Abstract
We have estimated, for hippocampal neurons in culture, the size of the autaptic readily releasable pool before and after stimulation of the sort that produces culture long term depression (LTD). This stimulation protocol causes a decrease in the pool size that is proportional to the depression of synaptic currents. To determine if depression in this system is synapse specific rather than general, we have also monitored synaptic transmission between pairs of cultured hippocampal neurons that are autaptically and reciprocally interconnected. We find that the change in synaptic strength is restricted to the synapses on the target neuron that were active during LTD induction. When viewed from the perspective of the presynaptic neuron, however, synapse specificity is partial rather than complete: synapses active during induction that were not on the target neuron were partially depressed.
The “readily releasable pool” refers to a relatively small number of quanta that are available for release at a synapse at any moment; because this pool is refilled over a period of seconds, the readily releasable pool is usually defined as the maximum number of quanta that can be released over a brief period (≈1 s). This concept originated in the classical work on the neuromuscular junction by Liley and North (1) and Del Castillo and Katz (2) and was extended by Takeuchi (3), Hubbard (4), Thies (5), and Betz (6). More recent investigations have started to apply the concept to mammalian central synapses (7–10). Other newer studies have examined properties of the readily releasable pool (11–16) and have started to explore its computational implications (17–21). At hippocampal synapses the readily releasable pool contains about 10 quanta (13, 16) whereas its size in the giant ribbon synapses of retinal bipolar cells is in the thousands (15). For both of these synapse types, however, the pool refills, once it has been emptied, with a time constant of about 8 s. Because modern concepts of synaptic transmission hold that only those vesicles docked to the active zone can be released, the readily releasable pool must represent a subset of the docked vesicle pool. Microanatomical studies have revealed, however, that the number of vesicles in the docked vesicle pool is very close to the size of the readily releasable pool for both ribbon synapses and hippocampal synapses (22, 23). For this reason, the docked vesicle pool has been identified with the readily releasable pool.
One must distinguish between the size of the readily releasable pool and its degree of fullness, but in any case the probability that a synapse will release neurotransmitter is approximately proportional to the number of quanta in the pool (16). Although the size of the pool varies from one synapse to the next, it remains constant in size at a given synapse over time (16). The fullness of a pool varies dramatically, however, with synapse use and several recent studies have investigated the role of the pool fullness in the dynamic control of synaptic strength (18, 20, 21). The size of a readily releasable pool thus is a key determinant of a synapse’s resting efficacy, and fluctuations in pool fullness—together with phenomena such as facilitation and augmentation—alter synaptic strength on the short (10 s) time scale. Although pool size is of prime importance in setting synaptic strength, none of the factors responsible for controlling the resting size of the readily releasable pool have yet been identified. Here we report that the history of synaptic use can alter the size of the readily releasable pool: the stimulation protocol that produces long term depression (LTD) in cultured hippocampal neurons (culture LTD) also causes parallel changes in the size of the pool. Because release probability is directly related to pool size, this correlation between LTD and changes in the size of the readily releasable pool provides a possible cell biological mechanism for this form of long term synaptic plasticity in culture.
LTD induction involves prolonged use of synapses and one thus must worry about the extent to which LTD represents some nonspecific “tiring out” of the depressed synapses. Probably because only about a half of the synapses in our cultures are on spines (C. Boyer, T. Schikorski, and C.F.S., unpublished data), LTD is only partially and variably blocked by the standard pharmacological and membrane potential manipulations (24). These manipulations are therefore of limited usefulness in evaluating the contribution of “tiring out” to culture LTD. In a effort to obtain additional and independent evidence on the contribution of nonspecific effects, we have carried out experiments to characterize the synapse specificity of LTD under the conditions of our experiments.
Synapse specificity traditionally refers to the extent to which LTD is limited to those synapses on the postsynaptic neuron that were active during LTD induction. In discussing this issue we need, however, to distinguish two types of synapse specificity. Postsynaptic synapse specificity will be taken to mean that LTD occurs only in those synapses on the postsynaptic neuron that were active during the LTD induction. This type of synapse specificity has been examined in hippocampal slice experiments by using two afferent pathways. If LTD were to spread from synapses in which depression was induced to neighboring, inactive synapses, then postsynaptic specificity would not hold; this sort of spread has been termed heterosynaptic LTD and has been reported to occur in hippocampal slices (reviewed in Bear and Abraham, ref. 25).
