In this article we discuss thromboprophylaxis for patients with atrial fibrillation.
Valvular atrial fibrillation
Patients with atrial fibrillation and valvular heart disease (valvular atrial fibrillation) have a substantially greater risk of stroke and other thromboembolic events. Specifically, the presence of mitral valve stenosis has been shown to be a substantial risk for stroke and thromboembolism, with these events occurring in 9–20% of patients, up to 75% of whom have cerebral emboli.1,2 Indeed, patients with mitral stenosis in sinus rhythm who develop atrial fibrillation have a 3–7 times increased risk of thromboembolism.2,3 Owing to the risk of stroke and thromboembolism, it has been considered unethical to conduct placebo‐controlled trials of antithrombotic treatment in patients with mitral valve disease.
Non‐valvular atrial fibrillation
The presence of atrial fibrillation without any valve disease (non‐valvular atrial fibrillation) increases the risk of stroke and thromboembolism fivefold.4 Silent cerebral infarction is also common,5 and there is also a clustering of stroke events at the time of onset of the atrial fibrillation.6 In contrast with valvular atrial fibrillation, however, many large randomised trials in the past two decades have examined the value of antithrombotic treatment in non‐valvular atrial fibrillation.7
Nonetheless, anticoagulation remains generally underused in clinical practice.8,9,10,11,12 Despite the higher risk associated with stroke in elderly people, as well as the greatest benefit of anticoagulation, elderly people (>75 years) are a group of patients in whom there is suboptimal use of thromboprophylaxis for atrial fibrillation. The many reasons for poor uptake of anticoagulants include the view that major randomised trials were not representative of clinical practice because of tight inclusion criteria and bias towards younger patients,8 as well as fears that the level of anticoagulation control, therapeutic efficacy and low complication rates seen in randomised trials may not be matched in clinical practice.13 In addition, reanalyses of pooled data suggested that the benefits of anticoagulation may have been overestimated, with little absolute reduction in stroke rates, but increased bleeding risk in patients with lower risk of stroke.14,15 There is also a perceived increase in bleeding risk associated with comorbidity, interactions with concomitant drug treatments and bleeding in elderly patients.16 Suboptimal use of thromboprophylaxis may also be due to patient factors, where some patients choose the option of informed dissent and decline an effective treatment option.17
Several studies over the past few years have shown that stroke and major haemorrhage rates after anticoagulation in clinical practice are comparable to data from randomised controlled studies.18,19,20 Reassuringly, more recent studies suggest that the intake of anticoagulants for prevention of stroke in patients with atrial fibrillation may have improved,20,21,22,23 with the reported intake in eligible patients being as high as 65–80% according to some studies.20,21,22 This optimism has been tempered by other recent observations, which show that only 56.5% of patients at very high risk of stroke in the community are taking anticoagulants,23 and a large proportion of eligible patients with atrial fibrillation in hospitals remain untreated.12 This suggests that many patients who might benefit from anticoagulation still do not receive it, especially if they are old, female, or have multiple comorbidities.23
Permanent atrial fibrillation
The risk of stroke in patients with chronic atrial fibrillation can be reduced substantially and consistently by thromboprophylaxis. Adjusted‐dose warfarin (box 1) reduces the relative risk of ischaemic stroke or systemic embolism by nearly two‐thirds compared with placebo.24 Although this benefit was seen for both primary and secondary prevention, the absolute risk reduction for secondary events was markedly greater than for primary events.24 In addition, anticoagulation is safe in randomised controlled trials, particularly with international normalised ratio targets of <3.25,26 For example, the pooled analysis of the first five primary prevention trials reported an annual rate of major bleeding of 1% in control patients compared with 1.3% in warfarin‐treated patients, which included an annual intracranial haemorrhage rate of 0.1% in controls and 0.3% in warfarin users.27 The major predictors of bleeding risk in clinical trials were advancing age and international normalised ratio levels >328 but later community‐based studies have shown a lack of association between advancing age and bleeding risk after adjusting for sex and comorbidities.29
