Abstract
In animal cells, small interfering RNAs (siRNA), when exogenously provided, have been reported to be capable of inhibiting replication of several different viruses. In preliminary studies, siRNA species were designed and tested for their ability to act on the protein expressed in Huh7 cells transfected with DNA-directed mRNA constructs containing hepatitis delta virus (HDV) target sequences. The aim was to achieve siRNA specific for each of the three RNAs of HDV replication: (i) the 1,679-nucleotide circular RNA genome, (ii) its exact complement, the antigenome, and (iii) the less abundant polyadenylated mRNA for the small delta protein. Many of the 16 siRNA tested gave >80% inhibition in this assay. Next, these three classes of siRNA were tested for their ability to act during HDV genome replication. It was found that only siRNA targeted against HDV mRNA sequences could interfere with HDV genome replication. In contrast, siRNA targeted against genomic and antigenomic RNA sequences had no detectable effect on the accumulation of these RNAs. Reconstruction experiments with nonreplicating HDV RNA sequences support the interpretation that neither the potential for intramolecular rod-like RNA folding nor the presence of the delta protein conferred resistance to siRNA. In terms of replicating HDV RNAs, it is considered more likely that the genomic and antigenomic RNAs are resistant because their location within the nucleus makes them inaccessible to siRNA-mediated degradation.
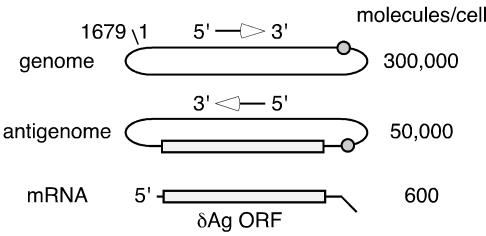
Human hepatitis delta virus (HDV) has a 1,679-nucleotide (nt) single-stranded circular RNA genome that is replicated by RNA-directed RNA synthesis, most probably involving host RNA polymerase II (25). During this replication, three RNA species accumulate (5), as represented in Fig. 1. The genome and its exact complement, the antigenome, are considered unit length. They exist primarily in a circular conformation but also in a linear conformation; these two conformations can be resolved using appropriate conditions of gel electrophoresis (2). The third RNA species consists of relatively lower amounts of an 800-nt polyadenylated RNA (of the same polarity as the antigenome), which is translated to produce a 195-amino-acid protein, known as the delta antigen (δAg-S), and is essential for HDV genome replication (14).
FIG. 1.
Representation of three main species of HDV RNA. Also indicated are the genomic and antigenomic ribozymes (cleavage site is shown as a circle) and the open reading frame (ORF) for the δAg (25). At the right is the number of molecules of each RNA per average liver cell for an infected woodchuck and chimpanzee, as previously reported (5). Indicated on the 1,679-nt genomic RNA is the origin for the nucleotide numbering, according to the sequence of Kuo et al. (15).
An increasing number of reports have shown that small interfering RNAs (siRNA) can be exogenously provided to cells undergoing animal virus replication and achieve inhibition (6, 8, 9, 12, 13, 17, 23). For the following reasons, we were specifically interested in the possible susceptibility to siRNA of HDV RNAs. (i) The HDV genomic and antigenomic RNAs can fold into an unbranched rod-like structure with 74% of the bases paired (15), and this folding might interfere with siRNA action. (ii) The delta protein has the ability to bind double-stranded RNA (4) and thus might also interfere. (iii) While several reports indicate that HDV genomic and antigenomic RNAs are predominantly located in the cell nucleus (7, 21, 24), two recent studies using cell fractionation indicated that much of the genomic RNA (but not the antigenomic RNA) might be cytoplasmic (10, 18) and thus possibly accessible to siRNA attack (10, 18).
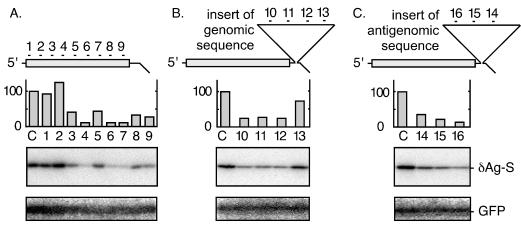
With these questions in mind, our first objective was to design and test siRNA species specific for sequences on HDV mRNA, genomic RNA, and antigenomic RNA. As represented in Fig. 2, the initial strategy was to use expression vectors to produce within Huh7 cells (20) DNA-directed mRNA species that contain these three sequences. As indicated, a total of 16 siRNA, each containing a 21-nt region based on the nucleotide sequence of Kuo et al. (15), were designed and constructed using a Silencer siRNA construction kit (Ambion). The locations and sequences of the target sites are listed in Table 1. The assay in each case was for the translation and accumulation of the delta protein, as detected by immunoblotting. The results and their quantitation are presented in Fig. 2. Also shown is the immunoblot for the internal control, green fluorescent protein (GFP). An expression vector for GFP was cotransfected, and as expected, the siRNA directed against HDV sequences did not inhibit the accumulation of GFP protein.
