Abstract
To infer similarities and differences in terminal pattern formation in insects, we analyzed several of the key genes of this process in the beetle Tribolium castaneum. We cloned two genes of the terminal pattern cascade, namely tailless (tll) and forkhead (fkh), from Tribolium and studied their expression patterns. In addition, we analyzed the pattern of MAP kinase activation at blastoderm stage as a possible signature for torso-dependent signaling. Further, we analyzed the late expression of the previously cloned Tribolium caudal (Tc-cad) gene. Finally, we used the upstream region of Tc-tll to drive a reporter gene construct in Drosophila. We find that this construct is activated at the terminal regions in Drosophila, suggesting that the torso-dependent pathway is conserved between the species. We show that most of the expression patterns of the genes studied here are similar in Drosophila and Tribolium, suggesting conserved functions. There is, however, one exception, namely the early function of Tc-tll at the posterior pole. In Drosophila, the posterior tll expression is involved in the direct regulation of the target genes of the terminal pathway. In Tribolium, posterior Tc-tll expression occurs only for a short time and ceases before the target genes known from Drosophila are activated. Thus, we infer that Tc-tll does not function as a direct regulator of segmentation genes at the posterior end. It is more likely to be involved in the early specification of a group of “terminal” cells, which begin to differentiate only at a later stage of embryogenesis, when much of the abdominal segmentation process is complete. Thus, there appears to have been a major shift in tll function during the evolutionary transition from short germ to long germ embryogenesis.
Keywords: tailless, forkhead, caudal, torso, short germ insect
Terminal pattern formation in Drosophila is maternally initiated at both poles of the egg through the torso-mediated receptor tyrosine kinase pathway (1). Two zygotic gap genes, namely tailless (tll) and huckebein (hkb), are activated in a concentration-dependent manner in response to the signaling activity of torso (tor) (2, 3). Both tll and hkb code for transcription factors that regulate further target genes, among them forkhead (fkh), brachyenteron (byn), and the terminal hunchback (hb) and wingless (wg) domains (4–8). fkh and wg are also activated by caudal (cad) (8), which is expressed maternally as well as zygotically in the posterior half of the embryo (9, 10). Together these genes are required for patterning the terminal cuticle structures, such as the most posterior segments, the anal pads, and the telson, as well as the stomodaeum, the hindgut, and the Malpighian tubules.
Embryogenesis in the flour beetle Tribolium represents a more ancestral form of embryogenesis in insects (11). Nevertheless, it was possible to clone most of the early segmentation genes known from Drosophila also from Tribolium, and the analysis of their expression patterns has suggested that they may have similar functions in both species (12, 13). At the same time, because of the different modes of embryogenesis between the two species, one has to expect that differences must have evolved, with respect either to regulatory interactions or to primary function of genes. We have shown previously that the primary regulation of hunchback has undergone an evolutionary transition from dependence on caudal in Tribolium to dependence on bicoid in Drosophila (14). Our results presented here suggest for the posterior expression domain of tll that there has also been a major transition in function.
Materials and Methods
Cloning the tll and fkh Orthologues.
Redundant primers for the fkh domain (gift of S. Farrington, Biological Laboratories, Harvard) and for the tll DNA-binding domain were used to initially amplify fragments from genomic DNA (fkh primers: forward -ckd atd swr tty tgc ca, reverse -aar ccn ccn tay wsh ta; tll primers: forward -ggn ath tay gcn tgy gay gg, reverse -ytg nac ngc rtc ytt rtt cat). The respective fragments were then used to screen a cDNA library (fkh) or a genomic library (tll). Because only truncated sequences could be recovered, 5′ and 3′ rapid amplification of cDNA ends (using the Marathon or the SMART Kit, CLONTECH) was used to find the 5′ end (tll and fkh) and the 3′end (tll) of these genes. The GenBank database accession no. for Tc-fkh is AF217810 and for Tc-tll is AF219117. To isolate the upstream sequence of the Tc-tll gene, the initially isolated genomic fragment was used to screen a genomic library. Of the eight recovered λ phages, one (phage no. 6.1), containing approximately 10 kb upstream region, was chosen for further analysis. SalI and EcoRI fragments were isolated, subcloned in Bluescript KS+ (Stratagene), and sequenced to get a 9.8-kb upstream sequence (accession no. AF225975).
Transgenic Flies.
In a two-step cloning strategy (details available on request), the complete 9.8-kb upstream region was cloned in the original orientation in front of the lacZ reporter by using the pCasperAUG vector (15). The resulting construct was called pTc-tll-9.8. P-element-mediated transformation was done as described (16). The recipient strain was y w67c23.