A recent Cochrane systematic review suggests that in atrial fibrillation, compared with placebo, aspirin reduces stroke risk modestly, by about 25%,30 although this is largely driven by results from one clinical trial, the Stroke Prevention in Atrial Fibrillation (SPAF‐1) Trial.31 The latter was composed of two separately randomised cohorts, one consisting of individuals who could not be randomised to warfarin (aspirin v placebo), and one for individuals who could be randomised to warfarin (in this trial there was also a warfarin arm); in the first cohort, the RRR afforded by aspirin was 94% (p<0.001), while in the second cohort the comparable RRR was 8% (p = 0.75)—the pooled analysis of events in these two cohorts (with the inconsistency between the two groups) gives the 42%% risk reduction with aspirin for the whole SPAF‐1 trial (p = 0.02).31 Of note, the stroke risk reduction with aspirin in atrial fibrillation is broadly similar to the reduction in vascular events (22%) for antiplatelet treatment in high‐risk patients with vascular disease versus controls, with no differences between different aspirin doses.32 However, higher aspirin doses may be associated with more adverse effects. Atrial fibrillation commonly coexists with vascular disease, and the benefits of aspirin in atrial fibrillation may simply relate to the effect on vascular disease rather than on thrombogenesis in atrial fibrillation.32 This is supported by the thrombi associated with atrial fibrillation being fibrin rich (“red clot”) and more related to coagulation abnormalities. In view of the controversy over trial evidence in relation to the pathophysiological data, the Guideline Development Group recommended the use of aspirin at a dose of 75–300 mg/day if used as thromboprophylaxis in low‐risk subjects.
Box 1: Recommendations for permanent and paroxysmal atrial fibrillation
In patients with permanent atrial fibrillation, a risk–benefit assessment should be carried out and discussed with the patients to inform them about the decision whether or not to give antithrombotic treatment.
-
In patients with permanent atrial fibrillation to whom antithrombotics are given to prevent strokes or thromboembolism:
-
-
adjusted‐dose warfarin should be given as the most effective treatment
-
-
adjusted‐dose warfarin should reach a target international normalised ratio of 2.5 (range 2–3)
-
-
where warfarin is not appropriate, aspirin should be given at a dose of 75–300 mg/day
-
-
where warfarin is appropriate, aspirin should not be coadministered with warfarin purely as thromboprophylaxis, as it provides no additional benefit
-
-
Decisions on the need for antithrombotic treatment in patients with paroxysmal atrial fibrillation should not be based on the frequency or duration of paroxysms (symptomatic or asymptomatic) but on appropriate risk stratification, as for permanent atrial fibrillation.
Comparisons between aspirin and warfarin show that reduction in risk of stroke using aspirin is considerably less than that seen using warfarin in high‐risk patients with atrial fibrillation. Adjusted‐dose warfarin reduced the risk of stroke nearly twofold compared with aspirin for both primary and secondary prevention.24 This was associated with a non‐significant trend to increased risk of haemorrhagic stroke with warfarin compared with aspirin.
Evidence suggesting that combinations of warfarin in any dosage regimen with aspirin is superior to adjusted‐dose warfarin for stroke prevention are not available ; indeed, such combinations increase the risk of bleeding.33 The use of low adjusted‐dose anticoagulant with acenocoumarol in combination with the antiplatelet agent triflusal was more effective than conventional adjusted‐dose anticoagulant with acenocoumarol alone for the prevention of stroke.34 However, trifusal is not used in the UK, and previous studies on warfarin and aspirin combination treatment have shown it to be less effective or to have more adverse effects than treatment with warfarin alone. Thus, where warfarin is appropriate, aspirin should not be given along with warfarin purely as thromboprophylaxis, as it provides no additional benefit. The appropriateness, duration and safety (eg, risk of bleeding) of the concomitant administration of warfarin with aspirin is considered a matter for clinical judgement.7
Persistent and paroxsymal atrial fibrillation
Studies have shown that paroxysmal atrial fibrillation may progress to more sustained forms of atrial fibrillation and carry a similar risk of stroke in patients with persistent or permanent atrial fibrillation and, thus, paroxysmal atrial fibrillation should be managed similarly to permanent atrial fibrillation.35 The decisions on the need for antithrombotic treatment in patients with paroxysmal atrial fibrillation should not be based on the frequency or duration of paroxysms (symptomatic or asymptomatic) but on appropriate risk stratification, as for patients with permanent atrial fibrillation.