FIG. 2.
Transfected siRNAs could target HDV mRNA species. Huh7 cells were transfected with one of three plasmids that express an HDV mRNA species. (A) pDL444 (16) expressed an mRNA equivalent to normal HDV mRNA. (B and C) pDL444 was modified to contain 657 nt of extra sequences in the 3′ untranslated region, which led to the transcription of either partial genomic HDV RNA sequences (position 4 to 660) (B) or antigenomic RNA sequences (position 660 to 4) (C). The constructs were cotransfected with a plasmid expressing GFP. After 2 days the total protein was extracted and examined by immunoblotting to detect both delta protein and GFP. Detection and quantitation were with a bioimager (Fuji). The 16 HDV-specific siRNA indicated in the figure were designed and delivered as a cotransfection using Lipofectamine 2000 (Invitrogen). As a negative control (lanes C) we used siRNA against glyceraldehyde-3-phosphate dehydrogenase.
TABLE 1.
Locations and sequences of siRNA targets
| siRNA no.a | Location of target (5′-3′)b | Sequence of target (5′-3′)c |
|---|---|---|
| 1 | 1530-1510 | AAGAAAGAAGUUAGAGGAACU |
| 2 | 1472-1452 | AAGAUAGAGGACGAAAAUCCC |
| 3 | 1374-1354 | AACGGACCAGAUGGAGGUAGA |
| 4 | 1287-1267 | AAGGAAGGCCCUCGAGAACAA |
| 5 | 1271-1251 | AACAAGAAGAAGCAGCUAUCG |
| 6 | 1241-1221 | AAGAACCUCAGCAAGGAGGAA |
| 7 | 1219-1199 | AAGAGGAACUCAGGAGGUUGA |
| 8 | 1292-1272 | AAGACGAGAGAAGGGAAAGAA |
| 9 | 1029-1009 | AAACCAGGGAUUUCCAUAGGA |
| 10 | 67-87 | AAAGAGCAUUGGAACGUCGGA |
| 11 | 248-268 | AAGGGUUGAGUAGCACUCAGA |
| 12 | 399-419 | AAGCGAGGAGGAAAGCAAAGA |
| 13 | 486-506 | AACUCGACUUAUCGUCCCCAU |
| 14 | 76-56 | AAUGCUCUUUACCGUGACAUC |
| 15 | 339-319 | AAGCGCCUCUUGUUCGCUGAA |
| 16 | 495-475 | AAGUCGAGUUCCCCGGGAUAA |
The numbers of the siRNA species are as in Fig. 2.
The positions are presented using the HDV genome numbering of Kuo et al. (15). The numbers go “up” in the 5′ to 3′ direction of the genomic strand but “down” in the 5′ to 3′ direction of the antigenomic strand.
Each siRNA was assembled as a short double-stranded RNA. The two strands were transcribed and assembled using a Silencer siRNA construction kit (Ambion) with chemically synthesized DNAs as a template. The 21-nt sequences shown, all of which begin with AA, represent the target on the HDV RNA, be it genomic or antigenomic, that is targeted by the double-stranded siRNA. Each siRNA has 19 nt of double-strandedness with two unpaired nucleotides, UU, at the 3′ end. It has 21 nt complementary to the target sequence and only 19 nt complementary to the opposite strand.
Consistent with the siRNA experience of others (19), we found that some of the designed siRNA were able to give 80 to 95% inhibition of the HDV mRNA sequences (Fig. 2A, lanes 4, 6, and 7). Furthermore, as planned, it was possible to obtain siRNA that attacked the genomic and antigenomic RNA sequences (Fig. 2B and C, respectively). It should be noted that in Fig. 2C, the insert of 657 nt of antigenomic sequence (from position 660 to 4) should thus have been able in large part to fold with the 585 nt spanning the delta antigen open reading frame (at position 1596 to 1011) into an extensive amount of unbranched rod-like structure. Even if this potential folding did occur in vivo, it did not confer resistance to functioning siRNA (lanes 14, 15, and 16).