Presynaptic synapse specificity will refer to the limitation of LTD to only those synapses on the postsynaptic neuron involved in the LTD induction without spreading to synapses made by the presynaptic cell on other neurons (not meeting the conditions for induction). Long term potentiation (LTP) in slices has been reported not to exhibit presynaptic specificity (26, 27), and postsynaptic specificity for LTP is also not obeyed at short distances (28). Although presynaptic specificity of LTD has not been examined in slices, Fitzsimonds et al. (29) have reported that LTD in culture is not presynaptically specific (termed lateral propagation). We find that, under the conditions of our experiments, culture-LTD exhibits complete postsynaptic specificity and at least partial presynaptic specificity; these findings lend support to the argument that culture LTD is a specific rather than a generalized change in synaptic strength.
Because the application of hypertonic solution to synapses causes depletion of the readily releasable pool without participation of the usual calcium dependent mechanisms (14, 24), this method for assaying pool size is the preferred one. The difficulty with the assay, however, is that it measures the average pool size for all of the synapses on the dendritic branches to which the hypertonic solution is applied. To estimate pool size before and after LTD, then, we must make sure all synapses have undergone the plastic changes. The only situation in which all synaptic inputs to a neuron are identified and express the same (average) amount of LTD is an autaptic circuit in which an isolated neuron has only itself as a potential target (24). For this reason, we have restricted our investigation of pool size before and after LTD induction to autapses.
MATERIALS AND METHODS
Cultures of dissociated hippocampal neurons were prepared from neonatal rat pups and from wild-type mice embryos as described (24). We routinely obtained coverslips that had at least several glial islands containing a single pair of neurons by this procedure. Cells were used 8–14 days after plating. Both rat and mice cultures were used interchangeably for monitoring synaptic transmission; synaptic properties were indistinguishable between the cultures of two rodent species.
Patch-clamp recordings from paired and autaptic cells were carried out as previously described using either an ATP regenerating system or perforated patches to prevent run-down of responses (30, 24). The LTD induction protocol consisted of 1 second long episodes of 5 Hz stimulation given every other second for 6 min. The postsynaptic cell was held at −70 mV except during low frequency stimulation when it was at −50 mV. For inducing autaptic LTD the cell was held at −70 mV except during 1 ms step depolarizations to +30 mV to evoke an action potential in the unclamped axonal processes; the dendritic compartments also received step depolarizations. The external bath solution consisted of 137 mM NaCl, 5 mM KCl, 10 mM CaCl2, 0.5 mM MgCl2, 10 mM D-glucose, 5 mM Hepes-NaOH (pH 7.3), and 100 μM picrotoxin. Concentrations of external CaCl2 and MgCl2 were varied as indicated. The extent of LTD was not sensitive to changes in external Ca2+ and Mg2+ concentrations tested (24). Hypertonic solution was extracellular recording solution with 0.5 M sucrose added, and a small island containing the entire dendritic field was sampled. Recordings were performed with Axopatch 200 (Axon Instruments, Foster City, CA). Signals were filtered at 2 kHz, digitized at 5kHz and analyzed with programs written in AXOBASIC and C. D(−)-2-amino-5-phosphovaleric acid, nitrendipine, nifedipine, and ((+)-α-methyl-4-carboxyphenylglycine) were from Research Biochemicals Inc., and amphotericin B was from Sigma.
The peak amplitudes of excitatory postsynaptic currents (EPSCs) were measured for analysis of LTD. LTD was expressed as the mean EPSC amplitude monitored ≈20–30 min (or ≈15–25 min) after LTD induction divided by the mean baseline EPSC amplitude; this quantity is termed synaptic strength ratio. The washout rate was obtained by determining the slope of the best linear fit of normalized EPSC responses, and is expressed as normalized unit response per minute. In cases when the rate of washout fluctuated—for example, before and after LTD—the best linear fit was determined for each time segments which were normalized separately, and the average rate of washout was obtained for the entire duration of recording.
RESULTS
The Readily Releasable Pool Is Decreased in Parallel with LTD.
We first sought to determine the effect of the LTD induction protocol on the size of the readily releasable pool. The rationale for this approach is, briefly, that our earlier work indicated LTD results (under the conditions of our experiments) from a change in the release mechanisms prior to the final calcium sensitive step; since the size of the readily releasable pool seems to be the major determinant of synaptic strength prior to the vesicle fusion controlling steps, regulation of pool size is a reasonable candidate mechanism. A more detailed explanation of this rationale follows.
A comparison of the frequency and the size of miniature ESPCs (mEPSCs) before and after LTD induction in our earlier work (24) indicated a specific change in the transmitter release during culture-LTD: whereas mEPSC frequency was most markedly decreased for large LTD, the amplitude of quantal events was unaltered irrespective of the size of LTD. A presynaptic mechanism for LTD has similarly been reported for LTD in hippocampal slices (refs. 31–33, but also see refs. 34 and 35). Our 1996 study (24), in addition, restricted the possible target of presynaptic alteration to a step prior to the final Ca2+-sensitive fusion step of transmitter release, or the actual fusion reaction itself. This conclusion was based on the following: (i) LTD is not only expressed for the synchronous, fast Ca2+-triggered phase of release but is also paralleled by the slow, asynchronous phase of release, and (ii) the rate of spontaneous vesicle fusion which is largely independent of extracellular Ca2+, is decreased following LTD induction.
