Abstract
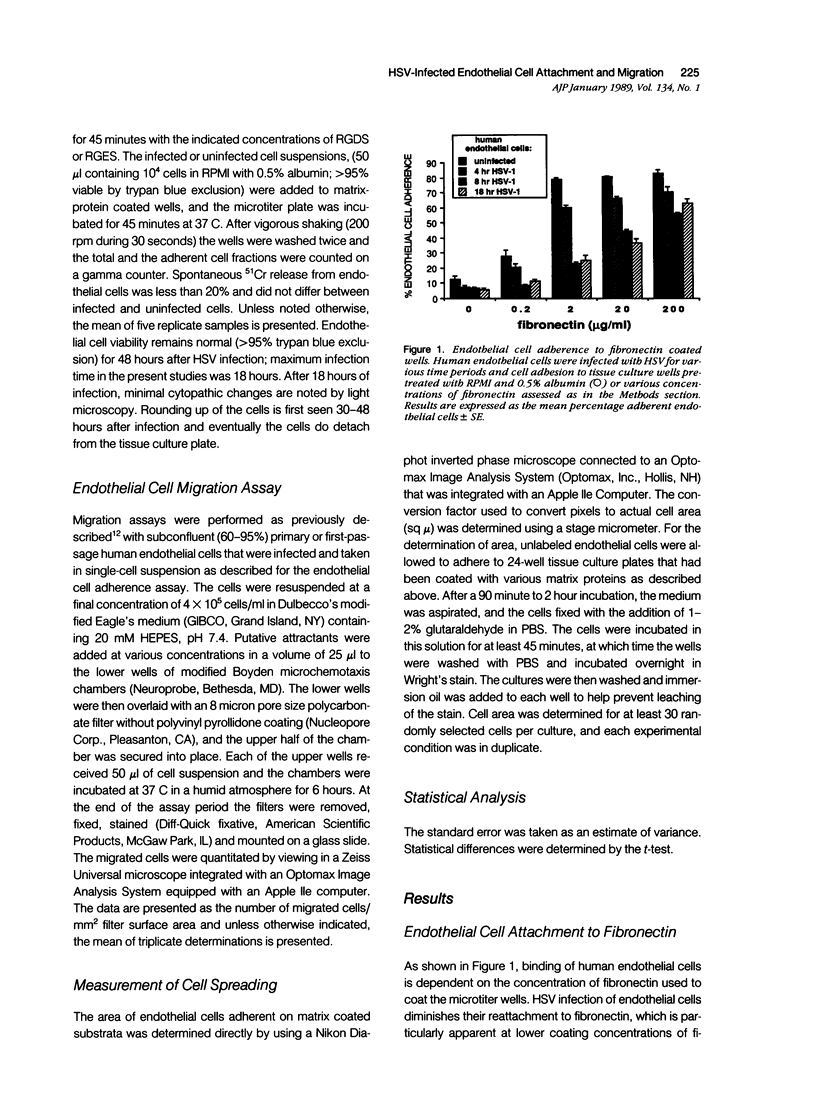
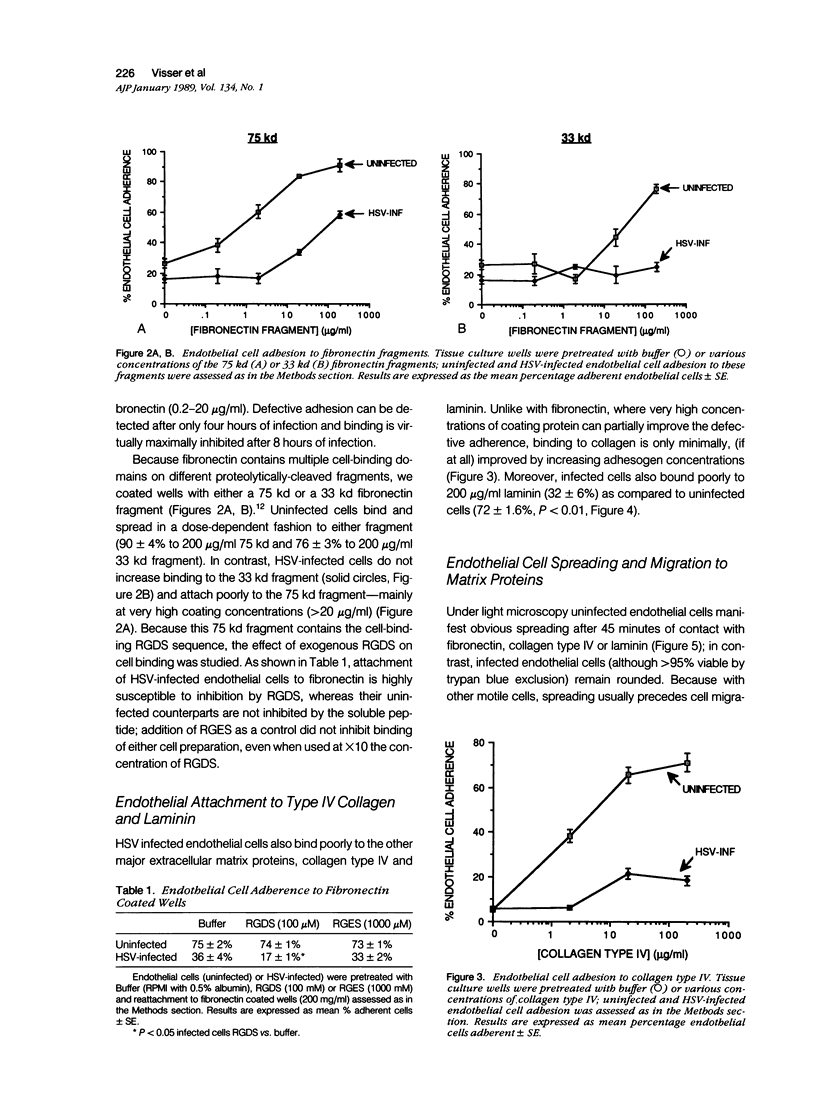
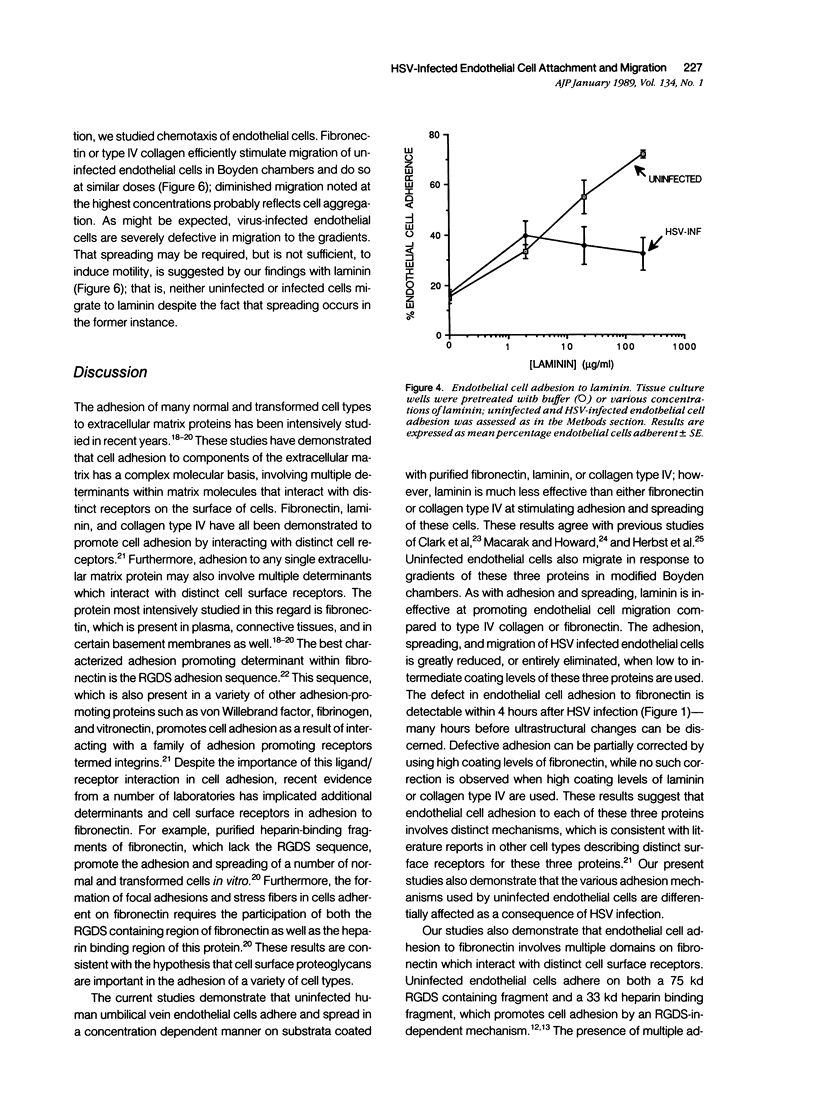
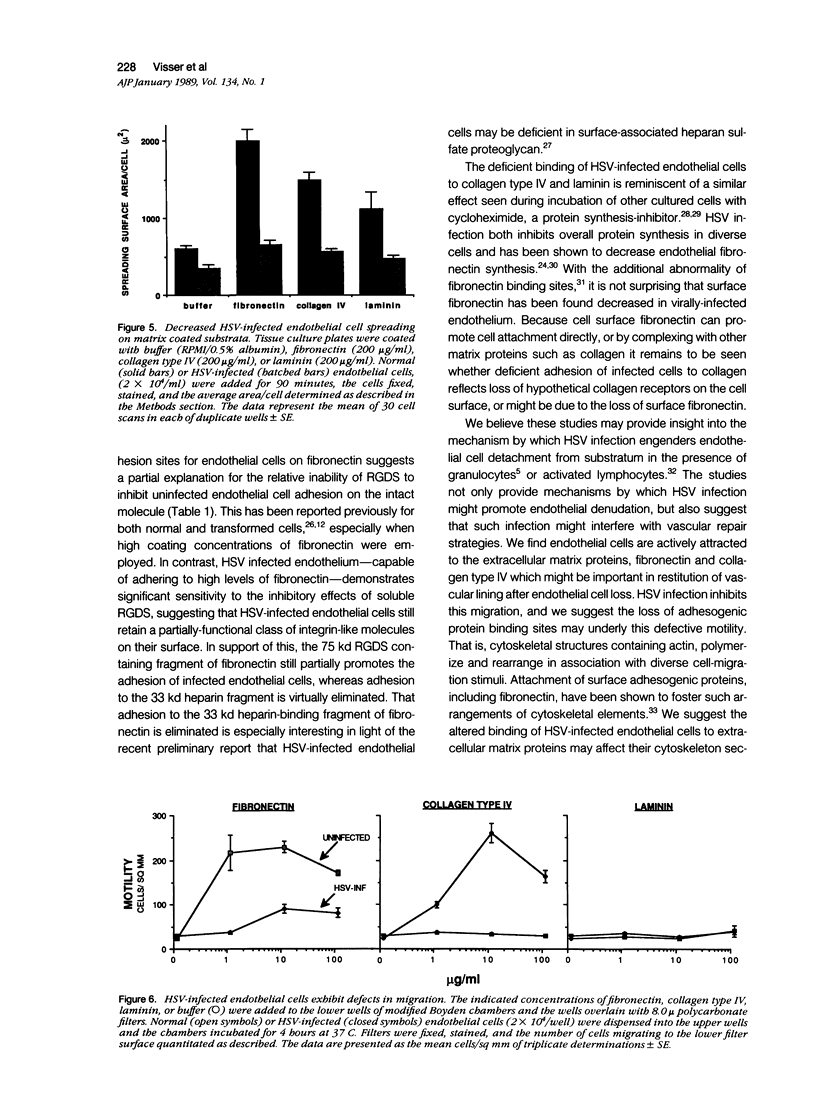
Herpes simplex virus (HSV) infection may be involved in various endothelial-injury syndromes, including vasculitis and atherosclerosis. In a previous study, it was reported that HSV-infected human umbilical endothelial cells are more vulnerable to detachment mediated by granulocyte-secreted proteases. To elucidate the molecular basis of this observation, the authors examined the interaction of infected endothelial cells with the purified basement membrane proteins, fibronectin, laminin, and type IV collagen. HSV-infected endothelial cells exhibited defects in their ability to adhere, spread, and migrate on all three matrix components. This defective adhesion could be partially overcome by increasing concentrations of fibronectin; in contrast, no abrogation of deficient binding occurs with increased levels of laminin or collagen type IV. This suggests that endothelial cells may use different surface constituents for binding to the three proteins and use multiple "receptors" for adhesion to the fibronectin molecule--"receptors" that are variably affected by HSV infection. The authors investigated this supposition by assaying adhesion of normal and infected endothelial cells to two non-overlapping cell-adhesion promoting fragments of fibronectin: 1) a 75 kd motility-promoting fragment which contains the arginyl-glycyl-aspartylserine (RGDS) adhesion sequence, and 2) a 33 kd carboxyl-terminal heparin binding fragment, which promotes cell adhesion by an RGDS-independent mechanism. Normal endothelial cells adhered and spread on both purified fragments. In contrast, while infected endothelial cells could adhere, albeit rather poorly, to high coating concentrations of the 75 kd fragment, these cells did not bind to the 33 kd heparin binding fragment of fibronectin at all. These results