Abstract
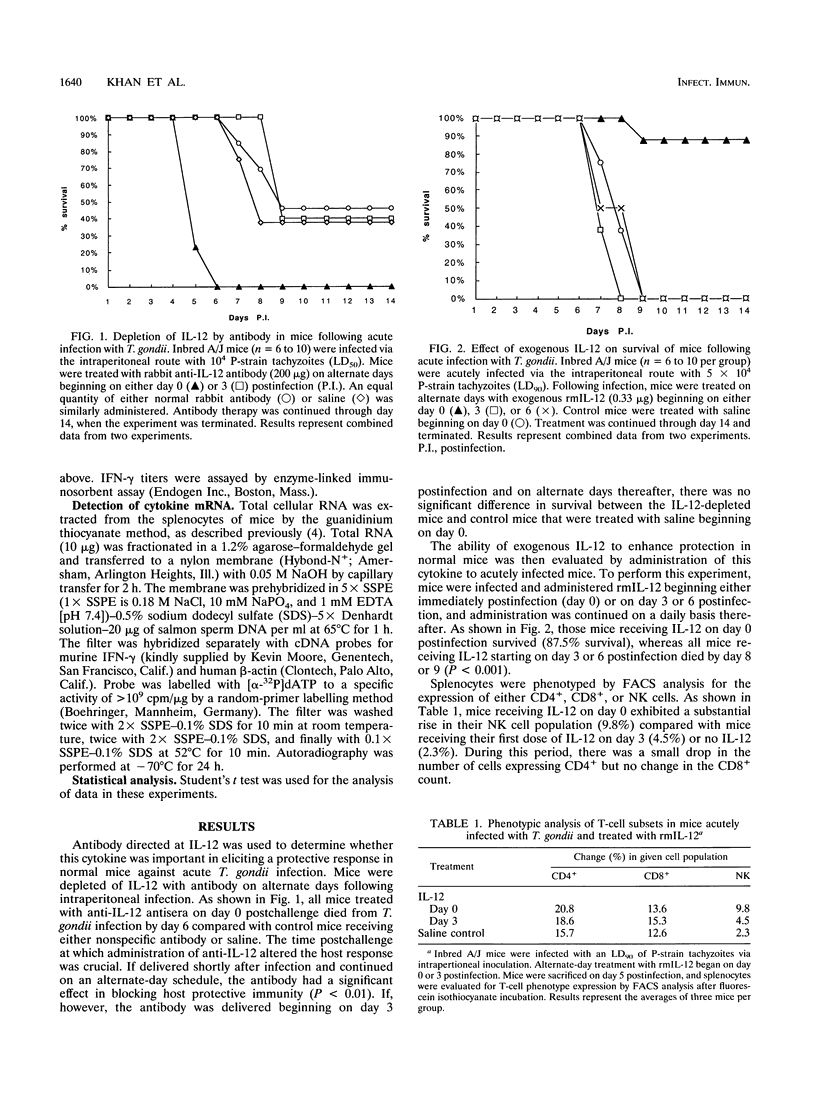
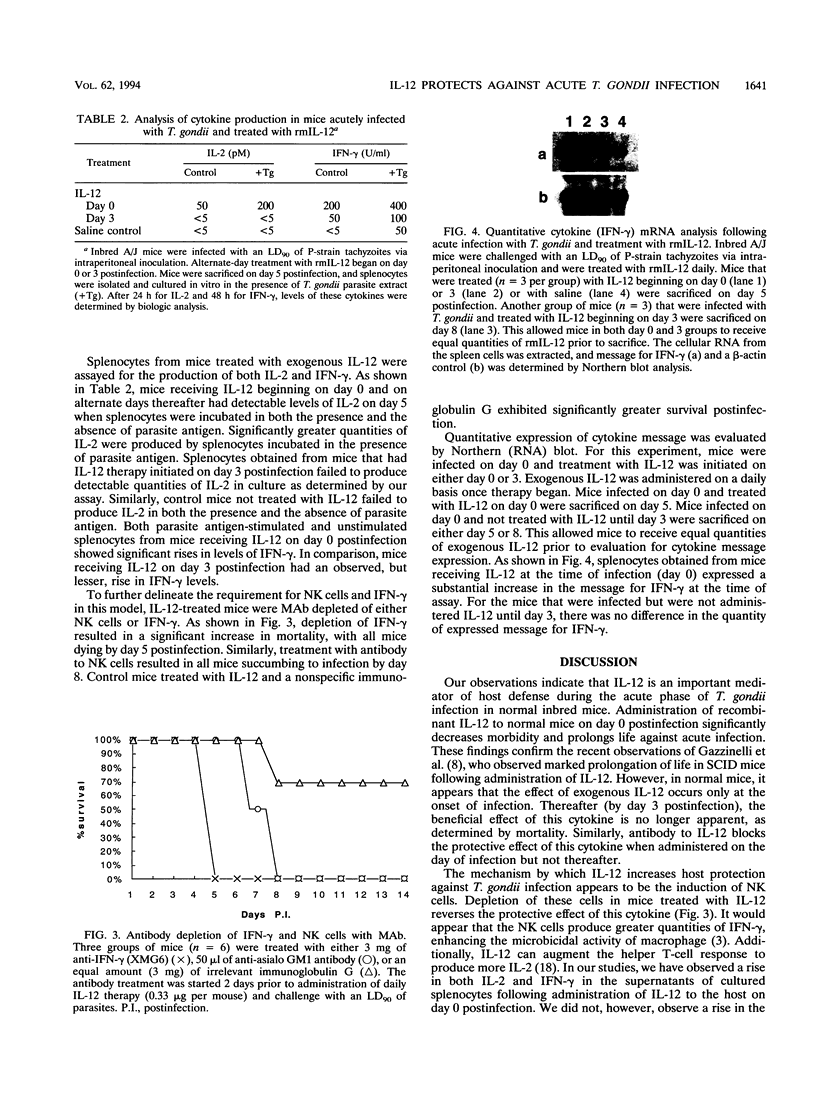
Protective immunity against Toxoplasma gondii is mediated by the host cellular immune response. Interleukin-12 (IL-12), a recently described cytokine that stimulates NK cells to produce gamma interferon (IFN-gamma), is able to enhance host protection against this parasite in SCID mice. Administration of IL-12 to A/J mice significantly increased survival over that of control mice when IL-12 was delivered early in the course of acute infection. If it was administered at day 3 or thereafter, there was no observed difference in mortality between treated and control mice. Antibody depletion of IL-12 increased susceptibility to infection, as measured by mortality, only when the IL-12 was administered before day 3 postinfection. Mice treated with IL-12 at day 0 postinfection exhibited a significant rise above the control in both IL-2 and IFN-gamma production. Once infection has been established in the host (3 days), administration of exogenous IL-12 is unable to alter parasite-induced downregulation of IFN-gamma production. Thus, IL-12 appears to play an important, but transitory, role in protection against acute infection with T. gondii in the normal murine host.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chan J., Siegel J. P., Luft B. J. Demonstration of T-cell dysfunction during acute toxoplasma infection. Cell Immunol. 1986 Apr 1;98(2):422–433. doi: 10.1016/0008-8749(86)90301-1. [DOI] [PubMed] [Google Scholar]
