Abstract
Understanding cellular repair mechanisms in vivo has been advanced through the use of well-defined injury and repair models and their application to knockout and transgenic animals, primarily mice generated in a variety of background strains. However, little is known concerning the effect that mouse strain itself has on the interpretation and comparability of observations when the strain used for genetic manipulation is not the strain used to develop the model. We compared acute bronchiolar injury and repair in three strains of mice used in knockout mouse development (C57BL/6, 129/TerSv, and 129/SvEv) to the model strain (Swiss Webster) after treatment with the same dose of naphthalene and sacrificed at 1, 2, 4, 7, and 14 days after treatment. Extent of Clara cell toxicity and exfoliation was identical in the distal airways of all strains. There were significant strain-related differences in ciliated cell squamation, initiation and duration of proliferation, epithelial differentiation, and time to completion of epithelial repair. We conclude that ciliated cells play a prominent role in repair of distal airway injury, but that all phases of the repair process differ by strain. In addition, our findings reinforce that control animals must be of the same strain, ideally litter mates, when transgenic or knockout mice are used for the study of airway repair processes and mechanisms.
Defining the cellular mechanisms of acute tracheobronchial airway epithelial injury and subsequent repair has been hampered by the architectural and cellular complexity of the pulmonary conducting airways and their inaccessibility to external manipulation. One approach to better define the role of specific proteins in repair mechanisms and cellular responses to injury is the use of transgenic animals that overexpress proteins or knockout animals that have a protein deleted. Transgenic animals have successfully been used in defining the role of Clara cell secretory protein in oxidative stress 1 and toxicant bioaccumulation, 2 the role of epithelial T lymphocytes in infectious airway inflammation, 3 the role of αvβ6 integrin in airway inflammation, 4 the role of gelatinase B in bleomycin-induced fibrosing alveolitis and bronchiolization, 5 and the role of metalloproteinases (matrilysin) in tracheal epithelial repair. 6 These transgenic animals, however, are derived from several different strains of mice. Confounding the use of transgenic and knockout mice is the lack of unified studies to compare the differences in injury or disease response between the wild-type animals of the strains in which these genetic manipulations are made. Some of the most common strains of mice used for transgenic manipulation include the C57BL/6, 129/TerSv, and 129/SvEv mouse strains. Many of these strains of mice were originally developed based on their susceptibility to specific disease entities, such as cancer, or for their differences in sensitivity or metabolic response to specific xenobiotics. For example, the responsiveness to acetylcholine after oxidative stress is markedly different between C57BL/6, 129/J, and DBA/2J mice. 7 These types of strain-related differences raise the question as to the use of genetically manipulated mice and the wild-type strains from which they were derived, in defining molecular, cellular, and biochemical mechanisms of injury and repair.
In this study, we compared the injury and repair response of three mouse strains commonly used as platforms for genetic manipulation, C57BL/6, 129/TerSv, and 129/SvEv, to a well-defined model of bronchiolar epithelial injury and repair originally developed in the male Swiss Webster mouse. 8-11 The distal airways are frequent sites of epithelial injury because of several factors, including distribution of the toxicant through the branching airway structure, cellular composition of the bronchiolar epithelium, and the capability of cells in this region to activate and/or detoxify chemicals. We, and others, have exploited Clara cell metabolic activation of the simple polycyclic aromatic hydrocarbon, naphthalene, to model distal airway injury and subsequent wound healing. 10,12-14 A single intraperitoneal dose of naphthalene is an acute Clara cell toxicant that is both airway level and cell-type-specific. 9,15 In Swiss Webster mice, acute Clara cell injury and death are followed by a clearly defined progression of epithelial events: ciliated cell squamation, proliferation, migration, and differentiation that progresses in a proximal to distal direction. 8,10 By 14 days after injury, epithelial repair is considered complete. However, the role of the ciliated cell in bronchiolar repair after acute Clara cell injury has not previously been examined in detail.
The purpose of this study was twofold: 1) to characterize the role of the ciliated cell in repair of distal airway Clara cell injury, and 2) to define strain-specific differences in the injury and repair responses of distal airway epithelium between Swiss Webster mice and C57BL/6, 129/TerSv, and 129/SvEv mice. These strains were compared with respect to the extent of injury and the progression and duration of epithelial repair, including the onset and duration of cell proliferation, changes in ciliated cell size and conformation, abundance at various phases of repair, and re-establishment of preinjury steady state epithelial organization and differentiation.
Materials and Methods
Animal Housing and Treatment
Nine-week-old, male Swiss Webster (Charles River Breeding Laboratory, Wilmington, MA), C57BL/6 (Charles River), 129/SvEv (Washington University, St. Louis, MO), and 129/TerSv (Jackson Laboratories, Bar Harbor, ME) were housed in a HEPA-filtered cage rack and maintained on a 12/12 hour light/dark cycle with free access to food and water for at least 7 days before use.
Airway Epithelial Injury Induction and Tissue Treatment
Naphthalene (Aldrich Chemical Co., Milwaukee, WI) was administered by intraperitoneal injection of 20 mg/ml of naphthalene in corn oil (Mazola, Best Foods/CPC International Inc., Englewood Cliffs, NJ) at a dose of 200 mg/kg body weight. Control animals received an equivalent volume of corn oil carrier. Mice of all strains were killed at each of the following times after naphthalene injection: 1, 2, 7, and 14 days (DPN). All mice were injected with 50 mg/kg of 5-bromo-2′-deoxyuridine (BrdU; Fitzgerald Industries International, Inc., Concord, MA) 1 hour before euthanasia. At the time of necropsy, all animals were anesthetized with an overdose of pentobarbital sodium and exsanguinated. After euthanasia, the trachea was exposed by a ventral midline cervical incision and cannulated at the larynx.
The lungs from three treated and one control animal for each time point were collected for paraffin tissue sections. The diaphragm was punctured and the lungs were infused in the thorax via intratracheal cannula for 1 hour at 30 cm pressure with 1% paraformaldehyde in 0.1 mol/L of phosphate buffer (pH 7.4). Once removed from the thorax, the right cranial lobe was processed for scanning electron microscopy (SEM) and the middle and caudal lobes were processed for paraffin embedding. For paraffin sectioning, the lobes were embedded whole with the mediastinal surface down. Paraffin blocks were sectioned at 5 μm on a Reichert-Jung Supercut microtome and placed on Silane-Prep glass slides (Sigma Diagnostics, St. Louis, MO).
The lungs from three treated and one control animal for each time point were collected for high-resolution light microscopy and SEM. The diaphragm was punctured and the lungs were infused while in the thorax for 1 hour at 30-cm pressure with a mixture of 0.7% paraformaldehyde and 0.9% glutaraldehyde in cacodylate buffer (pH 7.4, 330 mOsm). 16 Once removed from the thorax, the left lobe was transected into three equal segments cut perpendicular to the long axis of the lung lobe. The tissues were postfixed in 1% osmium tetroxide in Zetterquist’s buffer, processed by large block methodology, and embedded cut surface down in Araldite 502 (Electron Microscopy Sciences, Fort Washington, PA) epoxy resin. 16 Araldite blocks were sectioned at 1 μm on a Sorvall JB-4 Porter-Blum microtome (Dupont Company Biotechnology Systems, Wilmington, DE), and stained with methylene blue/azure II with 0.5% sodium borate.