Generation of the tll Antibody.
A fragment coding for part of the ligand-binding domain (position 181–305) was cloned in frame into an expression vector (pRSET C, Invitrogen). Overexpressed protein was affinity purified on a TALON column (CLONTECH). One rabbit was immunized with approximately 5 mg of protein (Eurogentec, Brussels) by using about 1 mg per boost. After the fifth boost, the serum was affinity purified by using the same protein.
Expression Analysis.
Whole-mount in situ hybridization and antibody stainings were done essentially as described for Drosophila (17, 18). The anti-active MAP-K antibody (Sigma) was used essentially as described (19).
Results
tll and fkh Orthologues from Tribolium.
Candidates for tll and fkh orthologues from Tribolium were first obtained by PCR amplification by using degenerate primers in conserved regions. Only one type of fragment was obtained in each case, which was subsequently used to screen a cDNA library. As no full length cDNAs could be recovered, rapid amplification of cDNA ends experiments were performed to obtain the full sequences. For tll, we also obtained the genomic region from a genomic λ library, approximately 10 kb of which were also sequenced.
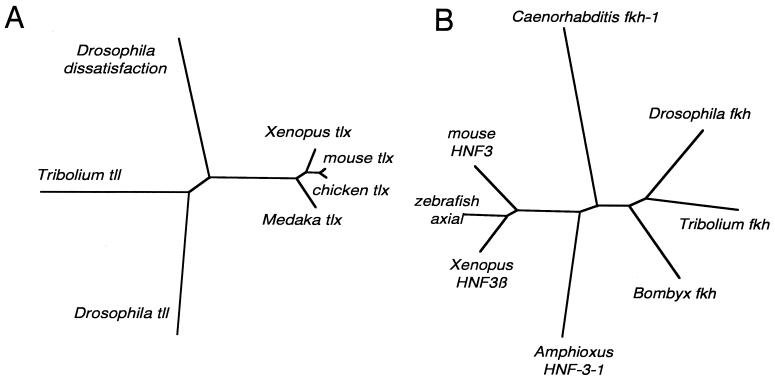
The tll gene displays two conserved protein motifs, namely a zinc finger DNA-binding domain and a ligand-binding domain (20). Both domains can be aligned to the respective orthologues from other species. The phylogram in Fig. 1A depicts the relationships between the known tll orthologues in different species.
Figure 1.
Phylograms for tll-related (A) and fkh-related genes (B). The trees were constructed with a maximum likelihood algorithm by using puzzle (48) based on a clustal alignment (49). All branches receive very high support. The accession numbers for the respective sequences are: (A) Drosophila tll M34639, Tribolium tll AF219117, Drosophila dissatisfaction (as a tll-related gene in Drosophila) AF106677, Xenopus tlx P70052, mouse tlx Q64104, chicken tlx Q91379, medaka tlx AJ131390, and (B) Drosophila fkh P14734, Tribolium fkh AF217810, Bombyx fkh Q17241, Caenorhabditis fkh-1 AAA96319, amphioxus HNF-3–1 CAA65368, Xenopus HNF-3β I51436, zebrafish Axial Q07342, and mouse HNF-3β P35583.
Transcription factors belonging to the forkhead class encode a winged-helix-type DNA-binding motif, the forkhead domain. Orthologues of the fkh gene are known from many different phyla. In a detailed sequence analysis, the different fkh genes were grouped into 10 different classes (21). fkh itself belongs to class 1 and shows diagnostic amino acids within the 110-aa-long fkh domain. All these amino acids can also be found in the Tc-fkh sequence. The phylogram resulting from the alignment with the known fkh orthologues from other species is shown in Fig. 1b.
Expression of Tc-tll.