The most likely site at which transmitter release is regulated prior to the final Ca2+-sensitive step is during docking and priming of synaptic vesicles at the plasma membrane. Moreover, events that occur during docking and priming must effectively regulate the efficiency of fusion reaction itself since there is very little time for enzymatic reaction to take place between the time of Ca2+ entry into the presynaptic terminal and when transmitter release is detected (reviewed in Goda and Südhof, ref. 36). We thus conjectured that the pool size of readily releasable quanta—which, as noted in the Introduction, presumably represents the fusion competent vesicles that are docked at the active zone and which is one of the prime factors controlling synaptic strength—might be altered by the LTD induction protocol.
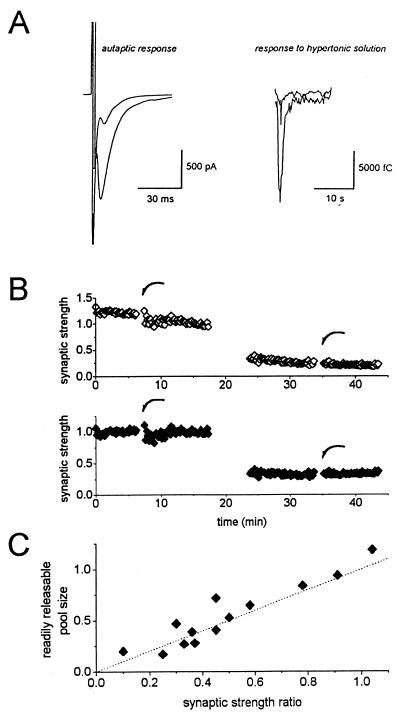
In the presence of hypertonic solution, synaptic vesicles rapidly fuse by a process not requiring Ca2+ and deplete the pool of vesicles available for nerve-evoked release (14). The number of quanta released during the transient hypertonic solution response defines the readily releasable vesicle pool. Fig. 1 A and B illustrates a typical experiment in which extracellular solution, made hyperosmolar with 0.5 M sucrose, was applied to the entire dendritic field of an autaptic cell before and after induction of LTD. Note that this series of experiments was carried out with small glial islands containing only a single autaptic neuron. Such recording condition ensures that the hyperosmolar medium applied with a large perfusion pipette covers all synapses contributing to the nerve-evoked autaptic response. For the particular cell shown in Fig. 1 A and B, the relative autaptic response decreased to 0.33 of baseline values, and hypertonic solution response—the readily releasable pool—was reduced to 0.27 of control.
Figure 1.
LTD accompanies a reduction in the pool size of readily releasable vesicles. (A, B) LTD was induced in an autapse in a small single cell island (B, Top); the synaptic strength ratio was 0.33 relative to the baseline synaptic responses. Note that autaptic responses gradually decreased over time before and after LTD induction: a response washout. (B, Bottom) The time course of the same experiment after correcting for the washout. The average rate of washout—i.e., the slope correction—was 0.017 of normalized synaptic strength per min. Hypertonic solution was applied to the entire autapse before and after LTD induction (B, arrows). LTD (synaptic strength ratio) of autaptic response was 0.33 of baseline values and the response to application of 0.5 M sucrose solution after LTD induction was 0.27 of the response elicited prior to LTD. Typical traces obtained before and after induction of LTD, in superposition, are shown for autapse (A, Left) and for hypertonic solution-induced response (A, Right). The autaptic cell was used after 9 days in culture, and extracellular recording solution contained 2 mM Mg2+ and 3 mM Ca2+. (C) Summary of LTD in single cell autapses in which the readily releasable pool size was monitored before and after induction of LTD (n = 13). Each point represents the relative autaptic response after LTD/before LTD (synaptic strength ratio) as function of the corresponding ratio of hypertonic solution-induced responses (after/before). The depression of the autaptic response and the hypertonic solution-evoked response paralleled each other, indicating that LTD involves an alteration in the mechanism(s) regulating the size of readily releasable vesicles.
For 13 such experiments, the relative synaptic strength after LTD induction (synaptic strength ratio) and the relative size of readily releasable pool after LTD were highly correlated as is shown in Fig. 1C (n = 13 autaptic cells). LTD is, therefore, closely paralleled by a corresponding decline in the pool size of readily releasable quanta. Because pool size can determine release probability, the cell biological mechanism for LTD could possibly be the down-regulation of the number of fusion competent vesicles.