Anticoagulation before cardioversion in persistent atrial fibrillation reduces the frequency of thromboembolic complications, and anticoagulation is recommended pre‐cardioversion (box 2).36 Even in patients after cardioversion, thromboembolic events occur after the anticoagulation is stopped or is subtherapeutic.37 Thus, in patients with persistent atrial fibrillation, anticoagulation should be considered long term in those who are at risk of recurrence of atrial fibrillation or in those who have stroke risk factors.
Box 2: Recommendations for patients with persistent atrial fibrillation
Before cardioversion, patients should maintain therapeutic anticoagulation with warfarin (international normalised ratio (INR) 2.5, range 2–3) for a minimum of 3 weeks
After successful cardioversion, patients should continue therapeutic anticoagulation with warfarin (INR 2.5, range 2–3) for a minimum of 4 weeks
-
In patients with atrial fibrillation in whom cardioversion cannot be postponed for 3 weeks:
-
-
heparin should be given and the cardioversion performed
-
-
warfarin should then be given for a minimum of 4 weeks after cardioversion
-
-
Anticoagulation should be continued for the long term in patients with atrial fibrillation who have undergone cardioversion where there is a high risk of atrial fibrillation recurrence or where it is recommended by the Stroke Risk Stratification Algorithm. (Factors indicating a high risk of recurrence of atrial fibrillation include a history of failed attempts at cardioversion, structural heart disease (mitral valve disease, left ventricular dysfunction or an enlarged left atrium), a prolonged history of atrial fibrillation (>12 months) and previous recurrence of atrial fibrillation)
In patients undergoing cardioversion and who have an atrial fibrillation of confirmed duration of <48 h, anticoagulation after successful restoration of sinus rhythm is not required
Patients with atrial flutter should be given antithrombotic treatment in the same manner as those with atrial fibrillation
Asymptomatic and newly diagnosed atrial fibrillation
Lack of symptoms may not confer lower thromboembolic risk; asymptomatic patients shared the same risks as those with symptoms when adjusted for baseline clinical parameters in the Atrial Fibrillation Follow‐up Investigation of Rhythm Management Study.38 Thus, patients with asymptomatic atrial fibrillation should receive thromboprophylaxis, as for symptomatic atrial fibrillation.
In patients with newly diagnosed atrial fibrillation for whom antithrombotic treatment is indicated, such treatment should be initiated with minimal delay after the appropriate management of comorbidities.
Box 3: Recommendations for antithrombotic treatment in acute‐onset atrial fibrillation
-
In patients with acute atrial fibrillation who are receiving no treatment, or are receiving subtherapeutic anticoagulation treatment:
-
-
in the absence of contraindications, heparin should be started at initial presentation
-
-
heparin should be continued until full assessment and appropriate antithrombotics started, based on risk stratification
-
-
-
In patients with a confirmed diagnosis of acute atrial fibrillation (<48 h after onset), oral anticoagulants should be administered if:
-
-
stable sinus rhythm is not successfully restored within the same 48 h period following onset of acute atrial fibrillation, or
-
-
there are factors indicating a high risk of atrial fibrillation recurrence. (Factors indicating a high risk of recurrence of atrial fibrillation include a history of failed attempts at cardioversion, structural heart disease (mitral valve disease, left ventricular dysfunction or an enlarged left atrium), a prolonged history of atrial fibrillation (>12 months) and previous recurrences of atrial fibrillation)
-
-
it is recommended by the Stroke Risk Stratification Algorithm
-
-
In patients with acute atrial fibrillation in whom there is uncertainty over the precise onset, oral anticoagulation should be used as for persistent atrial fibrillation
In cases of acute atrial fibrillation where the patient is haemodynamically unstable any emergency intervention should be performed as soon as possible and the initiation of anticoagulation should not delay any emergency intervention
Acute atrial fibrillation
In many instances, acute atrial fibrillation may be self‐limiting, especially if associated with infective or metabolic disturbances, and has a low potential for recurrence.6 Although clinical studies have shown that there is low risk of thromboembolism in most patients with atrial fibrillation of <48 h duration,39 transoesophageal echocardiography has shown the presence of cardiac thromboembolism in 15% of patients with atrial fibrillation of <3 days' duration,40 suggesting that there is a possibility of increased stroke risk even in these patients. However, there are no randomised trials that have specifically considered the issue of acute‐onset atrial fibrillation. The majority consensus is that cardioversion can be safely carried out without the need for oral anticoagulation if atrial fibrillation has been present for <48 h.