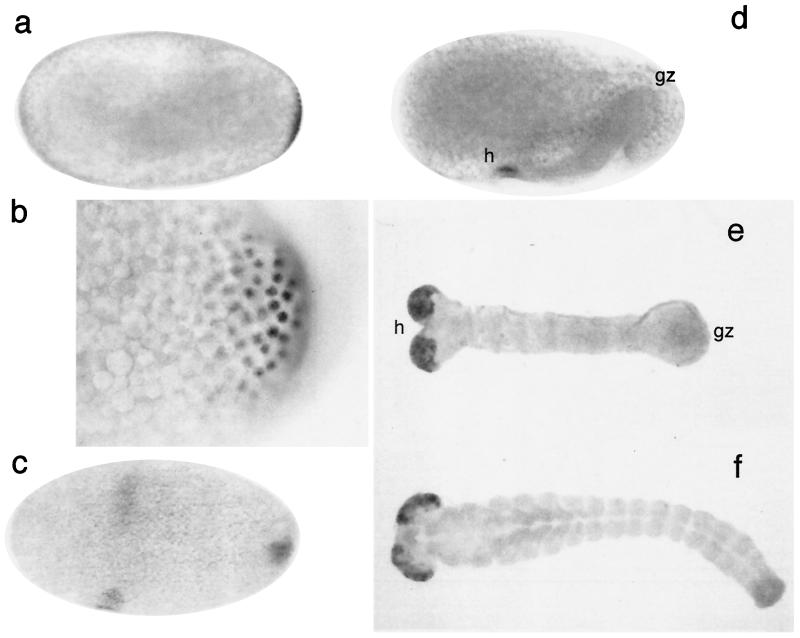
Tailless protein expression in Tribolium can be detected already at blastoderm stage, where a small group of cells at the posterior pole express the gene (Fig. 2a). Staining is seen in the nuclei, and a short posterior-to-anterior gradient is evident (Fig. 2b). It was shown previously that there is also a very early expression domain of Tc-wingless at the posterior pole of the Tribolium embryo at blastoderm stage (22), which is reminiscent of the terminal wg domain in Drosophila (23). This suggests that at least part of the posterior terminal structures are determined already at blastoderm stage in Tribolium. This is also in general accord with classical fate mapping experiments, which could localize the anlagen for the hindgut in short germ insects at the posterior end of the blastoderm embryo (24).
Figure 2.
RNA and protein expression of Tc-tll. Embryos were stained either with a Tc-tll-specific antibody (a–d) or by whole-mount in situ hybridization (e, f). a–f represent progressively older stages of embryogenesis. Anterior is to the left, a, b, and d are lateral views (dorsal is up), and c, e, and f are ventral views. No staining is detected in the growth zone (gz) with the beginning of germband growth (d and e), whereas the expression in the head (h) persists. See text for further details.
There is no expression of Tc-tll at the anterior pole of the Tribolium embryo, which is in contrast to Drosophila (20). However, at late blastoderm stage, Tc-tll expression appears in the developing head region, which lies at the ventral side of the embryo (Fig. 2c). It seems likely that this expression is homologous to the known expression of tll in the head of Drosophila (20, 25). With the beginning of gastrulation, tll expression ceases at the posterior end (Fig. 2d) at both the RNA and protein levels, whereas the head-specific expression domains become stronger and persist throughout development (Fig. 2 e and f).
Expression of Tc-fkh.
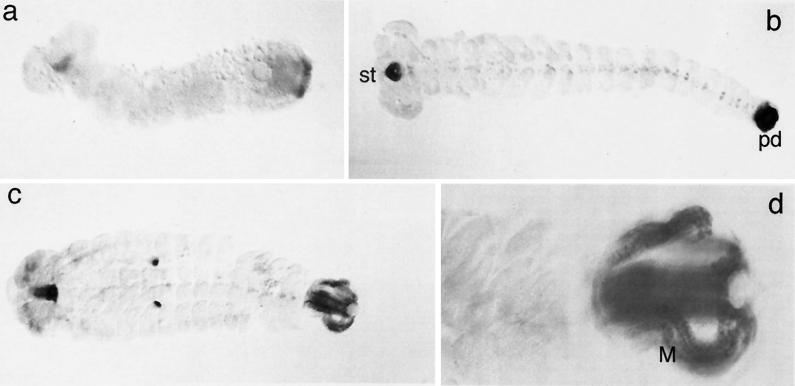
The first expression of Tc-fkh RNA can be seen in yolk nuclei (not shown). A similar expression is also known from Drosophila (4) although much later in development. As the germband elongates, Tc-fkh expression becomes visible in the primordia of the stomodaeum and the proctodaeum (Fig. 3 a and b). Staining in these regions persists until the end of embryogenesis (Fig. 3 b and c). In the late embryo, Tc-fkh is expressed in the primordium of the central nervous system, in parts of the brain, and in the Malpighian tubules (Fig. 3 b and c). All these expression aspects are very similar to Drosophila, where fkh acts as a homeotic gene for specifying the stomodaeum and the proctodaeum (4). In Drosophila, however, the first expression is seen at blastoderm stage (4), which is not evident in Tribolium. But this can be reconciled with the fact that fkh is required not for initiating pattern formation but only for later differentiation steps.