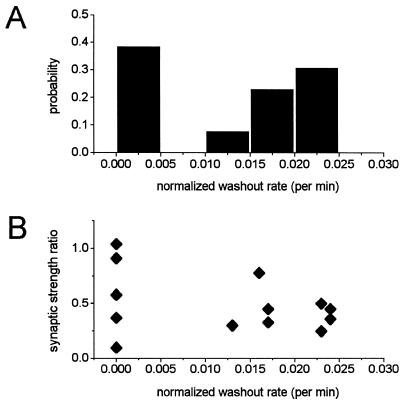
When one monitors synaptic transmission in cultured hippocampal neurons, a gradual reduction in EPSCs—i.e., a response washout—cannot be entirely avoided despite the use of perforated patches and optimizing culture conditions to improve neuronal viability. Washout, like LTD, is associated with a decline in pool size but does not depend on stimulation (not shown). To verify that LTD in culture is not simply an artifact that occurs in cells whose synaptic response is declining, we examined the relationship between the rate of washout and extent of LTD for each LTD-hypertonic solution experiment. The washout rate was represented as slope correction (see Materials and Methods; an example time course before and after slope correction is shown in Fig. 1B). Although experiments were not performed with cells that exhibited a noticeable rate of washout (the acceptable level of normalized washout rate was <0.025 per min; Fig. 2A), even a slight washout effect accumulates significantly during these experiments with a duration of 30 min. The depression observed after LTD induction and the inherent rate of washout were not correlated (Fig. 2B). Both LTD and washout accompany a decline in the readily releasable pool size; nevertheless, the reduction in synaptic strength induced by LTD protocol is not associated with the washout rate or extent.
Figure 2.
A slow rate of reduction in the amplitude of evoked response is not correlated with the extent of LTD. (A) Probability histogram of washout rate. The average rate of washout was determined after normalizing the response for each cell shown in Fig. 1C (n = 13; see Experimental Procedures). The bin size was 0.005 per min. (B) A comparison of the extent LTD (synaptic strength ratio) to the normalized washout rate is shown for each cell.
LTD in Culture Exhibits Complete Postsynaptic Synapse Specificity and at Least Partial Presynaptic Synapse Specificity.
One is always concerned that the depressive effect of the LTD stimulation protocol is general rather than specific, and especially so in culture where the usual manipulations for blocking LTD are of incomplete and variable effectiveness (24). In an effort to separate a generalized “rundown” of synaptic function from specific plasticity mechanisms, we have investigated the effects of the LTD stimulation protocol on different populations of synapses made by the same neuron. To examine the synapse specificity of culture-LTD we took advantage of the neural circuitry that is unique to low density cultures of hippocampal cells. When pairs of neurons are grown on isolated microislands of glial cells, they not only make reciprocal synapses onto each other but also form autapses. Thus each cell in a pair participates in a relatively complex circuit with both feed-forward and feed-back excitatory connections.
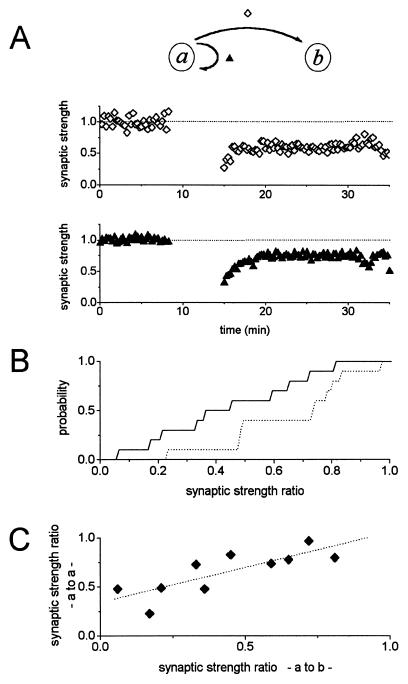
To simplify the description of these circuits we define one neuron, a, as the presynaptic cell of the pair—this is the neuron that we stimulate to induce LTD—and designate the other neuron, the postsynaptic cell from which we record postsynaptic currents and hold partly depolarized during LTD induction, as b (see Fig. 3, Inset). By patch-clamping both cells in a pair we can then sample all four different circuit components: (i) the forward input—the usual EPSCs—from cell a to cell b, (ii) autapses made by cell a onto itself, (iii) autapses made by b onto itself, and (iv) the reverse of the forward input, the synapses that cell b makes on cell a. The need for these distinctions will become clear as we discuss the rules for synapse specificity of LTD in this simple neuronal circuit. Briefly, we find that when LTD is induced in cell a’s synapses on cell b, cell a’s synapses on itself also express LTD to a lesser extent (partial presynaptic synapse specificity), but the synaptic strength of the b → a and the b → b connections are unchanged (complete postsynaptic synapse specificity).