For SEM, the apical lobe of the right lung was affixed to a 1.2-cm 2 Corning cover glass (Corning Glass Works, Palo Alto, CA), mediastinal side down, with cyanoacrylate tissue glue (Nexaband; Veterinary Products, Phoenix, AZ). The lumena of the mediastinal airways extending from the lobar bronchus to the terminal bronchioles were opened by removing the dorsal half of the airway while immersed in phosphate-buffered saline. Microdissection was aided by the use of a Wild Heerbrugg dissecting microscope (Technical Instruments, San Francisco, CA). While affixed to the coverslip, the microdissected lungs were dehydrated in 10-minute washes of a graded ethanol series of 70%, 85%, 95%, and 100%. To remove airway secretions from the epithelial surfaces, the lobes were agitated briefly in a 50/50 solution of 100% alcohol and toluene, then taken to 100% toluene. The process was reversed until the tissue was back in the 100% alcohol. The dehydrated lobes were then bathed in hexamethyldisilizane (Electron Microscopy Sciences, Fort Washington, PA) for 5 minutes at room temperature. The lung lobes were glued to SEM chucks with Nexaband and allowed to air-dry overnight. The lungs were sputter-coated for 2 minutes with gold using a Polaron II ES100 sputter-coater (acceleration voltage 2.5 kV, 10 mA current in argon) (Energy Beam Sciences, Agawan, MA). The microdissected lobes were viewed and imaged with a Philips SEM 501 microscope (FEI Corporation, Hillsboro, OR).
Transmission Electron Microscopy
Thin sections (60 to 90 nm) were cut using a diamond knife on a LKB Nova ultramicrotome (LKB Bromma, Sweden). Sections were stained with uranyl acetate and lead citrate, and visualized with a Zeiss EM10 at 80 kV (Zeiss Microimaging, Thornwood, NY).
Definition of Airway Levels
The microdissected lungs of the four different mouse strains were examined by SEM (Figure 1) ▶ and the histological changes are described and quantified by airway level. Summary data are reported for three animals per group, with more than 10 distal airways examined per animal. The distal airways were defined as the three most distal generations of bronchioles. All airways were examined in detail including airway bifurcations and airway segments between bifurcations.
Figure 1.

A: This low-magnification SEM map illustrates the defined distal airway levels examined in this study using a microdissected apical lung lobe. B: A high magnification of a distal airway bifurcation (asterisk). The spatial relationship between ciliated and Clara cells is similar at both airway bifurcations and between bifurcations. Scale bars: 1 mm (A); 50 μm (B).
Immunohistochemistry
The avidin-biotin-peroxidase procedure as outlined by the supplier (Vector Laboratories, Burlingame, CA) was used to identify BrdU antibody-binding sites. In addition, before the peroxidase block, all sections were digested for 10 minutes at 60°C with HCl, followed by a 10-minute neutralization step in borate buffer. Sections were then exposed to 0.05% proteinase K (Sigma Chemical Co.) for 3 minutes followed by a nonspecific protein-blocking step with bovine serum albumin. The sections were incubated with a 1:100 dilution of polyclonal sheep anti-BrdU (Fitzgerald Industries International, Inc.) as the primary antibody in a humidity chamber at 4°C overnight. Secondary biotinylated rabbit anti-sheep IgG (H+L) antibody (Vector Laboratories) was used at 1:500 and 3,3′-diaminobenzidine (Sigma Chemical Co.) was used as the chromagen.
Proliferation
Cross-sections of whole lung, from each of three animals from each time point, including carrier controls were examined for BrdU-labeled nuclei. At least 10 longitudinal cross-sections from the distal airways of each animal of every strain were categorized by the number of BrdU-positive nuclei within the airway: 1, 2, 3, or >4. Distal airways were defined by their proximity to or connection with a terminal bronchiole. The percentage of positive-labeled nuclei per category was calculated for each airway level and for each of the following days after naphthalene: 0, 1, 2, 4, 7, and 14. The data were entered into Cricket Graph III, version 1.0 (Computer Associates International, Inc.) for graphical analysis.
Ciliated Cell Size and Number of Ciliated Cells per Unit Surface Area
Scanning electron microscopic images were collected from one surface of specific airway segments between bifurcations for the distal three airway generations of three animals at each of the following time points: 0 (corn oil control), 2, 7, and 14 DPN. The time points selected represent the following phases of response in Swiss Webster mice: steady state, the phase of maximal squamation, near complete repair, and complete repair. The airways were imaged at ×750 magnification and surface area of 10 ciliated cells selected at random was measured using Scion Image (NIH). Omitted from counting were those ciliated cells present on any curved surface. For numeric density calculations, the same SEM images were used. The total airway surface area of each image was kept constant and the total number of ciliated cells per image was counted. The images were counted in random order to decrease bias based on mouse strain, airway level, or days after injury.
Statistics
All data from ciliated cell surface area and number of ciliated cells per unit surface area were imported into Stat View (Abacus Concepts, Berkeley, CA) for analysis of variance and Bonferroni/Dunn post hoc analysis. Differences between values within the same mouse strain and differences between Swiss Webster mice and C57BL/6, 129/TerSv, or 129/SvEv mice were assessed with significance determined by Bonferroni/Dunn at P < 0.05.
All proliferation data were also imported into Stat View for analysis. Association between strains and the number of BrdU-positive cells by airway level were assessed by chi-square analysis. 17 For all analyses a P value of <0.05 was considered statistically significant.
Results
Pathology
Control Mice All Strains
The epithelium lining distal bronchioles was similar in all four mouse strains (Figure 2) ▶ . Clara cells were arranged in slightly irregular longitudinal rows no more than two cells wide, and oriented parallel to the long axis of the airway (not shown). The rows of Clara cells were separated by discontinuous rows of ciliated cells no more than one cell wide. All Clara cells appeared to have at least one surface contact with a ciliated cell (Figure 1B) ▶ . Clara cells had large luminal surface areas and prominent apical projections that protruded into the lumen, whereas ciliated cells were angular with much smaller surface areas and had short cilia (Figure 2 ▶ ; A, B, C, E, and G). There was no phenotypic difference between the surface epithelium of airway bifurcations and the epithelium of airway segments between bifurcations (Figure 1B) ▶ .
Figure 2.
High-resolution light micrographs of the strain differences in response of ciliated cells to acute Clara cell-specific injury at 2 DPN. Before injury (A, C, E, and G), Clara cells can be identified by their apical domes that are raised above the epithelial surface (arrows). Two days after naphthalene injury (B, D, F, and H), there are notable strain-related differences in the terminal bronchiolar epithelial response after the same dose of naphthalene per body weight. Although complete Clara cell exfoliation occurs in all strains, the extent of squamation by the surviving ciliated cells varies by strain but not by the extent of basement membrane exposure. Ciliated cell squamation is less extensive in 129/SvEv mice (H), which have extensive areas of denuded basement membrane (arrowhead) than in 129/TerSv mice that have less basement membrane exposure (F). Scale bar, 20 μm (H).