- Chan S. H., Perussia B., Gupta J. W., Kobayashi M., Pospísil M., Young H. A., Wolf S. F., Young D., Clark S. C., Trinchieri G. Induction of interferon gamma production by natural killer cell stimulatory factor: characterization of the responder cells and synergy with other inducers. J Exp Med. 1991 Apr 1;173(4):869–879. doi: 10.1084/jem.173.4.869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chomczynski P., Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987 Apr;162(1):156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- D'Andrea A., Aste-Amezaga M., Valiante N. M., Ma X., Kubin M., Trinchieri G. Interleukin 10 (IL-10) inhibits human lymphocyte interferon gamma-production by suppressing natural killer cell stimulatory factor/IL-12 synthesis in accessory cells. J Exp Med. 1993 Sep 1;178(3):1041–1048. doi: 10.1084/jem.178.3.1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gazzinelli R. T., Hakim F. T., Hieny S., Shearer G. M., Sher A. Synergistic role of CD4+ and CD8+ T lymphocytes in IFN-gamma production and protective immunity induced by an attenuated Toxoplasma gondii vaccine. J Immunol. 1991 Jan 1;146(1):286–292. [PubMed] [Google Scholar]
- Gazzinelli R. T., Hieny S., Wynn T. A., Wolf S., Sher A. Interleukin 12 is required for the T-lymphocyte-independent induction of interferon gamma by an intracellular parasite and induces resistance in T-cell-deficient hosts. Proc Natl Acad Sci U S A. 1993 Jul 1;90(13):6115–6119. doi: 10.1073/pnas.90.13.6115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes H. P., Kasper L. H., Little J., Dubey J. P. Absence of a role for natural killer cells in the control of acute infection by Toxoplasma gondii oocysts. Clin Exp Immunol. 1988 Jun;72(3):394–399. [PMC free article] [PubMed] [Google Scholar]
- Khan I. A., Ely K. H., Kasper L. H. Antigen-specific CD8+ T cell clone protects against acute Toxoplasma gondii infection in mice. J Immunol. 1994 Feb 15;152(4):1856–1860. [PubMed] [Google Scholar]