Figure 3.
LTD exhibits partial presynaptic specificity in cultured hippocampal neurons. (A) An example cell pair in which LTD was induced from cell a → b. Synaptic strength ratio in cell b following LTD induction protocol was 0.59 relative to the baseline responses (⋄, Top). Autapse of presynaptic cell a also depressed to 0.74 of baseline responses (▴, Bottom). The cells were used at 11 days in culture, and external solution contained 0.5 mM Mg2+ and 10 mM Ca2+. (B) Summary of LTD experiments from 10 cell pairs. The mean synaptic strength ratio was 0.44 ± 0.08 (±SEM) between cell pairs a → b, and 0.65 ± 0.07 (±SEM) for autapses a → a. Autapses displayed LTD to a lesser degree (P < 0.06, Student’s t test). (C) Correlation of extent LTD (synaptic strength ratio) between forward (a → b) and autaptic (a → a) circuit for each cell pair represented in B. Linear regression results in a line shown with slope of 0.71 and y intercept of 0.34 (⋅⋅⋅··, correlation coefficient = 0.81, P < 0.005). The autapses consistently expressed smaller LTD.
A specific example of the LTD in the a → a connection that accompanies a → b LTD is shown in Fig. 3A. In contrast to the postsynaptic cell b, which was voltage clamped at −50 mV during the LTD induction protocol, the presynaptic cell a was continuously held at −70 mV except during the 1 ms step depolarizations to evoke an action potential. Thus, the conditions for optimal LTD induction were met only for the a → b connection because efficient LTD induction normally requires slight depolarization of the postsynaptic neuron (24). Nevertheless, the a → a synapses were also depressed. Note that the amount of LTD expressed by the autaptic a → a connections is less than that of the a → b connection.
For 10 experiments the mean synaptic strength ratio after LTD induction between cell pairs a → b was 0.44 ± 0.08 (±SEM; n = 10), and for presynaptic autapses a → a, the mean synaptic strength ratio was 0.65 ± 0.07 (±SEM; n = 10). The LTD expressed in autapses was always significantly weaker—about 25% on average—(P < 0.06, Student’s t test; Fig. 3B) than the depression observed in the forward direction (Fig. 3C). Thus, LTD exhibits at least partial presynaptic synapse specificity.
We previously demonstrated that LTD between a cell pair requires depolarization of the postsynaptic cell in that LTD is blocked by hyperpolarizing the postsynaptic cell during the low frequency stimulation of the presynaptic cell to induce LTD (24). Because cell a was held at −70 mV during LTD induction, the conditions for a → a LTD induction were not optimal, and we would have expected little LTD to result from the brief depolarizations associated with the presynaptic action potentials. Nevertheless, we cannot fully control the presynaptic cell’s membrane potential during induction of LTD in the a → b pathway and so cannot distinguish between weak LTD induction in the a → a input. At least then, we can conclude that our culture-LTD exhibits partial presynaptic synapse specificity and cannot exclude the possibility that it is completely synapse specific.
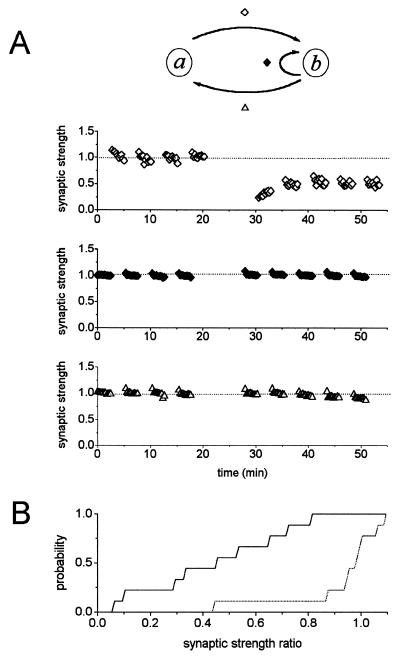
The effect of inducing LTD in a → b direction on the reciprocal connection (b → a) and also in the autaptic pathway (b → b) when present, was investigated next by sampling evoked responses in the b → a and in the b → b direction. As illustrated in an example cell pair (Fig. 4A), the inverse input b → a, and postsynaptic autapses b → b, were unchanged after induction of LTD in the forward a → b direction. For nine experiments, the mean synaptic strength ratio in the LTD pathway a → b, was 0.44 ± 0.09 (±SEM, n = 9; Fig. 4B), whereas the reciprocal input b → a, was essentially unchanged: the mean ratio of synaptic strength was 0.92 ± 0.07 (±SEM, n = 9; Fig. 4B). The postsynaptic autapse (b → b) present in five cell pairs examined, also remained unaltered [mean synaptic strength ratio was 1.00 ± 0.01 (±SEM), n = 5]. In summary, LTD exhibits complete postsynaptic synapse specificity under the conditions of our experiments.
