Abstract
Disulfiram has been studied as a treatment for cocaine dependence. We report results of a randomized, double-blind, placebo-controlled, within-subject study to examine the interaction of disulfiram with intravenous cocaine.
Methods
Non-treatment-seeking, cocaine-dependent, volunteers participated in serial experiments in which they received disulfiram placebo, 62.5 or 250 mg/day on days 1–6. On days 4–6, participants received a morning disulfiram dose two hours prior to a scheduled session in which they were administered intravenous cocaine placebo, 0.25 mg/kg (n=9), or 0.5 mg/kg (n=3) over 1 minute. Blood, cardiovascular, and subjective measures were collected. Seven days of washout occurred between disulfiram conditions.
Results
Following active disulfiram treatments and cocaine 0.25mg/kg administration, plasma cocaine AUC(0–480 min) was increased (p=0.003 and 0.001) and cocaine clearance decreased (p<0.001). Disulfiram treatments also decreased cocaine clearance for the 0.5 mg/kg cocaine dose (p=0.002 and < 0.001). Neither disulfiram dose with cocaine altered cardiovascular responses relative to cocaine alone. Following cocaine 0.25 mg/kg, ‘any high’ (p=0.021 and 0.019),’cocaine high’(p=0.017 and 0.018) and ‘rush’(p=0.013 and 0.047) significantly decreased with either disulfiram dose.
Conclusions
Disulfiram decreased cocaine clearance without toxicity. Cocaine ‘high’ and ‘rush’ were diminished. Disulfiram may be a promising pharmacotherapy in selected cocaine dependent individuals.
1. Introduction
Cocaine abuse and dependence continues to be a significant public health problem in the United States. The National Institute on Drug Abuse (NIDA) reported in 2002, approximately 1.5 million Americans could be classified as dependent on or abusing cocaine in the past 12 months. Between 1995 and 2002, cocaine-related emergency department visits increased 33% (NIDA 2004). Additionally, cocaine users often simultaneously ingest alcohol to modulate the unpleasant psychological and physical side effects of anxiety and agitation during cocaine binges. The consumption of cocaine and alcohol together results in the formation of cocaethylene, an active metabolite with cocaine-like properties which could contribute to toxicity during a cocaine binge and may play a role in reinforcement (Hearn et al., 1991; Woodward et al., 1991; Perez-Reyes and Jeffcoat, 1992; Farre et al., 1993; McCance-Katz et al., 1995; McCance-Katz et al., 1998a). Sixty to ninety percent of cocaine dependent individuals also abuse or are dependent on alcohol (Higgins et al., 1994; Wiseman and McMillan, 1996; Martin et al., 1996; Heil et al., 2001). A pharmacotherapy that could target both cocaine and alcohol use in those with cocaine dependence would be of benefit.
Currently, there are no medications which are U.S. Food and Drug Administration (U.S. FDA)-approved for the treatment of cocaine dependence. Disulfiram (Antabuse) inhibits aldehyde dehydrogenase, an enzyme necessary in alcohol metabolism (Brien and Loomis, 1983; Yourick and Faiman, 1989; Yourick and Faiman, 1991; Hart and Faiman, 1992) and is approved by the U.S. FDA for the treatment of alcoholism. Alcohol use with disulfiram produces a noxious physical reaction resulting from acetaldehyde accumulation that includes hypotension, diaphoresis, flushing, nausea and vomiting (Haley, 1979). Data from clinical settings indicating that alcohol use can be a behavioral antecedent to cocaine use (Crosby et al., 1997), led to clinical trials exploring the efficacy of disulfiram as a treatment for cocaine dependence. Carroll and colleagues evaluated disulfiram in combination with various psychotherapies in cocaine-dependent individuals with concurrent alcohol abuse or dependence. Disulfiram treatment was associated with significantly better retention in treatment and longer duration of abstinence from cocaine and alcohol use (Carroll et al., 1998). At one year follow-up those who had received disulfiram met fewer dependence criteria showing durability of the clinical benefit of disulfiram treatment (Carroll et al., 2000). More recently, however, researchers (Carroll et al., 2004) explored the use of disulfiram in combination with cognitive behavioral therapy or interpersonal psychotherapy among cocaine-dependent patients with or without an alcohol use disorder. Those assigned to disulfiram reduced their cocaine use significantly more than those assigned to placebo. Further, disulfiram in combination with cognitive behavioral therapy was most beneficial in subjects who were not alcohol dependent at baseline or who fully abstained from consuming alcohol during treatment. It was concluded that disulfiram must exert a direct effect on cocaine use rather than through reducing concomitant alcohol use as was earlier hypothesized.
The results of a human laboratory-based study (McCance-Katz et al., 1998b) which examined the effect of a chronic disulfiram administration (250 or 500 mg daily) on intranasally administered cocaine has been previously reported. This study showed an interaction between disulfiram and cocaine in that cocaine concentrations over time were greater with disulfiram treatment. Cardiovascular responses (systolic and diastolic blood pressure and heart rate) over time were also greater with disulfiram administration than those for cocaine alone. No significant effect on cocaine ‘high’, ‘rush’ or ‘craving’ was observed when subjects received disulfiram. A limitation of that study was the intranasal route of cocaine administration which introduced the possibility of first pass effects, consequent to gastrointestinal absorption of swallowed drug and/or prolonged absorption of cocaine from the nasal mucosa that might have been affected by disulfiram treatment that may have altered cocaine’s disposition.
This report describes the results of a randomized, double-blind, placebo-controlled, within-subject study of the effect of chronic disulfiram treatment on intravenous cocaine administration. This study aimed to answer the following questions: 1) Does disulfiram alter the pharmacokinetics of intravenous (i.v.) cocaine?; 2) Does disulfiram alter physiological parameters (heart rate and blood pressure) and/or subjective responses to intravenously administered cocaine?; 3) Is there a dose-related effect of disulfiram when cocaine is administered intravenously? Answers to these questions could make a significant contribution to optimizing disulfiram treatment for cocaine-dependent patients.