Antithrombotic treatment for atrial fibrillation in patients with acute stroke
Evidence supporting the use of anticoagulation in patients with acute cardioembolic stroke (boxes 3, 4) is based on limited data. Patients within the first 2 weeks of having had a cardioembolic stroke are at greatest risk of having stroke recurrence because of further embolisation, and intracranial haemorrhage or haemorrhagic transformation of the infarct. It is generally considered safe to withhold anticoagulation for 2 weeks to allow the injury to settle before anticoagulation, in accordance with the guidelines for secondary prevention.27 This delay in starting anticoagulation is particularly important for patients with large infarcts, extensive small‐vessel disease or uncontrolled hypertension—those who are at the greatest risk of intracranial bleeding. Although atrial fibrillation may not be responsible for all strokes occurring in atrial fibrillation, prevention of secondary stroke is warranted and all patients with atrial fibrillation who have had an ischaemic stroke will benefit from suitable antithrombotic treatment after appropriate risk assessment.
Box 4: Recommendations for anticoagulation in acute stroke patients
In all patients with acute stroke or transient ischaemic attack (TIA), uncontrolled hypertension should be appropriately managed before antithrombotic treatment is started.
In patients with atrial fibrillation and an acute stroke:
Imaging (computed tomographic (CT) scan or magnetic resonance imaging (MRI)) should be performed to exclude cerebral haemorrhage
In the absence of haemorrhage, anticoagulation should begin after 2 weeks
In the presence of haemorrhage, anticoagulation should not be given
In the presence of a large cerebral infarction, the initiation of anticoagulation should be delayed
In patients with atrial fibrillation and an acute TIA:
Imaging (CT scan or MRI) should be performed to exclude recent cerebral infarction or haemorrhage
In the absence of cerebral infarction or haemorrhage, anticoagulation treatment should begin as soon as possible
Assessment and stratification for stroke risk
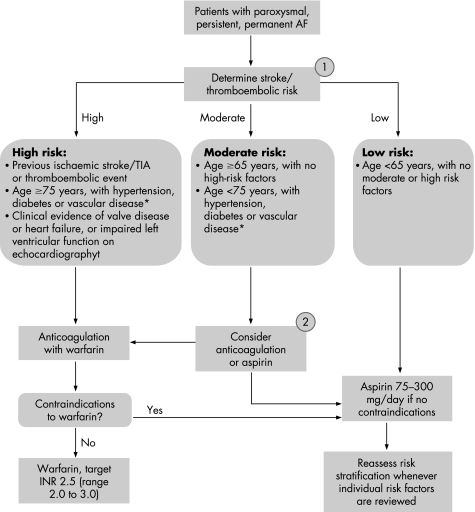
The overall annual risk of stroke in patients with atrial fibrillation varies from 2% to 12%, depending on age, sex, presence of other cardiovascular comorbidities, structural changes to the heart and evidence of previous thromboembolic events.7 The evidence for benefits of adjusted‐dose warfarin over placebo or aspirin is the strongest for those at highest risk and may be equivocal for those who have a lower annual risk for stroke.14,15 Hence, various risk stratification models of differing complexity based on clinical or echocardiographic criteria have been developed to help guide clinical decisions.7 Many of these schema have been derived from regression analyses of pooled data from the original trials on anticoagulation and, despite considerable overlap between different stratification schemes, there is often disagreement about the need for anticoagulation, especially for patients at moderate risk of stroke.11,41 Figure 1 shows the risk stratification algorithm given by the National Institute for Health and Clinical Excellence (NICE) to aid clinical decisions on thromboprophylaxis. The NICE clinical risk stratification defines subjects into low, moderate and high risk categories, and this clinical risk stratification scheme has been shown to be broadly similar to the CHADS2 scheme for predicting stroke and vascular event rates.42
Figure 1 Stroke risk stratification algorithm. (1) The risk factors are not mutually exclusive, and are additive to each other in producing a composite risk. Since the incidence of stroke and thromboembolic events in patients with thyrotoxicosis appears similar to other aetiologies of atrial fibrillation (AF), antithrombotic treatments should be chosen based on the presence of validated stroke risk factors. (2) Owing to lack of sufficient clear‐cut evidence, treatment may be decided on an individual basis, and the physician must balance the risk and benefits of warfarin versus aspirin. As stroke risk factors are cumulative, warfarin may, for example, be used in the presence of two or more moderate stroke risk factors. Referral and echocardiography may help in cases of uncertainty. *Coronary artery disease or peripheral artery disease. †An echocardiogram is not needed for routine assessment, but refines clinical risk stratification in the case of moderate or severe left ventricular dysfunction and valve disease. INR, international normalised ratio; TIA, transient ischaemic attack.