- Khan I. A., Smith K. A., Kasper L. H. Induction of antigen-specific parasiticidal cytotoxic T cell splenocytes by a major membrane protein (P30) of Toxoplasma gondii. J Immunol. 1988 Nov 15;141(10):3600–3605. [PubMed] [Google Scholar]
- McLeod R., Beem M. O., Estes R. G. Lymphocyte anergy specific to Toxoplasma gondii antigens in a baby with congenital toxoplasmosis. J Clin Lab Immunol. 1985 Jul;17(3):149–153. [PubMed] [Google Scholar]
- Sher A., Oswald I. P., Hieny S., Gazzinelli R. T. Toxoplasma gondii induces a T-independent IFN-gamma response in natural killer cells that requires both adherent accessory cells and tumor necrosis factor-alpha. J Immunol. 1993 May 1;150(9):3982–3989. [PubMed] [Google Scholar]
- Subauste C. S., Koniaris A. H., Remington J. S. Murine CD8+ cytotoxic T lymphocytes lyse Toxoplasma gondii-infected cells. J Immunol. 1991 Dec 1;147(11):3955–3959. [PubMed] [Google Scholar]
- Suzuki Y., Joh K., Kobayashi A. Macrophage-mediated suppression of immune responses in Toxoplasma-infected mice. III. Suppression of antibody responses to parasite itself. Cell Immunol. 1987 Dec;110(2):218–225. doi: 10.1016/0008-8749(87)90117-1. [DOI] [PubMed] [Google Scholar]
- Suzuki Y., Remington J. S. The effect of anti-IFN-gamma antibody on the protective effect of Lyt-2+ immune T cells against toxoplasmosis in mice. J Immunol. 1990 Mar 1;144(5):1954–1956. [PubMed] [Google Scholar]
- Trinchieri G. Interleukin-12 and its role in the generation of TH1 cells. Immunol Today. 1993 Jul;14(7):335–338. doi: 10.1016/0167-5699(93)90230-I. [DOI] [PubMed] [Google Scholar]
- Wing E. J., Boehmer S. M., Christner L. K. Toxoplasma gondii: decreased resistance to intracellular bacteria in mice. Exp Parasitol. 1983 Aug;56(1):1–8. doi: 10.1016/0014-4894(83)90090-5. [DOI] [PubMed] [Google Scholar]




