Abstract
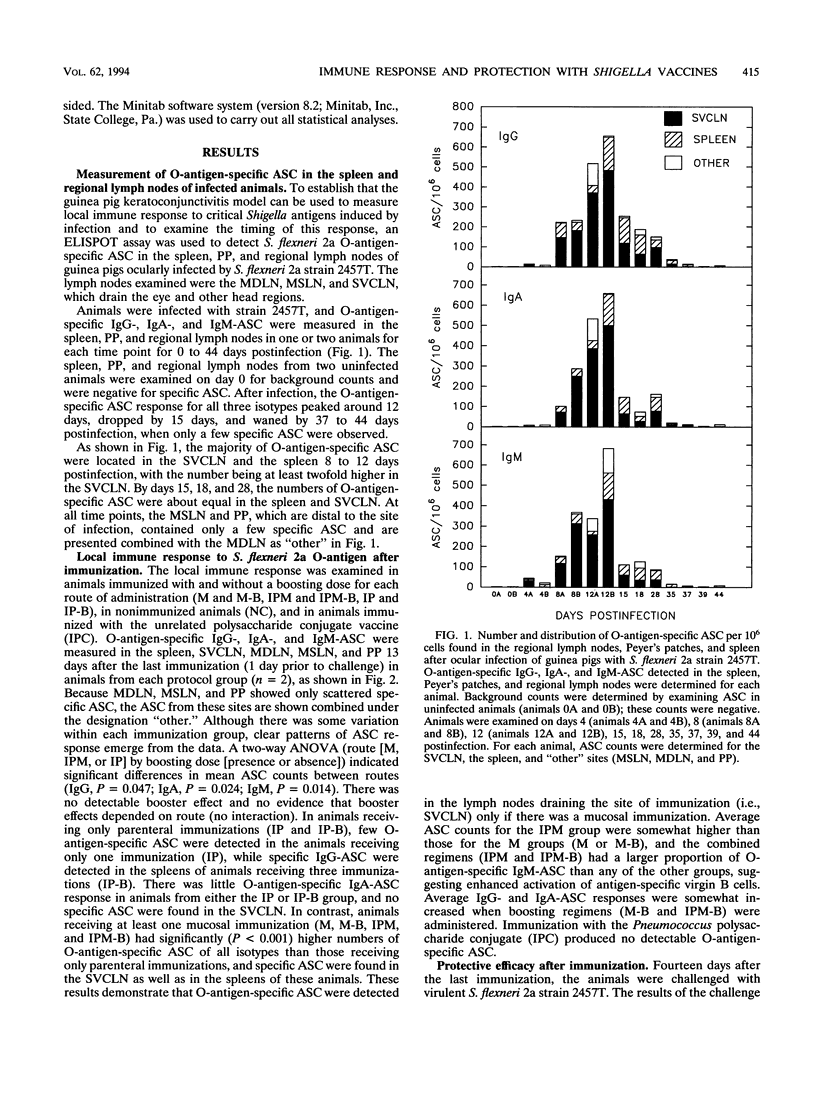
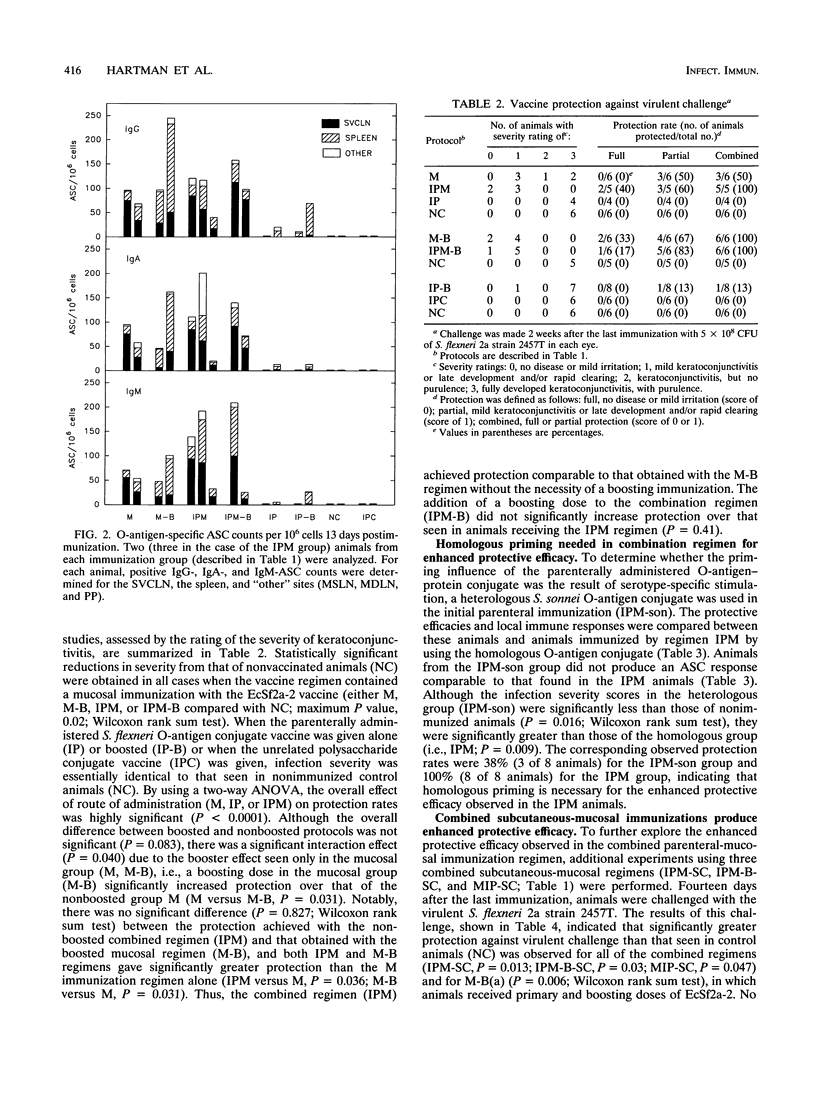
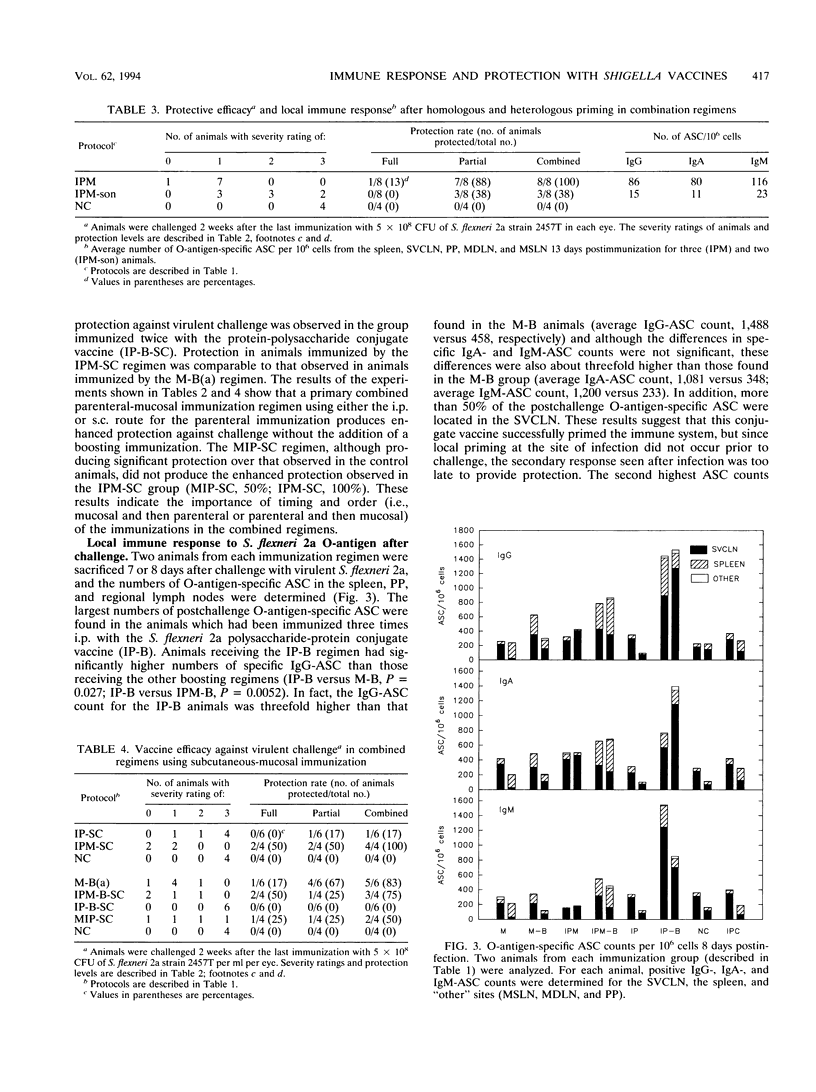
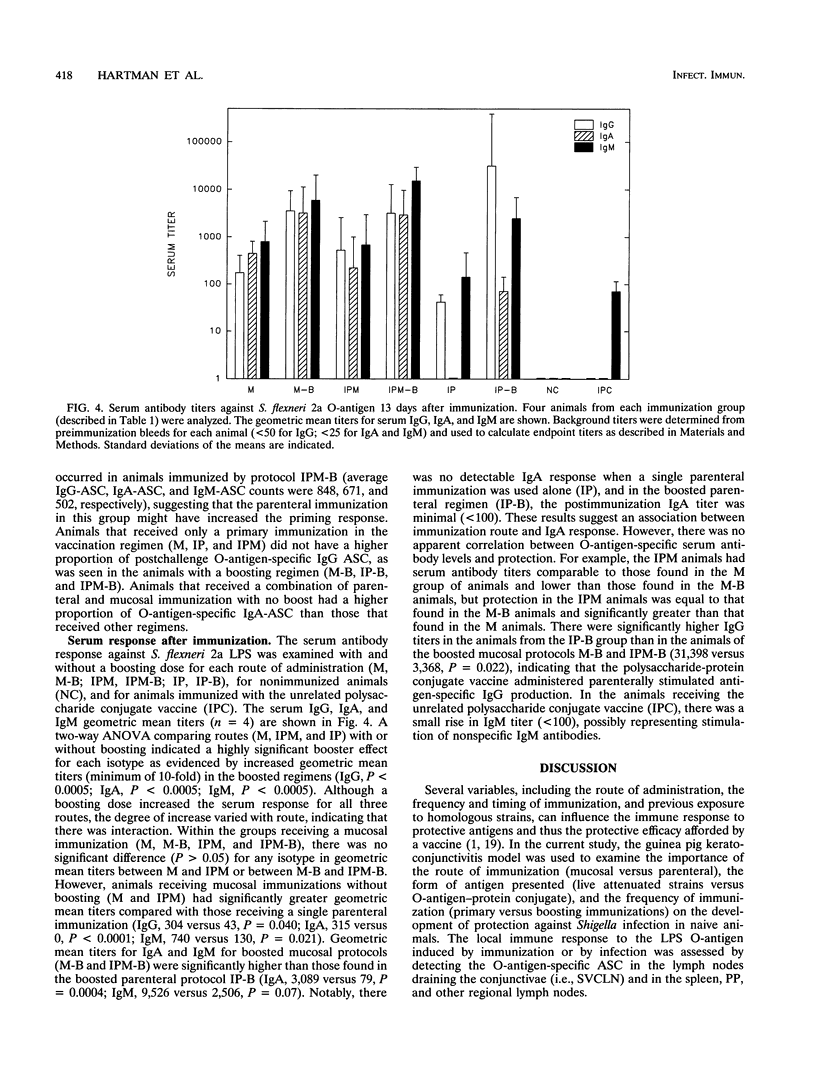
This study used the guinea pig keratoconjunctivitis model to examine the importance of route of administration (mucosal versus parenteral), frequency and timing of immunization (primary versus boosting immunization), and form of antigen given (live attenuated vaccine strain versus O-antigen-protein conjugate) on the production of protective immunity against Shigella infection. Since local immune response to the lipopolysaccharide (LPS) O-antigen of Shigella spp. is thought to be important for protection against disease, O-antigen-specific antibody-secreting cells (ASC) in the spleen and regional lymph nodes of immunized animals were measured by using an ELISPOT assay. Results indicated that protective efficacy was associated with a strong O-antigen-specific ASC response, particularly in the superficial ventral cervical lymph nodes draining the conjunctivae. In naive animals, a strong ASC response in the cervical lymph nodes and protection against challenge were detected only in animals that received a mucosal immunization. Protection in these animals was increased by a boosting mucosal immunization. While parenteral immunization alone with an O-antigen-protein conjugate vaccine did not protect naive animals against challenge, a combined parenteral-mucosal regimen elicited enhanced protection without the addition of a boosting immunization. Although O-antigen-specific serum immunoglobulin A titers were significantly higher in animals receiving a mucosal immunization, there was no apparent correlation between levels of serum antibody and protection against disease.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Black R. E., Levine M. M., Clements M. L., Losonsky G., Herrington D., Berman S., Formal S. B. Prevention of shigellosis by a Salmonella typhi-Shigella sonnei bivalent vaccine. J Infect Dis. 1987 Jun;155(6):1260–1265. doi: 10.1093/infdis/155.6.1260. [DOI] [PubMed] [Google Scholar]
