Abstract
Evidence supports the central role of endothelium and inflammation in all phases of the atherosclerotic process. Clinical studies have shown their prognostic potential for the development of ischaemic events and for adverse outcome after acute coronary syndromes. Reduction in inflammatory levels and improving endothelial function by traditional and novel treatment strategies were associated with a proportional reduction in cardiovascular events. However, randomised controlled trials are required to explore further whether drugs targeting the inflammatory process and endothelial function will constitute a reasonable adjunctive treatment for patients with coronary artery disease.
Keywords: atherosclerosis, endothelium, inflammation, stable angina
Coronary artery disease (CAD) remains the principal cause of death and disability leading to increased burden in health care costs in developed countries. CAD is characterised by a long asymptomatic phase of development, which starts as early as the first decade of life and progresses eventually to the formation of atherosclerotic plaques. These plaques, when they are unstable or become obstructive, lead to ischaemic syndromes, the clinical manifestations of atherosclerosis and CAD.
Over the past few decades, our understanding of the vascular biology of atherogenesis and its clinical presentations has evolved enormously. It is now clear that inflammatory processes have a key role not only in the initiation and progression of atherosclerosis but also in the stability of the established atherosclerotic plaques.1,2,3 In the long preclinical phase the vascular endothelium has been a major focus of research. Traditional and novel risk factors for cardiovascular disease trigger a chronic inflammatory process, which is accompanied by loss of vasodilatory and antithrombotic properties of the vascular endothelium.4,5,6
In this review endothelial function and inflammatory processes are assessed as important determinants of the development of CAD. In addition the exciting potential for treatments that target this disturbed pathology are discussed.
ENDOTHELIUM IN CAD
The endothelium has an important role in vascular homeostasis. It secretes numerous mediators such as nitric oxide, prostacyclin, and endothelin that regulate vascular tone, platelet activity, and coagulation factors but also influence vascular inflammation and cell migration.7
Risk factors and vascular endothelium
A variety of insults may damage endothelial structure and function, which include physical injuries, biochemical injury, and immune mediated damage. These insults cause alteration in endothelial physiology resulting in impairment or loss of its normal functions. The development of an imbalance in the release of vasoconstrictor and vasodilator agents from the endothelium results in impaired endothelium dependent vasodilatation, the hallmark of endothelial dysfunction.8 Over the past decade, the study of nitric oxide dependent regulation of vascular tone in response to pharmacological and physical stimuli in different vascular beds has been made possible. Various invasive and non‐invasive techniques have shown that patients with CAD and those with increased cardiovascular risk factor profile present with dysfunctional endothelium.
Some cardiovascular risk factors have been consistently associated with disturbances in normal endothelial physiology. Non‐modifiable risk factors include older age and male sex. Modifiable risk factors include hypercholesterolaemia, which appears as one of the most important contributors to atherosclerotic disease progression, and others such as smoking, hypertension, and diabetes, which have been shown to be major risk factors for both coronary and cerebral events irrespective of age and sex.4,9,10
New risk factors associated with the westernised way of life have also been described. Among them obesity, the introduction of an atherogenic diet, and physical inactivity are emerging as important players in this process.11,12,13 In addition, epidemiological data have suggested that infection and chronic inflammation may trigger endothelial dysfunction.14 Studies of adults documented that serological evidence of exposure to multiple intracellular pathogens, particularly in the context of a low grade inflammatory response, is associated with increased coronary atherosclerosis. Inflammatory vascular disease is associated with persistent endothelial dysfunction. More recently mild non‐specific viral infections have been found to have an apparent detrimental effect on the vascular endothelium.15,16
Identifying the aetiology of endothelial dysfunction and CAD has increased the list of candidate risk factors to encompass rise of serum homocysteine concentrations, birth weight, and polymorphisms that affect the expression of genes regulating endothelial biology.
PROGNOSTIC ROLE OF ENDOTHELIAL FUNCTION IN CAD
Endothelial vasodilator function was assessed as a test that can integrate the impact of multiple environmental and genetic influences on the vasculature and thus serve as a useful diagnostic prognostic tool but also as a therapeutic target to improve outcome.
