Abstract
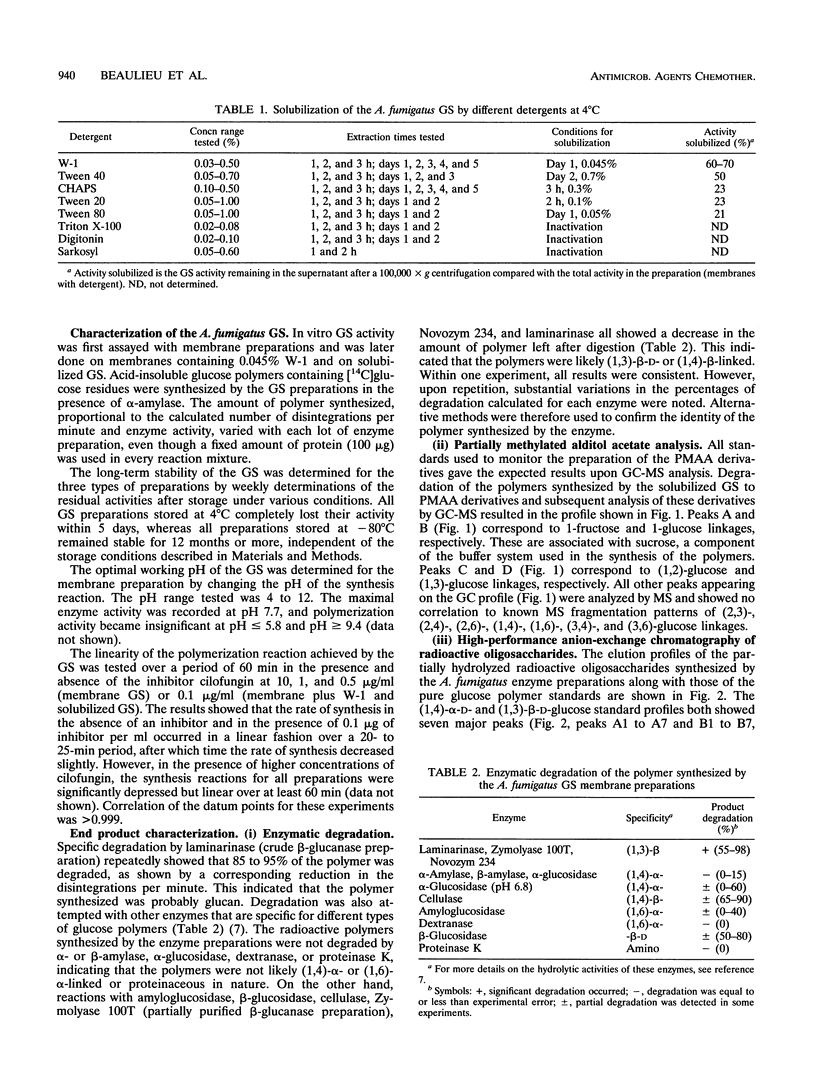
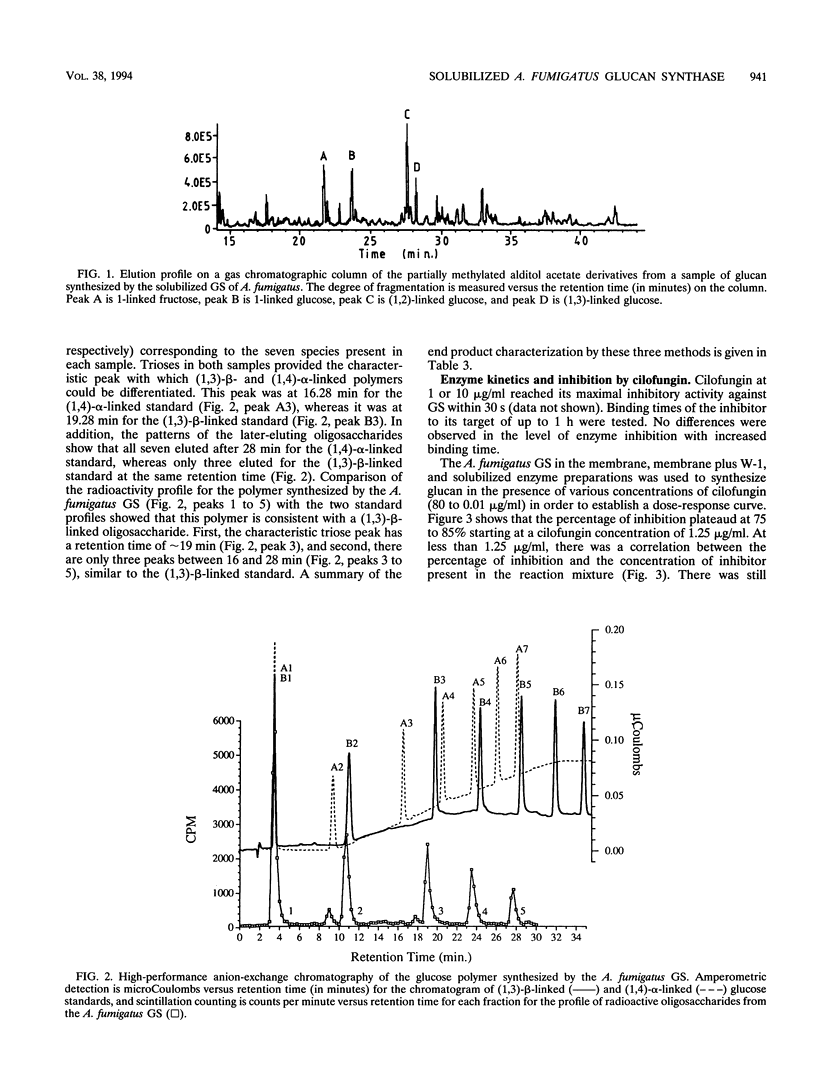
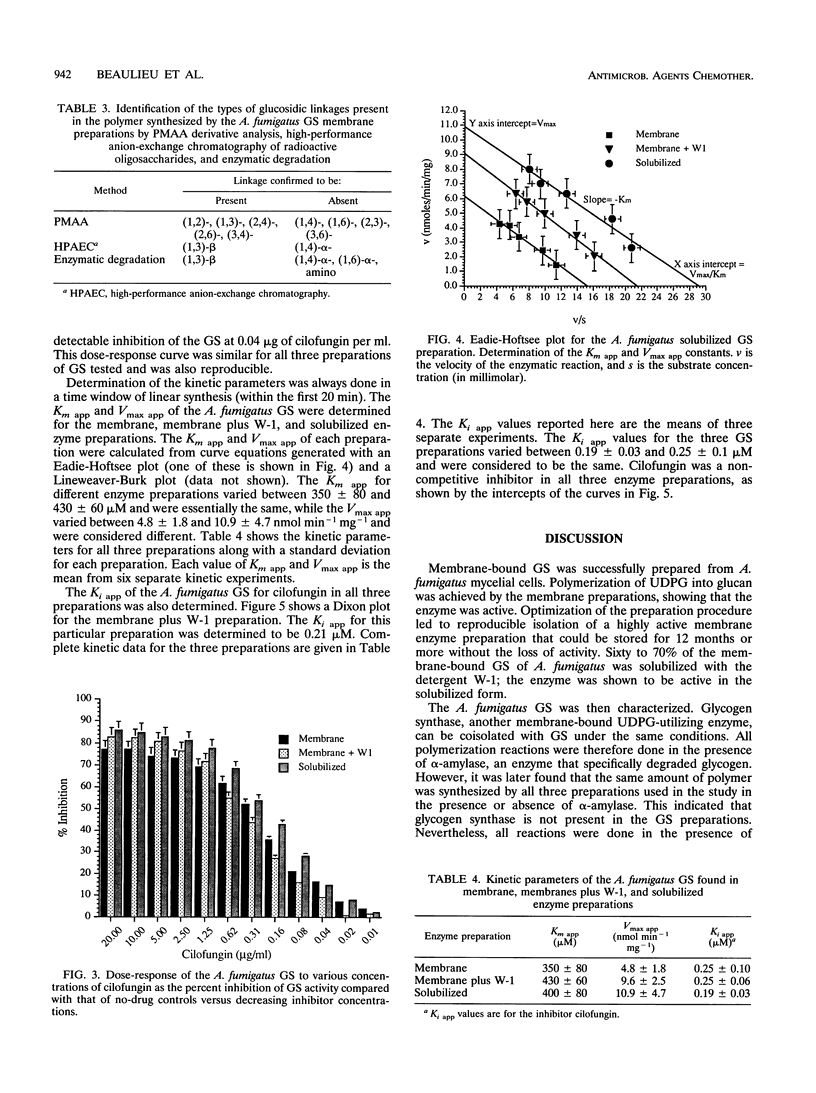
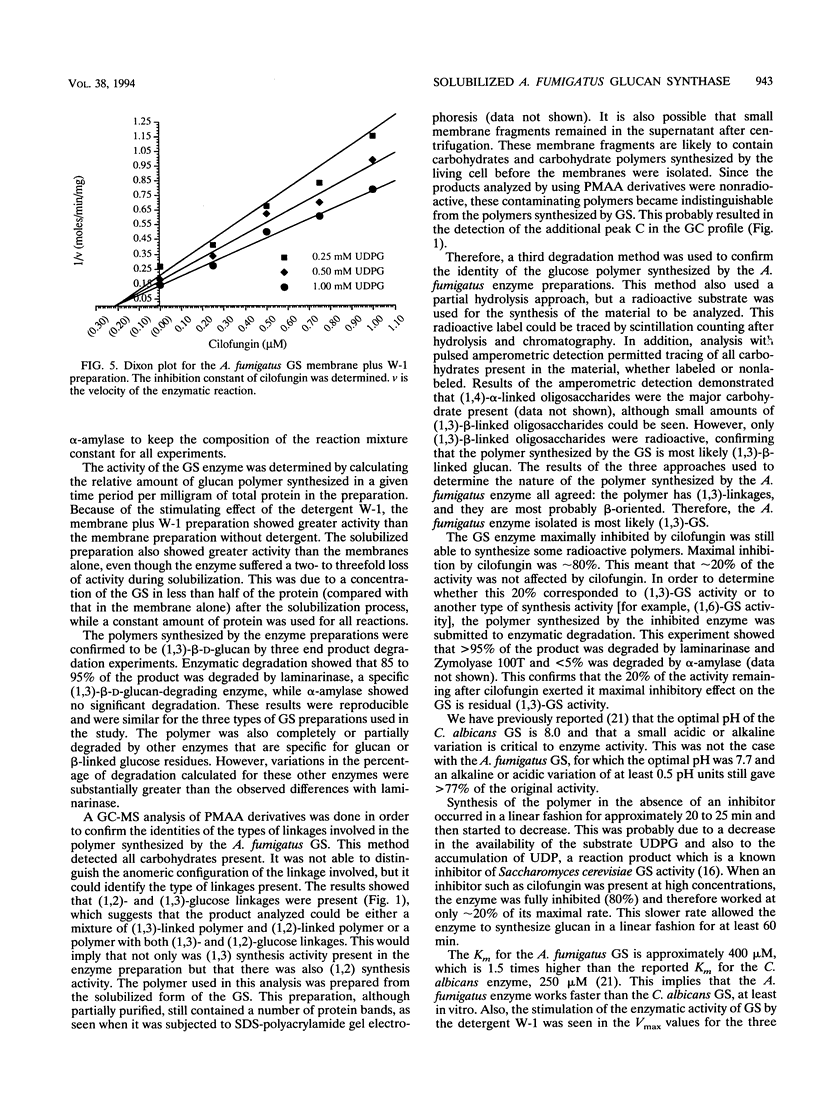
(1,3)-beta-D-Glucan synthase, a major cell wall synthesis enzyme, is the target of antifungal drugs of the lipopeptide class. Aspergillus fumigatus (1,3)-beta-D-glucan synthase was prepared and its activity was measured by incorporation of [14C]glucose from UDP-[U-14C]glucose into an insoluble polymer in the presence of alpha-amylase. Solubilization of the (1,3)-beta-D-glucan synthase was attempted with several detergents, and the maximum percent solubilization was obtained with a polyoxyethylene ether detergent, W-1. Up to 70% of enzyme activity and 50% of total protein were recovered when 1-mg/ml membrane preparations were extracted with 0.045% W-1 at 4 degrees C overnight. Confirmation of the presence of a (1,3)-beta-D-glucose polymer synthesized by this glucan synthase was done by three methods. The first was enzymatic end product degradation by alpha-amylase (no degradation) and beta-glucanase (85 to 95% degradation). The second was gas chromatography-mass spectroscopy analysis of the partially methylated alditol acetate derivatives prepared from total carbohydrate polymers present in the sample. This