Abstract
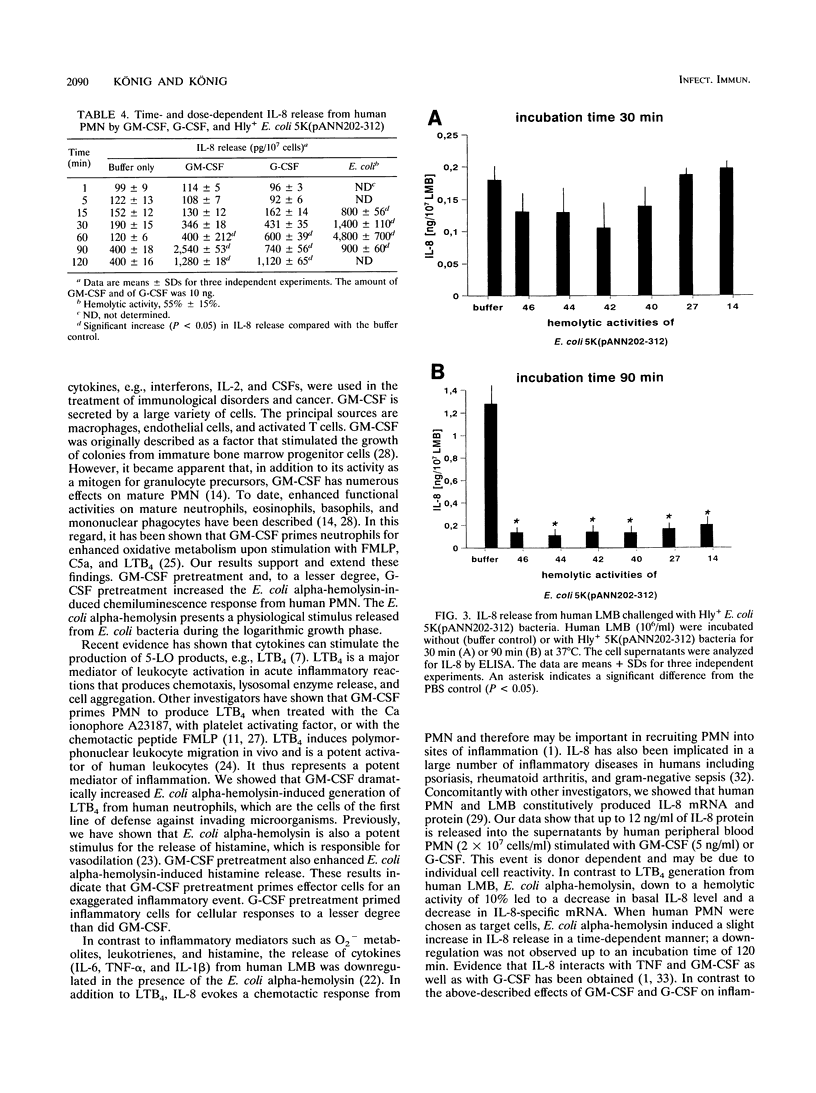
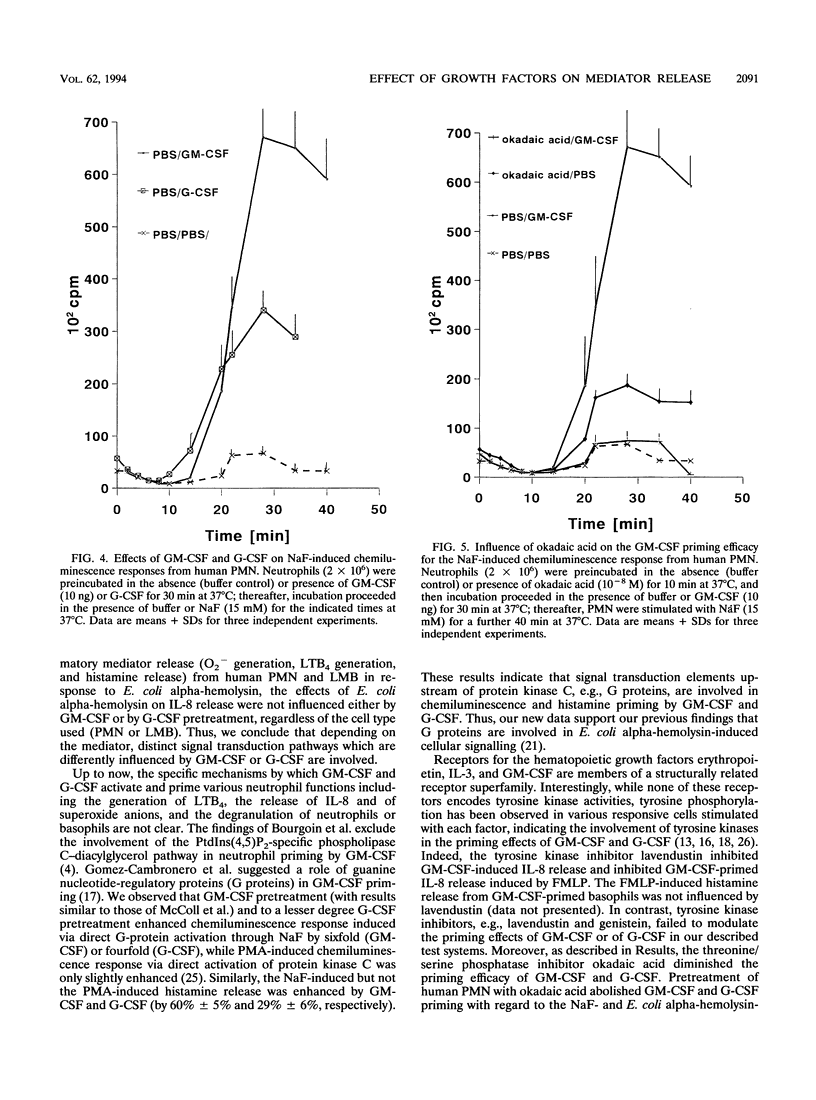
Previously, we have shown that Escherichia coli alpha-hemolysin represents a potent stimulus for inflammatory mediator release (O2- release, beta-glucuronidase release, and leukotriene generation) from human polymorphonuclear granulocytes (PMN) as well as for histamine release from a human lymphocyte-monocyte-basophil cell suspension (LMB). In contrast, the E. coli alpha-hemolysin leads to a downregulation of cytokine release (interleukin 6 [IL-6], tumor necrosis factor alpha, and IL-1 beta) from human LMB. This study was undertaken (i) to analyze the priming efficacy of growth factors (granulocyte-macrophage colony-stimulating factor [GM-CSF] and granulocyte CSF [G-CSF]) on inflammatory mediator release from human PMN and LMB challenged with hemolysin-producing E. coli bacteria as well as with cell-free E. coli alpha-hemolysin and (ii) to identify major components involved in GM-CSF and G-CSF priming. GM-CSF pretreatment led to an increased chemiluminescence response from human PMN by up to 100%, leukotriene B4 generation was enhanced up to fivefold, and histamine release from human LMB increased from 45% +/- 15% to 75% +/- 5% (mean +/- standard distribution) of the total histamine content. G-CSF priming induced an increase in the chemiluminescence response by up to 50% +/- 5% from human PMN and an increase in histamine release from human LMB by 20% +/- 5%. The growth factors, GM-CSF and G-CSF, modulated neither beta-glucuronidase release from human PMN nor IL-8 release from human PMN and LMB challenged with the E. coli alpha-hemolysin. GM-CSF and G-CSF pretreatment increased the fluoride (NaF)-induced chemiluminescence response by up to 10-fold; the serine/threonine phosphatase inhibitor okadaic acid inhibited GM-CSF- and G-CSF-induced priming. NaF-induced histamine release was enhanced up to 60 and 30% by GM-CSF and G-CSF priming, respectively. GM-CSF and G-CSF pretreatment did not modulate phorbol 12-myristate 13-acetate-induced chemiluminescence response or histamine release. GM-CSF by itself induced an increase in 5-lipoxygenase-specific mRNA expression within 5 min. Our results indicate that (i) GM-CSF and G-CSF interact with inflammatory cells via distinct cellular signalling, (ii) the signal transduction pathway is dependent on the cellular mediator, and (iii) the use of growth factors may be a potent tool to influence the clinical outcome in infectious diseases.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baggiolini M., Walz A., Kunkel S. L. Neutrophil-activating peptide-1/interleukin 8, a novel cytokine that activates neutrophils. J Clin Invest. 