Figure 3.
RNA expression of Tc-fkh. Embryos were stained by whole-mount in situ hybridization. a–c represent progressively older stages, and d is an enlargement of the posterior end of an embryo that is at the same stage as the embryo in c. st, stomodaeum; pd, proctodaeum; M, Malpighian tubules. Note that the dots seen in the middle of the embryo in c are unspecifically staining cells in the pleuropodia on segment A1.
Late Expression of Tc-cad.
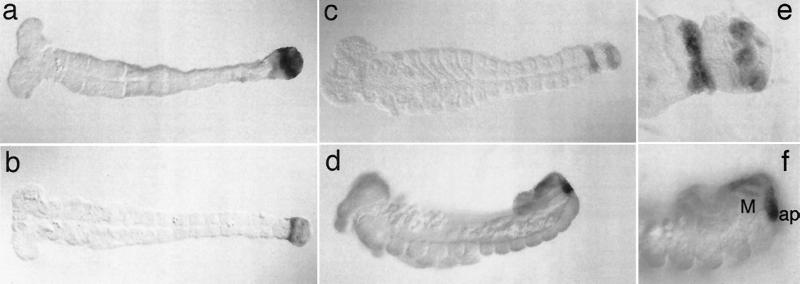
The early stages of Tc-cad expression have been described previously (26). We have looked here more closely at those expression pattern aspects that can be related to the terminal function of cad. Tc-cad expression is present throughout development in the growth zone (26), but at the end of the segmentation process, Tc-cad expression becomes restricted to a subterminal stripe (Fig. 4 a and b), which occurs similarly in Drosophila (9, 10). Some groups of cells at the most posterior end remain Tc-cad positive (Fig. 4 c and e). These might correspond to the primordia of the anal plates, as described for Drosophila (9, 10) and the silk moth Bombyx (27). With the beginning of germ band retraction and hindgut formation, Tc-cad expression becomes detectable in the Malpighian tubules (Fig. 4 d and f), which is again in line with the function of cad in Drosophila (28).
Figure 4.
RNA expression of Tc-cad during late stages of embryogenesis. Embryos were stained by whole-mount in situ hybridization with a probe for Tc-cad (26). (a–d) Progressively older stages of embryogenesis. (e and f) Enlarged views of the posterior ends of the embryos in c and d, respectively. ap, anal plates; M, Malpighian tubules.
Regulation by torso.
The tor signal transduction pathway acts via a MAP kinase, whose activity can be monitored with a specific antibody against active MAP-K (29). We used this antibody to see whether one can detect possible signs of tor activity in the region where tll is expressed. Fig. 5a shows that there is indeed staining at the posterior end of the embryo at early blastoderm stage, in the region where tll is activated (compare Fig. 2a). Interestingly, there is also staining in a broad anterior domain, which corresponds roughly to the region of the future serosa cells, which are probably specified at this stage (30, 31).
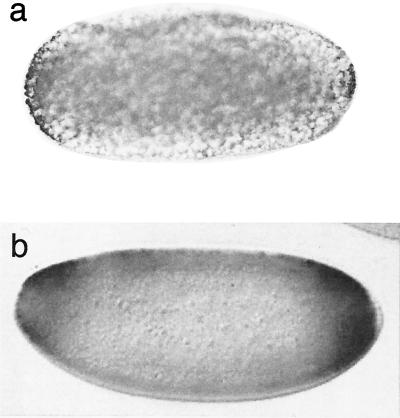
Figure 5.
Evidence for torso activity. (a) Staining of Tribolium early blastoderm stage embryo with an antibody against active MAP kinase, an intermediate protein in the receptor tyrosine kinase pathway that is activated by the torso-signaling cascade. (b) Staining of an early blastoderm stage embryo of Drosophila against lacZ as reporter. The embryo is transformed with a construct containing the upstream sequences of Tc-tll in front of the lacZ gene. See text for further details.
To test whether Tribolium tll responds to the terminal activation pathway in Drosophila, we fused the genomic upstream region of Tc-tll to a lacZ reporter and transformed this in Drosophila embryos. This reporter construct is indeed activated at the terminal regions of the embryo at blastoderm stage (Fig. 5b), comparable to similar constructs with the Drosophila tll upstream region (25). This indicates that this activation pathway is conserved between Drosophila and Tribolium.