- Cohen D., Green M. S., Block C., Rouach T., Ofek I. Serum antibodies to lipopolysaccharide and natural immunity to shigellosis in an Israeli military population. J Infect Dis. 1988 May;157(5):1068–1071. doi: 10.1093/infdis/157.5.1068. [DOI] [PubMed] [Google Scholar]
- Cohen D., Green M. S., Block C., Slepon R., Lerman Y. Natural immunity to shigellosis in two groups with different previous risks of exposure to Shigella is only partly expressed by serum antibodies to lipopolysaccharide. J Infect Dis. 1992 Apr;165(4):785–787. doi: 10.1093/infdis/165.4.785. [DOI] [PubMed] [Google Scholar]
- Czerkinsky C. C., Nilsson L. A., Nygren H., Ouchterlony O., Tarkowski A. A solid-phase enzyme-linked immunospot (ELISPOT) assay for enumeration of specific antibody-secreting cells. J Immunol Methods. 1983 Dec 16;65(1-2):109–121. doi: 10.1016/0022-1759(83)90308-3. [DOI] [PubMed] [Google Scholar]
- Czerkinsky C., Russell M. W., Lycke N., Lindblad M., Holmgren J. Oral administration of a streptococcal antigen coupled to cholera toxin B subunit evokes strong antibody responses in salivary glands and extramucosal tissues. Infect Immun. 1989 Apr;57(4):1072–1077. doi: 10.1128/iai.57.4.1072-1077.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DuPont H. L., Hornick R. B., Snyder M. J., Libonati J. P., Formal S. B., Gangarosa E. J. Immunity in shigellosis. II. Protection induced by oral live vaccine or primary infection. J Infect Dis. 1972 Jan;125(1):12–16. doi: 10.1093/infdis/125.1.12. [DOI] [PubMed] [Google Scholar]