Studies performed at the time of cardiac catheterisation, although somewhat limited by small number of studied patients or by the lack of a thorough multivariable assessment of other potentially important independent predictors of risk, clearly show a direct association between endothelial dysfunction and event rates (table 1). (To view tables 1 to 5, visit the Heart website—www.heartjnl.com/supplemental.) On the other hand, peripheral endothelial assessment gave contradictory results (table 2). Perticone et al17 and Heitzer et al,18 by using invasive forearm venous occlusion plethysmography, highlighted the prognostic importance of endothelial dysfunction. Three other groups by means of non‐invasive methods shared similar findings whereas others failed to do so in detailed multivariate analysis. Differences in the population studied, differences in treatment regimens used, the underlying inflammatory response, or differences in the methods used possibly account for these discrepancies.
INFLAMMATORY MECHANISMS IN ATHEROSCLEROSIS
In the presence of risk factors the vascular endothelium responds, and the activation of protein kinase C and transcriptional messenger nuclear factor κB are the primary events. This leads to upregulation of genes that code for and induce angiotensin converting enzyme activity, local production of angiotensin II, and expression of endothelial cell surface adhesion molecules. These events in turn initiate and amplify cellular and subcellular responses in conduit coronary arteries that lead to endothelial dysfunction. This may lead to intimal thickening, plaque formation, and ultimately disruption of plaque and clinical events.19
Three cellular components of the circulation—monocytes, platelets, and lymphocytes—together with endothelial and smooth muscle cells interact in multiple ways in concert with low density lipoprotein in generating atherosclerotic lesions. In addition, proinflammatory cytokines and cellular adhesion molecules, involved in the attachment of monocytes to the endothelial wall, appear to be critical in atherogenesis.
Adhesion molecules and monocytes
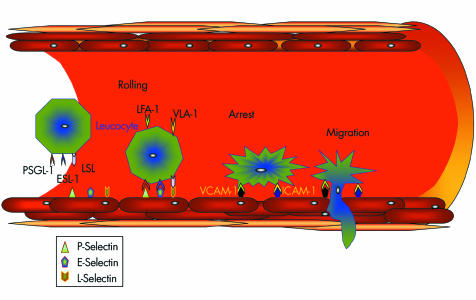
One early phase of atherosclerosis involves the recruitment of inflammatory cells from the circulation and their transendothelial migration. This process is mediated by cellular adhesion molecules, which are expressed on the vascular endothelium and on circulating leucocytes in response to several inflammatory stimuli. Adhesion is a multistep process that starts with leucocyte rolling on the endothelial surface. This is due to selectin ligation, whereas the subsequent firm adhesion depends on interactions between immunoglobulin‐like molecules (vascular cell adhesion molecule 1, intercellular cell adhesion molecule 1) on the endothelium and integrins on the leucocyte surface (fig 1).20
Figure 1 Leucocyte–endothelial cell interactions in atherosclerosis. Selectins mediate the initial rolling of leucocytes on the endothelial surface. Subsequent adhesion requires interaction between integrins expressed on leucocytes (leucocyte function associated antigen (LFA) 1 and very late antigen (VLA) 4) and immunoglobulin‐like molecules (vascular cell adhesion molecule (VCAM) 1 and intercellular cell adhesion molecule (ICAM) 1) on the endothelium. The final step is the transmigration of leucocytes in the intima. ESL‐1, E‐selectin ligand 1; LSL, L‐selectin ligand; PSGL‐1, P‐selectin glycoprotein ligand 1.
Monocytes/macrophages have key roles both in the initiation and progression of atherosclerosis. Recruitment of monocytes into the arterial wall is one of the earliest events in atherosclerosis. Intimal monocytes develop into macrophages, which are important mediators of inflammation and the innate immune response in atherosclerotic lesions. Macrophages contribute to the local inflammatory responses through production of cytokines, free oxygen radicals, proteases, and complement factors. The uptake of modified lipoproteins by macrophages leads to the accumulation of cholesterol esters and formation of macrophage derived foam cells, the hallmark of the fatty streak. Macrophages also contribute to lesion remodelling and to plaque rupture by secreting matrix metalloproteinases and thus contribute to the evolution of atherosclerosis in diverse ways.
B and T lymphocytes
A diverse lymphocyte population is found in atherosclerotic lesions with substantial number of T lymphocytes detected. T lymphocytes may enter the vessel wall before monocytes during the earliest stages of lesion formation and become activated as shown by the presence of activation markers on these cells.21 The presence of activated lymphocytes at each stage of human lesion formation provides compelling evidence for a role of this cell type in the orchestration of the disease process.
