Abstract
Aims
To determine factors predictive for leflunomide drug survival in an outpatient population with rheumatoid arthritis in a setting of care-as-usual.
Methods
A standard dataset was collected from medical records of consecutive outpatients on leflunomide treatment for rheumatoid arthritis between January 2000 and June 2003. The dataset consisted of patient, disease and treatment characteristics at the start of leflunomide treatment, and data on leflunomide use.
Results
Leflunomide was started in 279 patients and 173 patients (62.0%) withdrew from treatment during follow-up. From univariate analysis, concomitant systemic corticosteroid use [hazard ratio (HR) (95% confidence interval) 1.35 (1.00, 1.83)] and an erythrocyte sedimentation rate <35 mm h−1[HR 1.38 (1.01, 1.88)] at start of leflunomide were found to be predictive for better leflunomide survival. Furthermore, the attending rheumatologist was correlated with leflunomide drug survival. Hazard ratios varied, depending on the individual rheumatologist, from 0.60 to 2.66. Multivariate analysis suggested attending rheumatologist (HR varying from 0.54 to 2.30 depending on the individual rheumatologist), concomitant systemic corticosteroid use [HR 1.58 (1.14 2.21)] and erythrocyte sedimentation rate <35 mm h−1[HR 1.42 (1.03, 1.96)] at start of leflunomide to be associated with leflunomide survival.
Conclusions
Concomitant systemic corticosteroid use, erythrocyte sedimentation rate at the start of treatment and attending rheumatologist were found to be predictive for leflunomide survival. Information on these predictors at the start of leflunomide therapy may offer information on which patients are at an increased risk of withdrawal from leflunomide. Whether this information leads to optimization of leflunomide treatment outcomes remains to be studied.
Keywords: drug survival, leflunomide, predictors, rheumatoid arthritis
Introduction
The view that randomized controlled trials are the ‘gold standard’ for evaluation and that observational methods have little additional value is widely accepted. According to some experts, the results of observational studies should not be used for defining evidence-based medical care. The major criticism of observational studies is that unrecognized confounding factors may influence the results [1]. However, results from a study comparing the results of observational and randomized, controlled studies in 19 different fields of medicine suggest that observational studies usually do provide valid additional information [2].
One of the major reasons to conduct observational studies is the potentially limited external validity of the results of the randomized controlled trial. To what extent are the results of the trial generalizable to a population not treated in the setting of a specific trial? Several potential reasons for limited external validity can be recognized. For example, healthcare providers in the setting of clinical trials may be unrepresentative, e.g. because they are innovators. Or the patients included in the trials may have characteristics different from the patients treated outside the setting of the trial due to the inclusion criteria applicable to the trial. Furthermore, the treatment in the trial may be atypical. For example, patients included in trials may receive different care due to intensified follow-up [1]. These aspects may introduce bias in the results of the trial, resulting in limited external validity.
Besides limited external validity, prescription of new drugs and treatment outcomes may be influenced by channelling bias. Channelling bias occurs when a patient is directed away from or toward a specific therapy. Several reasons for channelling bias can be recognized, such as underlying medical conditions or perceptions of the provider. Besides, when a new drug comes onto the market, a large pool of subjects with insufficient response or adverse drug reactions on existing medications is present. These patients will be prone to be treated with the new medication and thereby introduce a bias in the population treated with that medication and the treatment outcomes. Therefore, channelling bias has to be taken into account when extrapolating the results from randomized controlled trials (RCT) to the setting of care-as-usual. Channelling bias is not specific to one field of pharmacotherapy but may occur, for example, in the case of pharmacotherapy for treatment of rheumatoid arthritis (RA).
In January 2000 leflunomide was registered in the Netherlands for the treatment of active RA on the basis of results from RCTs including over 1000 patients [3–5]. Results of an observational study in patients with RA on leflunomide treatment showed that the characteristics of patients treated in a setting of care-as-usual differ from characteristics of the populations in RCTs, suggesting channelling of the application of leflunomide [6]. Compared with the population included in the RCTs on leflunomide, the population in this study had a higher age, had longer disease duration, and was more intensively treated with disease-modifying antirheumatic drugs (DMARDs) before starting leflunomide.