- Formal S. B., Maenza R. M., Austin S., LaBrec E. H. Failure of parenteral vaccines to protect monkeys against experimental shigellosis. Proc Soc Exp Biol Med. 1967 Jun;125(2):347–349. doi: 10.3181/00379727-125-32087. [DOI] [PubMed] [Google Scholar]
- Formal S. B., Oaks E. V., Olsen R. E., Wingfield-Eggleston M., Snoy P. J., Cogan J. P. Effect of prior infection with virulent Shigella flexneri 2a on the resistance of monkeys to subsequent infection with Shigella sonnei. J Infect Dis. 1991 Sep;164(3):533–537. doi: 10.1093/infdis/164.3.533. [DOI] [PubMed] [Google Scholar]
- Forrest B. D. Identification of an intestinal immune response using peripheral blood lymphocytes. Lancet. 1988 Jan 16;1(8577):81–83. doi: 10.1016/s0140-6736(88)90284-x. [DOI] [PubMed] [Google Scholar]
- Forrest B. D., LaBrooy J. T., Dearlove C. E., Shearman D. J. Effect of parenteral immunization on the intestinal immune response to Salmonella typhi Ty21a. Infect Immun. 1992 Feb;60(2):465–471. doi: 10.1128/iai.60.2.465-471.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HIGGINS A. R., FLOYD T. M., KADER M. A. Studies in shigellosis. III. A controlled evaluation of a monovalent Shigella vaccine in a highly endemic environment. Am J Trop Med Hyg. 1955 Mar;4(2):281–288. [PubMed] [Google Scholar]
- Hamilton S. R., Yardley J. H., Brown G. D. Suppression of local intestinal immunoglobulin A immune response to cholera toxin by subcutaneous administration of cholera toxoids. Infect Immun. 1979 May;24(2):422–426. doi: 10.1128/iai.24.2.422-426.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartman A. B., Powell C. J., Schultz C. L., Oaks E. V., Eckels K. H. Small-animal model to measure efficacy and immunogenicity of Shigella vaccine strains. Infect Immun. 1991 Nov;59(11):4075–4083. doi: 10.1128/iai.59.11.4075-4083.