Figure 4.
LTD in culture displays complete postsynaptic synapse specificity. (A) EPSCs of the forward (a → b) and reverse (b → a) circuits, and autapses onto the postsynaptic cell (b → b) were monitored before and after LTD induction in the a → b pathway. Although the a → b input displayed mean synaptic strength ratio of 0.53 after LTD induction (Top), the mean synaptic strength ratio in b → b (Middle) and b → a (Bottom) remained at 1.01 and 0.95 relative to the baseline values, respectively. The cultures were 10 days old, and the recordings were performed in 0.5 mM Mg2+ and 10 mM Ca2+. (B) Cumulative probability distributions of LTD responses from nine reciprocally connected cell pairs. Mean LTD responses for the forward circuit to which LTD protocol was applied, expressed as synaptic strength ratio, was 0.44 ± 0.09 (±SEM, n = 9; —); in contrast, the mean synaptic strength ratio of the reverse circuit was 0.92 ± 0.07 (±SEM, n = 9; ···⋅⋅⋅). Note that not all cell pairs examined had all four inputs: a → a, a → b, b → b, and b → a.
DISCUSSION
We have discovered that the LTD induction protocol produces a change in the size of the readily releasable pool that parallels the amount of LTD. Further, we find that culture LTD follows specific rules that govern the pattern of synaptic strength changes and is thus not simply a generalized and nonspecific reduction in the efficacy of all synapses made by a particular neuron.
The reduction of pool size that accompanies LTD is the first instance for neurons of a manipulation that can change the size of the readily releasable pool. Earlier work has shown that a host of factors that alter neurotransmitter release have no effect on the size of the readily releasable pool. For example, pool size is unchanged at synapses lacking synaptotagmin 1 and Rab3a; the synaptogagmin deletion produces a profound decrease in transmitter release and the Rab3A causes a 2-fold increased (30, 37). Decreases in extracellular calcium concentration dramatically reduce release (38) whereas the size of the readily releasable pool remains unchanged when the extracellular calcium concentration is reduced to nearly zero (14, 39–42). Furthermore, pool size is unaffected by buffering the internal calcium concentration with bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetate (75 mM), or by applying extracellular cadmium (0.2 mM), caffine, andryanodine (14). Nor is pool size changed by application of baclofen, hydroxysaclofen, trans-(1S, 3R)-l-aminocyclopentane-1,3-dicarboxilic acid, and (14, 16). That the LTD induction protocol can produce pool size changes thus comes as a surprise.
We find partial presynaptic synapse specificity for culture LTD, but technical difficulties prevent us from determining the mechanism of this effect. The partial specificity might simply be a weaker induction of LTD in the autaptic circuit or it could reflect some more complicated mechanism that involves a intracellular signaling mechanism. The important point is, however, that LTD in these circuit exhibits at least partial presynaptic synapse specificity because the LTD in the a → b circuit was always larger than that in the a → a circuit.
Various forms of heterosynaptic LTP and LTD have been reported. The term “heterosynaptic” refers to the fact that alterations in synaptic strength spreads to synapses that were inactive during the test pulse to evoke LTP or LTD, and the existence of heterosynaptic LTD (or LTP) typically means that the phenomenon is not, in our present terminology, postsynaptically synapse specific. The most commonly studied forms of heterosynaptic LTD are those in which inactive synapses converging onto the same postsynaptic cell undergo LTD (reviewed in Bear and Abraham, ref. 25). Nevertheless, in the simplest reciprocal circuit in culture, convergent inputs onto the postsynaptic cell do not undergo LTD but rather LTD spreads to synapses sharing the same presynaptic cell. This result suggests that there may be a presynaptic factor(s) for LTD expression that bypasses postsynaptic induction cues and can spread between presynaptic terminals in the presynaptic cell. Such presynaptic spread of synaptic depression has been observed at developing neuromuscular synapses in Xenopus nerve-muscle cultures (43). Our result is also in accord with lateral propagation of LTD described recently in a similar dissociated hippocampal culture preparation (29).