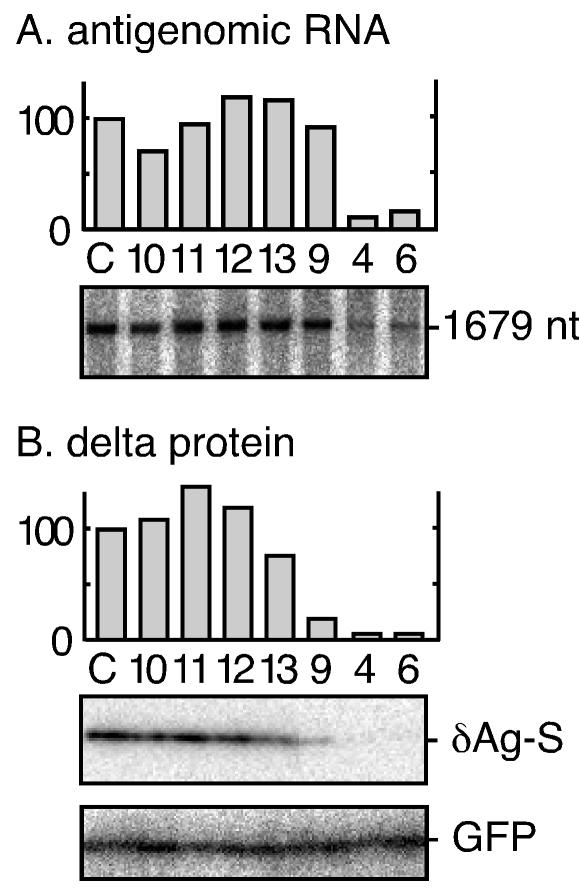
We next examined whether those siRNA with proven activity could interfere with HDV genome replication, as assayed by the accumulation of unit-length HDV RNA species. Some results are shown in Fig. 3, along with quantitation. We observed that inhibition of RNA accumulation (Fig. 3A) occurred only with siRNA 4 and 6, which targeted the mRNA sequences and caused a significant reduction of delta protein accumulation (Fig. 3B). (In panel A, the data shown are for antigenomic RNA; however, similar results were obtained for genomic RNA [data not shown]. Also, siRNA 14, 15, and 16 failed to reduce HDV RNA accumulation.)
FIG. 3.
Effect of transfected siRNA on the accumulation of replicating HDV RNAs. Cells were transfected with pDL553 (16) to initiate HDV genome replication. siRNA species (at 30 nM) were cotransfected as indicated and as previously described in Fig. 2. At day 2, total RNA was extracted, glyoxalated, and analyzed by electrophoresis in a 1% agarose gel. HDV antigenomic RNA was then detected by Northern assay (A). Similarly, total protein was examined by immunoblotting to detect δAg-S (B). For both panels, bioimager data were subjected to quantitation, expressing the amount of signal detected relative to that obtained for the control transfection. As a negative control (lane C), we used siRNA against endogenous glyceraldehyde-3-phosphate dehydrogenase (GAPDH); treatment with this siRNA reduced GAPDH mRNA but had no effect on HDV RNA levels (data not shown). Also shown in panel B is the immunoblot assay for expression of GFP, which was cotransfected as a control.
In the above-described experiment, the gel electrophoresis conditions used do not separate the circular from the linear conformations of unit-length HDV RNAs. However, if the siRNA treatment led to single endonucleolytic cuts on unit-length circles to produce linears, we expect that this would have inhibited further RNA accumulation (3). Therefore, to further test this preliminary interpretation that unit-length genomic and antigenomic RNAs were resistant to siRNA, we carried out the following additional experiment.
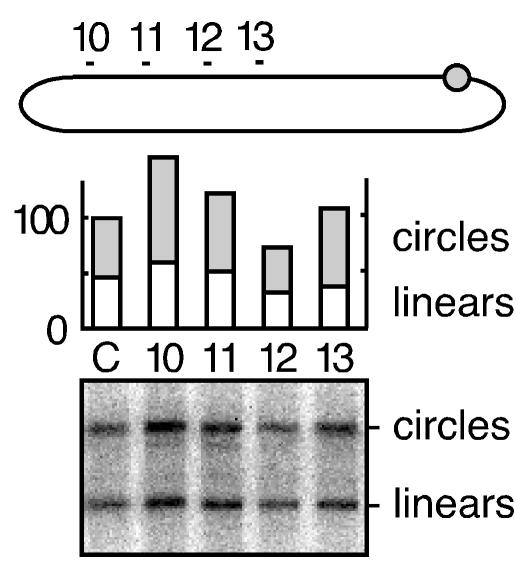
We used transfection to express HDV RNA multimers by DNA-directed RNA transcription. From previous studies, we knew that these would be posttranscriptionally processed to form unit-length RNA circles and yet, because of a 2-nt deletion in the open reading frame for the small delta protein, would be unable to make the essential delta protein and undergo RNA-directed transcription and replication (16). We then used gel analysis conditions capable of separating both linear and circular conformations of unit-length HDV RNA. Some data are shown in Fig. 4. As can be seen from the quantitation, cotransfection with siRNA 10, 11, 12, and 13, specific for genomic RNA sequences, did not reduce the accumulation of unit-length genomic RNA. Furthermore, the presence of this siRNA did not cause a reduction in the fraction of RNAs with a circular conformation (lanes 10 to 13 relative to lane C). In similar experiments, we expressed antigenomic nonreplicating RNAs and found that they were not sensitive to siRNA 4, 6, and 9 (data not shown). We interpret these data as evidence against siRNA action, even for inducing a single nick on the nonreplicating HDV RNAs. In addition, the delta protein was not present and thus could not be the basis for the observed resistance.