Cytokines and inflammatory markers in atherosclerosis
Cytokines differentially affect atherogenesis with distinct cytokines directing proatherogenic and antiatherogenic processes, and modulating plaque characteristics and clinical outcomes.22 The classic proinflammatory cytokines, interleukin 1 and tumour necrosis factor α, typically mediate proatherogenic processes while interleukin 10 mediates antiatherogenic pathways.
In addition recent evidence suggests that C reactive protein (CRP), a marker of underlying inflammation, may have a direct role in the pathophysiology of atherosclerosis. Thus, in the presence of CRP, uptake of low density lipoprotein cholesterol by macrophages is increased and contributes to foam cell formation. CRP can activate complement in atherosclerotic plaques leading potentially to plaque instability.23,24,25 It can induce adhesion molecule expression on human coronary endothelial cells.26 Lastly, increased CRP is also associated with endothelial dysfunction and the progression of atherosclerosis.27
PROGNOSTIC ROLE OF INFLAMMATION IN CAD
In identifying a reliable and independent predictor of the risk of an acute coronary event, the endothelium has not been the only candidate. Inflammatory mediators, which are instrumental in the pathology of CAD, have also been thoroughly tested. The inflammatory response may influence the prognosis of patients with CAD by accelerating atherosclerosis but also by sudden development of instability in already established atherosclerotic plaques. Furthermore, in the presence of an acute event inflammatory processes can modulate the consequences of ischaemia and necrosis. However, the contribution of each of these secondary and primary mechanisms of inflammation to prognosis may vary in different groups of patients.
The total white blood cell count is an inexpensive, reliable, easy to interpret inflammatory marker, which has been associated with adverse prognosis in patients who have stable coronary heart disease after a myocardial infarction.28 Experimental evidence suggests numerous mechanisms through which leucocytes may affect the stability of plaques in acute coronary syndromes.29,30,31 One potential participant in this process is the leucocyte enzyme myeloperoxidase. Myeloperoxidase concentrations are increased in persons with angiographically documented cardiovascular disease and within culprit lesions prone to rupture.32 Myeloperoxidase has been linked to the development of lipid laden soft plaque, the activation of protease cascades affecting the stability and thrombogenicity of plaque, the production of cytotoxic and prothrombogenic oxidised lipids, and the consumption of nitric oxide leading to vasoconstriction. Given its pathophysiological role, myeloperoxidase seems to be a good prognostic marker of the incidence of a coronary event (table 3).32,33
Increased concentrations of other circulating inflammatory markers, such as CRP, serum amyloid A protein, interleukin 6, and interleukin 1 receptor antagonist, are also commonly found in acute coronary syndromes (table 3). These markers have been shown to reflect the extent of myocardial necrosis and ischaemia/reperfusion damage; they have also been considered to be triggers of coronary instability.1,22
At the moment, however, the most attractive candidate marker of cardiovascular outcome is CRP. Commercially available high sensitivity assays allow the accurate and reproducible determination of CRP concentrations both in fresh and in frozen plasma. In addition the absence of circadian variation makes CRP determination applicable for comparisons in large population studies. Liuzzo et al34 were the first to report a direct association between increased CRP concentrations and adverse short term prognosis independently of necrosis and ischaemia.
Since then, several population based studies have shown that baseline CRP concentrations predict future cardiovascular events and implied that CRP testing may thus have a major adjunctive role in the global assessment of cardiovascular risk above the already used Framingham risk score (tables 4 and 5).35 However, this strong association does not seem to hold in all studies after adjustment for potential confounders. These discrepancies still possibly reflect differences in the studied end points or the confounding effect of medications usually administered to patients with CAD. Indeed, in the physicians health study aspirin reduced the concentrations of plasma CRP in apparently healthy men and this effect was associated with a reduction in the risk of a first myocardial infarction.35 In addition, data from the CARE (cholesterol and recurrent events) trial showed that patients with evidence of low grade vascular inflammation as measured by increased concentrations of CRP and serum amyloid A had a more than 50% reduction in recurrent events while receiving pravastatin.36 Randomised controlled trials are required to explore further whether a drug approach targeting CRP will be a reasonable adjunctive treatment for patients with CAD.