method identified the presence of (1,3)- and (1,2)-glucosidic linkages. The third was high-performance anion exchange chromatography of radioactive oligosaccharides. This method allowed differentiation of the newly synthesized, radioactive polymers from the contaminating carbohydrates already present in the preparation. The results showed that the polymer synthesized comprised oligosaccharides consistent with beta-(1,3)-linked sugars. Maximal inhibition of the (1,3)-beta-D-glucan synthase by the lipopeptide antifungal agent cilofungin was 80%. Dose-response experiments with this inhibitor showed that the solubilized enzyme was maximally inhibited at a cilofungin concentration of 1.25 microgram/ml and showed <5% inhibition at 0.02 microgram/ml. The apparent K(m) (K(m app)) for the solubilized glucan synthase was 400 +/- 80 microM, and the apparent K(i) (K(i app)) for cilofungin was 0.19 +/- 0.03 microM. Inhibition of A.fumigatus (1,3)-beta-D-glucan synthase with cilofungin was noncompetitive, as it was for the Candida albicans (1,3)-beta-D-glucan synthase.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bartnicki-Garcia S. Cell wall chemistry, morphogenesis, and taxonomy of fungi. Annu Rev Microbiol. 1968;22:87–108. doi: 10.1146/annurev.mi.22.100168.000511. [DOI] [PubMed] [Google Scholar]
