentrations were measured by tandem mass spectrometry before and after ERT.7
Table 1 Patient characteristics.
| Patient | Age at study (years) | Chest pain | NYHA class | Syncope | Palpitations | Max LVWT (mm) | FS (%) | LVMI (g/m2) | CFR |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 46 | + | II | + | + | 25 | 40 | 263 | 1.58 |
| 2* | 58 | − | I | + | + | 26 | 26 | 407 | 2.60 |
| 3* | 81 | − | III | − | + | 18 | 23 | 228 | 2.31 |
| 4 | 49 | + | II | − | − | 17 | 35 | 192 | 2.09 |
| 6* | 55 | + | II | − | + | 15 | 36 | 222 | 2.24 |
| 7* | 49 | + | II | − | + | 16 | 39 | 180 | 2.74 |
| 8 | 45 | + | I | − | + | 20 | 51 | 248 | 1.27 |
| 9* | 54 | − | I | − | − | 24 | 42 | 314 | 2.27 |
| 10 | 43 | − | I | − | − | 15 | 33 | 93 | 1.28 |
| 12 | 58 | + | II | + | + | 20 | 27 | 407 | 1.83 |
*Five patients with follow up positron emission tomography after enzyme replacement therapy.
+, present; −, absent.
CFR, coronary flow reserve; FS, fractional shortening; LVMI, left ventricular mass index; Max LVWT, maximum left ventricular wall thickness; NYHA, New York Heart Association.
The study was approved by the local research ethics committees and all participants gave written informed consent. Radiation exposure was approved by the UK Administration of Radioactive Substances Advisory Committee.
Positron emission tomography
All patients and controls underwent PET to measure MBF at rest and during adenosine induced hyperaemia (140 μg/kg/min intravenously). Images were recorded with an ECAT 931‐08/12 15 slice tomograph with a 10.5 cm axial field of view (CTI/Siemens, Knoxville, Tennessee, USA). Resting and hyperaemic MBF were measured with oxygen‐15 labelled water (H215O) as previously reported.8 Arterial blood pressure was recorded by automatic cuff sphygmomanometer at one minute intervals and the ECG was monitored continuously throughout the procedure.
PET data analysis
All emission and transmission data were reconstructed with a Hanning filter with a cut off frequency of 0.5 units of the reciprocal of the sampling interval of the projection data resulting in an image resolution of 8.4 × 8.3 × 6.6 mm full width at half maximum at the centre of the field of view. Myocardial and blood pool images were then generated directly from the dynamic H215O study as previously reported.9 Regions of interest were drawn within the left atrium and ventricular myocardium on consecutive image planes. These were projected on to the dynamic H215O images to generate blood and tissue time activity curves. Arterial and tissue activity curves were fitted to a single tissue compartment tracer kinetic model to give values of MBF (ml/g/min).9 Coronary resistance (mm Hg/ml/min/g) was calculated as the ratio of mean arterial pressure to MBF and CFR was calculated as the ratio of hyperaemic MBF to resting MBF.
Statistical analysis
Continuous variables were compared by the paired Student's t test (Statview version 5.0; SAS institute Inc, Cary, North Carolina, USA). Linear regression was determined to assess the relation between cholesterol concentrations, LV mass index, MBF, and CFR. Data are reported as mean (SD) values. A probability value of p< 0.05 was considered significant.
RESULTS
Patient characteristics
Table 1 shows the clinical characteristics of the patients. Six (60%) patients had exertional chest pain. None of the nine patients had angiographically significant coronary artery disease. Three patients had rate responsive dual chamber permanent pacemakers: one for LV outflow tract gradient reduction and two for heart block. None of the patients had a PR interval less than 120 ms. Two patients had non‐sustained ventricular tachycardia on Holter monitoring (patients 2 and 3) and two were in atrial fibrillation (patients 5 and 11). Seven years before the diagnosis of AFD, patient 1 had coronary sinus pH assessed during dipyridamole infusion.10 The peak change in coronary sinus pH during hyperaemia was −0.086 units, indicating severe myocardial ischaemia.
Serum cholesterol was higher in the patients than in controls (mean difference 0.97 mmol/l, 95% confidence interval 0.3 to 1.6, p = 0.006). Patients also had higher high density lipoprotein cholesterol concentrations than did controls (1.7 (0.5) mmol/l v 1.1 (0.4) mmol/l, respectively, p = 0.002). Low density lipoprotein cholesterol (3.1 (0.8) mmol/l v 3.7 (1.3) mmol/l, respectively, p = 0.2) and triglyceride concentrations (1.4 (0.7) mmol/l v 1.8 (1.6) mmol/l, respectively, p = 0.5) did not differ between patients and controls.