support the concept that endothelial cells adhere to multiple domains of fibronectin, and that HSV infection preferentially abrogates binding to the heparin-binding domain, while leaving relatively intact receptors for the RGDS-containing domain. In support, soluble RGDS significantly blocked fibronectin adhesion of infected, but not control, endothelial cells. It is concluded that HSV infection inhibits the interaction of endothelial cells with basement membrane proteins and weakens their tethering to substratum. This tethering is inadequate for proper cell spreading or movement to occur and may result in both excessive endothelial lift-off and impaired vascular repair in HSV infections.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Benditt E. P., Barrett T., McDougall J. K. Viruses in the etiology of atherosclerosis. Proc Natl Acad Sci U S A. 1983 Oct;80(20):6386–6389. doi: 10.1073/pnas.80.20.6386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bretscher M. S. Endocytosis: relation to capping and cell locomotion. Science. 1984 May 18;224(4650):681–686. doi: 10.1126/science.6719108. [DOI] [PubMed] [Google Scholar]
- Buck C. A., Horwitz A. F. Cell surface receptors for extracellular matrix molecules. Annu Rev Cell Biol. 1987;3:179–205. doi: 10.1146/annurev.cb.03.110187.001143. [DOI] [PubMed] [Google Scholar]
- Chen W. T., Wang J., Hasegawa T., Yamada S. S., Yamada K. M. Regulation of fibronectin receptor distribution by transformation, exogenous fibronectin, and synthetic peptides. J Cell Biol. 1986 Nov;103(5):1649–1661. doi: 10.1083/jcb.103.5.1649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark R. A., Folkvord J. M., Nielsen L. D. Either exogenous or endogenous fibronectin can promote adherence of human endothelial cells. J Cell Sci. 1986 Jun;82:263–280. doi: 10.1242/jcs.82.1.263. [DOI] [PubMed] [Google Scholar]
- Dienes H. P., Knoblich A., Falke D. Loss of surface fibronectin after infection of cultured cells by HSV-1 and 2. Arch Virol. 1985;86(3-4):223–237. doi: 10.1007/BF01309827. [DOI] [PubMed] [Google Scholar]