Many studies have also shown that despite the increasing availability of sophisticated investigations to quantify risk for stroke, clinical criteria remain a robust method for identifying those at greatest risk and most likely to benefit from anticoagulation.43 In all patients with atrial fibrillation, a risk–benefit assessment should be carried out and discussed with the patients to inform them about the decision whether or not to give antithrombotic treatment.
Bleeding risk
Major risk factors for intracranial haemorrhage are advanced patient age, raised blood pressure, intensity of anticoagulation and previous cerebral ischaemia.44 Combining antiplatelet agents with anticoagulation and the combined use of aspirin and clopidogrel appear to increase the risk for intracerebral haemorrhage, but modest lowering of the blood pressure halves the frequency of intracerebral haemorrhage during antiplatelet treatment.44 Evidence suggests that the combined use of warfarin with antiplatelet agents such as aspirin and clopidogrel markedly increases the risk of bleeding beyond that associated with the single use of any one of these agents.45 This raises a particular dilemma in those patients with both atrial fibrillation and coronary or peripheral artery disease who are often coprescribed warfarin and antiplatelet drugs. With the increasing use of drug‐eluting stents, for which 6–12 months of aspirin–clopidogrel use has recently been recommended,46 information on how best to manage patients who have both atrial fibrillation and a drug‐eluting stent who also require anticoagulation is currently lacking. Indeed, there is a lack of any coordinated strategy in the prevention of thrombotic or thromboembolic events in patients with AF and a recent percutaneous coronary intervention, especially in the setting of acute coronary syndromes, although suggested management guidelines have been published.47
Thus, the guidelines recommend that both antithrombotic benefits and the potential bleeding risks of long‐term anticoagulation should be explained to and discussed with the patient. Particular attention should be paid to the following groups of patients:
Those aged >75 years
Those taking antiplatelet drugs (such as aspirin or clopidogrel) or non‐steroidal anti‐inflammatory drugs
Those receiving multiple drug treatments (polypharmacy)
Those with uncontrolled hypertension
Those with a history of bleeding (eg, peptic ulcer or cerebral haemorrhage)
Those with a history of poorly controlled anticoagulation treatment.
Many of the risk factors for bleeding (eg, age >75 years, hypertension) were also risk factors for stroke. Thus, it may be appropriate to undertake an assessment of bleeding risk as part of the clinical assessment of patients before starting antithrombotic treatment. The utility of bleeding risk stratification schemes for anticoagulated patients with atrial fibrillation also requires further prospective data.48
Box 5: Recommendations for self‐monitoring of anticoagulation
In patients with atrial fibrillation who require long‐term anticoagulation, self‐monitoring should be considered if preferred by the patient and if the following criteria are met:
The patient is both physically and cognitively able to perform the self‐monitoring test, or in cases in which the patient is not physically or cognitively able to perform self‐monitoring, a designated carer is able to do so
An adequate supportive educational programme is in place to train patients and/or carers
The patient's ability to self‐manage is regularly reviewed
The equipment for self‐monitoring is regularly checked via a quality control programme
Anticoagulation Self‐monitoring
The evidence suggests that patient self‐monitoring of oral anticoagulation is more effective in terms of patient satisfaction than supervised management.49 The use of self‐monitoring needs to be balanced between patient preference and the ability of local services to provide support (eg, patient education programmes). Guidelines regarding patient self‐monitoring of oral anticoagulation have been published.50 These recommend that patients undertaking anticoagulation self‐monitoring be trained by a competent healthcare professional and remain in contact with a named clinician. They also highlight the need for self‐monitoring devices that have been adequately quality assured (box 5).