Swiss Webster Mice
Two days after naphthalene treatment (2 DPN), the distal airways were lined by a diffuse sheet of large polygonal cells with short surface microvilli and prominent intercellular demarcation. The cells were attenuated to low cuboidal with intercellular borders depressed from the luminal surface plasma membrane (Figure 2B) ▶ . The majority of the cells had central tufts of cilia, whereas a few cells lacked surface differentiation or had central, slightly raised blebs. Those cells lacking surface differentiation had short, scattered, individual, peripheralized cilia or had a central circular area that was smooth and lacked surface microvilli. The mean ciliated cell surface area at 2 DPN was 1.5-fold greater than the surface area found in control animals (Figure 3) ▶ . The mean number of ciliated cells per unit surface area was half that of steady state (Figure 4) ▶ .
Figure 3.

Changes in surface area of individual ciliated cells throughout the course of repair in four strains of mice treated with corn oil (CO, not injured) and naphthalene in corn oil (2, 7, or 14 days after treatment). The ciliated cells of all four stains of mice have a similar surface area before injury (CO). However, immediately after injury, the surface area of ciliated cells in Swiss Webster mice change little, whereas the change in surface area in 129/TerSv mice is extreme. C57BL/6 and 129/SvEv mice had similar changes in ciliated cell surface area but 129/SvEv mice had extensive basement membrane exposure. This suggests that the ability of ciliated cells to squamate in response to Clara cell exfoliation is related to strain and not to the extent of injury. Also note that the pattern of ciliated cell surface area change is similar among all four strains, with the greatest spreading being attained directly after Clara cell exfoliation and decreasing to the surface area of control epithelium at the end of repair (14 days). Data reported are the mean ± 1 SD for three animals per time point.
Figure 4.

Number of ciliated cells per unit of distal airway surface area in four strains of mice treated with corn oil (CO) and naphthalene in corn oil (2, 7, or 14 days after treatment). Number of ciliated cells per unit of surface is similar for all four strains of mice at steady state conditions (CO). However, the pattern of change in the number of ciliated cells per unit surface area during the course of repair varies by both the amount of basement membrane exposure and the plasticity of ciliated cells. In all four strains of mice, the number of ciliated cells per unit surface area was a good measure of the return to steady state (14 days). The period of low ciliated cell number in each of the strains corresponds to the period of maximal proliferation. Data reported are the mean ± 1 SD for three animals per time point.
At 7 DPN, the distal airways were lined by integrated Clara and ciliated cells interspersed with patches of ciliated cells arranged in small clusters to linear cords three to four cells wide. In comparison to the ciliated cells that were integrated with Clara cells, these cords of ciliated cells had a larger surface area and the luminal surface was completely covered by cilia and microvilli. At several sites, individual ciliated cells with a large surface area were surrounded by Clara cells. The mean ciliated cell surface area had decreased compared to 2 DPN, to a surface area equal to controls and the number of ciliated cells per unit surface area had increased more than 2.5-fold compared to 2 DPN and remained at that number at 14 DPN.
At 14 DPN, the majority of ciliated cells lining the distal airways were integrated with Clara cells. A few clusters of ciliated cells with prominent, elongate cilia were scattered randomly within the distal airways, and several of the ciliated cells still had a large surface area. The mean ciliated cell surface area decreased two-thirds below that found at 7 DPN and in controls, to an area averaging 24% smaller than the ciliated cell surface area of epithelium at steady state. The mean number of ciliated cells per unit surface area was 1.3-fold greater than the number at steady state.
C57BL/6 Mice
When compared to the mean ciliated cell surface area of Swiss Webster mice at steady state, the surface area of the C57BL/6 was smaller (Figure 3) ▶ . In addition, C57BL/6 mice had 1.6-fold more ciliated cells per unit of airway surface area when compared to Swiss Webster mice (Figure 4) ▶ . Statistically significant differences in mean ciliated cell surface area and in number of ciliated cells per unit of airway surface area in comparison to Swiss Webster mice are summarized in Tables 1 and 2 ▶ ▶ . At 2 DPN, the diffuse sheets of ciliated cells had fewer cells that lacked surface differentiation and had fewer with reduced or scattered cilia. The majority of ciliated cells had central membrane invaginations. By high-resolution light microscopy, foci of epithelial hyperplasia were present primarily at airway bifurcations and fewer were at airway segments between bifurcations. Some cells at airway segments between bifurcations had swollen ciliated cells that were raised above the surrounding epithelium. The mean ciliated cell surface area increased approximately threefold compared to the surface area at steady state and 1.8-fold greater than in Swiss Webster mice at 2 DPN. In addition, the number of ciliated cells per unit surface area increased compared to control animals, a difference of more than fourfold between Swiss Webster and C57BL/6 mouse strains.
Table 1.
Summary of Statistical Significance—Differences in Ciliated Cell Surface Area for All Strains when Compared to Swiss Webster Mice Distal Airways at the Same Time Point
| Day 0 | Day 2 | Day 7 | Day 14 | |
|---|---|---|---|---|
| C57BL/6 | + | + | + | + |
| 129/TerSv | + | + | + | + |
| 129/SvEv | + | + | + | + |
+, Bonferroni-Dunn, P ≤ 0.05; −, not significant.
Table 2.
Summary of Statistical Significance—Differences in Ciliated Cell Number Surface Area for All Strains when Compared to Swiss Webster Mice Distal Airways at the Same Time Point
| Day 0 | Day 2 | Day 7 | Day 14 | |
|---|---|---|---|---|
| C57BL/6 | + | − | + | − |
| 129/TerSv | + | + | − | − |
| 129/SvEv | + | − | − | + |
+, Bonferroni-Dunn, P ≤ 0.05; −, not significant.
At 7 DPN, the majority of the epithelium was composed of ciliated cells integrated with Clara cells. These areas were multifocally broken by cords or small clusters of ciliated cells with large surface areas. The cords of cells ranged from two to three cells wide and five to eight cells long. The ciliated cell surface area decreased compared to the surface area at 2 DPN, but was still approximately twofold greater than controls and in Swiss Webster mice at 7 DPN. The number of ciliated cells per unit airway surface area was decreased by two-thirds compared to 2 DPN, and was similar to Swiss Webster mice at 7 DPN.
At 14 DPN, the distal airways were lined by integrated ciliated and Clara cells and no ciliated cell cords or clusters were present. The mean ciliated cell surface area was two-thirds of that at 7 DPN, but still one-third larger than the ciliated cells at steady state and twofold greater than in Swiss Webster mice at 14 DPN. The mean number of ciliated cells per unit surface area was greater than at 7 DPN, and similar to the number at steady state. The number of ciliated cells per unit surface area at 14 DPN in C57BL/6 was greater than in Swiss Webster mice at 14 DPN.