Two patients had acroparaesthesia, one had hypohidrosis, and one had renal dysfunction (creatinine clearance 52 ml/min). None of the patients was hypertensive and one had non‐insulin dependent diabetes mellitus. No angiokeratomas were noted in any of the patients. All patients had a maximum LV wall thickness ⩾ 13 mm (17 (4.0) mm, range 14–26 mm). Nine patients had concentric LV hypertrophy and one had asymmetric septal hypertrophy. At the time of study no patient had LV outflow tract obstruction; the mean LV end systolic, end diastolic, and left atrial diameters were 33 (0.9), 5.2 (0.8), and 4.7 (1.1) mm, respectively. The mean LV mass index was 234 (81.3) g/m2 (range 141–407 g/m2).
MBF and CFR
Heart rate and mean arterial blood pressure were similar in controls and AFD patients at rest and during adenosine infusion. The rate pressure product (systolic blood pressure × heart rate) was similar in the controls and patients during all study conditions.
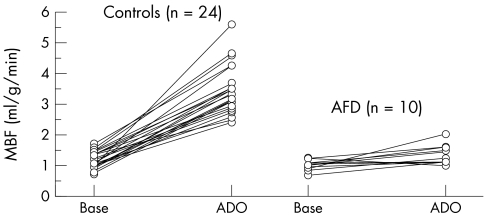
Resting and hyperaemic MBF and CFR were significantly reduced in AFD patients compared with controls (0.99 (0.17) v 1.17 (0.25) ml/g/min, p < 0.05; 1.37 (0.32) v 3.44 (0.78) ml/g/min, p < 0.0001; and 1.41 (0.39) v 3.03 (0.85), p < 0.0001, respectively) (fig 1, table 1). This reduction in coronary microvascular function was independent of cholesterol and high density lipoprotein cholesterol concentrations. Resting coronary resistance was comparable in patients and controls (82.2 (27.8) v 89.3 (31.2) mm Hg/ml/min/g, respectively, not significant). The minimum resistance during hyperaemia was higher in AFD patients than in controls (65.1 (16.9) v 26.5 (6.0) mm Hg/ml/min/g, p < 0.0001). No correlation was found between LV mass index and resting or hyperaemic MBF or CFR.
Figure 1 Myocardial blood flow (MBF) at rest (Base) and during adenosine induced hyperaemia (ADO) in patients with Anderson‐Fabry disease (AFD) and controls.
Response to ERT
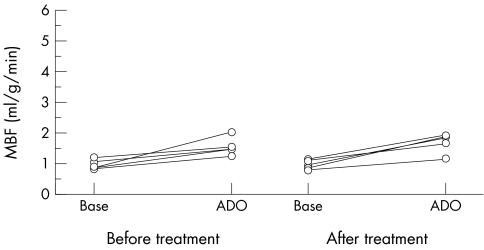
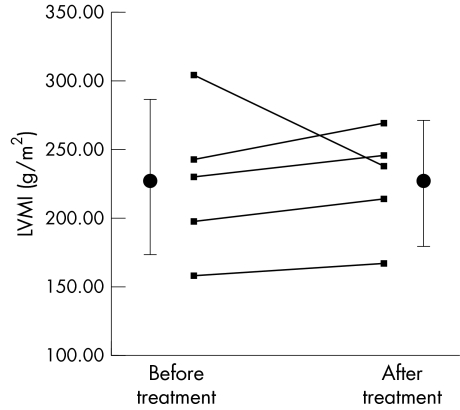
In patients who underwent a repeat PET plasma globotriaosylceramide had decreased with ERT (mean change 5.3 μg/ml, 95% confidence interval 0.5 to 10.1, p = 0.04). Resting and hyperaemic MBF and CFR before and after 10.1 (2.3) months of ERT were similar (0.99 (0.16) v 0.99 (0.16) ml/g/min; 1.56 (0.29) v 1.71 (0.3) ml/g/min; and 1.6 (0.37) v 1.74 (0.28), respectively; all not significant) (fig 2). Resting coronary resistance was similar before and after ERT (83.9 (18.9) v 90.2 (26.5) mm Hg/ml/min/g, respectively, not significant). The minimum resistance during hyperaemia was unchanged after ERT (56.9 (14.7) v 52.8 (18.7) mm Hg/ml/min/g, not significant). LV mass index did not change (mean change 0.4 (37.8) g/m2, range −66.3 to +26.4, not significant) (fig 3). LV cavity dimensions did not change (mean change in end systolic cavity dimension 0.1 (0.3) mm; mean change in end diastolic cavity dimension 0.1 (0.2) mm; both not significant). Fractional shortening tended to decease (mean pre‐ERT fractional shortening 34.8 (6.7)%, mean post‐ERT fractional shortening 31.4 (6.3)%, p = 0.08). Maximum LV wall thickness did not change (mean change 0.02 (0.3) mm, not significant).