2. Materials and methods
2.1. Subjects
This study was reviewed and approved by the internal institutional review board at the university-affiliated medical center. Nine volunteers participated (7 African-American, 1 Caucasian, 1 Hispanic; 3 women; age 40 ± 2.5 years (mean ± S.E.)). Subjects were non-treatment-seeking and recruited by word-of-mouth or newspaper advertisement. All subjects reported actively abusing cocaine at the time of study entry and met Diagnostic and Statistical Manual of Mental Disorders, 2000 (DSM-IV) criteria for cocaine dependence. Six participants met diagnostic criteria for alcohol abuse and one for alcohol dependence (not physiological dependence). Cocaine use was confirmed by urine toxicology screens which were collected at each visit during the evaluation process prior to study entry. Subjects were paid for participation in the studies. All identified cocaine as their preferred drug and were experienced with the smoked route of administration. The mean amount of street cocaine used was 3.9 ± 0.9 g/week. Subjects received a comprehensive clinical psychiatric and medical evaluation prior to entry into the study and had no other concurrent medical or mental illnesses. Five subjects admitted occasional marijuana use (1–2 joints/week), but did not meet abuse or dependence criteria for any other illicit drugs.
2.2. Procedures
This study was conducted under IND 57173 (cocaine) and the design was reviewed and approved by the U.S. FDA. The study utilized a double-blind, randomized, counter-balanced, placebo-controlled, within-subject design for both disulfiram and cocaine assignment. After giving written, voluntary, informed consent, the participants were assigned to the first of three disulfiram study drug regimens (disulfiram 62.5 mg, 250 mg or placebo daily). The duration of each disulfiram study block was 6 days for the participants that completed three cocaine doses (0, 0.25 mg/kg and 0.5 mg/kg) (n = 3) and 5 days for the participants that completed two cocaine doses (0, 0.25 mg/kg) (n = 9). Participants were given one disulfiram or placebo dose as outpatients and were admitted the following day to the inpatient research unit where they resided for the remaining duration of the study block (days 2 through 5 or 6). Subjects received 4 disulfiram doses prior to the first cocaine challenge session (day 4). Cocaine doses were blinded to the subject and research staff. A different cocaine dose was given each day on days 4 through 5 or 6. A single cocaine dose was administered in each study session and only one session occurred in a single day.
On cocaine study session days, disulfiram doses were administered two hours before cocaine administration. All disulfiram ingestion was oral and was witnessed by study staff. Cocaine administration studies began on the day following receipt of the forth disulfiram dose to account for the time necessary for the disulfiram to inhibit aldehyde dehydrogenase (Helander and Carlsson, 1990). Cocaine study drugs included cocaine hydrochloride (0.25 mg/kg or 0.5 mg/kg in a total volume of 10 ml of 0.9% normal saline or placebo (10 ml of 0.9% normal saline)). Due to safety concerns, the initial 5 participants received only cocaine 0.25 mg/kg or placebo as suggested by FDA reviewers. Following reporting of findings for the cocaine 0.25 mg/kg dose in combination with disulfiram to FDA, the higher 0.5 mg/kg cocaine dose was added to each block of disulfiram treatment. Cocaine was administered intravenously by the study physician over 1 minute immediately followed by a 10 ml 0.9% normal saline flush over 1 minute. Completion of each block of cocaine study drug administration sessions was followed by at least 7 days of disulfiram washout which occurred on an outpatient basis. This allowed adequate time for the elimination of disulfiram and its major metabolite (> 90% eliminated within 3 days of drug discontinuation) and for new enzyme synthesis (Helander and Carlsson, 1990).
2.3. Cocaine study drug administration sessions
Cocaine administration sessions began in the morning, with baseline physiological and subjective assessments at −30 and −15 minutes before cocaine administration. Blood sampling (by means of an intravenous catheter placed in an arm vein) occurred at baseline prior to cocaine administration and over the next 8 hours at time points 2.5, 10, 30, 60, 90, 120, 180, 240, 300, 360, 420, and 480 minutes. Cardiac rhythm monitoring and physiological measures of heart rate, systolic and diastolic blood pressure were assessed using automated equipment (Hewlett-Packard 43200A). Cardiac rhythm monitoring occurred until heart rate and blood pressure returned to baseline. Physiological monitoring was performed throughout the initial 3 hours at time points 2.5, 5, 7.5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 75, 90, 120, 150, and 180 minutes. A series of measures designed to assess subjective drug effects were administered. Visual analog scales consisted of lines anchored at 0 mm = minimal and 100 mm = maximal and measured ‘cocaine high’, ‘any high’, ‘rush’, ‘sleepy’, ‘pleasant’, ‘nervous’, ‘paranoid’, ‘sad/depressed’, ‘I want cocaine’, ‘good effects’, and ‘bad effects’.
2.4. Laboratory analysis
Blood samples for analysis of cocaine were collected in gray-stoppered vacutainer tubes which contained sufficient sodium fluoride to prevent degradation of the cocaine by cholinesterase. Samples were immediately centrifuged, the plasma separated and stored at −70 degrees C until the time of analysis. The cocaine concentrations were determined by reverse-phase high performance liquid chromatography with ion pairing, as previously described (Jatlow et al., 1991). The reproducibility (coefficient of variation) for cocaine was less than 5% at concentrations of 100 ng/ml.
2.5. Data analysis
Estimates of sample size were calculated based on the results of a previous study (McCance-Katz et al., 1998b). One of the main reasons for undertaking the present study was to determine if the drug interaction observed with disulfiram and intranasally administered cocaine which included large differences in observed plasma cocaine concentrations in the presence or absence of disulfiram, would also occur with intravenous cocaine administration. Therefore, the power analysis was based on cocaine plasma concentrations from which cocaine clearance was derived. Power analysis was calculated based on mean and standard deviation for cocaine clearance in those receiving cocaine alone versus cocaine following chronic disulfiram administration (250 mg daily ≥ 3 days). In order to show a 40% decrease in cocaine clearance (effect seen in our previous report for intranasal cocaine with disulfiram) in the current study, with alpha set at 0.05 (two-tailed) and beta set at 0.9, a sample size of 3 subjects was required.
The pharmacokinetic parameters for cocaine were evaluated for each subject after each intravenous administration of cocaine alone and again after each administration of the same cocaine dose after 4 or 5 days of either disulfiram dose. Standard noncompartmental methods were used to estimate cocaine pharmacokinetic parameters (WinNonLin 4.1, Pharsight Corporation, Mountain View, CA) (Gibaldi and Perrier, 1982). These parameters included the area under the plasma concentration-time curve (AUC), clearance (Cl) and volume (Vss). The elimination rate constant was derived from the computer fit and from this half-life was computed. AUC values were also computed for each dependent cardiovascular and subjective measure under each of the study conditions.