129/TerSv Mice
When compared to the mean ciliated cell surface area of Swiss Webster mice at steady state, ciliated cells in 129/TerSv mice were smaller (Figure 3) ▶ . In addition, 129/TerSv mice had 1.5-fold more ciliated cells per unit surface area than Swiss Webster mice at steady state (Figure 4) ▶ . Statistically significant differences in mean ciliated cell surface area and in number of ciliated cells per unit of airway surface area in comparison to Swiss Webster mice are summarized in Tables 1 and 2 ▶ ▶ . At 2 DPN, the sheets of ciliated cells were extremely attenuated (Figure 2F) ▶ and tufts of cilia were extremely reduced in length. In some areas intercellular borders were poorly delineated. These areas were identified by high-resolution light microscopy and transmission electron microscopy as areas of denuded basement membrane (Figures 5 and 6) ▶ ▶ . At some bifurcations, approximately four cells on either side of the crest between airways were cuboidal cells that were either ciliated or slightly domed (Figure 5) ▶ . The ciliated cell surface area increased 12.5-fold over the surface area at steady state, which was 6.5-fold greater than in Swiss Webster mice at the same period after injury (Figure 3) ▶ . In addition, the number of ciliated cells per unit of airway surface area decreased to 20% of the number of ciliated cells at steady state, resulting in fewer cells when compared to Swiss Webster mice at the 2 DPN time point (Figure 4) ▶ .
Figure 5.
Surviving nonciliated cell populations at airway bifurcations. A: At 2 DPN, when all injured Clara cells had exfoliated, 129/TerSv and 129/SvEv mice had areas of basement membrane exposure, not present in Swiss Webster and C57BL/6 mice (arrow). In all four strains, but most evident in mice with extensive squamation of ciliated cells (129/TerSv), a small population of nonciliated cells located at airway bifurcations survived the acute injury phase (B). These cells were often missed by cross-section analysis (A) because of their random location around bifurcations. Although increases in proliferating cell abundance as evidenced by cellular hyperplasia (asterisk) can be centered around these sites at bifurcations (C), increases in proliferating cell abundance is not limited to bifurcations (D). Scale bar, 10 μm (D).
Figure 6.
Electron and light micrographs of the basement membrane exposure in 129/TerSv (A and C) and 129/SvEv (B and D) mice at 2 DPN. During the acute injury phase, basement membrane is exposed in 129/TerSv and 129/SvEv mouse strains only. The exposure is most severe in 129/SvEv mice, which demonstrates edematous separation (E) of the basement membrane (arrowhead) from the underlying layer of fibroblasts (F). Note the elongated cytoplasmic extensions of the fibroblasts that extend to and make contact with the basement membrane. The arrow denotes the leading edge of the ciliated cell pseudopodia. The asterisk denotes squamated ciliated cells. TEM bar, 3.5 μm; LM bar, 10 μm.
At 7 DPN, the majority of the epithelial cells lacked surface differentiation, whereas others were ciliated but had peripheralized microvilli or had large cytoplasmic invaginations at the cilia tuft (Figure 7) ▶ . By high-resolution light microscopy the airway epithelium was attenuated, and had multifocal areas of hyperplasia at airway bifurcations and airway segments between bifurcations (Figure 5) ▶ . At airway segments between bifurcations, some areas of basement membrane were covered by thin cytoplasmic extensions from adjacent cells. The ciliated cell surface area was decreased slightly from the 2 DPN time point, which was greater than eightfold larger than the surface area of the ciliated cells of Swiss Webster mice at 7 DPN. In addition, the number of ciliated cells per unit airway surface area was low at 7 DPN (similar to 2 DPN) to a number one-fourth of that in Swiss Webster mice at the same time after injury (Figure 4) ▶ .
Figure 7.
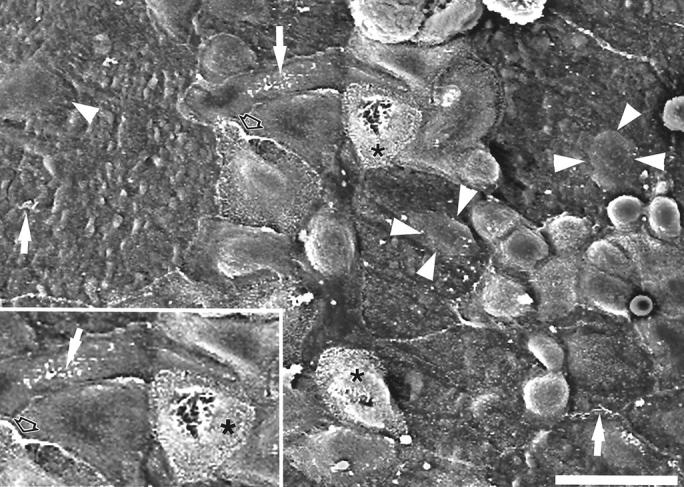
Scanning electron micrograph of ciliated cell dedifferentiation as seen in 129/TerSv mice at 7 DPN. The appearance of ciliated cells during the different phases of repair is dynamic and consistent for all strains of mice. Ciliated cells with smaller surface areas developed cytoplasmic invaginations of the surface plasma membrane, partially internalizing the cilia tuft (asterisk, in inset). Before and during the proliferation phase, ciliated cells lost or greatly reduced their surface microvilli and the cilia became short and disorganized on the surface membrane (arrows) and often migrated to the edges of the cell (open arrows). Note the extreme ciliated cell surface area of some cells and the prominence of their nuclei (arrowheads). Scale bar, 20 μm.
At 14 DPN, the airways were lined by integrated ciliated and Clara cells and all lacked ciliated cell cords or clusters. The terminal bronchioles were often lined by low cuboidal epithelium and few areas of epithelial hyperplasia were scattered multifocally at airway segments between bifurcations. Multiple small clusters of mucous cells were scattered throughout the distal airways in some mice. The mean ciliated cell surface area dropped eightfold, restoring the surface area to levels similar to the surface area at steady state and greater than the ciliated cells of Swiss Webster mice (Figure 3) ▶ . In addition, the mean number of ciliated cells per unit surface area increased fivefold, to a number 18% greater than the number of ciliated cells at steady state and 1.4-fold greater than the number of ciliated cells in Swiss Webster mice at 14 DPN (Figure 4) ▶ .
129 SvEv Mice
When compared to the mean ciliated cell surface area of Swiss Webster mice at steady state, ciliated cells of 129/SvEv mice were 50% smaller (Figure 3) ▶ . In addition, 129/SvEv mice had 1.6-fold more ciliated cells per unit surface area than Swiss Webster mice at steady state (Figure 4) ▶ . Statistically significant differences in mean ciliated cell surface area and in number of ciliated cells per unit of airway surface area in comparison to Swiss Webster mice are summarized in Tables 1 and 2 ▶ ▶ . At 2 DPN, the ciliated cells were extremely attenuated and there was extensive basement membrane exposure (Figure 2H ▶ and Figure 6 ▶ ). Tufts of cilia were extremely reduced in length. Rare, small clusters of epithelial cells at bifurcations were less squamated and had prominent intercellular borders. The ciliated cell surface area increased 6.2-fold from steady state surface area, which was twofold greater than the ciliated cell surface area in Swiss Webster mice (Figure 3) ▶ . In addition, the number of ciliated cells per unit airway surface area decreased 4.2-fold over the number of ciliated cells at steady state, resulting in little difference when compared to Swiss Webster mice at the same time point (Figure 4) ▶ .
