Abstract
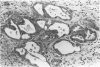
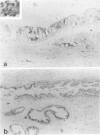
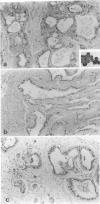
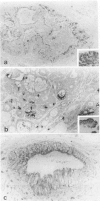
Our previous studies have shown that pancreatic cancer induced in Syrian hamsters by N-nitrosobis(2-oxopropyl)amine (BOP) shows remarkable similarities with the human disease in morphologic and biologic characteristics. Moreover, both human and hamster pancreatic cancer share expression of some tumor-associated antigens, such as those with blood group specificities, including A, B, H, Leb, Lex, and Ley. By examining other antigens commonly expressed in human pancreatic cancer, we have found that monoclonal antibodies CO17-1A (recognizing 17-1A antigen), OC 125 (recognizing CA 125 antigen), B72.3 (recognizing TAG-72 and DU-PAN-2 react with induced pancreatic cancer in a pattern similar to that seen in human pancreatic cancer. Remarkably, although the epitopes of the antigens recognized by these three antibodies are different, many tumor cells were reactive with all these antibodies. However, in contrast to the human cancer, none of these antigens were expressed in the normal hamster pancreatic tissue, except for 17-1A. However, all of these antigens were expressed in some hamster tissues showing the same cellular localization as pancreatic cancer cells and corresponded, to a great extent, with findings in human tissue. Expression of these antigens was diminished in vitro (cell culture) but was regained in vivo (homologous transplantation). The results emphasize the usefulness of this experimental model for studying some aspects of tissue antigenicity, particularly as it relates to pancreatic cancer.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bast R. C., Jr, Feeney M., Lazarus H., Nadler L. M., Colvin R. B., Knapp R. C. Reactivity of a monoclonal antibody with human ovarian carcinoma. J Clin Invest. 1981 Nov;68(5):1331–1337. doi: 10.1172/JCI110380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colcher D., Esteban J. M., Carrasquillo J. A., Sugarbaker P., Reynolds J. C., Bryant G., Larson S. M., Schlom J. Quantitative analyses of selective radiolabeled monoclonal antibody localization in metastatic lesions of colorectal cancer patients. Cancer Res. 1987 Feb 15;47(4):1185–1189. [PubMed] [Google Scholar]
- Colcher D., Hand P. H., Nuti M., Schlom J. A spectrum of monoclonal antibodies reactive with human mammary tumor cells. Proc Natl Acad Sci U S A. 1981 May;78(5):3199–3203. doi: 10.1073/pnas.78.5.3199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis H. M., Zurawski V. R., Jr, Bast R. C., Jr, Klug T. L. Characterization of the CA 125 antigen associated with human epithelial ovarian carcinomas. Cancer Res. 1986 Dec;46(12 Pt 1):6143–6148. [PubMed] [Google Scholar]
- Egami H., Takiyama Y., Cano M., Houser W. H., Pour P. M. Establishment of hamster pancreatic ductal carcinoma cell line (PC-1) producing blood group-related antigens. Carcinogenesis. 1989 May;10(5):861–869. doi: 10.1093/carcin/10.5.861. [DOI] [PubMed] [Google Scholar]
- Göttlinger H. G., Funke I., Johnson J. P., Gokel J. M., Riethmüller G. The epithelial cell surface antigen 17-1A, a target for antibody-mediated tumor therapy: its biochemical nature, tissue distribution and recognition by different monoclonal antibodies. Int J Cancer. 1986 Jul 15;38(1):47–53. doi: 10.1002/ijc.2910380109. [DOI] [PubMed] [Google Scholar]
- Herlyn D., Koprowski H. IgG2a monoclonal antibodies inhibit human tumor growth through interaction with effector cells. Proc Natl Acad Sci U S A. 1982 Aug;79(15):4761–4765. doi: 10.1073/pnas.79.15.4761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herlyn M., Steplewski Z., Herlyn D., Koprowski H. Colorectal carcinoma-specific antigen: detection by means of monoclonal antibodies. Proc Natl Acad Sci U S A. 1979 Mar;76(3):1438–1442. doi: 10.1073/pnas.76.3.1438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horan Hand P., Colcher D., Salomon D., Ridge J., Noguchi P., Schlom J. Influence of spatial configuration of carcinoma cell populations on the expression of a tumor-associated glycoprotein. Cancer Res. 1985 Feb;45(2):833–840. [PubMed] [Google Scholar]
- Hsu S. M., Raine L., Fanger H. Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem. 1981 Apr;29(4):577–580. doi: 10.1177/29.4.6166661. [DOI] [PubMed] [Google Scholar]
- Kabawat S. E., Bast R. C., Jr, Bhan A. K., Welch W. R., Knapp R. C., Colvin R. B. Tissue distribution of a coelomic-epithelium-related antigen recognized by the monoclonal antibody OC125. Int J Gynecol Pathol. 1983;2(3):275–285. doi: 10.1097/00004347-198303000-00005. [DOI] [PubMed] [Google Scholar]