Discussion
Previous comparative studies of segmentation genes in Drosophila and Tribolium have shown that differences in expression patterns can usually be correlated with the differences in the early fate map (12–14). Thus, it would seem that the general function of most segmentation genes is conserved between the species. This is also likely to be the case for the genes studied here, with the exception of posterior tll function. If this tll function were comparable in Drosophila and Tribolium, we would expect tll in Tribolium to remain expressed at the posterior pole until the target genes are activated. We find, however, that posterior Tc-tll expression ceases quickly at both the RNA and protein levels, long before the first expression of the target genes is visible. Thus, it appears that the early function of tll has undergone a major evolutionary shift between Tribolium and Drosophila.
Changing Role of Posterior tll Function.
There are at least three known direct target genes of tll that are required for the formation of the posterior terminal structures of the Drosophila embryo, namely fkh, byn, and the terminal hb stripe (4–7, 32). All three are thought to be regulated directly by tll, although this has been shown formally only for the terminal hb stripe (6). In Tribolium, all three of these genes (or expression domains) start to be expressed only after tll expression has ceased at the posterior end. This late onset of expression of the target genes suggests that tll could not be their direct activator. A similar inference applies to another target gene of tll, namely Krüppel (Kr). tll acts as a repressor of Kr in Drosophila (33, 34). In Tribolium, Tc-Kr expression occurs at the posterior pole of the blastoderm embryo (35), overlapping with the early Tc-tll expression. This coexpression suggests that Tc-tll does not act as a repressor of Tc-Kr. Thus, Tc-tll does not appear to play the same role as tll in Drosophila as a direct regulator of other segmentation genes.
Nonetheless, Tc-tll is likely to be involved in determining the posterior terminal fate in Tribolium. We can infer this from the observations presented here, as well as from classic fate mapping experiments. We find that a receptor tyrosine kinase pathway is active at the same time and location where Tc-tll is activated at the posterior pole, suggesting that the torso-mediated induction of tll is conserved between the two species. Accordingly, we find that a reporter gene construct carrying the Tc-tll upstream region in Drosophila is activated in a very similar pattern as that known for the endogenous tll pattern in Drosophila (25). The other clue comes from the general fate mapping experiments in short germ insects. These fatemaps suggest that the future terminus, represented by the hindgut anlage, is specified already at blastoderm stage, although the structure itself develops only much later (reviewed in ref. 24). This is in contrast to the abdominal segments, for which there is no representation in the blastoderm fate map. They are sequentially generated from the posterior growth zone only after blastoderm stage. To reconcile these observations, we propose that there is a group of cells within the growth zone that is determined at blastoderm stage to produce the terminal structures, but which remains quiescent until much of the abdominal segmentation has been completed. In this scenario, the most likely role for Tc-tll would be an involvement in the differentiation of cells that could be called “terminal cells.”
There is further evidence that such “terminal cells” do indeed exist at the posterior end of the Tribolium embryo. This comes from the observation of a very early activation of a Tribolium wingless (Tc-wg) expression domain in this region (22). Tc-wg may be involved in the differentiation process, because wg is also required for terminal structure formation in Drosophila (8). However, different from Tc-tll, Tc-wg remains expressed in the terminal region until late in embryogenesis (22), suggesting that its primary function may be conserved.
The Role of the torso-Signaling Pathway.
The torso-signaling pathway acts symmetrically at both ends of the embryo in Drosophila. The same appears to apply for torso in Tribolium, as is shown by the fairly symmetrical staining for the activated MAP kinase. One might not have expected this outcome, however, because of the differences in the fate map in the two species. Although the anterior terminal structures become specified at the anterior end of the blastoderm in Drosophila, this is different for Tribolium. In Tribolium the extraembryonic serosa is specified at the anterior end (30, 31), a structure that does not exist in this form in Drosophila. In Drosophila, the extraembryonic cells are determined at the dorsal side and consist of remnants of both the serosa and the amnion (36). Still, a molecular similarity between these structures is suggested by the fact that zerknüllt (zen) and decapentaplegic (dpp) are expressed in both species in these groups of cells (30, 31, 37, 38).
One can speculate that the role of the anterior torso pathway in Tribolium is the activation of Tc-zen and Tc-dpp. In this case, one would have to propose another major functional shift, because zen and dpp are activated primarily by the dorsoventral system in Drosophila (36). There are, however, indications that in both species zen and dpp are under the control of both the terminal and the dorsoventral system. In Tribolium, the expression of Tc-dpp and Tc-zen occurs initially at the anterior pole, but it shifts quickly toward the dorsal side, together with the expansion of the serosa. This shift is likely to be under the control of the dorsoventral system. In Drosophila, it is known that polar expression of zen and dpp require activation by the terminal system (39).