At 7 DPN, the majority of the epithelium was composed of cells lacking surface differentiation. Clara cells with prominent apical domes were present multifocally in small clusters and ciliated cells were often individual and scattered among the undifferentiated cells. By high-resolution light microscopy, sheets of swollen cells were multifocal and separated by fine cytoplasmic extensions from adjacent cells. The ciliated cell surface area decreased four-fifths of the 2 DPN time point to an area 2.5-fold greater than Swiss Webster mice (Figure 3) ▶ . In addition, ciliated cell numbers decreased to two-thirds of 2 DPN, to a number that was 20% of the number of ciliated cells in Swiss Webster mice at the same time after injury (Figure 4) ▶ .
At 14 DPN, the airways were lined by integrated ciliated and Clara cells and no ciliated cell cords or clusters were present. The mean ciliated cell surface area decreased more than sixfold, to an area two-thirds that of Swiss Webster ciliated cells and similar to the 129/SvEv ciliated cell surface area at steady state (Figure 3) ▶ . In addition, the mean number of ciliated cells per unit airway surface area increased sixfold, to a number approximately equal to the number of ciliated cells at steady state and 1.3-fold greater than the number of ciliated cells in Swiss Webster mice (Figure 4) ▶ .
Proliferative Response
Swiss Webster Mice
In carrier controls, 94% of the distal airways lacked BrdU-positive nuclei (Figure 8) ▶ . The remaining 6% had no more than one positive nucleus per airway profile. At 1 DPN, 38% of the distal airways had BrdU-positive nuclei and 17% had two or more positive nuclei per airway. At 2 DPN, 100% of the airways had BrdU-positive nuclei, and 43% had four or more positive nuclei per airway. BrdU-positive nuclei were distributed throughout the bronchioles, including airway bifurcations as well as airway segments between bifurcations. At 4 DPN, only 18% of the distal airways had BrdU-positive nuclei and 15% had no more than one positive nucleus per airway. At 7 DPN, 17% of the airways had BrdU-positive nuclei and 2% had two to three positive nuclei per airway. At 14 DPN, none of the airways had BrdU-positive nuclei.
Figure 8.

Distribution plots comparing the changes in proliferating cell abundance during the course of repair measured as the percentage of airway profiles with various numbers of BrdU-positive cells. The temporal pattern, peak, and duration of the increase in proliferating cells was different in each of the four strains of mice examined. Proliferating cell abundance increased and peaked earliest in Swiss Webster mice and the peak was later and proliferation had a longer duration in 129/TerSv mice. The delay in proliferating cell abundance in 129/TerSv mice was associated with the greater surface area achieved by the ciliated cells of this strain of mouse. This figure illustrates the sum of observations for all three animals for each strain of mouse.
C57BL/6 Mice
In carrier controls, 94% of the distal airways lacked BrdU-positive nuclei and the 6% that were positive had no more than one per airway (Figure 8) ▶ . At 1 DPN, 9% of the airways had BrdU-positive nuclei and all airways had no more than one positive nucleus per airway. By 2 DPN, 72% of the airways had positive nuclei, and 49% had two or more positive nuclei per airway. At 4 DPN, 91% of the distal airways had BrdU-positive nuclei and 38% had four or more BrdU-positive nuclei per airway. BrdU-positive nuclei were distributed throughout the bronchioles, including airway bifurcations as well as airway segments between bifurcations. By 7 DPN, 40% of the airways had positive nuclei and 13% had two or more positive nuclei per airway. At 14 DPN, 9% of the airways had BrdU-positive nuclei and none had more than one positive nucleus per airway. The differences in the extent of proliferation compared to Swiss Webster mice were statistically significant at 1, 2, 4, and 7 DPN (Table 3) ▶ .
Table 3.
Summary of Statistical Significance—Differences in Proliferation for All Strains when Compared to Swiss Webster Mice Distal Airways at the Same Time Point
| Day 0 | Day 1 | Day 2 | Day 4 | Day 7 | Day 14 | |
|---|---|---|---|---|---|---|
| C57BL/6 | − | + | + | + | + | − |
| 129/TerSv | − | + | + | + | + | + |
| 129/SvEv | − | + | + | + | + | − |
+, chi-squared, P ≤ 0.05; −, not significant.
129/TerSv Mice
In carrier controls, 95% of the distal airways had no BrdU-positive nuclei and of the 5% that were positive, none had more than one positive nucleus per airway (Figure 8) ▶ . At 1 DPN, 2% of the distal airways had BrdU-positive nuclei and none had more than one positive nucleus per airway. At 2 DPN, 38% of the airways had BrdU-positive nuclei and 17% had two or more positive nuclei per airway. At 4 DPN, 58% of the airways had BrdU-positive nuclei, and 32% had two or more positive nuclei per airway. At 7 DPN, 94% of the distal airways had BrdU-positive nuclei, and 47% of those had more than two positive nuclei per airway. BrdU-positive nuclei were distributed throughout the bronchioles, including airway bifurcations as well as airway segments between bifurcations. At 14 DPN, 34% of the airways had BrdU-positive nuclei and 7% had two or more positive nuclei per airway. The differences in the extent of proliferation compared to Swiss Webster mice were significant at 1, 2, 4, 7, and 14 DPN (Table 3) ▶ .
129/SvEv Mice
In carrier controls, 97% of the distal airways had no BrdU-positive-labeled nuclei, and the 3% of airways that did, all had two positive nuclei per airway (Figure 8) ▶ . At 1 DPN, 7% of the airways had BrdU-positive nuclei, and none had more than one BrdU-positive nucleus per airway. At 2 DPN, 71% of the distal airways had positive labeled nuclei and 22% had four or more positive nuclei per airway. At 4 DPN, 88% of the distal airways had BrdU-positive nuclei with 61% having four or more positive nuclei per airway. BrdU-positive nuclei were distributed throughout the bronchioles, including airway bifurcations as well as airway segments between bifurcations. At 7 DPN, 51% of the airways had BrdU-positive nuclei with 28% having four or more positive nuclei per airway. At 14 DPN, 12% of distal airways had BrdU-positive nuclei and 10% had no more than one positive labeled nucleus per airway. The differences in the extent of proliferation compared to Swiss Webster mice were statistically significant at 1, 2, 4, and 7 DPN (Table 3) ▶ .
Discussion
The purpose of this study was twofold: 1) to define the ciliated cell response subsequent to Clara cell injury, and 2) to identify strain-specific differences in the repair of acute bronchiolar epithelial injury between Swiss Webster and C57BL/6, 129/TerSv, and 129/SvEv mice. These four mouse strains were identical with respect to the extent of Clara cell injury to the distal airways after a single intraperitoneal dose of naphthalene, but varied greatly in the temporal pattern of epithelial repair, including the onset and duration of cell proliferation; changes in ciliated cell size; conformation and abundance at various phases of repair; and re-establishment of preinjury steady state epithelial organization, density, and differentiation (summarized in Table 4 ▶ ). We used a well-defined model of acute airway epithelial injury that is both cell-type- and airway level-specific and has a well-characterized pattern of repair in Swiss Webster mice that includes cell squamation, proliferation, migration, and cytodifferentiation. 8 The naphthalene-induced injury/repair model uses a Clara cell-specific toxicant that targets the distal bronchioles and spares the ciliated cell. In addition to significant differences in the temporal pattern of airway epithelial injury and repair between all four strains of mice, we found commonalities in the sequence of events and in the plasticity of the ciliated cell response.