- Kjeldsen T., Clausen H., Hirohashi S., Ogawa T., Iijima H., Hakomori S. Preparation and characterization of monoclonal antibodies directed to the tumor-associated O-linked sialosyl-2----6 alpha-N-acetylgalactosaminyl (sialosyl-Tn) epitope. Cancer Res. 1988 Apr 15;48(8):2214–2220. [PubMed] [Google Scholar]
- Klug T. L., Sattler M. A., Colcher D., Schlom J. Monoclonal antibody immunoradiometric assay for an antigenic determinant (CA 72) on a novel pancarcinoma antigen (TAG-72). Int J Cancer. 1986 Nov 15;38(5):661–669. doi: 10.1002/ijc.2910380508. [DOI] [PubMed] [Google Scholar]
- Lan M. S., Bast R. C., Jr, Colnaghi M. I., Knapp R. C., Colcher D., Schlom J., Metzgar R. S. Co-expression of human cancer-associated epitopes on mucin molecules. Int J Cancer. 1987 Jan 15;39(1):68–72. doi: 10.1002/ijc.2910390112. [DOI] [PubMed] [Google Scholar]
- Magnani J. L., Nilsson B., Brockhaus M., Zopf D., Steplewski Z., Koprowski H., Ginsburg V. A monoclonal antibody-defined antigen associated with gastrointestinal cancer is a ganglioside containing sialylated lacto-N-fucopentaose II. J Biol Chem. 1982 Dec 10;257(23):14365–14369. [PubMed] [Google Scholar]
- Metzgar R. S., Gaillard M. T., Levine S. J., Tuck F. L., Bossen E. H., Borowitz M. J. Antigens of human pancreatic adenocarcinoma cells defined by murine monoclonal antibodies. Cancer Res. 1982 Feb;42(2):601–608. [PubMed] [Google Scholar]
- Metzgar R. S., Rodriguez N., Finn O. J., Lan M. S., Daasch V. N., Fernsten P. D., Meyers W. C., Sindelar W. F., Sandler R. S., Seigler H. F. Detection of a pancreatic cancer-associated antigen (DU-PAN-2 antigen) in serum and ascites of patients with adenocarcinoma. Proc Natl Acad Sci U S A. 1984 Aug;81(16):5242–5246. doi: 10.1073/pnas.81.16.5242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pour P. M., Runge R. G., Birt D., Gingell R., Lawson T., Nagel D., Wallcave L., Salmasi S. Z. Current knowledge of pancreatic carcinogenesis in the hamster and its relevance to the human disease. Cancer. 1981 Mar 15;47(6 Suppl):1573–1589. doi: 10.1002/1097-0142(19810315)47:6+<1573::aid-cncr2820471420>3.0.co;2-k. [DOI] [PubMed] [Google Scholar]
- Pour P. M., Tempero M. M., Takasaki H., Uchida E., Takiyama Y., Burnett D. A., Steplewski Z. Expression of blood group-related antigens ABH, Lewis A, Lewis B, Lewis X, Lewis Y, and CA 19-9 in pancreatic cancer cells in comparison with the patient's blood group type. Cancer Res. 1988 Oct 1;48(19):5422–5426. [PubMed] [Google Scholar]
- Pour P. M., Uchida E., Burnett D. A., Steplewski Z. Blood-group antigen expression during pancreatic cancer induction in hamsters. Int J Pancreatol. 1986 Dec;1(5-6):327–340. doi: 10.1007/BF02801865. [DOI] [PubMed] [Google Scholar]
- Pour P., Althoff J., Krüger F. W., Mohr U. A potent pancreatic carcinogen in Syrian hamsters: N-nitrosobis(2-oxopropyl)amine. J Natl Cancer Inst. 1977 May;58(5):1449–1453. doi: 10.1093/jnci/58.5.1449. [DOI] [PubMed] [Google Scholar]
- Pour P., Mohr U., Cardesa A., Althoff J., Krüger F. W. Pancreatic neoplasms in an animal model: morphological, biological, and comparative studies. Cancer. 1975 Aug;36(2):379–389. doi: 10.1002/1097-0142(197508)36:2<379::aid-cncr2820360213>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- Sears H. F., Herlyn D., Steplewski Z., Koprowski H. Phase II clinical trial of a murine monoclonal antibody cytotoxic for gastrointestinal adenocarcinoma. Cancer Res. 1985 Nov;45(11 Pt 2):5910–5913. [PubMed] [Google Scholar]
- Takasaki H., Tempero M. A., Uchida E., Büchler M., Ness M. J., Burnett D. A., Metzgar R. S., Colcher D., Schlom J., Pour P. M. Comparative studies on the expression of tumor-associated glycoprotein (TAG-72), CA 19-9 and DU-PAN-2 in normal, benign and malignant pancreatic tissue. Int J Cancer. 1988 Nov 15;42(5):681–686. doi: 10.1002/ijc.2910420508. [DOI] [PubMed] [Google Scholar]
- Takasaki H., Uchida E., Tempero M. A., Burnett D. A., Metzgar R. S., Pour P. M. Correlative study on expression of CA 19-9 and DU-PAN-2 in tumor tissue and in serum of pancreatic cancer patients. Cancer Res. 1988 Mar 15;48(6):1435–1438. [PubMed] [Google Scholar]
- Takiyama Y., Tempero M. A., Takasaki H., Onda M., Tsuchiya R., Büchler M., Ness M., Colcher D., Schlom J., Pour P. M. Reactivity of CO17-1A and B72.3 in benign and malignant pancreatic diseases. Hum Pathol. 1989 Sep;20(9):832–838. doi: 10.1016/0046-8177(89)90093-2. [DOI] [PubMed] [Google Scholar]
- Tempero M. A., Pour P. M., Uchida E., Herlyn D., Steplewski Z. Monoclonal antibody CO17-1A and leukapheresis in immunotherapy of pancreatic cancer. Hybridoma. 1986 Jul;5 (Suppl 1):S133–S138. [PubMed] [Google Scholar]
- Thor A., Ohuchi N., Szpak C. A., Johnston W. W., Schlom J. Distribution of oncofetal antigen tumor-associated glycoprotein-72 defined by monoclonal antibody B72.3. Cancer Res. 1986 Jun;46(6):3118–3124. [PubMed] [Google Scholar]