- Fabricant C. G., Fabricant J., Litrenta M. M., Minick C. R. Virus-induced atherosclerosis. J Exp Med. 1978 Jul 1;148(1):335–340. doi: 10.1084/jem.148.1.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman H. M., Macarak E. J., MacGregor R. R., Wolfe J., Kefalides N. A. Virus infection of endothelial cells. J Infect Dis. 1981 Feb;143(2):266–273. doi: 10.1093/infdis/143.2.266. [DOI] [PubMed] [Google Scholar]
- Gyorkey F., Melnick J. L., Guinn G. A., Gyorkey P., DeBakey M. E. Herpesviridae in the endothelial and smooth muscle cells of the proximal aorta in arteriosclerotic patients. Exp Mol Pathol. 1984 Jun;40(3):328–339. doi: 10.1016/0014-4800(84)90050-9. [DOI] [PubMed] [Google Scholar]
- Hajjar D. P., Pomerantz K. B., Falcone D. J., Weksler B. B., Grant A. J. Herpes simplex virus infection in human arterial cells. Implications in arteriosclerosis. J Clin Invest. 1987 Nov;80(5):1317–1321. doi: 10.1172/JCI113208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hebbel R. P., Visser M. R., Goodman J. L., Jacob H. S., Vercellotti G. M. Potentiated adherence of sickle erythrocytes to endothelium infected by virus. J Clin Invest. 1987 Nov;80(5):1503–1506. doi: 10.1172/JCI113233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herbst T. J., McCarthy J. B., Tsilibary E. C., Furcht L. T. Differential effects of laminin, intact type IV collagen, and specific domains of type IV collagen on endothelial cell adhesion and migration. J Cell Biol. 1988 Apr;106(4):1365–1373. doi: 10.1083/jcb.106.4.1365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaffe E. A., Hoyer L. W., Nachman R. L. Synthesis of antihemophilic factor antigen by cultured human endothelial cells. J Clin Invest. 1973 Nov;52(11):2757–2764. doi: 10.1172/JCI107471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kefalides N. A., Ziaie Z. Herpes simplex virus suppression of human endothelial matrix protein synthesis is independent of viral protein synthesis. Lab Invest. 1986 Sep;55(3):328–336. [PubMed] [Google Scholar]