Footnotes
Competing interests: None.
References
- 1.Fleming H A, Bailey S M. Mitral valve disease, systemic embolism and anticoagulants. Postgrad Med J 197147599–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Salem D N, Stein P D, Al‐Ahmad A.et al Antithrombotic therapy in valvular heart disease—native and prosthetic: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004126(Suppl)S457–S482. [DOI] [PubMed] [Google Scholar]
- 3.Szekely P. Systemic embolism and anticoagulant prophylaxis in rheumatic heart disease. BMJ 196453921209–1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wolf P A, Abbott R D, Kannel W B. Atrial fibrillation as an independent risk factor for stroke: the Framingham study. Stroke 199122983–988. [DOI] [PubMed] [Google Scholar]
- 5.Petersen P, Madsen E B, Brun B.et al Silent cerebral infarction in chronic atrial fibrillation. Stroke 1987181098–1100. [DOI] [PubMed] [Google Scholar]
- 6.Wolf P A, Kannel W B, McGee D L.et al Duration of atrial fibrillation and imminence of stroke: the Framingham study. Stroke 198314664–667. [DOI] [PubMed] [Google Scholar]
- 7.Lip G Y, Boos C J. Antithrombotic treatment in atrial fibrillation. Heart 200692155–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sudlow M, Thomson R, Thwaites B.et al Prevalence of atrial fibrillation and eligibility for anticoagulants in the community. Lancet 19983521167–1171. [DOI] [PubMed] [Google Scholar]
- 9.Brass L M, Krumholz H M, Scinto J M.et al Warfarin use among patients with atrial fibrillation. Stroke 1997282382–2389. [DOI] [PubMed] [Google Scholar]
- 10.Kalra L, Perez I, Melbourn A. Risk assessment and anticoagulation for primary stroke prevention in atrial fibrillation. Stroke 1999301218–1222. [DOI] [PubMed] [Google Scholar]
- 11.Perez I, Melbourn A, Kalra L. Use of antithrombotic measures for stroke prevention in atrial fibrillation. Heart 199982570–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Waldo A L, Becker R C, Tapson V F.et al Hospitalized patients with atrial fibrillation and a high risk of stroke are not being provided with adequate anticoagulation. J Am Coll Cardiol 2005461729–1736. [DOI] [PubMed] [Google Scholar]
- 13.Gottlieb L K, Salem S S. Anticoagulation in atrial fibrillation: does effectiveness in clinical trials translate into effective practice? Arch Intern Med 19941541945–1953. [DOI] [PubMed] [Google Scholar]
- 14.Green C J, Hadorn D C, Bassett K.et al Anticoagulation in chronic non‐valvular atrial fibrillation: a critical appraisal and meta‐analysis. Can J Cardiol 199713811–815. [PubMed] [Google Scholar]
- 15.Taylor F C, Cohen H, Ebrahim S. Systematic review of long term anticoagulation or antiplatelet treatment in patients with non‐rheumatic atrial fibrillation. BMJ 2001322321–326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lane D, Lip G Y H. Anti‐thrombotic therapy for atrial fibrillation and patients' preferences for treatment. Age Ageing 2005341–3. [DOI] [PubMed] [Google Scholar]
- 17.Howitt A, Armstrong D. Implementing evidence based medicine in general practice: audit and qualitative study of antithrombotic treatment for atrial fibrillation. BMJ 19993181324–1327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kalra L, Yu G, Perez I.et al A prospective cohort study of anticoagulation for stroke prevention in atrial fibrillation: does trial efficacy translate into clinical effectiveness? BMJ 20003201236–1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Evans A, Kalra L. Are results of RCT on anticoagulation in AF generalisable to clinical practice. Arch Intern Med 20011611443–1447. [DOI] [PubMed] [Google Scholar]