1989 Oct;84(4):1045–1049. doi: 10.1172/JCI114265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bazzoni F., Cassatella M. A., Rossi F., Ceska M., Dewald B., Baggiolini M. Phagocytosing neutrophils produce and release high amounts of the neutrophil-activating peptide 1/interleukin 8. J Exp Med. 1991 Mar 1;173(3):771–774. doi: 10.1084/jem.173.3.771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhakdi S., Muhly M., Korom S., Schmidt G. Effects of Escherichia coli hemolysin on human monocytes. Cytocidal action and stimulation of interleukin 1 release. J Clin Invest. 1990 Jun;85(6):1746–1753. doi: 10.1172/JCI114631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourgoin S., Plante E., Gaudry M., Naccache P. H., Borgeat P., Poubelle P. E. Involvement of a phospholipase D in the mechanism of action of granulocyte-macrophage colony-stimulating factor (GM-CSF): priming of human neutrophils in vitro with GM-CSF is associated with accumulation of phosphatidic acid and diradylglycerol. J Exp Med. 1990 Sep 1;172(3):767–777. doi: 10.1084/jem.172.3.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brom J., König W. Cytokine-induced (interleukins-3, -6 and -8 and tumour necrosis factor-beta) activation and deactivation of human neutrophils. Immunology. 1992 Feb;75(2):281–285. [PMC free article] [PubMed] [Google Scholar]
- Bøyum A. Isolation of lymphocytes, granulocytes and macrophages. Scand J Immunol. 1976 Jun;Suppl 5:9–15. [PubMed] [Google Scholar]
- Carroll M. P., Clark-Lewis I., Rapp U. R., May W. S. Interleukin-3 and granulocyte-macrophage colony-stimulating factor mediate rapid phosphorylation and activation of cytosolic c-raf. J Biol Chem. 1990 Nov 15;265(32):19812–19817. [PubMed] [Google Scholar]
- Chomczynski P., Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987 Apr;162(1):156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- Colditz I. G., Zwahlen R. D., Baggiolini M. Neutrophil accumulation and plasma leakage induced in vivo by neutrophil-activating peptide-1. J Leukoc Biol. 1990 Aug;48(2):129–137. doi: 10.1002/jlb.48.2.129. [DOI] [PubMed] [Google Scholar]
- Dahinden C. A., Zingg J., Maly F. E., de Weck A. L. Leukotriene production in human neutrophils primed by recombinant human granulocyte/macrophage colony-stimulating factor and stimulated with the complement component C5A and FMLP as second signals. J Exp Med. 1988 Apr 1;167(4):1281–1295. doi: 10.1084/jem.167.4.1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans J. P., Mire-Sluis A. R., Hoffbrand A. V., Wickremasinghe R. G. Binding of G-CSF, GM-CSF, tumor necrosis factor-alpha, and gamma-interferon to cell surface receptors on human myeloid leukemia cells triggers rapid tyrosine and serine phosphorylation of a 75-Kd protein. Blood. 1990 Jan 1;75(1):88–95. [PubMed] [Google Scholar]