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrington D. A., Van de Verg L., Formal S. B., Hale T. L., Tall B. D., Cryz S. J., Tramont E. C., Levine M. M. Studies in volunteers to evaluate candidate Shigella vaccines: further experience with a bivalent Salmonella typhi-Shigella sonnei vaccine and protection conferred by previous Shigella sonnei disease. Vaccine. 1990 Aug;8(4):353–357. doi: 10.1016/0264-410x(90)90094-3. [DOI] [PubMed] [Google Scholar]
- Holmgren J., Czerkinsky C., Lycke N., Svennerholm A. M. Mucosal immunity: implications for vaccine development. Immunobiology. 1992 Feb;184(2-3):157–179. doi: 10.1016/S0171-2985(11)80473-0. [DOI] [PubMed] [Google Scholar]
- Hone D., Hackett J. Vaccination against enteric bacterial diseases. Rev Infect Dis. 1989 Nov-Dec;11(6):853–877. doi: 10.1093/clinids/11.6.853. [DOI] [PubMed] [Google Scholar]
- Keren D. F., McDonald R. A., Carey J. L. Combined parenteral and oral immunization results in an enhanced mucosal immunoglobulin A response to Shigella flexneri. Infect Immun. 1988 Apr;56(4):910–915. doi: 10.1128/iai.56.4.910-915.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klipstein F. A., Engert R. F., Clements J. D. Arousal of mucosal secretory immunoglobulin A antitoxin in rats immunized with Escherichia coli heat-labile enterotoxin. Infect Immun. 1982 Sep;37(3):1086–1092. doi: 10.1128/iai.37.3.1086-1092.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kärnell A., Stocker B. A., Katakura S., Sweiha H., Reinholt F. P., Cam P. D., Trach D. D., Lindberg A. A. An auxotrophic live oral Shigella flexneri vaccine: development and testing. Rev Infect Dis. 1991 Mar-Apr;13 (Suppl 4):S357–S361. doi: 10.1093/clinids/13.supplement_4.s357. [DOI] [PubMed] [Google Scholar]
- Lindberg A. A., Kärnell A., Stocker B. A., Katakura S., Sweiha H., Reinholt F. P. Development of an auxotrophic oral live Shigella flexneri vaccine. Vaccine. 1988 Apr;6(2):146–150. doi: 10.1016/s0264-410x(88)80018-5. [DOI] [PubMed] [Google Scholar]