The above described calculated values for each disulfiram condition (placebo, 62.5 and 250 mg/day) were compared with one another within the cocaine conditions (0.25 and 0.5 mg/kg and placebo). All values were analyzed in a series of comparisons done within a mixed-model repeated measures analysis of variance (ANOVA). In the analysis of primary interest, the simple main effect of the disulfiram within each cocaine dose was determined. Significant main effects allowed for the comparison of the disulfiram 62.5 or 250 mg/day treatment with that of the placebo disulfiram treatment, as well as for comparison of the two active disulfiram doses (62.5 mg versus 250 mg/day). All F ratios (simple main effects tests) with P < 0.05 (two-tailed) were considered statistically significant. Student’s paired t-test was used to test for significant differences and was considered statistically significant if P ≤ 0.05. JMP (version 5.0.5 SAS Institute Inc., Cary, NC) software was used to analyze data.
Nine subjects completed study sessions with 0.25 mg/kg cocaine challenges. Three subjects completed study sessions with both 0.25 and 0.5 mg/kg cocaine challenges. The lesser number in the study that included three doses of cocaine were, as indicated, a response to safety concerns. With the decision to expand the study cocaine doses, it was decided to run the minimum number of subjects to examine the pharmacokinetic interaction between cocaine and disulfiram.
3. Results
3.1. Cocaine pharmacokinetics
Following administration of either active disulfiram dose (62.5 mg or 250 mg daily), cocaine AUCs were significantly increased and cocaine clearances were correspondingly decreased following 0.25 mg/kg cocaine (Table 1, Figure 1A). There was also an increase, sometimes substantial (30–140%) in cocaine plasma AUC following the 0.5 mg/kg cocaine dose when subjects received either dose of disulfiram versus placebo disulfiram (i.e.: cocaine alone administration) (Table 1, Figure 1B). This main effect did not reach statistical significance (p = 0.073) likely due to the small sample size (n = 3). Disulfiram administration was associated with an increase in cocaine half-life ranging from 112 to 193% (Table 1). Administration of cocaine 0.5 mg/kg was associated with a significant decrease in cocaine clearance when disulfiram 250 mg daily was given versus disulfiram 62.5 mg daily (p = 0.0156). Cocaine Cmax and Vd were not altered following disulfiram administration.
Table 1.
Pharmacokinetics of cocaine administered intravenously following treatment with disulfiram (DS) 62.5 or 250 mg/d (cocaine 0.25 mg/kg [n=9]; cocaine 0.5 mg/kg [n=3])
| Variable | Cocaine dose (mg/kg) | Placebo | 62.5 mg/day | DS 62.5 mg/day vs. Placebo (t, p)* | 250 mg/day | DS 250 mg/day vs. Placebo (t, p)* | Main effect (F, p) |
|---|---|---|---|---|---|---|---|
| AUC(inf) (ng.min/ml) | 0.25
0.50 |
10830 (788)*
23717 (2309) |
18108 (1842)
30793 (6715) |
3.17, 0.006 | 19938 (2687)
57267 (11444) |
3.97, 0.001 | 8.84, 0.003
5.38, 0.073 |
| AUC (0–480) (ng.min/ml) | 0.25
0.50 |
10320 (773)
22630 (2078) |
16602 (1321)
29231 (6954) |
3.42, 0.003 | 17658 (2113)
49330 (8094) |
4.00, 0.001 | 9.32, 0.002
5.0, 0.082 |
| T1/2 (min) | 0.25
0.50 |
71.4 (6.6)
89 (16) |
119 (23)
100 (14) |
2.33, 0.033 | 138 (17.3)
169 (26) |
3.29, 0.005 | 5.72, 0.013
5.0, 0.082 |
| Cl (L/min per kg) | 0.25
0.50 |
0.024 (0.002)
0.024 (0.002) |
0.015 (0.001)
0.015 (0.001) |
4.96, <0.001
7.13, 0.002 |
0.014 (0.002)
0.010 (0.002) |
5.39, <0.001
11.16, <0.001 |
17.94, <0.001
63.92, <0.001 |
| Vdss (l) | 0.25
0.50 |
2.1 (1.73)
2.3 (0.46) |
2.0 (0.21)
2.0 (0.51) |
2.3 (1.8)
2.0 (0.2) |
1.10, 0.356
0.76, 0.525 |
||
| Cmax (ng/ml) | 0.25
0.50 |
222 (57.4)
376 (103) |
180 (24.4)
383 (113) |
160 (31.5)
421 (114) |
1.49, 0.256
2.41, 0.205 |
Mean (S.E.M.)
Fig. 1.

(A) Mean plasma cocaine concentrations over time (min.) following cocaine (Coc) 0.25 mg/kg (i.v.) (n=9) administration during treatment with disulfiram (DS) placebo, 62.5 mg/d or 250 mg/d. (B) Mean plasma cocaine concentrations over time (min.) following cocaine 0.5 mg/kg (i.v.) (n=3) administration during treatment with disulfiram placebo, 62.5 mg/d or 250 mg/d.
3.2. Cardiovascular responses to cocaine alone and following disulfiram treatment
The physiological parameters monitored following the cocaine study drug administration sessions included heart rate (Figure 2, A and B), systolic (Figure 3, A and B) and diastolic blood pressure (Figure 4, A and B). The administration of cocaine alone produced significant increases in heart rate (main effect: F = 9.27, p = 0.005, cocaine 0.25 mg/kg versus placebo cocaine/placebo disulfiram t = 3.56, p = 0.005, cocaine 0.5 mg/kg versus placebo cocaine/placebo disulfiram t = 3.50, p = 0.006). Cocaine significantly increased systolic blood pressure relative to cocaine placebo/disulfiram placebo administration (main effect: F = 22.00, p = 0.0003, cocaine 0.25 mg/kg versus placebo t = 4.00, p = 0.003, cocaine 0.5 mg/kg versus placebo t = 6.11, p = 0.0002). Diastolic blood pressure significantly increased following active cocaine administration (main effect: F = 5.99, p = 0.022, cocaine 0.25 mg/kg versus placebo t = 2.57, p = 0.030, cocaine 0.5 mg/kg versus placebo t = 2.92, p = 0.017). A statistically significant difference was also observed for the comparison of cocaine 0.25 mg/kg versus 0.5 mg/kg systolic blood pressure (t = 3.313, p = 0.009), but not for heart rate and diastolic blood pressure. Disulfiram 62.5 and 250 mg/day did not significantly increase heart rate, systolic or diastolic blood pressure relative to that of cocaine 0.25 mg/kg or cocaine 0.5 mg/kg administered singly. Neither dose of disulfiram was associated with a significant difference in the time necessary for physiological measures to return to baseline following cocaine administration.