Table 4.
Summary of Strain Differences in Repair Response of Bronchiolar Epithelium Following Naphthalene-Induced Acute Clara Cell Toxicity
| Day 0 | Day 1 | Day 2 | Day 4 | Day 7 | Day 14 | |
|---|---|---|---|---|---|---|
| Clara cell exfoliation | ||||||
| Swiss-Webster | Complete* | |||||
| C57BL/6 | Complete | |||||
| 129/TerSv | Complete | |||||
| 129/SvEv | Complete | |||||
| Ciliated cell squamation | ||||||
| Swiss-Webster | − | ND | ↑ | ND | − | − |
| C57BL/6 | − | ND | ↑↑ | ND | ↑ | − |
| 129/TerSv | − | ND | ↑↑↑↑ | ND | ↑↑↑↑ | − |
| 129/SvEv | − | ND | ↑↑ | ND | ↑↑ | − |
| Cell proliferation | ||||||
| Swiss-Webster | − | ↑↑ | ↑↑↑ | ↑ | ↑ | − |
| C57BL/6 | − | − | ↑↑ | ↑↑↑ | ↑↑ | − |
| 129/TerSv | − | − | ↑↑ | ↑↑ | ↑↑↑ | ↑↑ |
| 129/SvEv | − | − | ↑↑ | ↑↑↑ | ↑↑ | ↑ |
| Ciliated cell density | ||||||
| Swiss-Webster | − | ND | ↓ | ND | − | − |
| C57BL/6 | − | ND | ↑ | ND | − | − |
| 129/TerSv | − | ND | ↓↓↓ | ND | ↓↓↓ | − |
| 129/SvEv | − | ND | ↓↓↓ | ND | ↓↓↓ | − |
*, Clara cell exfoliation did not differ in the distal airways by strain.
↑ indicates degree of change from control (Day 0) for that strain; − not different from control (day 0); ND, no data at this time point.
Several studies have focused on strain-related differences in the biological functions of the airway epithelium in mice, including responses to inhaled gases, 7,18,19 particles, 20 airway hypersensitivity, 21 and inflammation. 19 However, none of these studies have focused on the impact that mouse strain has on the biological activities of a surviving cell population throughout the course of repair. All of these studies have identified marked strain-dependent differences in the biological responses to lung injurants. A comparison of the studies emphasizes that the relative sensitivity of any one strain varies with the class of injurants, the analytical approach, and the time point during the response when the analysis was undertaken. We have found that the same surviving cell phenotype has a very different temporal pattern and extent of response depending on the strain. These differences occur not only in the extent of proliferation of individual cells, but also the time course throughout which proliferation occurs and the rate at which the epithelial cells repair the injured site.
Based on SEM, all Clara cells were removed from the most distal three airway generations, with the exception of branch points, in all four strains of mice. A similar surviving cell population at branch points has also been described by Stripp and colleagues. 12 In their study, a population of cells at airway bifurcations expressed CC10 mRNA before proliferation suggesting that these cells were either Clara cells that survived injury or an immature Clara cell population. In our study, we were able to demonstrate by SEM that some of these cells maintain a Clara cell phenotype although most squamate in response to the surrounding cell loss.
Mouse strain-related differences were present in the response of the surviving cell population, ciliated cells, to Clara cell injury and exfoliation. In all four strains of mice, ciliated cells demonstrated dynamic changes in terms of their ability to alter their shape in response to cell loss or increases in cell density, as well as their ability to alter their surface appearance through changes in the plasma membrane, microvilli, and cilia. Surviving ciliated cells in Swiss Webster mice respond to cytotoxic Clara cell injury and exfoliation by squamating and sending cytoplasmic pseudopodia beneath necrotic Clara cells before their exfoliation, leaving the basement membrane undenuded as previously described. 8 The response of ciliated cells in C57BL/6 mice was similar to that of Swiss Webster mice, but large areas of basement membrane were left denuded in 129/TerSv and 129/SvEv mice. The amount of basement membrane exposure in both the 129/TerSv and 129/SvEv strains of mice seemed excessive relative to the abundance and size of the ciliated cell population at steady state when compared to the Swiss Webster and C57BL/6 mice where denudation was not apparent. It has been well established that the Clara cell is the cellular target in naphthalene-induced injury, 9,15 however it is possible that the loss of Clara cells from the basement membrane was too rapid in 129/TerSv and 129/SvEv mice for the ciliated cells to completely respond and maintain an intact epithelial barrier. In all four strains of mice, ciliated cells demonstrated extreme plasticity in their ability to squamate, especially the ciliated cells of 129/TerSv mice (12.5-fold increase in surface area compared to steady state). Yet the basement membrane was left exposed. A possible explanation is that some ciliated cells were lost as bystanders of acute Clara cell injury. Regardless, it seems that ciliated cells do not have the same cytokinetic ability to squamate and cover the basement membrane in response to Clara cell loss in all strains of mice. Additionally, the increase in number of cells lacking surface differentiation was proportional to the increased abundance of proliferating cells in these strains of mice. These phenotypic changes in the ciliated cell population during repair suggest that the ciliated cell is capable of undergoing biological changes that allow it to proliferate and take an active role in epithelial repair.
In this study ciliated cells demonstrated a remarkable plasticity in their efforts to form an epithelial barrier after acute exfoliation of injured Clara cells. A novel finding in this study, facilitated by the coupling of microdissection with SEM, is that ciliated cell plasticity also includes resorption of cilia to yield a distal airway epithelium that lacks the characteristics of the two predominant differentiated cell types found in the distal airways: apical protrusions and cilia. We observed directly, using SEM, large membrane invaginations in the center of squamating ciliated cells that engulfed the cilia present on the cell surface. Resorption of specialized membrane structures is not a new concept. Several diverse organisms, including protozoa, 22, 23 sea urchins, 24 and vertebrate rat kangaroo cells 25 resorb cilia. Rieder and colleagues, 25 using the rat Kangaroo cell line PtK1, documented that resorption of cilia occurs before mitosis and speculated that the ciliary tubules were “re-used” during cell division. When sea urchin embryo cells are disaggregated and then allowed to reaggregate, the cilia are resorbed and the epithelial cells take on the appearance of a less differentiated cell as the epithelium sorts and the embryo reassembles. 24 Ciliary resorption is hypothesized to be related to the ability of the sea urchin epithelial cells to migrate. It has been speculated that resorption of the tubulin-rich ciliary structure is necessary before cell division can occur. Presumably the tubulin is recycled for use in spindles. We suggest that a similar phenomenon may be occurring in squamated bronchiolar ciliated cells, perhaps triggered by their change in shape in response to Clara cell injury.