- Kleinman H. K., McGarvey M. L., Liotta L. A., Robey P. G., Tryggvason K., Martin G. R. Isolation and characterization of type IV procollagen, laminin, and heparan sulfate proteoglycan from the EHS sarcoma. Biochemistry. 1982 Nov 23;21(24):6188–6193. doi: 10.1021/bi00267a025. [DOI] [PubMed] [Google Scholar]
- MacGregor R. R., Friedman H. M., Macarak E. J., Kefalides N. A. Virus infection of endothelial cells increases granulocyte adherence. J Clin Invest. 1980 Jun;65(6):1469–1477. doi: 10.1172/JCI109811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macarak E. J., Howard P. S. Adhesion of endothelial cells to extracellular matrix proteins. J Cell Physiol. 1983 Jul;116(1):76–86. doi: 10.1002/jcp.1041160112. [DOI] [PubMed] [Google Scholar]
- Madri J. A., Stenn K. S. Aortic endothelial cell migration. I. Matrix requirements and composition. Am J Pathol. 1982 Feb;106(2):180–186. [PMC free article] [PubMed] [Google Scholar]
- McCarthy J. B., Chelberg M. K., Mickelson D. J., Furcht L. T. Localization and chemical synthesis of fibronectin peptides with melanoma adhesion and heparin binding activities. Biochemistry. 1988 Feb 23;27(4):1380–1388. doi: 10.1021/bi00404a044. [DOI] [PubMed] [Google Scholar]
- McCarthy J. B., Hagen S. T., Furcht L. T. Human fibronectin contains distinct adhesion- and motility-promoting domains for metastatic melanoma cells. J Cell Biol. 1986 Jan;102(1):179–188. doi: 10.1083/jcb.102.1.179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McSorley J., Shapiro L., Brownstein M. H., Hsu K. C. Herpes simplex and varicella-zoster: comparative histopathology of 77 cases. Int J Dermatol. 1974 Mar-Apr;13(2):69–75. doi: 10.1111/j.1365-4362.1974.tb01769.x. [DOI] [PubMed] [Google Scholar]
- Minick C. R., Fabricant C. G., Fabricant J., Litrenta M. M. Atheroarteriosclerosis induced by infection with a herpesvirus. Am J Pathol. 1979 Sep;96(3):673–706. [PMC free article] [PubMed] [Google Scholar]