Fig. 2.
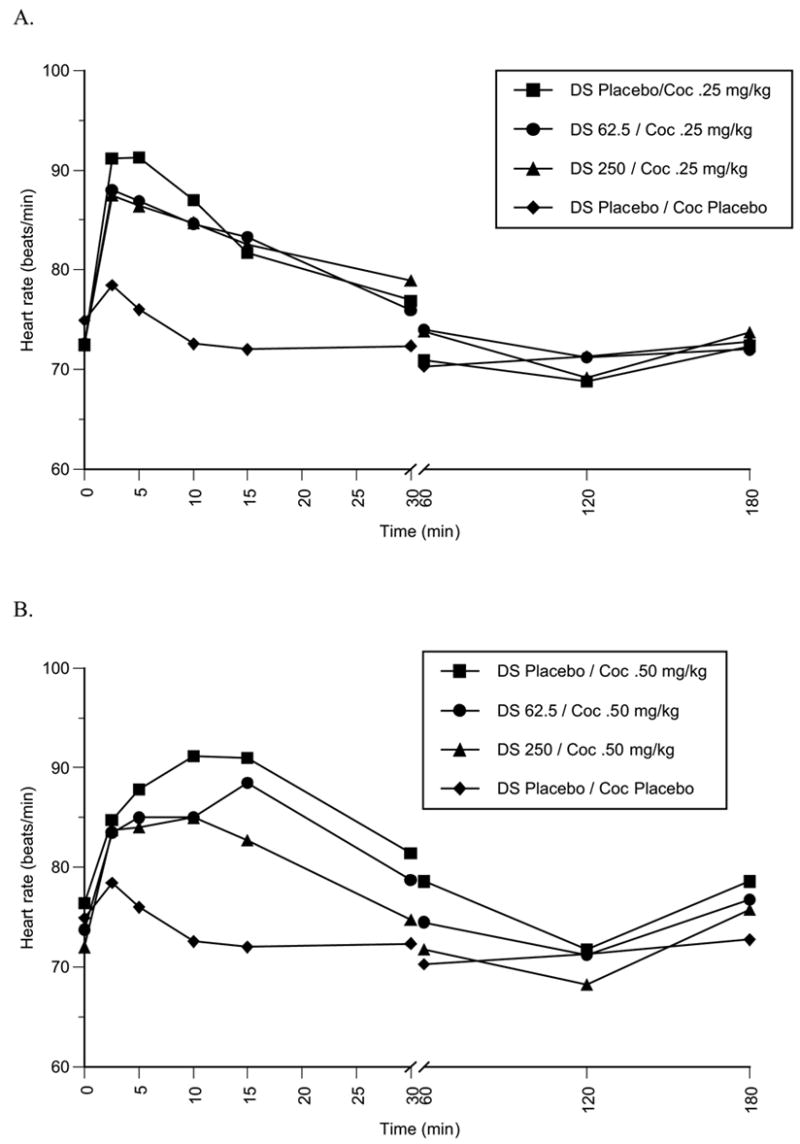
(A): Mean heart rate (beats/min) over time (min.) following cocaine0.25 mg/kg (i.v.) (n=9) administration during treatment with disulfiram placebo, 62.5 mg/d or 250 mg/d. (B) Mean heart rate (beats/min) over time (min.) following cocaine 0.5 mg/kg (i.v.) (n=3) administration during treatment with disulfiram placebo, 62.5 mg/d or 250 mg/d.
Fig 3.

(A): Mean systolic blood pressure (mmHg) over time (min.) following cocaine 0.25 mg/kg (i.v.) (n=9) administration during treatment with disulfiram placebo, 62.5 mg/d or 250 mg/d. (B) Mean systolic blood pressure (mmHg) over time (min.) following cocaine 0.5 mg/kg (i.v.) (n=3) administration during treatment with disulfiram placebo, 62.5 mg/d or 250 mg/d.
Fig 4.

(A): Mean diastolic blood pressure (mmHg) over time (min.) following cocaine 0.25 mg/kg (i.v.) (n=9) administration during treatment with disulfiram placebo, 62.5 mg/d or 250 mg/d. (B) Mean diastolic blood pressure (mmHg) over time (min.) following cocaine 0.5 mg/kg (i.v.) (n=3) administration during treatment with disulfiram placebo, 62.5 mg/d or 250 mg/d.
3.3. Behavioral effects
Following cocaine administration, analyses of subjective ratings for visual analog scale (VAS) items, ‘craving’, ‘sleepy’, ‘pleasant’, ‘nervous’, ‘paranoid’, ‘sad’, ‘depressed’, ‘good effects’ and ‘bad effects’ revealed no statistically significant findings. There was a significant reduction in ‘any high’, ‘cocaine high’ and ‘rush’ following the 0.25 mg/kg cocaine administration when subjects received either disulfiram 62.5 or 250 mg/day (Table 2, Figures 5A and B). Following the 0.25 mg/kg cocaine injection, mean ‘any high’ and ‘cocaine high’ AUC (0–60) decreased approximately 50 % when subjects received either disulfiram 62.5 or 250 mg/day (Table 2, Figure 5A). Mean ‘rush’ AUC (0–60) decreased 60% when subjects received disulfiram 62.5 mg/day and 46% with disulfiram 250 mg/day following the 0.25 mg/kg cocaine injection (Table 2, Figure 5B). None of the subjective data comparisons for the cocaine 0.5 mg/kg condition reached statistical significance due to the small sample (n = 3).
Table 2.