Compared with RCTs, reporting withdrawal rates that varied from 30 to 47 per 100 patient years [4, 5], observational studies consistently showed higher withdrawal rates, suggesting that channelling influences treatment outcomes in clinical practice. Failure rates have been reported between 55.5% and 56.2% per 100 patient years’ follow-up [6, 7]. Furthermore, Geborek et al. [8] and Siva [9] reported withdrawal from leflunomide treatment of 78% of patients after 20 months’ and 63% after 6 months’ follow-up, respectively. The results from these observational studies suggested that the withdrawal rate from leflunomide treatment is higher in the setting of care-as-usual compared with RCT.
Time remaining on therapy is a relevant factor in obtaining treatment effects, particularly in chronic diseases such as RA. To optimize leflunomide therapy, early recognition of factors predictive for longer treatment survival is of clinical importance. For this reason we performed a study to determine predictors for leflunomide withdrawal in an outpatient population with RA.
Methods
Population
In two regions in the Netherlands (Friesland and Twente) with a total of approximately 1200 000 inhabitants, all consecutive patients starting leflunomide for RA were followed. The study period started in January 2000, the moment leflunomide became available for prescription in the Netherlands, and ended in June 2003.
Leflunomide prescription
At the time this study was conducted the place of leflunomide in the sequence of DMARD therapy in RA was not established. The launch of leflunomide preceded the launch of tumour necrosis factor (TNF) blockers with only 8 months in the Netherlands. The anti-TNF-α therapy was reimbursed only after approval by a central, national adjudication procedure. Reimbursement was possible only in cases with higher disease activity (DAS28 > 3.2) with proven failure of at least two DMARDs including methotrexate. So, generally, methotrexate was regarded as the option of first choice, and anti-TNF-α therapy was available. Prescription of leflunomide therefore was left to the judgement of the attending rheumatologist, with anti-TNF-α therapy being the most important alternative. Leflunomide was prescribed in a dose as recommended by the manufacturer, i.e. a loading dose of 100 mg daily for 3 days, followed by 20 mg daily. In case of adverse events, dose reduction to 10 mg daily was an option.
Data collection
Data were collected at every routine visit of the patient to the rheumatologist. The standard dataset, using outpatient medical records, consisted of patient characteristics, disease characteristics, laboratory values and data on leflunomide prescription. Intensity of follow-up of the patients during this study was similar to nonstudy patients, reflecting care-as-usual.
Withdrawal
Some discontinuations of leflunomide use are temporary, e.g. in case of intercurrent disease or surgery, and are not related to adverse drug reactions or ineffectiveness of therapy. For this reason the withdrawal from leflunomide was defined as a reported discontinuation of leflunomide for a period longer than 12 weeks. Continuation of leflunomide therapy within this time frame was considered to be a continuation of the first treatment episode and patients were not recorded as ‘withdrawn from therapy’. In case of restarting leflunomide after a period over 12 weeks, patients were not eligible for re-entry into this study.
Predictors
Possible predictors for survival of leflunomide treatment that are continuous variables were studied as continuous and as dichotomous variables. Translation to a dichotomous variable was performed by dividing the population at the median value of the variable under study.
Statistical analysis
SPSS 12.0.1. was used for data collection, data validation, data selection and statistical analysis. Kolmogorov–Smirnov analysis was used to test for normality. T-test and Mann–Whitney tests were used where appropriate to test for differences between group means of continuous variables. For comparison with respect to categorical variables, Fisher's exact test was used. Cumulative probability of survival was estimated using Kaplan–Meier curves.
Differences in time-to-withdrawal for certain factors were investigated using log-rank tests or Cox proportional hazard models, for continuous and categorical variables, respectively. Forward stepwise conditional regression analysis was used for studying variable contribution in multivariate analysis. The independent variables in this analysis were selected on the basis of the univariate analysis. Variables with P < 0.10 in univariate analysis were selected for the Cox regression by means of a forward stepwise selection procedure. P-values of 0.05 are considered significant.
Results
Inclusion
Twelve rheumatologists included a total of 279 patients, 140 from the Friesland and 139 from the Twente region, with a median of 21 (range 2–49) patients starting leflunomide per rheumatologist. Baseline demographic and clinical characteristics of the population are shown in Table 1. The dataset was complete, except for 16 and 41 patients for whom no erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) at start of leflunomide treatment were recorded, respectively.
Table 1.