Figure 2 MBF at rest and during ADO before and after enzyme replacement therapy with Fabrazyme.
Figure 3 Left ventricular mass index (LVMI) before (226.3 (54.1) g/m2) and after (226.7 (39.0) g/m2, not significant) enzyme replacement therapy with Fabrazyme. Cumulative data are presented as mean (SD).
DISCUSSION
This study shows that patients with AFD have a substantial reduction in hyperaemic MBF and CFR compared with normal controls. The significance of these findings for individual patients remains to be determined, but the severity of the microvascular dysfunction suggests that these abnormalities may have a substantial influence on the natural history of AFD.
Mechanisms of microvascular dysfunction
Symptoms and signs suggestive of myocardial ischaemia in the absence of coronary disease are common in patients with cardiomyopathies and in patients with LV hypertrophy secondary to pressure overload. Reductions in CFR similar to those seen in this study have been observed in all these conditions, although different mechanisms are involved. In patients with hypertrophic cardiomyopathy, remodelling of the intramural arterioles is important,11,12 whereas in aortic stenosis increased perivascular fibrosis and intramyocardial pressure and reduced diastolic filling are the dominant mechanisms for microvascular dysfunction.13,14
In AFD, several mechanisms may contribute to microvascular dysfunction. AFD cardiomyopathy is characterised by globotriaosylceramide deposition in myocytes, conduction tissue, vascular endothelium, and valve tissue. This is accompanied by secondary changes such as myocyte hypertrophy and fibrosis,15 which cause raised coronary vascular resistance and increased myocardial oxygen demand. Although endothelial globotriaosylceramide deposits may lead to microvascular dysfunction, a recent publication has shown that patients with AFD have enhanced nitric oxide independent endothelial function measured by forearm venous plethysmography.16 It is unknown whether similar abnormalities would be seen the in coronary microvasculature.
Response to ERT
In this study plasma concentrations of globotriaosylceramide were significantly reduced with ERT but no improvements in coronary microvascular function or LV mass were seen. However, interpretation of this observation is limited by the small size of the cohort and by the fact that the cohort was considerably older and had more severe cardiac disease than did patients in previous trials of ERT.17,18,19 Along with an improvement in AFD symptoms these trials have shown a reduction in LV mass and an improvement in systolic function with ERT. To understand the impact of ERT on coronary microvascular function a larger study conducted over a longer time is required.
Conclusion
This study showed that microvascular dysfunction is important in the pathophysiology of AFD cardiomyopathy. Further studies are required to assess the impact of ERT on microvascular function.
ACKNOWLEDGEMENTS
We thank Dr Elizabeth Young and her colleagues at the Institute of Child Health, London, and Amy Cole at The Charles Dent Metabolic Unit, The National Hospital for Neurology and Neurosurgery, London. WJM, BS, RT, and MTT were supported by the British Heart Foundation. JSS was supported by an educational grant from TKT (Transkaryotic Therapies) Europe‐5S.
Abbreviations
AFD - Anderson‐Fabry disease
CFR - coronary flow reserve
ERT - enzyme replacement therapy
LV - left ventricular
MBF - myocardial blood flow
PET - positron emission tomography
References
- 1.Desnick R J, Ioannou Y A, Eng C M. a‐Galactosidase A deficiency: Fabry disease. In: Scriver CR, Beaudet AL, Sly WS, et al, eds. The metabolic and molecular bases of inherited disease. 8th ed. New York: McGraw‐Hill, 20013733–3774.