- Mel D. M., Arsić B. L., Nikolić B. D., Radovanić M. L. Studies on vaccination against bacillary dysentery. 4. Oral immunization with live monotypic and combined vaccines. Bull World Health Organ. 1968;39(3):375–380. [PMC free article] [PubMed] [Google Scholar]
- Newland J. W., Hale T. L., Formal S. B. Genotypic and phenotypic characterization of an aroD deletion-attenuated Escherichia coli K12-Shigella flexneri hybrid vaccine expressing S. flexneri 2a somatic antigen. Vaccine. 1992;10(11):766–776. doi: 10.1016/0264-410x(92)90512-i. [DOI] [PubMed] [Google Scholar]
- Pal S., Taylor H. R., Huneke R. B., Prendergast R. A., Whittum-Hudson J. A. Frequency of antigen-specific B cells during experimental ocular Chlamydia trachomatis infection. Infect Immun. 1992 Dec;60(12):5294–5297. doi: 10.1128/iai.60.12.5294-5297.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce N. F., Cray W. C., Jr, Sircar B. K. Induction of a mucosal antitoxin response and its role in immunity to experimental canine cholera. Infect Immun. 1978 Jul;21(1):185–193. doi: 10.1128/iai.21.1.185-193.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce N. F. Induction of optimal mucosal antibody responses: effects of age, immunization route(s), and dosing schedule in rats. Infect Immun. 1984 Jan;43(1):341–346. doi: 10.1128/iai.43.1.341-346.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce N. F., Koster F. T. Priming and suppression of the intestinal immune response to cholera toxoid/toxin by parenteral toxoid in rats. J Immunol. 1980 Jan;124(1):307–311. [PubMed] [Google Scholar]