- Fabian I., Kletter Y., Mor S., Geller-Bernstein C., Ben-Yaakov M., Volovitz B., Golde D. W. Activation of human eosinophil and neutrophil functions by haematopoietic growth factors: comparisons of IL-1, IL-3, IL-5 and GM-CSF. Br J Haematol. 1992 Feb;80(2):137–143. doi: 10.1111/j.1365-2141.1992.tb08890.x. [DOI] [PubMed] [Google Scholar]
- Goebel W., Hedgpeth J. Cloning and functional characterization of the plasmid-encoded hemolysin determinant of Escherichia coli. J Bacteriol. 1982 Sep;151(3):1290–1298. doi: 10.1128/jb.151.3.1290-1298.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomez-Cambronero J., Huang C. K., Bonak V. A., Wang E., Casnellie J. E., Shiraishi T., Sha'afi R. I. Tyrosine phosphorylation in human neutrophil. Biochem Biophys Res Commun. 1989 Aug 15;162(3):1478–1485. doi: 10.1016/0006-291x(89)90841-3. [DOI] [PubMed] [Google Scholar]
- Isfort R. J., Ihle J. N. Multiple hematopoietic growth factors signal through tyrosine phosphorylation. Growth Factors. 1990;2(2-3):213–220. doi: 10.3109/08977199009071507. [DOI] [PubMed] [Google Scholar]
- Kawakami M., Tsutsumi H., Kumakawa T., Abe H., Hirai M., Kurosawa S., Mori M., Fukushima M. Levels of serum granulocyte colony-stimulating factor in patients with infections. Blood. 1990 Nov 15;76(10):1962–1964. [PubMed] [Google Scholar]
- Knöller J., Schönfeld W., Köller M., Hensler T., König W. Arachidonic acid metabolites from polymorphonuclear leukocytes of healthy donors, severely burned patients and children with cystic fibrosis--routine monitoring by high-performance liquid chromatography. J Chromatogr. 1988 Jun 3;427(2):199–208. doi: 10.1016/0378-4347(88)80122-1. [DOI] [PubMed] [Google Scholar]
- König B., König W. Induction and suppression of cytokine release (tumour necrosis factor-alpha; interleukin-6, interleukin-1 beta) by Escherichia coli pathogenicity factors (adhesions, alpha-haemolysin). Immunology. 1993 Apr;78(4):526–533. [PMC free article] [PubMed] [Google Scholar]
- König B., König W. Roles of human peripheral blood leukocyte protein kinase C and G proteins in inflammatory mediator release by isogenic Escherichia coli strains. Infect Immun. 1991 Oct;59(10):3801–3810. doi: 10.1128/iai.59.10.3801-3810.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- König B., König W., Scheffer J., Hacker J., Goebel W. Role of Escherichia coli alpha-hemolysin and bacterial adherence in infection: requirement for release of inflammatory mediators from granulocytes and mast cells. Infect Immun. 1986 Dec;54(3):886–892. doi: 10.1128/iai.54.3.886-892.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- König W., Schönfeld W., Raulf M., Köller M., Knöller J., Scheffer J., Brom J. The neutrophil and leukotrienes--role in health and disease. Eicosanoids. 1990;3(1):1–22. [PubMed] [Google Scholar]