- Beaulieu D., Tang J., Zeckner D. J., Parr T. R., Jr Correlation of cilofungin in vivo efficacy with its activity against Aspergillus fumigatus (1,3)-beta-D-glucan synthase. FEMS Microbiol Lett. 1993 Apr 1;108(2):133–137. doi: 10.1111/j.1574-6968.1993.tb06088.x. [DOI] [PubMed] [Google Scholar]
- Borgia P. T., Dodge C. L. Characterization of Aspergillus nidulans mutants deficient in cell wall chitin or glucan. J Bacteriol. 1992 Jan;174(2):377–383. doi: 10.1128/jb.174.2.377-383.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calderone R. A., Braun P. C. Adherence and receptor relationships of Candida albicans. Microbiol Rev. 1991 Mar;55(1):1–20. doi: 10.1128/mr.55.1.1-20.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang M. S., Cabib E. Regulation of fungal cell wall growth: a guanine nucleotide-binding, proteinaceous component required for activity of (1----3)-beta-D-glucan synthase. Proc Natl Acad Sci U S A. 1986 Aug;83(16):5808–5812. doi: 10.1073/pnas.83.16.5808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawistowska-Schröder E. T., Kerridge D., Perry H. Echinocandin inhibition of 1,3-beta-D-glucan synthase from Candida albicans. FEBS Lett. 1984 Jul 23;173(1):134–138. doi: 10.1016/0014-5793(84)81032-7. [DOI] [PubMed] [Google Scholar]