Summary of subjective responses to cocaine 0.25 mg/kg (i.v.) following treatment with disulfiram (DS) 62.5 or 250 mg daily (N = 9)
| Variable | Placebo | 62.5 mg/day | DS 62.5 mg/day vs. Placebo (t, p) * | 250 mg/day | DS 250 mg/day vs. Placebo (t, p) | Main effect (F, p) |
|---|---|---|---|---|---|---|
| Cocaine ‘high’ AUC (0–60) | 799 (208)* | 381 (134) | 2.65, 0.017 | 384 (83) | 2.63, 0.018 | 4.65, 0.02 |
| ‘Any high’ AUC (0–60) | 790 (202) | 391 (136) | 2.56, 0.021 | 384 (83) | 2.60, 0.019 | 4.45, 0.03 |
| ‘Rush’ AUC (0–60) | 466 (113) | 189 (45) | 2.74, 0.013 | 251 (65) | 2.12, 0.047 | 4.24, 0.03 |
Mean (S.E.M.)
Abbreviations
AUC: area under the curve
T1/2: elimination half-life
Cl: Clearance
Vdss: Volume of distribution at steady state
Cmax: maximum plasma concentration
Fig. 5.

(A) Mean cocaine ‘high’ over time (min.) following cocaine 0.25 mg/kg (i.v.) (n=9) administration during treatment with disulfiram placebo, 62.5 mg/d or 250 mg/day. (B) Mean ‘rush’ measurements over time (min.) following cocaine 0.25 mg/kg (i.v.) (n=9) administration during treatment with disulfiram placebo, 62.5 mg/d or 250 mg/day.
4. Discussion
Theses results show that disulfiram, even in doses well below those used for the treatment of alcoholism, are associated with increased cocaine plasma concentration versus time and decreased cocaine clearance following intravenous cocaine administration. Disulfiram had no effect on cocaine-associated cardiovascular responses and diminished cocaine ‘high’ and ‘rush’ although this was found to be statistically significant only at the lower disulfiram dose, potentially related to the small sample who received cocaine 0.5 mg/kg in conjunction with chronic disulfiram administration. The behavioral findings are particularly notable in view of the increased cocaine exposure associated with disulfiram treatment.
In previous studies (Hameedi et al., 1995; McCance-Katz et al., 1998b) of the effect of disulfiram on cocaine responses, disulfiram appeared to increase cocaine exposure, but we initially considered that this may have been related to the intranasal route of administration. Disulfiram decreases the tissue content of catecholamines at the synaptic nerve endings and therefore may reduce vasoconstriction as a consequence of reduced local norepinephrine concentration. A reduction of vasoconstriction might enhance cocaine absorption via the intranasal route. In the current study, an intravenous route of administration was selected to eliminate first-pass effects on swallowed cocaine, and any alterations in absorption secondary to disulfiram administration. The current study revealed significant increases in cocaine AUC and half-life, and correspondingly reduced clearance for cocaine at the 0.25 mg/kg dose. The magnitude of changes observed may possibly be related to the disulfiram dose, as there was a significant difference between cocaine clearances when participants were administered disulfiram 62.5 mg daily versus 250 mg/day following cocaine 0.25 mg/kg and 0.5 mg/kg administration.
These findings show that a pharmacokinetic interaction, most likely metabolic in origin, occurs when cocaine is administered in the presence of disulfiram. Changes in clearance and AUC, but not Vdss or Cmax are consistent with this interpretation. Disulfiram and its major metabolite, diethyldithiocarbamate, probably inhibit cocaine metabolism. Disulfiram is a general inhibitor of aldehyde dehydrogenases (Faiman, 1979; Haley, 1979; Wright and Moore, 1990) and has also been reported to inhibit both plasma and microsomal carboxylesterases and plasma cholinesterase (Faiman, 1979) which are the primary pathways for cocaine metabolism (Stewart et al., 1979). In addition, a minor oxidative pathway of N-demethylation of cocaine to norcocaine has been described (Stewart et al., 1979). Oxidative pathways account for 2–6 % of the cocaine metabolites (Inaba et al., 1978). Disulfiram has been reported to inhibit drug demethylation as a result of its ability to competitively inhibit hepatic microsomal drug oxidizing enzymes (Honjo and Netter, 1969). Inhibition of this minor pathway may make a small contribution to the observed decreased cocaine clearance, although the estaerase pathways are more important. This confirmation of the metabolic interaction between cocaine and disulfiram should be considered in patients who might receive disulfiram as a cocaine dependence pharmacotherapy and who are at risk for relapse to binge cocaine use. The metabolic interaction between cocaine and disulfiram could potentially be associated with cocaine accumulation and toxicity. It is also notable, however, that several clinical trials have been conducted in cocaine-dependent outpatients without reports of serious adverse events (Carroll et al., 1998; Petrakis et al., 2000; George et al., 2000; Carroll et al., 2004). It may be that disulfiram alters subjective effects of cocaine such that binge use of the drug is curtailed if relapse occurs, but this remains to be explored in further clinical trials.
In contrast to findings when cocaine was administered by nasal insufflation (McCance-Katz et al., 1998b), cocaine administered by the intravenous route in the presence of chronic disulfiram administration did not further increase cardiovascular responses. There are several possible explanations for the lack of significant cardiovascular findings in this study. Cocaine doses in this study were associated with lower cocaine exposure probably related to a difference in doses for the two studies. In comparing the lower cocaine doses in each study (current study: cocaine 0.25 mg/kg intravenous versus previous study: cocaine 1 mg/kg intranasal), the lower cocaine dose in the current study resulted in a higher cocaine Cmax (222 versus 146 ng/ml), but approximately half the cocaine AUCinf (10,830 versus 23,448 ng.min/ml) as compared to the cocaine alone dose (1 mg/kg) in the previous study (McCance-Katz et al., 1998b). The higher cocaine alone dose in the current study (0.5 mg/kg, intravenous) resulted in a higher cocaine Cmax (376 versus 146 ng/ml) and approximately the same AUCinf (23,717 versus 23,448 ng.min/ml) as observed in the previous study for the 1 mg/kg intranasal cocaine dose. A 20% increase in heart rate AUC (p = 0.048) was observed in the earlier study when the lower dose of cocaine (1 mg/kg) was administered following chronic disulfiram 250 mg/day administration, but no significant effect on systolic or diastolic blood pressure was observed in that study for the 1 mg/kg intranasal cocaine dose. In evaluating these two studies, differences in dosages as well as route dependent differences in bioavailability including variation in nasal absorption impacts pharmacodynamics and Cmax. In the present study, the equivalent cocaine (0.5 mg/kg) and disulfiram (250 mg/day) doses did not produce a significant increase in heart rate or blood pressure over that of cocaine alone administration. Another caveat to our results in the current study, however, is the small sample size, particularly in the 0.5 mg/kg cocaine study group. The sample size of three, probably gave us inadequate power to detect a statistically significant finding.