In the adult lung the ciliated cell is regarded as terminally differentiated. This is based primarily on the low labeling index of this cell type in long-term labeling studies and on the pattern of proliferation described in previous injury models that injure primarily ciliated cells (such as NO2 and O3). 26-28 Our current study found that the ciliated cell is a much more plastic phenotype than was previously appreciated. When distal airway Clara cells are injured by naphthalene and exfoliate, the remaining ciliated cells resorb their cilia to return to a less-differentiated cell phenotype (dedifferentiation) and may even be capable of transdifferentiation. Dedifferentiation of conducting airway epithelial cells during wound healing in the lung has been previously described in detail by Erjefalt and colleagues. 29 Using the tracheal scrape model in guinea pigs, they clearly describe the presence of large flattened epithelial cells that migrate from the edge of the wound and form a barrier as early as 8 hours after wounding. These cells lack markers of cell surface differentiation. Although it is clear that the ciliated cells remaining after Clara cell injury undergo a similar phenotypic shift to an undifferentiated cell type, what is less clear is what role this flattened epithelial cell type plays in repopulation of the distal airways. Erjefalt and colleagues 29 were unable to determine whether squamated cells were also the actual proliferating cell population because of the presence of basal cells, which are known to be capable of proliferation. In the distal airways, basal cells are not present and proliferation is not restricted to airway bifurcations where neuroendocrine cells have been shown to congregate. 14,30 We hypothesize that the ciliated cell is not terminally differentiated and plays a dynamic role in the repair of airway epithelium through the process of transdifferentiation. A similar switch in phenotype has previously been described for another lung epithelial cell type thought to be terminally differentiated: alveolar type 1 cells that convert to type 2 cells in vitro. 31 Indeed, the issue of bronchiolar-ciliated cell transdifferentiation has interesting parallels with alveolar cells in that extracellular factors and changes in cell shape may play a prominent role in signaling phenotype shifts in the bronchiolar cells as suggested by Danto and colleagues 31 for alveolar cells. Clearly further studies are needed to clarify the cellular events involved in transdifferentiation of airway epithelium and to identify changes in the composition of the basement membrane in the airways of mice injured by naphthalene.
The onset and duration of proliferation was different for each of the four strains of mice, although the extent of Clara cell exfoliation was the same. In those strains whose ciliated cell response resulted in complete coverage of the basement membrane, proliferation began and ended more rapidly than in those strains whose ciliated cells squamated more or where ciliated cells failed to completely cover the basement membrane. In addition, although 129/TerSv mice exhibited the greatest increase in ciliated cell surface area during squamation, they subsequently exhibited lower numbers of proliferating cells initially, a delayed peak in proliferative rate, and greater numbers of proliferating cells later in repair when compared to 129/SvEv mice that also had exposed basement membrane. Although extensive cell stretch has been shown to have a positive effect on endothelial cell survival, 32 our study suggests the opposite effect for airway epithelial cells because the strain whose ciliated cells achieved the greatest surface area (129/TerSv), was also the strain with the greatest delay and prolongation of the proliferation phase. It seems that the greater the extent of cell squamation the greater the delay in onset of cell proliferation. The strain difference in the ability of ciliated cells to squamate has an inverse regulatory effect on the proliferative rate in the injury target zone, the distal airways.
The repair phases of migration and cytodifferentiation were marked by the return of ciliated cell abundance and surface area, as well as their integration with Clara cells, to a phenotypic organization consistent with steady state conditions. In all four strains of mice, an epithelial organization consistent with steady state for each strain was reached by 14 DPN, but for one strain, Swiss Webster, it was earlier. This occurred regardless of the varied responses during squamation and proliferation.
In summary, phenotype-specific acute bronchiolar epithelial injury exposed profound differences in the temporal pattern of injury and repair of four strains of mice: Swiss Webster, C57BL/6, 129/TerSv, and 129/SvEv. These strain-related differences centered on the biology of one cell type, the ciliated cell. This study demonstrates the importance of defining the phases of injury and repair by strain of mouse and in using like strains of mice for defining mechanisms of acute airway epithelial injury and repair. The varied responses in different strains of mice greatly emphasizes the need to base assumptions regarding parameters of lung injury on a clear understanding of the degree of resistance or sensitivity of the specific mouse strain under experimentation. The impact that strain differences have on the pattern of airway epithelial repair after acute injury can have a profound impact on the interpretation of biological responses in genetically modified mice. The potential for redundancies in genes regulating biological processes present significant difficulties in the interpretation of their role in airway epithelial repair when using genetically modified mice. Strain differences in the pattern of responses, such as those demonstrated here will only compound interpretive problems if not taken into account. Further, this study shows that the ciliated cell is capable of remarkable plasticity of cell shape and phenotypic expression and documents that ciliated cells play a prominent but strain-variable role in distal bronchiolar repair. Defining repair parameters for additional mouse strains that have not been previously evaluated will become more important with the broader use of transgenic and knockout mouse variants. Our findings provide support for good laboratory practice: that control animals must be of the same strain, ideally litter mates, when transgenic or knockout mice are used for the study of airway repair processes and mechanisms.
Footnotes
Address reprint requests to Charles G. Plopper, Ph.D., Dept. of Anatomy, Physiology, and Cell Biology, School of Veterinary Medicine, University of California at Davis, Davis, CA 95616. E-mail: cgplopper@ucdavis.edu.
Supported by grants from the National Institute of Environmental Health Sciences (ES04311, ES04699, ES06700, ES05707, NHLBI, HL07013, and HL47328 to R. M. S.), the American Lung Association Research Training Fellowship Program (to G. W. L.), the National Institute of Environmental Health Sciences Training Fellowship Program (to G. W. L.), a California Tobacco-Related Diseases Research Program (grant 6KT-0306 to L. S. V.), and the Alan W. and Edith L. Wolff Charitable Trust (to R. M. S.).