- Shematek E. M., Braatz J. A., Cabib E. Biosynthesis of the yeast cell wall. I. Preparation and properties of beta-(1 leads to 3)glucan synthetase. J Biol Chem. 1980 Feb 10;255(3):888–894. [PubMed] [Google Scholar]
- Shepherd M. G., Poulter R. T., Sullivan P. A. Candida albicans: biology, genetics, and pathogenicity. Annu Rev Microbiol. 1985;39:579–614. doi: 10.1146/annurev.mi.39.100185.003051. [DOI] [PubMed] [Google Scholar]
- Taft C. S., Selitrennikoff C. P. LY121019 inhibits Neurospora crassa growth and (1-3)-beta-D-glucan synthase. J Antibiot (Tokyo) 1988 May;41(5):697–701. doi: 10.7164/antibiotics.41.697. [DOI] [PubMed] [Google Scholar]
- Taft C. S., Stark T., Selitrennikoff C. P. Cilofungin (LY121019) inhibits Candida albicans (1-3)-beta-D-glucan synthase activity. Antimicrob Agents Chemother. 1988 Dec;32(12):1901–1903. doi: 10.1128/aac.32.12.1901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang J., Parr T. R., Jr W-1 solubilization and kinetics of inhibition by cilofungin of Candida albicans (1,3)-beta-D-glucan synthase. Antimicrob Agents Chemother. 1991 Jan;35(1):99–103. doi: 10.1128/aac.35.1.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young L. S. Aspergillus infection in the neutropenic host. Hosp Pract (Off Ed) 1989 May 30;24(5A):37–43. [PubMed] [Google Scholar]