Thus, if the genes involved in extraembryonic membrane formation are regulated by the terminal and the dorsoventral system in both species, the apparent difference could be explained by heterochronic shifts and differences in the relative importance of the maternal axis determining systems for their expression. In this case, it would not be necessary to propose a major evolutionary transition to explain these differences.
Anterior tll Expression.
In contrast to Drosophila, we do not observe an initial expression of tll at the anterior pole in Tribolium. But this is in line with the changed fate map, as discussed above. On the other hand, the brain-specific expression of Drosophila occurs also in Tribolium, albeit at somewhat later stages. At this time, the expression is at the ventral side and is therefore unlikely to be under the control of the terminal system. In Drosophila, the regulation of the anterior tll expression is also under the control of bicoid (40). It is, however, still unclear whether there is a homologue of bicoid in Tribolium (41), although functional evidence has suggested that it could be present (14). Still, it seems premature to make any speculation on whether this anterior expression of tll has a conserved regulation in both species. However, at least its function may be conserved. In Drosophila, the brain-specific expression is required for the formation of the anterior-most portions of the brain, including the optic lobes (25). A similar function is likely in Tribolium, as tll staining persists in the most anterior head region and also covers the ocular region. An involvement for the formation of the eyes has also been suggested for the Xenopus homologue of tll (42). Thus, at least parts of the brain-specific functions of tll may have been conserved even between phyla.
Conclusions
If the above interpretations are correct, then one would ascribe the most distinctive evolutionary changes in the terminal patterning systems of Tribolium and Drosophila to the function of the posterior tll expression. If we take Tribolium as a representative of the more ancestral form of embryogenesis in insects, then we would conclude that the ancestral function of tll was to differentiate the terminal cells. In Drosophila, tll has apparently lost this function but has acquired the direct control of the activation of key genes in this process, such as byn and fkh. In addition, tll in Drosophila must have acquired new regulatory interactions with the other gap genes, as well as with the enhancers driving the posterior pair rule stripes of hairy. tll acts mainly as a repressor in this context, and these interactions are direct (43, 44). Thus, at least for one expression aspect of tll, one has to infer a major transition in regulatory interactions in the evolution of the long germ insects. A more detailed analysis of this evolutionary transition will become possible with the help of the emerging genetic (45, 46) and transgenic (47) techniques in Tribolium.
Acknowledgments
We thank S. Farrington (Biological Laboratories, Harvard University) for providing the initial primers to screen for forkhead homologues, S. Brown (Kansas State University, Manhattan, KS) for providing a genomic Tribolium library, G. Büttner for help with sequencing and embryo staining, and M. Klingler, E. Wimmer, and H. Dove for discussions and suggestions on the manuscript. This work was supported by the Deutsche Forschungsgemeinschaft, with grants to R.S. and D.T., and by the Fonds der Chemischen Industrie.
Footnotes
This paper was submitted directly (Track II) to the PNAS office.
Data deposition: The sequences reported in this paper have been deposited in the GenBank database (accession nos. AF217810, AF219117, and AF225975).
Article published online before print: Proc. Natl. Acad. Sci. USA, 10.1073/pnas.100005497.