CONCLUSION
In recent years understanding of the pathophysiology of CAD has evolved enormously. Current evidence supports the central role of endothelium and inflammation in all phases of the atherosclerotic process. Clinical studies have shown their prognostic potential for the development of ischaemic events and for adverse outcome after acute coronary syndromes. Reduction in inflammatory levels and improving endothelial function by traditional and novel treatment strategies were associated with a proportional reduction in cardiovascular events. However, the identification of a single putative trigger for acute coronary syndrome seems unlikely in a disease with diverse aetiology and such a complex presentation of clinical symptoms. Further studies are needed to evaluate whether a combination of treatments will provide additional anti‐inflammatory effects and protection from cardiovascular events. (Additional references are available on the Heart website—www.heartjnl.com/supplemental.)
Tables 1–5 and additional references appear on the Heart website—http://www.heartjnl.com/supplemental
Supplementary Material
Abbreviations
CAD - coronary artery disease
CARE - cholesterol and recurrent events
CRP - C reactive protein
Footnotes
Tables 1–5 and additional references appear on the Heart website—http://www.heartjnl.com/supplemental
References
- 1.Libby P, Ridker P M, Maseri A. Inflammation and atherosclerosis. Circulation 20021051135–1143. [DOI] [PubMed] [Google Scholar]
- 2.Economou E, Tousoulis D, Katinioti A.et al Chemokines in patients with ischaemic heart disease and the effect of coronary angioplasty. Int J Cardiol 20018055–60. [DOI] [PubMed] [Google Scholar]
- 3.Tousoulis D, Davies G J, Asimakopoulos G.et al Vascular cell adhesion molecule‐1 and intercellular adhesion molecule‐1 serum level in patients with chest pain and normal coronary arteries (syndrome X). Clin Cardiol 200124301–304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Antoniades C, Tousoulis D, Tountas C.et al Vascular endothelium and inflammatory process, in patients with combined type 2 diabetes mellitus and coronary atherosclerosis: the effects of vitamin C. Diabet Med 200421552–558. [DOI] [PubMed] [Google Scholar]
- 5.Tentolouris C, Tousoulis D, Antoniades C.et al Endothelial function and proinflammatory cytokines in patients with ischemic heart disease and dilated cardiomyopathy. Int J Cardiol 200494301–305. [DOI] [PubMed] [Google Scholar]
- 6.Tentolouris C, Tousoulis D, Davies G.et al The impact of risk factors for atherosclerosis on the vasomotor effects of inhibition of nitric oxide synthesis in patients with normal angiograms. Cardiology 20009426–30. [DOI] [PubMed] [Google Scholar]
- 7.Kharbanda R K, Deanfield J E. Functions of the healthy endothelium. Coron Artery Dis 200112485–491. [DOI] [PubMed] [Google Scholar]
- 8.Behrendt D, Ganz P. Endothelial function: from vascular biology to clinical applications. Am J Cardiol 20029040L–8L. [DOI] [PubMed] [Google Scholar]
- 9.Celermajer D S, Sorensen K E, Bull C.et al Endothelium‐dependent dilation in the systemic arteries of asymptomatic subjects relates to coronary risk factors and their interaction. J Am Coll Cardiol 1994241468–1474. [DOI] [PubMed] [Google Scholar]
- 10.Tousoulis D, Antoniades C, Tentolouris C.et al Effects of combined administration of vitamins C and E on reactive hyperemia and inflammatory process in chronic smokers. Atherosclerosis 2003170261–267. [DOI] [PubMed] [Google Scholar]
- 11.Sundell J, Laine H, Luotolahti M.et al Obesity affects myocardial vasoreactivity and coronary flow response to insulin. Obes Res 200210617–624. [DOI] [PubMed] [Google Scholar]
- 12.Higashi Y, Sasaki S, Kurisu S.et al Regular aerobic exercise augments endothelium‐dependent vascular relaxation in normotensive as well as hypertensive subjects: role of endothelium‐derived nitric oxide. Circulation 19991001194–1202. [DOI] [PubMed] [Google Scholar]
- 13.Panagiotakos D B, Pitsavos C, Chrysohoou C.et al Impact of lifestyle habits on the prevalence of the metabolic syndrome among Greek adults from the ATTICA study. Am Heart J 2004147106–112. [DOI] [PubMed] [Google Scholar]