- 20.Go A S, Hylek E M, Chang Y.et al Anticoagulation therapy for stroke prevention in atrial fibrillation: how well do randomized trials translate into clinical practice? JAMA 20032902685–2692. [DOI] [PubMed] [Google Scholar]
- 21.Bradley B C, Perdue K S, Tisdel K A.et al Frequency of anticoagulation for atrial fibrillation and reasons for its non‐use at a Veterans Affairs medical center. Am J Cardiol 200085568–572. [DOI] [PubMed] [Google Scholar]
- 22.Weisbord S D, Whittle J, Brooks R C. Is warfarin really underused in patients with atrial fibrillation? J Gen Intern Med 200116743–749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dewilde S, Carey I M, Emmas C.et al Trends in the prevalence of diagnosed atrial fibrillation, its treatment with anticoagulation, and predictors of such treatment in UK primary care. Heart 2005 [DOI] [PMC free article] [PubMed]
- 24.Lip G Y H, Edwards S J. Stroke prevention with aspirin, warfarin and ximelagatran in patients with non‐valvular atrial fibrillation: a systematic review and meta‐analysis. Thromb Res 2006118321–323. [DOI] [PubMed] [Google Scholar]
- 25.Atrial Fibrillation Investigators Risk factors for stroke and efficacy of antithrombotic treatment in atrial fibrillation: analysis of pooled data from five randomised controlled studies. Arch Intern Med 19941541449–1457. [PubMed] [Google Scholar]
- 26.Stroke Prevention in Atrial Fibrillation Investigators Bleeding during antithrombotic therapy in patients with atrial fibrillation. Arch Intern Med 1996156409–416. [PubMed] [Google Scholar]
- 27.Atrial Fibrillation Investigators Risk factors for stroke and efficacy of antithrombotic therapy in atrial fibrillation: analysis of pooled data from five randomized controlled trials. Arch Intern Med 19941541449–1457. [PubMed] [Google Scholar]
- 28.Hylek E M, Singer D E. Risk factors for intracranial hemorrhage in outpatients taking warfarin. Ann Intern Med 1994120897–902. [DOI] [PubMed] [Google Scholar]
- 29.Sam C, Massaro J M, D'Agostino R B., Sret al Warfarin and aspirin use and the predictors of major bleeding complications in atrial fibrillation (the Framingham Heart Study). Am J Cardiol 200494947–951. [DOI] [PubMed] [Google Scholar]
- 30.Aguilar M, Hart R. Antiplatelet therapy for preventing stroke in patients with non‐valvular atrial fibrillation and no previous history of stroke or transient ischemic attacks. Cochrane Database Syst Rev 200519CD001925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stroke Prevention in Atrial Fibrillation Investigators A differential effect of aspirin in prevention of stroke on atrial fibrillation. J Stroke Cerebrovasc Dis 19933181–188. [DOI] [PubMed] [Google Scholar]
- 32.Antithrombotic Trialists' Collaboration Collaborative metaanalysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ 200232471–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.DiMarco J P, Flaker G, Waldo A L.et al Factors affecting bleeding risk during anticoagulant therapy in patients with atrial fibrillation: observations from the Atrial Fibrillation Follow‐up Investigation of Rhythm Management (AFFIRM) Study. Am Heart J 2005149650–656. [DOI] [PubMed] [Google Scholar]
- 34.Perez‐Gomez F, Alegria E, Berjon J.et al Comparative effects of antiplatelet, anticoagulant, or combined therapy in patients with valvular and nonvalvular atrial fibrillation: a randomized multicenter study. J Am Coll Cardiol 2004441557–1566. [DOI] [PubMed] [Google Scholar]
- 35.Hart R G, Pearce L A, Rothbart R M.et al Stroke with intermittent atrial fibrillation: incidence and predictors during aspirin therapy. Stroke Prevention in Atrial Fibrillation Investigators. J Am Coll Cardiol 200035183–187. [DOI] [PubMed] [Google Scholar]