Article and publication date are at www.pnas.org/cgi/doi/10.1073/pnas.100005497
References
- 1.Lu X, Perkins L A, Perrimon N. Development (Cambridge, U.K.) 1993. Suppl., 47–56. [PubMed] [Google Scholar]
- 2.Greenwood S, Struhl G. Development (Cambridge, UK) 1997;124:4879–4886. doi: 10.1242/dev.124.23.4879. [DOI] [PubMed] [Google Scholar]
- 3.Ghiglione C, Perrimon N, Perkins L A. Dev Biol. 1999;205:181–193. doi: 10.1006/dbio.1998.9102. [DOI] [PubMed] [Google Scholar]
- 4.Weigel D, Jürgens G, Kuttner F, Seifert E, Jäckle H. Cell. 1989;57:645–658. doi: 10.1016/0092-8674(89)90133-5. [DOI] [PubMed] [Google Scholar]
- 5.Weigel D, Seifert E, Reuter D, Jäckle H. EMBO J. 1990;9:1199–1207. doi: 10.1002/j.1460-2075.1990.tb08227.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Margolis J S, Borowsky M L, Steingrimsson E, Shim C W, Lengyel J A, Posakony J W. Development (Cambridge, UK) 1995;121:3067–3077. doi: 10.1242/dev.121.9.3067. [DOI] [PubMed] [Google Scholar]
- 7.Singer J B, Harbecke R, Kusch T, Reuter R, Lengyel J A. Development (Cambridge, UK) 1996;122:3707–3718. doi: 10.1242/dev.122.12.3707. [DOI] [PubMed] [Google Scholar]
- 8.Wu L H, Lengyel J A. Development (Cambridge, UK) 1998;125:2433–2442. doi: 10.1242/dev.125.13.2433. [DOI] [PubMed] [Google Scholar]
- 9.Macdonald P M, Struhl G. Nature (London) 1986;324:537–545. doi: 10.1038/324537a0. [DOI] [PubMed] [Google Scholar]
- 10.Mlodzik M, Gehring W J. Cell. 1987;13:465–478. doi: 10.1016/0092-8674(87)90197-8. [DOI] [PubMed] [Google Scholar]
- 11.Tautz D, Friedrich M, Schröder R. Development (Cambridge, U.K.) 1994. Suppl., 193–199. [Google Scholar]
- 12.Tautz D, Sommer R. Trends Genet. 1995;11:23–27. doi: 10.1016/s0168-9525(00)88982-9. [DOI] [PubMed] [Google Scholar]
- 13.Brown S J, Denell R E. Semin Cell Dev Biol. 1996;7:553–560. [Google Scholar]
- 14.Wolff C, Schröder R, Schulz C, Tautz D, Klingler M. Development (Cambridge, UK) 1998;125:3645–3654. doi: 10.1242/dev.125.18.3645. [DOI] [PubMed] [Google Scholar]
- 15.Thummel C S, Boulet A M, Lipshitz H D. Gene. 1988;74:445–456. doi: 10.1016/0378-1119(88)90177-1. [DOI] [PubMed] [Google Scholar]
- 16.Rubin G M, Spradling A C. Science. 1982;218:348–353. doi: 10.1126/science.6289436. [DOI] [PubMed] [Google Scholar]
- 17.Tautz D, Pfeifle C. Chromosoma. 1989;98:81–85. doi: 10.1007/BF00291041. [DOI] [PubMed] [Google Scholar]
- 18.Lehmann R, Tautz D. Methods Cell Biol. 1994;44:575–598. doi: 10.1016/s0091-679x(08)60933-4. [DOI] [PubMed] [Google Scholar]
- 19.Peri F, Bökel C, Roth S. Mech Dev. 1999;81:75–88. doi: 10.1016/s0925-4773(98)00228-7. [DOI] [PubMed] [Google Scholar]
- 20.Pignoni F, Baldarelli R M, Steingrimsson E, Diaz R J, Patapoutian A, Merriam J R, Lengyel J A. Cell. 1990;62:151–163. doi: 10.1016/0092-8674(90)90249-e. [DOI] [PubMed] [Google Scholar]
- 21.Kaufmann E, Knöchel W. Mech Dev. 1996;57:3–20. doi: 10.1016/0925-4773(96)00539-4. [DOI] [PubMed] [Google Scholar]
- 22.Nagy L M, Carroll S. Nature (London) 1994;367:460–463. doi: 10.1038/367460a0. [DOI] [PubMed] [Google Scholar]
- 23.van den Heuvel M, Nusse R, Johnston P, Lawrence P A. Cell. 1989;59:739–749. doi: 10.1016/0092-8674(89)90020-2. [DOI] [PubMed] [Google Scholar]
- 24.Anderson D T. In: Developmental Sytems: Insects. Counce S J, Waddington C H, editors. London: Academic; 1972. pp. 96–242. [Google Scholar]
- 25.Rudolph K M, Liaw G J, Daniel A, Green P, Courey A J, Hartenstein V, Lengyel J A. Development (Cambridge, UK) 1997;124:4297–4308. doi: 10.1242/dev.124.21.4297. [DOI] [PubMed] [Google Scholar]