References
- 1.Mango GW, Johnston CJ, Reynolds SD, Finkelstein JN, Plopper CG, Stripp BR: Clara cell secretory protein deficiency increases oxidant stress response in conducting airways. Am J Physiol 1998, 275:L348-L356 [DOI] [PubMed] [Google Scholar]
- 2.Stripp BR, Lund J, Mango GW, Doyen KC, Johnston C, Hultenby K, Nord M, Whitsett JA: Clara cell secretory protein: a determinant of PCB bioaccumulation in mammals. Am J Physiol 1996, 271:L656-L664 [DOI] [PubMed] [Google Scholar]
- 3.King DP, Hyde DM, Jackson KA, Novosad DM, Ellis TN, Putney L, Stovall MY, Van Winkle LS, Beaman BL, Ferrick DA: Cutting edge: protective response to pulmonary injury requires gamma delta T lymphocytes. J Immunol 1999, 162:5033-5036 [PubMed] [Google Scholar]
- 4.Huang XZ, Wu JF, Cass D, Erle DJ, Corry D, Young SG, Farese Jr R, Sheppard D: Inactivation of the integrin beta 6 subunit gene reveals a role of epithelial integrins in regulating inflammation in the lung and skin. J Cell Biol 1996, 133:921–928 [DOI] [PMC free article] [PubMed]
- 5.Betsuyaku T, Fukuda Y, Parks WC, Shipley JM, Senior RM: Gelatinase B is required for alveolar bronchiolization after intratracheal bleomycin. Am J Pathol 2000, 157:525-535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dunsmore SE, Saarialho-Kere UK, Roby JD, Wilson CL, Matrisian LM, Welgus HG, Parks WC: Matrilysin expression and function in airway epithelium. J Clin Invest 1998, 102:1321-1331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhang LY, Levitt RC, Kleeberger SR: Differential susceptibility to ozone-induced airways hyperreactivity in inbred strains of mice. Exp Lung Res 1995, 21:503-518 [DOI] [PubMed] [Google Scholar]
- 8.Van Winkle LS, Buckpitt AR, Nishio SJ, Isaac JM, Plopper CG: Cellular response in naphthalene-induced Clara cell injury and bronchiolar epithelial repair in mice. Am J Physiol 1995, 269:L800-L818 [DOI] [PubMed] [Google Scholar]
- 9.Plopper CG, Suverkropp C, Morin D, Nishio S, Buckpitt A: Relationship of cytochrome P-450 activity to Clara cell cytotoxicity. I. Histopathologic comparison of the respiratory tract of mice, rats and hamsters after parenteral administration of naphthalene. J Pharmacol Exp Ther 1992, 261:353-363 [PubMed] [Google Scholar]
- 10.Van Winkle LS, Isaac JM, Plopper CG: Distribution of the epidermal growth factor receptor and ligands during bronchiolar epithelial repair from naphthalene-induced Clara cell injury in the mouse. Am J Pathol 1997, 151:443-459 [PMC free article] [PubMed] [Google Scholar]
- 11.Van Winkle LS, Johnson ZA, Nishio SJ, Brown CD, Plopper CG: Early events in naphthalene-induced acute Clara cell toxicity: comparison of membrane permeability and ultrastructure. Am J Respir Cell Mol Biol 1999, 21:44-53 [DOI] [PubMed] [Google Scholar]
- 12.Stripp BR, Maxson K, Mera R, Singh G: Plasticity of airway cell proliferation and gene expression after acute naphthalene injury. Am J Physiol 1995, 269:L791-L799 [DOI] [PubMed] [Google Scholar]
- 13.Stevens TP, McBride JT, Peake JL, Pinkerton KE, Stripp BR: Cell proliferation contributes to PNEC hyperplasia after acute airway injury. Am J Physiol 1997, 272:L486-L493 [DOI] [PubMed] [Google Scholar]
- 14.Reynolds SD, Giangreco A, Power JH, Stripp BR: Neuroepithelial bodies of pulmonary airways serve as a reservoir of progenitor cells capable of epithelial regeneration. Am J Pathol 2000, 156:269-278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Plopper CG, Macklin J, Nishio SJ, Hyde DM, Buckpitt AR: Relationship of cytochrome P-450 activity to Clara cell cytotoxicity. III. Morphometric comparison of changes in the epithelial populations of terminal bronchioles and lobar bronchi in mice, hamsters, and rats after parenteral administration of naphthalene. Lab Invest 1992, 67:553-565 [PubMed] [Google Scholar]
- 16.Plopper CG: Structural methods for studying bronchiolar epithelial cells. Gil J eds. Models of Lung Disease, Microscopy and Structural Methods. 1990, :pp 537-559 Marcel Dekker, Inc., New York [Google Scholar]
- 17.Glantz SA: Primer of Biostatistics, ed 3 1992, McGraw-Hill, New York
- 18.Kleeberger SR, Reddy S, Zhang LY, Jedlicka AE: Genetic susceptibility to ozone-induced lung hyperpermeability: role of toll-like receptor 4. Am J Respir Cell Mol Biol 2000, 22:620-627 [DOI] [PubMed] [Google Scholar]
- 19.Paquette NC, Tankersley CG, Zhang LY, Kleeberger SR: Repeated subacute ozone exposure of inbred mice: airway inflammation and ventilation. Exp Lung Res 1994, 20:579-594 [DOI] [PubMed] [Google Scholar]
- 20.Ohtsuka Y, Clarke RW, Mitzner W, Brunson K, Jakab GJ, Kleeberger SR: Interstrain variation in murine susceptibility to inhaled acid-coated particles. Am J Physiol 2000, 278:L469-L476 [DOI] [PubMed] [Google Scholar]
- 21.Longphre M, Kleeberger SR: Susceptibility to platelet-activating factor-induced airway hyperreactivity and hyperpermeability: interstrain variation and genetic control. Am J Respir Cell Mol Biol 1995, 13:586-594 [DOI] [PubMed] [Google Scholar]
- 22.Kormos J: Two types of ciliary resorption. Acta Biol 1971, 22:245-260 [PubMed] [Google Scholar]
- 23.Watanabe T: A scanning electron-microscopic study of the local degeneration of cilia during sexual reproduction in Paramecium. J Cell Sci 1978, 32:55-66 [DOI] [PubMed] [Google Scholar]
- 24.Hamada SH: Phagocytosis and resorption during the reassembly of dissociated embryonic cells of sea urchins. Exp Cell Biol 1979, 47:294-306 [DOI] [PubMed] [Google Scholar]
- 25.Rieder CL, Jensen CG, Jensen LC: The resorption of primary cilia during mitosis in a vertebrate (PtK1) cell line. J Ultrastruct Res 1979, 68:173-185 [DOI] [PubMed] [Google Scholar]
- 26.Evans MJ, Johnson LV, Stephens RJ, Freeman G: Renewal of the terminal bronchiolar epithelium in the rat following exposure to NO2 or O3. Lab Invest 1976, 35:246-257 [PubMed] [Google Scholar]
- 27.Evans MJ, Shami SG, Cabral-Anderson LJ, Dekker NP: Role of nonciliated cells in renewal of the bronchial epithelium of rats exposed to NO2. Am J Pathol 1986, 123:126-133 [PMC free article] [PubMed] [Google Scholar]
- 28.Evans MJ, Shami SG: Lung cell kinetics. Massaro D eds. Lung Cell Biology. 1989, :pp 1-36 Marcel Dekker, New York [Google Scholar]
- 29.Erjefalt JS, Erjefalt I, Sundler F, Persson CG: In vivo restitution of airway epithelium. Cell Tissue Res 1995, 281:305-316 [DOI] [PubMed] [Google Scholar]
- 30.Peake JL, Reynolds SD, Stripp BR, Stephens KE, Pinkerton KE: Alteration of pulmonary neuroendocrine cells during epithelial repair of naphthalene-induced airway injury. Am J Pathol 2000, 156:279-286 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Danto SI, Shannon JM, Borok Z, Zabski SM, Crandall ED: Reversible transdifferentiation of alveolar epithelial cells. Am J Respir Cell Mol Biol 1995, 12:497-502 [DOI] [PubMed] [Google Scholar]
- 32.Chen CS, Mrksich M, Huang S, Whitesides GM, Ingber DE: Geometric control of cell life and death. Science 1997, 276:1425-1428 [DOI] [PubMed] [Google Scholar]