- 14.Prasad A, Zhu J, Halcox J P.et al Predisposition to atherosclerosis by infections: role of endothelial dysfunction. Circulation 2002106184–190. [DOI] [PubMed] [Google Scholar]
- 15.Charakida M, Donald A E, Terese M.et al Endothelial dysfunction in childhood infection. Circulation 20051111660–1665. [DOI] [PubMed] [Google Scholar]
- 16.Dhillon R, Clarkson P, Donald A E.et al Endothelial dysfunction late after Kawasaki disease. Circulation 1996942103–2106. [DOI] [PubMed] [Google Scholar]
- 17.Perticone F, Ceravolo R, Pujia A.et al Prognostic significance of endothelial dysfunction in hypertensive patients. Circulation 2001104191–196. [DOI] [PubMed] [Google Scholar]
- 18.Heitzer T, Schlinzig T, Krohn K.et al Endothelial dysfunction, oxidative stress, and risk of cardiovascular events in patients with coronary artery disease. Circulation 20011042673–2678. [DOI] [PubMed] [Google Scholar]
- 19.Ross R. Atherosclerosis: an inflammatory disease. N Engl J Med 1999340115–126. [DOI] [PubMed] [Google Scholar]
- 20.Blankenberg S, Barbaux S, Tiret L. Adhesion molecules and atherosclerosis. Atherosclerosis 2003170191–203. [DOI] [PubMed] [Google Scholar]
- 21.Jonasson L, Holm J, Skalli O.et al Expression of class II transplantation antigen on vascular smooth muscle cells in human atherosclerosis. J Clin Invest 198576125–131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Young J L, Libby P, Schonbeck U. Cytokines in the pathogenesis of atherosclerosis. Thromb Haemost 200288554–567. [PubMed] [Google Scholar]
- 23.Tanaka A, Shimada K, Sano T.et al Multiple plaque rupture and C‐reactive protein in acute myocardial infarction. J Am Coll Cardiol 2005451594–1599. [DOI] [PubMed] [Google Scholar]
- 24.Pasceri V, Cheng J S, Willerson J T.et al Modulation of C‐reactive protein‐mediated monocyte chemoattractant protein‐1 induction in human endothelial cells by anti‐atherosclerosis drugs. Circulation 20011032531–2534. [DOI] [PubMed] [Google Scholar]
- 25.Bhakdi S, Torzewski M, Klouche M.et al Complement and atherogenesis: binding of CRP to degraded, nonoxidized LDL enhances complement activation. Arterioscler Thromb Vasc Biol 1999192348–2354. [DOI] [PubMed] [Google Scholar]
- 26.Pasceri V, Willerson J T, Yeh E T. Direct proinflammatory effect of C‐reactive protein on human endothelial cells. Circulation 20001022165–2168. [DOI] [PubMed] [Google Scholar]
- 27.Fichtlscherer S, Rosenberger G, Walter D H.et al Elevated C‐reactive protein levels and impaired endothelial vasoreactivity in patients with coronary artery disease. Circulation 20001021000–1006. [DOI] [PubMed] [Google Scholar]
- 28.Madjid M, Awan I, Willerson J T.et al Leukocyte count and coronary heart disease: implications for risk assessment. J Am Coll Cardiol 2004441945–1956. [DOI] [PubMed] [Google Scholar]
- 29.De Gaetano G, Cerletti C, Evangelista V. Recent advances in platelet‐polymorphonuclear leukocyte interaction. Haemostasis 19992941–49. [DOI] [PubMed] [Google Scholar]
- 30.Harlan J M, Killen P D, Harker L A.et al Neutrophil‐mediated endothelial injury in vitro mechanisms of cell detachment. J Clin Invest 1981681394–1403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mehta J, Dinerman J, Mehta P.et al Neutrophil function in ischemic heart disease. Circulation 198979549–556. [DOI] [PubMed] [Google Scholar]
- 32.Zhang R, Brennan M L, Fu X.et al Association between myeloperoxidase levels and risk of coronary artery disease. JAMA 20012862136–2142. [DOI] [PubMed] [Google Scholar]
- 33.Podrez E A, Schmitt D, Hoff H F.et al Myeloperoxidase‐generated reactive nitrogen species convert LDL into an atherogenic form in vitro. J Clin Invest 19991031547–1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liuzzo G, Biasucci L M, Gallimore J R.et al The prognostic value of C‐reactive protein and serum amyloid a protein in severe unstable angina. N Engl J Med 1994331417–424. [DOI] [PubMed] [Google Scholar]
- 35.Ridker P M, Cushman M, Stampfer M J.et al Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N Engl J Med 1997336973–979. [DOI] [PubMed] [Google Scholar]
- 36.Ridker P M, Rifai N, Pfeffer M A.et al Inflammation, pravastatin, and the risk of coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and recurrent events (CARE) investigators. Circulation 199898839–844. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.