- McColl S. R., Beauseigle D., Gilbert C., Naccache P. H. Priming of the human neutrophil respiratory burst by granulocyte-macrophage colony-stimulating factor and tumor necrosis factor-alpha involves regulation at a post-cell surface receptor level. Enhancement of the effect of agents which directly activate G proteins. J Immunol. 1990 Nov 1;145(9):3047–3053. [PubMed] [Google Scholar]
- McColl S. R., DiPersio J. F., Caon A. C., Ho P., Naccache P. H. Involvement of tyrosine kinases in the activation of human peripheral blood neutrophils by granulocyte-macrophage colony-stimulating factor. Blood. 1991 Oct 1;78(7):1842–1852. [PubMed] [Google Scholar]
- McColl S. R., Krump E., Naccache P. H., Poubelle P. E., Braquet P., Braquet M., Borgeat P. Granulocyte-macrophage colony-stimulating factor increases the synthesis of leukotriene B4 by human neutrophils in response to platelet-activating factor. Enhancement of both arachidonic acid availability and 5-lipoxygenase activation. J Immunol. 1991 Feb 15;146(4):1204–1211. [PubMed] [Google Scholar]
- Metcalf D. The granulocyte-macrophage colony stimulating factors. Cell. 1985 Nov;43(1):5–6. doi: 10.1016/0092-8674(85)90004-2. [DOI] [PubMed] [Google Scholar]
- Mielke V., Bauman J. G., Sticherling M., Ibs T., Zomershoe A. G., Seligmann K., Henneicke H. H., Schröder J. M., Sterry W., Christophers E. Detection of neutrophil-activating peptide NAP/IL-8 and NAP/IL-8 mRNA in human recombinant IL-1 alpha- and human recombinant tumor necrosis factor-alpha-stimulated human dermal fibroblasts. An immunocytochemical and fluorescent in situ hybridization study. J Immunol. 1990 Jan 1;144(1):153–161. [PubMed] [Google Scholar]
- Reid G. K., Kargman S., Vickers P. J., Mancini J. A., Léveillé C., Ethier D., Miller D. K., Gillard J. W., Dixon R. A., Evans J. F. Correlation between expression of 5-lipoxygenase-activating protein, 5-lipoxygenase, and cellular leukotriene synthesis. J Biol Chem. 1990 Nov 15;265(32):19818–19823. [PubMed] [Google Scholar]
- Van Zee K. J., DeForge L. E., Fischer E., Marano M. A., Kenney J. S., Remick D. G., Lowry S. F., Moldawer L. L. IL-8 in septic shock, endotoxemia, and after IL-1 administration. J Immunol. 1991 May 15;146(10):3478–3482. [PubMed] [Google Scholar]
- Willems J., Joniau M., Cinque S., van Damme J. Human granulocyte chemotactic peptide (IL-8) as a specific neutrophil degranulator: comparison with other monokines. Immunology. 1989 Aug;67(4):540–542. [PMC free article] [PubMed] [Google Scholar]
- Yoshimura T., Matsushima K., Tanaka S., Robinson E. A., Appella E., Oppenheim J. J., Leonard E. J. Purification of a human monocyte-derived neutrophil chemotactic factor that has peptide sequence similarity to other host defense cytokines. Proc Natl Acad Sci U S A. 1987 Dec;84(24):9233–9237. doi: 10.1073/pnas.84.24.9233. [DOI] [PMC free article] [PubMed] [Google Scholar]