- 2.Moon J C, Sachdev B, Elkington A G.et al Gadolinium enhanced cardiovascular magnetic resonance in Anderson‐Fabry disease: evidence for a disease specific abnormality of the myocardial interstitium. Eur Heart J 2003242151–2155. [DOI] [PubMed] [Google Scholar]
- 3.Shah J S, Lee P, Hughes D.et al The natural history of left ventricular systolic function in Anderson‐Fabry disease. Heart 200591533–534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sachdev B, Takenaka T, Teraguchi H.et al Prevalence of Anderson‐Fabry disease in male patients with late onset hypertrophic cardiomyopathy. Circulation 20021051407–1411. [DOI] [PubMed] [Google Scholar]
- 5.Maron B J, Gottdiener J S, Epstein S E. Patterns and significance of distribution of left ventricular hypertrophy in hypertrophic cardiomyopathy: a wide angle, two dimensional echocardiographic study of 125 patients. Am J Cardiol 198148418–428. [DOI] [PubMed] [Google Scholar]
- 6.Devereux R B, de Simone G, Ganau A.et al Left ventricular hypertrophy and hypertension. Clin Exp Hypertens 1993151025–1032. [DOI] [PubMed] [Google Scholar]
- 7.Mills K, Vellodi A, Morris P.et al Monitoring the clinical and biochemical response to enzyme replacement therapy in three children with Fabry disease. Eur J Pediatr 2004163595–603. [DOI] [PubMed] [Google Scholar]
- 8.Hermansen F, Rosen S D, Fath‐Ordoubadi F.et al Measurement of myocardial blood flow with oxygen‐15 labelled water: comparison of different administration protocols. Eur J Nucl Med 199825751–759. [DOI] [PubMed] [Google Scholar]
- 9.Hermansen F, Ashburner J, Spinks T J.et al Generation of myocardial factor images directly from the dynamic oxygen‐15‐water scan without use of an oxygen‐15‐carbon monoxide blood‐pool scan. J Nucl Med 1998391696–1702. [PubMed] [Google Scholar]
- 10.Elliott P M, Rosano G M, Gill J S.et al Changes in coronary sinus pH during dipyridamole stress in patients with hypertrophic cardiomyopathy. Heart 199675179–183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Camici P, Chiriatti G, Lorenzoni R.et al Coronary vasodilation is impaired in both hypertrophied and nonhypertrophied myocardium of patients with hypertrophic cardiomyopathy: a study with nitrogen‐13 ammonia and positron emission tomography. J Am Coll Cardiol 199117879–886. [DOI] [PubMed] [Google Scholar]
- 12.Maron B J, Wolfson J K, Epstein S E.et al Intramural (“small vessel”) coronary artery disease in hypertrophic cardiomyopathy. J Am Coll Cardiol 19868545–557. [DOI] [PubMed] [Google Scholar]
- 13.Rajappan K, Rimoldi O E, Dutka D P.et al Mechanisms of coronary microcirculatory dysfunction in patients with aortic stenosis and angiographically normal coronary arteries. Circulation 2002105470–476. [DOI] [PubMed] [Google Scholar]
- 14.Rajappan K, Rimoldi O E, Camici P G.et al Functional changes in coronary microcirculation after valve replacement in patients with aortic stenosis. Circulation 20031073170–3175. [DOI] [PubMed] [Google Scholar]
- 15.Von Scheidt W, Eng C M, Fitzmaurice T F.et al An atypical variant of Fabry's disease with manifestations confined to the myocardium. N Engl J Med 1991324395–399. [DOI] [PubMed] [Google Scholar]
- 16.Altarescu G M, David F, Pursley R.et al Enhanced endothelium‐dependent vasodilation in Fabry disease. Stroke 2001321559–1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Eng C M, Guffon N, Wilcox W R.et al Safety and efficacy of recombinant human alpha‐galactosidase A: replacement therapy in Fabry's disease. N Engl J Med 20013459–16. [DOI] [PubMed] [Google Scholar]
- 18.Weidemann F, Breunig F, Beer M.et al Improvement of cardiac function during enzyme replacement therapy in patients with Fabry disease: a prospective strain rate imaging study. Circulation 20031081299–1301. [DOI] [PubMed] [Google Scholar]
- 19.Beck M, Ricci R, Widmer U.et al Fabry disease: overall effects of agalsidase alfa treatment. Eur J Clin Invest 200434838–844. [DOI] [PubMed] [Google Scholar]