- 36.Gallagher M M, Hennessy B J, Edvardsson N.et al Embolic complications of direct current cardioversion of atrial arrhythmias: association with low intensity of anticoagulation at the time of cardioversion. J Am Coll Cardiol 200240926–933. [DOI] [PubMed] [Google Scholar]
- 37.Sherman D G, Kim S G, Boop B S.et al Occurrence and characteristics of stroke events in the Atrial Fibrillation Follow‐up Investigation of Sinus Rhythm Management (AFFIRM) Study. Arch Intern Med 20051651185–1191. [DOI] [PubMed] [Google Scholar]
- 38.Flaker G C, Belew K, Beckman K.et al Asymptomatic atrial fibrillation: demographic features and prognostic information from the atrial fibrillation follow‐up investigation of rhythm management (AFFIRM) Study. Am Heart J 2005149657–663. [DOI] [PubMed] [Google Scholar]
- 39.Weigner M J, Caulfield T A, Danias P G.et al Risk for clinical thromboembolism associated with conversion to sinus rhythm in patients with atrial fibrillation lasting less than 48 hours. Ann Intern Med 1997126615–620. [DOI] [PubMed] [Google Scholar]
- 40.Stoddard M F, Dawkins P R, Prince C R.et al Transesophageal echocardiographic guidance of cardioversion in patients with atrial fibrillation. Am Heart J 19951291204–1215. [DOI] [PubMed] [Google Scholar]
- 41.Rockson S G, Albers G W. Comparing the guidelines: anticoagulation therapy to optimize stroke prevention in patients with atrial fibrillation. J Am Coll Cardiol 200443929–935. [DOI] [PubMed] [Google Scholar]
- 42.Lip G Y H, Lane D, van Walraven C.et al The additive role of plasma von Willebrand factor levels to clinical factors for risk stratification in patients with atrial fibrillation. Stroke 2006372294–2300. [DOI] [PubMed] [Google Scholar]
- 43.Evans A, Perez I, Yu G.et al Secondary stroke prevention in atrial fibrillation: lessons from clinical practice. Stroke 2000322106–2111. [DOI] [PubMed] [Google Scholar]
- 44.Hart R G, Tonarelli S B, Pearce L A. Avoiding central nervous system bleeding during antithrombotic therapy: recent data and ideas. Stroke 2005361588–1593. [DOI] [PubMed] [Google Scholar]
- 45.Shireman T I, Howard P A, Kresowik T F.et al Combined anticoagulant‐antiplatelet use and major bleeding events in elderly atrial fibrillation patients. Stroke 2004352362–2367. [DOI] [PubMed] [Google Scholar]
- 46.Silber S, Albertsson P, Aviles F F.et al Guidelines for percutaneous coronary interventions: the task force for percutaneous coronary interventions of the European Society of Cardiology. Eur Heart J 200526804–847. [DOI] [PubMed] [Google Scholar]
- 47.Lip G Y H, Karpha M. Anticoagulant and antiplatelet therapy use in patients with atrial fibrillation undergoing percutaneous coronary intervention: the need for consensus and a management guideline. Chest 20061301823–1827. [DOI] [PubMed] [Google Scholar]
- 48.Kakar P, Lane D, Lip G Y H. Bleeding risk stratification models in deciding on anticoagulation in patients with atrial fibrillation: a useful complement to stroke risk stratification scheme. Chest (in press) [DOI] [PubMed]
- 49.Sawicki P T. A structured teaching and self‐management program for patients receiving oral anticoagulation: a randomized controlled trial. Working Group for the Study of Patient Self‐Management of Oral Anticoagulation. JAMA 1999281145–150. [DOI] [PubMed] [Google Scholar]
- 50.Fitzmaurice D A, Machin S J, British Society of Haematology Task Force for Haemostasis and Thrombosis Recommendations for patients undertaking self management of oral anticoagulation. BMJ 2001323985–989. [DOI] [PMC free article] [PubMed] [Google Scholar]