- Quiding M., Nordström I., Kilander A., Andersson G., Hanson L. A., Holmgren J., Czerkinsky C. Intestinal immune responses in humans. Oral cholera vaccination induces strong intestinal antibody responses and interferon-gamma production and evokes local immunological memory. J Clin Invest. 1991 Jul;88(1):143–148. doi: 10.1172/JCI115270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robbins J. B., Chu C., Watson D. C., Szu S. C., Daniels E. M., Lowe C. U., Schneerson R. O-specific side-chain toxin-protein conjugates as parenteral vaccines for the prevention of shigellosis and related diseases. Rev Infect Dis. 1991 Mar-Apr;13 (Suppl 4):S362–S365. doi: 10.1093/clinids/13.supplement_4.s362. [DOI] [PubMed] [Google Scholar]
- Robbins J. B., Schneerson R. Polysaccharide-protein conjugates: a new generation of vaccines. J Infect Dis. 1990 May;161(5):821–832. doi: 10.1093/infdis/161.5.821. [DOI] [PubMed] [Google Scholar]
- SERENY B. Experimental keratoconjunctivitis shigellosa. Acta Microbiol Acad Sci Hung. 1957;4(4):367–376. [PubMed] [Google Scholar]
- Stoll B. J., Glass R. I., Huq M. I., Khan M. U., Banu H., Holt J. Epidemiologic and clinical features of patients infected with Shigella who attended a diarrheal disease hospital in Bangladesh. J Infect Dis. 1982 Aug;146(2):177–183. doi: 10.1093/infdis/146.2.177. [DOI] [PubMed] [Google Scholar]
- Svennerholm A. M., Hanson L. A., Holmgren J., Lindblad B. S., Nilsson B., Quereshi F. Different secretory immunoglobulin A antibody responses to cholera vaccination in Swedish and Pakistani women. Infect Immun. 1980 Nov;30(2):427–430. doi: 10.1128/iai.30.2.427-430.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svennerholm A. M., Holmgren J., Hanson L. A., Lindblad B. S., Quereshi F., Rahimtoola R. J. Boosting of secretory IgA antibody responses in man by parenteral cholera vaccination. Scand J Immunol. 1977;6(12):1345–1349. doi: 10.1111/j.1365-3083.1977.tb00376.x. [DOI] [PubMed] [Google Scholar]