We did not study a drug combination in the current study that was equivalent to intranasally administered cocaine 2 mg/kg given in the presence of disulfiram 500 mg/day because the findings from our initial study led to a design in which it could be determined whether lower doses of disulfiram might be less likely to result in altered cocaine pharmacokinetics, and thereby might be safer and potentially more effective for the treatment of cocaine dependence.
Significant differences in subjective responses to cocaine occurred with disulfiram treatment. Disulfiram diminished cocaine 0.25 mg/kg i.v. responses for ‘high’ and ‘rush’. The results from this study do not show that the disulfiram-associated diminution of subjective effects is dose related. Each disulfiram dose either significantly decreased cocaine ‘high’ or ‘rush’ (cocaine 0.25 mg/kg) or showed a trend for a decrease in these subjective responses (cocaine 0.5 mg/kg). Further, there was no difference in subjective responses for ‘high’ and ‘rush’ when the two disulfiram doses were compared to each other.
It has been postulated that the ability of disulfiram to inhibit dopamine-β-hydroxylase (DβH), an enzyme that catalyzes the conversion of dopamine to norepinephrine in peripheral and central noradrenergic neurons (Vaccari et al., 1996) may be the mechanism by which disulfiram reduces cocaine use in clinical trials (Petrakis et al., 2000; George et al., 2000). Inhibition of DβH results in a decrease in norepinephrine in selected areas of the brain (Goldstein and Nakajima, 1967; Karamanakos et al., 2001) leading to a corresponding increase in dopamine in these neurons. Another consideration is that inhibition of this enzyme might be associated with increased ‘high’ and ‘rush’ as a result of increased dopamine concentrations in neurons in areas of the brain responsible for reinforcing effects of cocaine. However, another possibility is that when paired with increased dopamine-mediated anxiety and agitation that can occur with stimulant use, the overall result might be a reduction in cocaine use as has been observed in clinical trials (Carroll et al., 1998; Petrakis et al., 2000; George et al., 2000; Carroll et al., 2004). Further, in our earlier study utilizing an intranasal route of cocaine administration and higher treatment doses of disulfiram, no significant increases in euphoria were observed (McCance-Katz et al., 1998b).
An alternative explanation for the decreased cocaine-associated subjective effects with disulfiram treatment could lie in its effects on the serotonin system. Attenuation of positive subjective effects of cocaine with enhanced serotonin neurotransmission have been reported (Walsh et al., 1994; Walsh, 1995). Cocaine alters serotonergic function by blockade of serotonin reuptake. Disulfiram might inhibit the conversion of serotonin to its principal metabolite, 5-hydroxyindoleacetic acid through inhibition of aldehyde dehydrogenase leading to increased production of the serotonin metabolite 5-hydroxytryptohol (5-HTOL) (Beck et al., 1980; Beck et al., 1986; Beck et al., 1995). Disulfiram treatment has been shown to increase 5-HTOL when given in a single dose and after one week of chronic treatment (Beck et al., 1995). 5-HTOL has been shown to have biological activity; specifically it has been shown to induce sleep in preclinical studies and to produce vasoconstriction (Wang Fu et al., 1980). Increased 5-HTOL levels observed in the context of ingestion of alcohol and foods high in serotonin content have also been associated with adverse events including headache, fatigue and diarrhea (Helander and Some, 2000). Psychological and physiological effects of 5-HTOL in the context of cocaine ingestion might have negatively impacted perception of cocaine effects and contributed to the observed significant reductions in ‘high’ and ‘rush’ observed in this study. Although speculative, the effect of disulfiram on central aldehyde dehydrogenases might be differential; thus the disulfiram 62.5 mg daily dose might alter serotonin metabolism with little effect on DβH, but as the dose of disulfiram increased, the effect on DβH could become predominant. This might be a possible explanation for the greatest diminution of cocaine high and rush having been observed with the lower dose of disulfiram (62.5 mg/d) (Figures 5A and 5B). Future studies should consider inclusion of the measurement of urinary 5-HTOL as one approach to examining the possible contribution of 5-HTOL to understanding the effect of disulfiram on cocaine responses.
There are several limitations to this study. The sample size, while adequate for showing an effect of disulfiram on cocaine’s disposition and subjective effects, was too small to extend the findings for the 0.25 mg/kg cocaine dose to the 0.5 mg/kg cocaine dose. Another limitation is the lack of the use of smoked cocaine for this study. The hospital environment in which this study was conducted did not permit administration of smoked cocaine; however this is the route of administration used by the majority of those with primary cocaine dependence in the U.S. Whether the findings in this study are directly applicable to current smoked cocaine users is not certain, although many other investigators have produced findings from studies using an intravenous route of administration that have advanced the field of cocaine (principally smoked) dependence treatment (Foltin and Fischman, 1991; Foltin and Fischman, 1992; Walsh et al., 1994; Sofuoglu et al., 2001). Intravenous cocaine is a better paradigm for the smoked route than is intranasal administration.
In summary, the chronic administration of disulfiram was shown to have significant effects on intravenous cocaine administration including a metabolic interaction that produces increased cocaine exposure by inhibition of cocaine metabolism and significant diminution of cocaine-associated subjective effects (‘high’ and ‘rush’). These findings, in the context of a lack of effect on cardiovascular responses to cocaine, hold promise for the development of disulfiram in a dose range of 62.5–250 mg daily as a treatment for cocaine dependence. Further clinical trials with this medication appear warranted.