- Moldow C. F., Jacob H. S. Endothelial culture, neutrophil or enzymic generation of free radicals: in vitro methods for the study of endothelial injury. Methods Enzymol. 1984;105:378–385. doi: 10.1016/s0076-6879(84)05051-5. [DOI] [PubMed] [Google Scholar]
- Norrild B., Lehto V. P., Virtanen I. Organization of cytoskeleton elements during herpes simplex virus type 1 infection of human fibroblasts: an immunofluorescence study. J Gen Virol. 1986 Jan;67(Pt 1):97–105. doi: 10.1099/0022-1317-67-1-97. [DOI] [PubMed] [Google Scholar]
- Rosenthal K. S., Leuther M. D., Barisas B. G. Herpes simplex virus binding and entry modulate cell surface protein mobility. J Virol. 1984 Mar;49(3):980–983. doi: 10.1128/jvi.49.3.980-983.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruoslahti E., Hayman E. G., Pierschbacher M. D. Extracellular matrices and cell adhesion. Arteriosclerosis. 1985 Nov-Dec;5(6):581–594. doi: 10.1161/01.atv.5.6.581. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E., Pierschbacher M. D. Arg-Gly-Asp: a versatile cell recognition signal. Cell. 1986 Feb 28;44(4):517–518. doi: 10.1016/0092-8674(86)90259-x. [DOI] [PubMed] [Google Scholar]
- Yamada K. M. Cell surface interactions with extracellular materials. Annu Rev Biochem. 1983;52:761–799. doi: 10.1146/annurev.bi.52.070183.003553. [DOI] [PubMed] [Google Scholar]
- Yamada K. M., Kennedy D. W. Dualistic nature of adhesive protein function: fibronectin and its biologically active peptide fragments can autoinhibit fibronectin function. J Cell Biol. 1984 Jul;99(1 Pt 1):29–36. doi: 10.1083/jcb.99.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziaie Z., Friedman H. M., Kefalides N. A. Suppression of matrix protein synthesis by herpes simplex virus type 1 in human endothelial cells. Coll Relat Res. 1986 Oct;6(4):333–349. doi: 10.1016/s0174-173x(86)80004-8. [DOI] [PubMed] [Google Scholar]