FIG. 4.
Transfected siRNA did not target nonreplicating unit-length HDV genomic RNA. Cells were transfected with pDL542 (16) to achieve the transcription and accumulation of nonreplicating unit-length genomic HDV RNA circles. Cotransfected with this plasmid was either a glyceraldehyde-3-phosphate dehydrogenase control siRNA (lane C) or genomic HDV-specific siRNA (lanes 10 to 13), as indicated at the top of the figure and as previously described for Fig. 2. At day 2, total RNA was extracted, glyoxalated, and analyzed by electrophoresis in a 3% agarose gel. Linear and circular forms of HDV genomic RNA were then detected by Northern assay. After hybridization and quantitation using the bioimager, we deduced the amounts of linear and circular RNAs, as summarized in the histogram.
Our studies show that HDV circular RNAs, whether transcribed from an RNA template (Fig. 3) or from a DNA template (Fig. 4), were resistant to siRNA attack. In the case of the DNA-directed transcript, the expression vector was such that the primary (nonreplicating) multimeric transcript was via host RNA polymerase II and that RNA (prior to ribozyme processing and ligation to form unit-length circles) should have undergone both 5′-capping and 3′ polyadenylation. Others have shown that for a host mRNA precursor, the intronic regions are resistant to siRNA while the exons are sensitive (22). Our studies show that the unit-length circular RNA processed out of the multimeric transcript was not only stable but also resistant to siRNA attack.
In summary, these studies support the interpretation that during genome replication, the only HDV RNA directly susceptible to inhibition mediated by siRNA is the mRNA. At least under the conditions of these experiments, the resistance of the genomic and antigenomic RNAs was not dependent on RNA structure, RNA conformation, or the presence of the small delta protein. Further experiments will be needed to determine if this “resistance” was in fact due to inaccessibility based on nuclear localization of genomic and antigenomic unit-length RNAs. It remains possible that some of the genomic RNA is cytoplasmic but is somehow inaccessible to attack by siRNA. For example, it could be protected by a host RNA-binding protein. For this or maybe other reasons, these HDV RNAs are probably resistant to siRNA because they are simply not accessible to the RISC, a protein-RNA effector nuclease complex that recognizes and destroys target RNAs (11). In this respect, our findings with HDV are analogous to those reported for siRNA action on the replication of respiratory syncytial virus (1) and influenza virus (8). That is, siRNA cannot target the replicating viral RNA transcripts directly but only indirectly via action on the viral mRNA species.
Acknowledgments
Constructive comments on the manuscript were given by Severin Gudima, Chi Tarn, Glenn Rall, and Richard Katz.
This work was supported by grants AI-26522 and CA-06927 from the NIH and by an appropriation from the Commonwealth of Pennsylvania.