Acknowledgments
This work was supported by NIDA/NIH grants RO1 DA 09573, KO2 DA 00478, and M01RR00065 NCRR/NIH (General Clinical Research Center at Virginia Commonwealth University).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Reference List
- Beck O, Borg S, Holmstedt B, Kvande H, Shroder R. Acta Pharmacol Toxicol. Vol. 47. Copenh: 1980. Concentration of serotonin metabolites in the cerebrospinal fluid from alcoholics before and during disulfiram therapy; pp. 305–307. [DOI] [PubMed] [Google Scholar]
- Beck O, Eriksson CJ, Kiianmaa K, Lundman A. 5-Hydroxyindoleacetic acid and 5-hydroxytryptophol levels in rat brain: effects of ethanol, pyrazole, cyanamide and disulfiram treatment. Drug Alcohol Depend. 1986;16:303–308. doi: 10.1016/0376-8716(86)90064-5. [DOI] [PubMed] [Google Scholar]
- Beck O, Helander A, Carlsson S, Borg S. Changes in serotonin metabolism during treatment with the aldehyde dehydrogenase inhibitors disulfiram and cyanamide. Pharmacol Toxicol. 1995;77:323–326. doi: 10.1111/j.1600-0773.1995.tb01035.x. [DOI] [PubMed] [Google Scholar]
- Brien JF, Loomis CW. Disposition and pharmacokinetics of disulfiram and calcium carbimide (calcium cyanamide) Drug Metab Rev. 1983;14:113–126. doi: 10.3109/03602538308991384. [DOI] [PubMed] [Google Scholar]
- Carroll KM, Fenton LR, Ball SA, Nich C, Frankforter TL, Shi J, Rounsaville BJ. Efficacy of disulfiram and cognitive behavior therapy in cocaine-dependent outpatients: a randomized placebo-controlled trial. Arch Gen Psychiatry. 2004;61:264–272. doi: 10.1001/archpsyc.61.3.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carroll KM, Nich C, Ball SA, McCance-Katz EF, Frankforter TL, Rounsaville BJ. One-year follow-up of disulfiram and psychotherapy for cocaine-alcohol users: sustained effects of treatment. Addiction. 2000;95:1335–1349. doi: 10.1046/j.1360-0443.2000.95913355.x. [DOI] [PubMed] [Google Scholar]
- Carroll KM, Nich C, Ball SA, McCance-Katz EF, Rounsavile BJ. Treatment of cocaine and alcohol dependence with psychotherapy and disulfiram. Addiction. 1998;93:713–727. doi: 10.1046/j.1360-0443.1998.9357137.x. [DOI] [PubMed] [Google Scholar]
- Crosby R, Specker S, Borden J, Tarleton S, Hatsukami D. Relapse and coping skills of cocaine using women. NIDA Res Monogr. 1997;174:113. [Google Scholar]
- Diagnostic and Statistical Manual of Mental Disorders. 4. American Psychiatric Association; 2000. [Google Scholar]
- Faiman MD. Biochemistry and Pharmacology of Ehtanol. Plenum; New York: 1979. [Google Scholar]
- Farre M, de la Torre R, Llorente M, Lamas X, Ugena B, Segura J, Cami J. Alcohol and cocaine interactions in humans. J Pharmacol Exp Ther. 1993;266:1364–1373. [PubMed] [Google Scholar]
- Foltin RW, Fischman MW. Smoked and intravenous cocaine in humans: acute tolerance, cardiovascular and subjective effects. J Pharmacol Exp Ther. 1991;257:247–261. [PubMed] [Google Scholar]
- Foltin RW, Fischman MW. Self-administration of cocaine by humans: choice between smoked and intravenous cocaine. J Pharmacol Exp Ther. 1992;261:841–849. [PubMed] [Google Scholar]
- George TP, Chawarski MC, Pakes J, Carroll KM, Kosten TR, Schottenfeld RS. Disulfiram versus placebo for cocaine dependence in buprenorphine-maintained subjects: a preliminary trial. Biol Psychiatry. 2000;47:1080–1086. doi: 10.1016/s0006-3223(99)00310-8. [DOI] [PubMed] [Google Scholar]
- Gibaldi M, Perrier D. Pharmacokinetics. 2. Marcel Decker; New York: 1982. [Google Scholar]
- Goldstein M, Nakajima K. The effect of disulfiram on catecholamine levels in the brain. J Pharmacol Exp Ther. 1967;157:96–102. [PubMed] [Google Scholar]
- Haley TJ. Disulfiram (tetraethylthioperoxydicarbonic diamide): a reappraisal of its toxicity and therapeutic application. Drug Metab Rev. 1979;9:319–335. doi: 10.3109/03602537908993897. [DOI] [PubMed] [Google Scholar]
- Hameedi FA, Rosen MI, McCance-Katz EF, McMahon TJ, Price LH, Jatlow PI, Woods SW, Kosten TR. Behavioral, physiological, and pharmacological interaction of cocaine and disulfiram in humans. Biol Psychiatry. 1995;37:560–563. doi: 10.1016/0006-3223(94)00361-6. [DOI] [PubMed] [Google Scholar]
- Hart BW, Faiman MD. In vitro and in vivo inhibition of rat liver aldehyde dehydrogenase by S-methyl N,N-diethylthiolcarbamate sulfoxide, a new metabolite of disulfiram. Biochem Pharmacol. 1992;43:403–406. doi: 10.1016/0006-2952(92)90555-w. [DOI] [PubMed] [Google Scholar]
- Hearn WL, Flynn DD, Hime GW, Rose S, Cofino JC, Mantero-Atienza E, Wetli CV, Mash DC. Cocaethylene: a unique cocaine metabolite displays high affinity for the dopamine transporter. J Neurochem. 1991;56:698–701. doi: 10.1111/j.1471-4159.1991.tb08205.x. [DOI] [PubMed] [Google Scholar]
- Heil SH, Badger GJ, Higgins ST. Alcohol dependence among cocaine-dependent outpatients: demographics, drug use, treatment outcome and other characteristics. J Stud Alcohol. 2001;62:14–22. doi: 10.15288/jsa.2001.62.14. [DOI] [PubMed] [Google Scholar]
- Helander A, Carlsson S. Use of leukocyte aldehyde dehydrogenase activity to monitor inhibitory effect of disulfiram treatment. Alcohol Clin Exp Res. 1990;14:48–52. doi: 10.1111/j.1530-0277.1990.tb00445.x. [DOI] [PubMed] [Google Scholar]
- Helander A, Some M. Dietary serotonin and alcohol combined may provoke adverse physiological symptoms due to 5-hydroxytryptophol. Life Sci. 2000;67:799–806. doi: 10.1016/s0024-3205(00)00674-3. [DOI] [PubMed] [Google Scholar]