In addition to a failure of presynaptic synapse specificity for the LTD reported by Fitzsimonds et al. (29)—their “lateral propagation”—these authors also report a “backpropagation” of LTD to synapses made by a third cell on the presynaptic neuron. Such back-propagation of LTD was identified in serially connected triplets of cultured hippocampal pyramidal neurons. The spread of LTD was limited to the reverse direction, however; LTD did not propagate to the forward input. Interestingly, in our closed two-neuron circular circuit, the inverse input (b → a) does not undergo LTD. To confirm that the differences in LTD induction paradigms between our study and that of Fitzsimonds et al. (29) do not determine the spread of LTD to the inverse input, we stimulated the presynaptic cell during the LTD protocol in current clamp mode. Despite the use of stimulus conditions identical to that employed by Fitzsimonds et al. (29), LTD did not spread to reciprocal inputs (not shown). A closed circuit, therefore, displays properties distinct from serially connected neurons grown in culture. Moreover, a cellular mechanism that controls synapse-specificity postsynaptically is dominant over the presynaptic factor that permits spreading of LTD among synapses that share the same presynaptic cell. It remains to be determined what signaling machinery underlies such regulation.
Our characterization of synapse specificity and the identification of a cell biological mechanism that could account for LTD have been limited to the very special circumstances of cell culture. The relevance of our conclusions for synaptic plasticity as measured in slices or in vivo thus remains to be established. Whether or not changes in the size of the readily releasable pool participates in synaptic plasticity outside of culture, our experiments have revealed a long term regulatory mechanism that should operate in a more general context.
Acknowledgments
We thank Mu-ming Poo for comments on the manuscript and Chris Boyer for assistance with cell culture. This work was supported by Howard Hughes Medical Institute (C.F.S.), and National Institutes of Health Grant NS 12961 (C.F.S.).
ABBREVIATIONS
- LTD
long term depression
- LTP
long term potentiation
- EPSC
excitatory miniature postsynaptic current
- mEPSC
miniature EPSC
References
- 1.Liley A W, North K A K. J Neurophysiol. 1953;16:509. doi: 10.1152/jn.1953.16.5.509. [DOI] [PubMed] [Google Scholar]
- 2.Del Castillo J, Katz B. J Physiol (London) 1954;124:560. doi: 10.1113/jphysiol.1954.sp005129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Takeuchi A. Jpn J Physiol. 1958;8:102. doi: 10.2170/jjphysiol.8.102. [DOI] [PubMed] [Google Scholar]
- 4.Hubbard J I. J Physiol (London) 1963;169:641. doi: 10.1113/jphysiol.1963.sp007286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Thies R E. J Neurophysiol. 1965;28:427. doi: 10.1152/jn.1965.28.3.427. [DOI] [PubMed] [Google Scholar]
- 6.Betz W J. J Physiol (London) 1970;206:629–644. doi: 10.1113/jphysiol.1970.sp009034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Larkman A, Stratford K, Jack J. Nature (London) 1991;350:344–347. doi: 10.1038/350344a0. [DOI] [PubMed] [Google Scholar]
- 8.Thomson A M, Deuchars J, West D C. J Neurophysiol. 1993;70:2354–2369. doi: 10.1152/jn.1993.70.6.2354. [DOI] [PubMed] [Google Scholar]
- 9.Katz P S, Kirk M D, Govind C K. J Neurosci. 1993;13:3075–3089. doi: 10.1523/JNEUROSCI.13-07-03075.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Borst J G G, Helmchen F, Sakmann B. J Physiol (London) 1995;489:825–840. doi: 10.1113/jphysiol.1995.sp021095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liu G, Tsien R W. Nature (London) 1995;375:404–408. doi: 10.1038/375404a0. [DOI] [PubMed] [Google Scholar]
- 12.Liu G, Tsien R W. Neuropharmacology. 1995b;34:1407–1421. doi: 10.1016/0028-3908(95)00143-t. [DOI] [PubMed] [Google Scholar]
- 13.Stevens C F, Tsujimoto T. Proc Natl Acad Sci USA. 1995;92:846–849. doi: 10.1073/pnas.92.3.846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rosenmund C, Stevens C F. Neuron. 1996;16:1197–207. doi: 10.1016/s0896-6273(00)80146-4. [DOI] [PubMed] [Google Scholar]