REFERENCES
- 1.Bitko, V., and S. Barik. 2001. Phenotypic silencing of cytoplasmic genes using sequence-specific double-stranded short interfering RNA and its application in the reverse genetics of wild type negative-strand RNA viruses. BMC Microbiol. 1:34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chang, J., G. Moraleda, S. Gudima, and J. Taylor. 2000. Efficient site-specific non-ribozyme opening of hepatitis delta virus genomic RNA in infected liver. J. Virol. 74:9889-9894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chang, J., and J. Taylor. 2002. In vivo RNA-directed transcription, with template switching, by a mammalian RNA polymerase. EMBO J. 21:157-164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chao, M., S.-Y. Hsieh, and J. Taylor. 1991. The antigen of hepatitis delta virus: examination of in vitro RNA-binding specificity. J. Virol. 65:4057-4062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen, P.-J., G. Kalpana, J. Goldberg, W. Mason, B. Werner, J. Gerin, and J. Taylor. 1986. Structure and replication of the genome of hepatitis δ virus. Proc. Natl. Acad. Sci. USA 83:8774-8778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Coburn, G. A., and B. R. Cullen. 2002. Potent and specific inhibition of human immunodeficiency virus type 1 replication by RNA interference. J. Virol. 76:9225-9231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cunha, C., J. Monjardino, D. Chang, S. Krause, and M. Carmo-Fonseca. 1998. Localization of hepatitis delta virus RNA in the nucleus of human cells. RNA 4:680-693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ge, Q., M. T. McManus, T. Nguyen, C.-H. Shen, P. A. Sharp, H. N. Eisen, and J. Chen. 2003. RNA interference of influenza virus production by directly targeting mRNA for degradation and indirectly inhibiting all viral RNA transcription. Proc. Natl. Acad. Sci. USA 100:2718-2723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gitlin, L., S. Karelsky, and R. Andino. 2002. Short interfering RNA confers intracellular antiviral immunity in human cells. Nature 418:430-434. [DOI] [PubMed] [Google Scholar]
- 10.Gudima, S. O., J. Chang, G. Moraleda, A. Azvolinsky, and J. Taylor. 2002. Parameters of human hepatitis delta virus replication: the quantity, quality, and intracellular distribution of viral proteins and RNA. J. Virol. 76:3709-3719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hannon, G. J. 2002. RNA interference. Nature 418:244-251. [DOI] [PubMed] [Google Scholar]
- 12.Jacque, J. M., K. Triques, and M. Stevenson. 2002. Modulation of HIV-1 replication by RNA interference. Nature 418:435-438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jia, Q., and R. Sun. 2003. Inhibition of gammaherpesvirus replication by RNA interference. J. Virol. 77:3301-3306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kuo, M. Y.-P., M. Chao, and J. Taylor. 1989. Initiation of replication of the human hepatitis delta virus genome from cloned DNA: role of delta antigen. J. Virol. 63:1945-1950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kuo, M. Y.-P., J. Goldberg, L. Coates, W. Mason, J. Gerin, and J. Taylor. 1988. Molecular cloning of hepatitis delta virus RNA from an infected woodchuck liver: sequence, structure, and applications. J. Virol. 62:1855-1861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lazinski, D. W., and J. M. Taylor. 1994. Expression of hepatitis delta virus RNA deletions: cis and trans requirements for self-cleavage, ligation, and RNA packaging. J. Virol. 68:2879-2888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee, N. S., T. Dohjima, G. Bauer, H. Li, M. J. Li, A. Ehsani, P. Salvaterra, and J. Rossi. 2002. Expression of small interfering RNAs targeted against HIV-1 rev transcripts in human cells. Nat. Biotechnol. 20:500-505. [DOI] [PubMed] [Google Scholar]
- 18.Macnaughton, T. B., and M. M. Lai. 2002. Genomic but not antigenomic hepatitis delta virus RNA is preferentially exported from the nucleus immediately after synthesis and processing. J. Virol. 76:3928-3935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.McManus, M. T., and P. A. Sharp. 2002. Gene silencing in mammals by small interfering RNAs. Nat. Rev. Genet. 3:737-747. [DOI] [PubMed] [Google Scholar]
- 20.Nakabayashi, H., K. Taketa, K. Miyano, T. Yamane, and J. Sato. 1982. Growth of human hepatoma cell lines with differentiated functions in chemically defined medium. Cancer Res. 42:3858-3863. [PubMed] [Google Scholar]
- 21.Negro, F., F. Bonino, A. Di Bisceglie, J. Hoofnagle, and J. L. Gerin. 1989. Intrahepatic markers of hepatitis delta virus infection: a study by in situ hybridization. Hepatology 10:916-920. [DOI] [PubMed] [Google Scholar]
- 22.Ruiz, M. T., O. Voinnet, and D. C. Baulcombe. 1998. Initiation and maintenance of virus-induced gene silencing. Plant Cell 10:937-946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Seo, M. Y., S. Abrignani, M. Houghton, and J. H. Han. 2003. Small interfering RNA-mediated inhibition of hepatitis C virus replication in the human hepatoma cell line Huh-7. J. Virol. 77:810-812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Taylor, J., W. Mason, J. Summers, J. Goldberg, C. Aldrich, L. Coates, J. Gerin, and E. Gowans. 1987. Replication of human hepatitis delta virus in primary cultures of woodchuck hepatocytes. J. Virol. 61:2891-2895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Taylor, J. M. 2003. Replication of human hepatitis delta virus: recent developments. Trends Microbiol. 11:185-190. [DOI] [PubMed] [Google Scholar]