- Higgins ST, Budney AJ, Bickel WK, Foerg FE, Badger GJ. Alcohol dependence and simultaneous cocaine and alcohol use in cocaine-dependent patients. J Addict Dis. 1994;13:177–189. doi: 10.1300/j069v13n04_06. [DOI] [PubMed] [Google Scholar]
- Honjo T, Netter KJ. Inhibition of drug demethylation by disulfiram in vivo and in vitro. Biochem Pharmacol. 1969;18:2681–2683. doi: 10.1016/0006-2952(69)90201-9. [DOI] [PubMed] [Google Scholar]
- Inaba T, Stewart DJ, Kalow W. Metabolism of cocaine in man. Clin Pharmacol Ther. 1978;23:547–552. doi: 10.1002/cpt1978235547. [DOI] [PubMed] [Google Scholar]
- Jatlow P, Elsworth JD, Bradberry CW, Winger G, Taylor JR, Russell R, Roth RH. Cocaethylene: a neuropharmacologically active metabolite associated with concurrent cocaine-ethanol ingestion. Life Sci. 1991;48:1787–1794. doi: 10.1016/0024-3205(91)90217-y. [DOI] [PubMed] [Google Scholar]
- Karamanakos PN, Pappas P, Stephanou P, Marselos M. Differentiation of disulfiram effects on central catecholamines and hepatic ethanol metabolism. Pharmacol Toxicol. 2001;88:106–110. [PubMed] [Google Scholar]
- Martin CS, Clifford PR, Maisto SA, Earleywine M, Kirisci L, Longabaugh R. Polydrug use in an inpatient treatment sample of problem drinkers. Alcohol Clin Exp Res. 1996;20:413–417. doi: 10.1111/j.1530-0277.1996.tb01067.x. [DOI] [PubMed] [Google Scholar]
- McCance-Katz EF, Kosten TR, Jatlow PI. Concurrent use of cocaine and alcohol is more potent and potentially more toxic than use of either alone--a multiple-dose study. Biol Psychiatry. 1998a;44:250–259. doi: 10.1016/s0006-3223(97)00426-5. [DOI] [PubMed] [Google Scholar]
- McCance-Katz EF, Kosten TR, Jatlow PI. Disulfiram effects on acute cocaine administration. Drug Alcohol Depend. 1998b;52:27–39. doi: 10.1016/s0376-8716(98)00050-7. [DOI] [PubMed] [Google Scholar]
- McCance-Katz EF, Price LH, Kosten TR, Jatlow PI. Cocaethylene: pharmacology, physiology and behavioral effects in humans. J Pharmacol Exp Ther. 1995;274:215–223. [PubMed] [Google Scholar]
- NIDA. Cocaine abuse and addiction. http://www.nida.nih.gov/ResearchReports/Cocaine/Cocaine.html. 2004
- Perez-Reyes M, Jeffcoat AR. Ethanol/cocaine interaction: cocaine and cocaethylene plasma concentrations and their relationship to subjective and cardiovascular effects. Life Sci. 1992;51:553–563. doi: 10.1016/0024-3205(92)90224-d. [DOI] [PubMed] [Google Scholar]
- Petrakis IL, Carroll KM, Nich C, Gordon LT, McCance-Katz EF, Frankforter T, Rounsaville BJ. Disulfiram treatment for cocaine dependence in methadone-maintained opioid addicts. Addiction. 2000;95:219–228. doi: 10.1046/j.1360-0443.2000.9522198.x. [DOI] [PubMed] [Google Scholar]
- Sofuoglu M, Nelson D, Babb DA, Hatsukami DK. Intravenous cocaine increases plasma epinephrine and norepinephrine in humans. Pharmacol Biochem Behav. 2001;68:455–459. doi: 10.1016/s0091-3057(01)00482-8. [DOI] [PubMed] [Google Scholar]
- Stewart DJ, Inaba T, Lucassen M, Kalow W. Cocaine metabolism: cocaine and norcocaine hydrolysis by liver and serum esterases. Clin Pharmacol Ther. 1979;25:464–468. doi: 10.1002/cpt1979254464. [DOI] [PubMed] [Google Scholar]
- Vaccari A, Saba PL, Ruiu S, Collu M, Devoto P. Disulfiram and diethyldithiocarbamate intoxication affects the storage and release of striatal dopamine. Toxicol Appl Pharmacol. 1996;139:102–108. doi: 10.1006/taap.1996.0147. [DOI] [PubMed] [Google Scholar]
- Walsh SL, Preston KL, Sullivan JT, Fromme R, Bigelow GE. Fluoxetine effects on cocaine response: a double-blind laboratory assessment in humans. In: Harris LS, editor. Problems of drug dependence 1994. US Govt Printing Office; Washington, D.C: 1995. p. 310. [Google Scholar]
- Walsh SL, Preston KL, Sullivan JT, Fromme R, Bigelow GE. Fluoxetine alters the effects of intravenous cocaine in humans. J Clin Psychopharmacol. 1994;14:396–407. [PubMed] [Google Scholar]
- Wang Fu LH, Hayashi S, Toda N. Effects of 5-hydroxytryptophol, a 5-hydroxytryptamine metabolite, on isolated cerebral arteries of the dog. Br J Pharmacol. 1980;68:17–18. doi: 10.1111/j.1476-5381.1980.tb10692.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiseman EJ, McMillan DE. Combined use of cocaine with alcohol or cigarettes. Am J Drug Alcohol Abuse. 1996;22:577–587. doi: 10.3109/00952999609001682. [DOI] [PubMed] [Google Scholar]
- Woodward JJ, Mansbach R, Carroll FI, Balster RL. Cocaethylene inhibits dopamine uptake and produces cocaine-like actions in drug discrimination studies. Eur J Pharmacol. 1991;197:235–236. doi: 10.1016/0014-2999(91)90531-t. [DOI] [PubMed] [Google Scholar]
- Wright C, Moore RD. Disulfiram treatment of alcoholism. Am J Med. 1990;88:647–655. doi: 10.1016/0002-9343(90)90534-k. [DOI] [PubMed] [Google Scholar]
- Yourick JJ, Faiman MD. Disulfiram metabolism as a requirement for the inhibition of rat liver mitochondrial low Km aldehyde dehydrogenase. Biochem Pharmacol. 1991;42:1361–1366. doi: 10.1016/0006-2952(91)90446-c. [DOI] [PubMed] [Google Scholar]
- Yourick JJ, Faiman MD. Comparative aspects of disulfiram and its metabolites in the disulfiram-ethanol reaction in the rat. Biochem Pharmacol. 1989;38:413–421. doi: 10.1016/0006-2952(89)90380-8. [DOI] [PubMed] [Google Scholar]