- 15.Von Gersdorff H, Matthews G. J Neurosci. 1997;17:1919–1927. doi: 10.1523/JNEUROSCI.17-06-01919.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dobrunz L E, Stevens C F. Neuron. 1997;18:995–1008. doi: 10.1016/s0896-6273(00)80338-4. [DOI] [PubMed] [Google Scholar]
- 17.Thomson A M, Deuchars J. Trends Neurosci. 1994;17:119–126. doi: 10.1016/0166-2236(94)90121-x. [DOI] [PubMed] [Google Scholar]
- 18.Abbott L F, Sen K, Varela J A, Nelson S B. Science. 1997;275:220–222. doi: 10.1126/science.275.5297.221. [DOI] [PubMed] [Google Scholar]
- 19.Markram H, Tsodyks M V. Nature (London) 1996;382:807–810. doi: 10.1038/382807a0. [DOI] [PubMed] [Google Scholar]
- 20.Tsodyks M V, Markram H. Proc Natl Acad Sci USA. 1997;94:719–723. doi: 10.1073/pnas.94.2.719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Varela J A, Sen K, Gibson J, Fost J, Abbott L F, Nelson S B. J Neurosci. 1997;17:7926–7940. doi: 10.1523/JNEUROSCI.17-20-07926.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Von Gersdorff H, Vardi E, Matthews G, Sterling P. Neuron. 1996;16:1221–1227. doi: 10.1016/s0896-6273(00)80148-8. [DOI] [PubMed] [Google Scholar]
- 23.Schikorski T, Stevens C F. J Neurosci. 1997;17:5858–5867. doi: 10.1523/JNEUROSCI.17-15-05858.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Goda Y, Stevens C F. Neuron. 1996;16:103–111. doi: 10.1016/s0896-6273(00)80027-6. [DOI] [PubMed] [Google Scholar]
- 25.Bear M F, Abraham W C. Ann Rev Neurosci. 1996;19:437–462. doi: 10.1146/annurev.ne.19.030196.002253. [DOI] [PubMed] [Google Scholar]
- 26.Bonhoeffer T, Staiger V, Aertsen A. Proc Natl Acad Sci USA. 1989;86:8113–8117. doi: 10.1073/pnas.86.20.8113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schuman E M, Madison D V. Science. 1994;263:532–536. doi: 10.1126/science.8290963. [DOI] [PubMed] [Google Scholar]
- 28.Engert F, Bonhoeffer T. Nature (London) 1997;388:279–284. doi: 10.1038/40870. [DOI] [PubMed] [Google Scholar]
- 29.Fitzsimonds R M, Song H-j, Poo M-m. Nature (London) 1997;388:439–448. doi: 10.1038/41267. [DOI] [PubMed] [Google Scholar]
- 30.Geppert M, Goda Y, Hammer R E, Li C, Rosahl T W, Stevens C F, Südhof T C. Cell. 1994;79:717–727. doi: 10.1016/0092-8674(94)90556-8. [DOI] [PubMed] [Google Scholar]
- 31.Stevens C F, Wang Y. Nature (London) 1994;371:704–707. doi: 10.1038/371704a0. [DOI] [PubMed] [Google Scholar]
- 32.Bolshakov V Y, Siegelbaum S A. Science. 1994;264:1148–1152. doi: 10.1126/science.7909958. [DOI] [PubMed] [Google Scholar]
- 33.Xiao M-Y, Wigström H, Gustaffson B. Eur J Neurosci. 1994;6:1055–1057. doi: 10.1111/j.1460-9568.1994.tb00600.x. [DOI] [PubMed] [Google Scholar]
- 34.Isaac J T R, Nicoll R A, Malenka R C. Neuron. 1995;15:427–434. doi: 10.1016/0896-6273(95)90046-2. [DOI] [PubMed] [Google Scholar]
- 35.Liao D, Hessler N A, Malinow R. Nature (London) 1995;375:400–404. doi: 10.1038/375400a0. [DOI] [PubMed] [Google Scholar]
- 36.Goda Y, Südhof T C. Curr Opin Cell Biol. 1997;9:513–518. doi: 10.1016/s0955-0674(97)80027-0. [DOI] [PubMed] [Google Scholar]
- 37.Geppert M, Goda Y, Stevens C F, Südhof T C. Nature (London) 1997;387:810–814. doi: 10.1038/42954. [DOI] [PubMed] [Google Scholar]
- 38.Dodge F A, Jr, Rahamimoff R. J Physiol (London) 1967;193:419–432. doi: 10.1113/jphysiol.1967.sp008367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hubbard J I, Jones S F, Landau E M. J Physiol (London) 1968;197:639–657. doi: 10.1113/jphysiol.1968.sp008579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Blioch Z L, Glagoleva I M, Liberman E A, Nenashev V A. J Physiol (London) 1968;199:11–35. doi: 10.1113/jphysiol.1968.sp008637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Quastel D M J, Hackett J T, Cooke J D. Science. 1971;172:1034–1036. doi: 10.1126/science.172.3987.1034. [DOI] [PubMed] [Google Scholar]
- 42.Shimone Y, Alnaes E, Rahamimoff R. Nature (London) 1977;267:170–173. doi: 10.1038/267170a0. [DOI] [PubMed] [Google Scholar]
- 43.Cash S, Zucker R S, Poo M-m. Science. 1996;272:998–1001. doi: 10.1126/science.272.5264.998. [DOI] [PubMed] [Google Scholar]