- 26.Schulz C, Schröder R, Hausdorf B, Wolff C, Tautz D. Dev Genes Evol. 1998;208:283–289. doi: 10.1007/s004270050183. [DOI] [PubMed] [Google Scholar]
- 27.Xu X, Xu P X, Suzuki Y. Development (Cambridge, UK) 1994;120:277–285. doi: 10.1242/dev.120.2.277. [DOI] [PubMed] [Google Scholar]
- 28.Liu S, Jack J. Dev Biol. 1992;150:133–143. doi: 10.1016/0012-1606(92)90013-7. [DOI] [PubMed] [Google Scholar]
- 29.Gabay L, Seger R, Shilo B Z. Science. 1997;277:1103–1106. doi: 10.1126/science.277.5329.1103. [DOI] [PubMed] [Google Scholar]
- 30.Wolff C, Sommer R, Schröder R, Glaser G, Tautz D. Development (Cambridge, UK) 1995;121:4227–4236. doi: 10.1242/dev.121.12.4227. [DOI] [PubMed] [Google Scholar]
- 31.Falciani F, Hausdorf B, Schröder R, Akam M, Tautz D, Denell R, Brown S. Proc Natl Acad Sci USA. 1996;93:8479–8484. doi: 10.1073/pnas.93.16.8479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kispert A, Herrmann B G, Leptin M, Reuter R. Genes Dev. 1994;8:2137–2150. doi: 10.1101/gad.8.18.2137. [DOI] [PubMed] [Google Scholar]
- 33.Gaul U, Jäckle H. Cell. 1987;51:549–555. doi: 10.1016/0092-8674(87)90124-3. [DOI] [PubMed] [Google Scholar]
- 34.Hoch M, Gerwin N, Taubert H, Jäckle H. Science. 1992;256:94–97. doi: 10.1126/science.1348871. [DOI] [PubMed] [Google Scholar]
- 35.Sommer R J, Tautz D. Nature (London) 1993;361:448–450. doi: 10.1038/361448a0. [DOI] [PubMed] [Google Scholar]
- 36.Morisato D, Anderson K V. Annu Rev Genet. 1995;29:371–399. doi: 10.1146/annurev.ge.29.120195.002103. [DOI] [PubMed] [Google Scholar]
- 37.Doyle H J, Harding K, Hoey T, Levine M. Nature (London) 1986;323:76–79. doi: 10.1038/323076a0. [DOI] [PubMed] [Google Scholar]
- 38.St Johnston R D, Gelbart W M. EMBO J. 1987;6:2785–2791. doi: 10.1002/j.1460-2075.1987.tb02574.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ray R P, Arora K, Nüsslein-Volhard C, Gelbart W M. Development (Cambridge, UK) 1991;113:35–54. doi: 10.1242/dev.113.1.35. [DOI] [PubMed] [Google Scholar]
- 40.Liaw G J, Lengyel J A. Mech Dev. 1993;40:47–61. doi: 10.1016/0925-4773(93)90087-e. [DOI] [PubMed] [Google Scholar]
- 40a.Pignoni F, Steingrimsson E, Lengyel J A. Development (Cambridge, UK) 1992;115:239–251. doi: 10.1242/dev.115.1.239. [DOI] [PubMed] [Google Scholar]
- 41.Stauber M, Jäckle H, Schmidt-Ott U. Proc Natl Acad Sci USA. 1999;96:3786–3789. doi: 10.1073/pnas.96.7.3786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hollemann T, Bellefroid E, Pieler T. Development (Cambridge, UK) 1998;125:2425–2432. doi: 10.1242/dev.125.13.2425. [DOI] [PubMed] [Google Scholar]
- 43.Pankratz M J, Jäckle H. Trends Genet. 1990;6:287–292. doi: 10.1016/0168-9525(90)90234-w. [DOI] [PubMed] [Google Scholar]
- 44.Sauer F, Rivera-Pomar R, Hoch M, Jäckle H. Philos Trans R Soc London B. 1996;351:579–587. doi: 10.1098/rstb.1996.0057. [DOI] [PubMed] [Google Scholar]
- 45.Sulston I A, Anderson K V. Development (Cambridge, UK) 1996;122:805–814. doi: 10.1242/dev.122.3.805. [DOI] [PubMed] [Google Scholar]
- 46.Maderspacher F, Bucher G, Klingler M. Dev Genes Evol. 1998;208:558–568. doi: 10.1007/s004270050215. [DOI] [PubMed] [Google Scholar]
- 47.Berghammer A J, Klingler M, Wimmer E A. Nature (London) 1999;402:370–371. doi: 10.1038/46463. [DOI] [PubMed] [Google Scholar]
- 48.Strimmer K, von Haeseler A. Mol Biol Evol. 1996;13:964–969. [Google Scholar]
- 49.Higgins D G, Bleasby A J, Fuchs R. CABIOS. 1991;8:189–191. doi: 10.1093/bioinformatics/8.2.189. [DOI] [PubMed] [Google Scholar]