Abstract
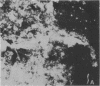
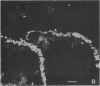
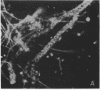
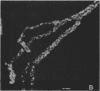
Fluorescent-antibody techniques using Zoogloea ramigera 106 antiserum were used to study fresh activated sludge flocs and finger-like zoogloeae in the microbial film that developed over stored samples of activated sludge. Few cells in fresh activated sludge reacted positively with the fluorescein-labeled antiserum. Finger-like zoogloeae containing reactive cells were readily observed in the microbial film layer over stored activated sludge. Certain of the natural finger-like projections were entirely composed of cells that reacted positively to the labeled Z. ramigera 106 antiserum, whereas other projections were devoid of reactive cells.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amin P. M., Ganapati S. V. Occurrence of zoogloea colonies and protozoans at different stages of sewage purification. Appl Microbiol. 1967 Jan;15(1):17–21. doi: 10.1128/am.15.1.17-21.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CRABTREE K., MCCOY E., BOYLE W. C., ROHLICH G. A. ISOLATION, IDENTIFICATION, AND METABOLIC ROLE OF THE SUDANOPHILIC GRANULES OF ZOOGLOEA RAMIGERA. Appl Microbiol. 1965 Mar;13:218–226. doi: 10.1128/am.13.2.218-226.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman B. A., Dugan P. R. Identification of Zoogloea species and the relationship to zoogloeal matrix and floc formation. J Bacteriol. 1968 May;95(5):1903–1909. doi: 10.1128/jb.95.5.1903-1909.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olson W. P. Maximal brightness and yield of fluorescent antibody. J Bacteriol. 1968 Mar;95(3):1176–1177. doi: 10.1128/jb.95.3.1176-1177.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prakasam T. B., Dondero N. C. Aerobic heterotrophic bacterial populations of sewage and activated sludge. I. Enumeration. Appl Microbiol. 1967 May;15(3):461–467. doi: 10.1128/am.15.3.461-467.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ROMANO A. H., GEASON D. J. PATTERN OF SHEATH SYNTHESIS IN SPHAEROTILUS NATANS. J Bacteriol. 1964 Oct;88:1145–1150. doi: 10.1128/jb.88.4.1145-1150.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unz R. F., Dondero N. C. The predominant bacteria in natural zoogloeal colonies. I. Isolation and identification. Can J Microbiol. 1967 Dec;13(12):1671–1682. doi: 10.1139/m67-217. [DOI] [PubMed] [Google Scholar]
- Unz R. F., Dondero N. C. The predominant bacteria in natural zoogloeal colonies. II. Physiology and nutrition. Can J Microbiol. 1967 Dec;13(12):1683–1694. doi: 10.1139/m67-218. [DOI] [PubMed] [Google Scholar]
- Unz R. F., Farrah S. R. Use of aromatic compounds for growth and isolation of Zoogloea. Appl Microbiol. 1972 Mar;23(3):524–530. doi: 10.1128/am.23.3.524-530.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]