Baseline demographic and clinical characteristics
| Age (years) | 62.6 [12.6] |
| Female (%) | 66.7 |
| IgM rheumatoid factor positive (%) | 81.4 |
| RA disease duration (year; median [range]) | 10.0 [0–60] |
| Leflunomide as first DMARD (%) | 9.7 |
| DMARDs prior to leflunomide (n) | 3 [0–11] |
| Concomitant systemic corticosteroids (%) | 43.0 |
| Median corticosteroid dose (mg) | 5 |
| ESR start (mm h−1) | 40 [26] |
| CRP start (mg l−1) | 38 [33] |
| Creatinine (micromoles l−1) | 78 [22] |
Values represent mean [SD] unless stated otherwise. CRP start, C-reactive protein at the start of leflunomide therapy; DMARD, disease-modifying antirheumatic drug; ESR start, erythrocyte sedimentation rate at the start of leflunomide therapy; RA, rheumatoid arthritis.
Patients were followed for a median period of 9.6 months (range 0.1–38.9 months). In only three patients was the 3-day 100-mg loading dose omitted – they started with the daily maintenance dose of 20 mg. Ten patients started leflunomide treatment in combination with methotrexate. None of the patients started leflunomide in combination with other DMARDs.
Populations in both centres differed with respect to age, RA disease duration, the number of DMARDs prescribed prior to the start of leflunomide, and the fraction of patients for whom the leflunomide dose was reduced to 10 mg during follow-up. Other variables did not show significant differences between the two centres. Centre is therefore taken into account as a covariate or stratifier for further analyses. Since attending rheumatologists work exclusively in one of the two centres, for analysis of the attending rheumatologist as potential predictor, centre is not taken into account as stratifier.
Leflunomide survival
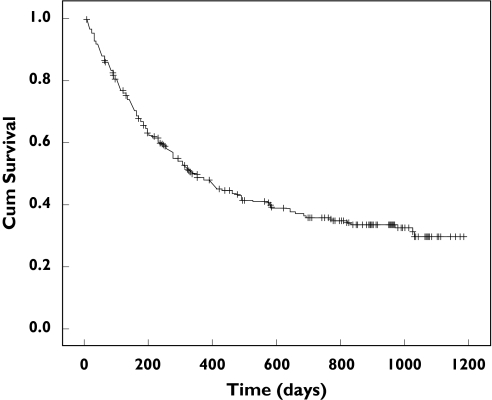
Of the 279 patients included, 173 (62.0%) were withdrawn from leflunomide therapy, a withdrawal rate of 54.3 per 100 patient years. Seventeen patients were lost to follow-up. Figure 1 shows the Kaplan–Meier cumulative survival curve for all included patients. No significant differences in survival rates between both centres could be found (P = 0.20).
Figure 1.
Kaplan-Meier estimate for withdrawal from leflunomide. += censored observation
Predictors for longer leflunomide drug survival
Independence of variables
Since the characteristics of the population treated per rheumatologist may differ, the correlation between rheumatologist and patient age, ESR at start of leflunomide, CRP at start of leflunomide, duration of RA or number of DMARDs prior to leflunomide were studied. Rheumatologist was found to be correlated with disease duration (P = 0.01) and number of previous DMARDs (P < 0.001).
Rheumatoid factor status was not correlated with systemic corticosteroid use at the start of leflunomide therapy.
Corticosteroids reduce inflammation and may influence ESR. We tested correlation of these variables to study whether ESR and corticosteroids may be treated as independent variables. Systemic corticosteroid use was not correlated with ESR at start (P = 0.08). Since no significant correlation between the variables is detected, both are judged to be independent and as such are used separately for uni- and multivariate analysis.
However, significant positive correlations were found between systemic corticosteroid use and IgM rheumatoid factor status (P = 0.44), CRP at start (P < 0.001, mean CRP at start with and without concomitant corticosteroids was 48 and 30 mg l−1, respectively).
Uni- and multivariate analysis
Overall withdrawal. Gender, age, disease duration, IgM rheumatoid factor status, the number of previous DMARDs, serum creatinine, ESR (continuous variable) and CRP (continuous and dichotomous variable) at start of leflunomide were found not to be predictive for leflunomide survival.
However, concomitant systemic corticosteroid use at the start of leflunomide, and ESR at start of leflunomide (dichotomous; <35 vs.≥35 mm h−1) were recognized as predicting variables for leflunomide survival (Table 2). Patients with concomitant systemic corticosteroid use at start of leflunomide showed better leflunomide survival [hazard ratio (HR) (95% CI) 1.35 (1.00, 1.83)] compared with patients without concomitant systemic corticosteroids. Patients with ESR <35 mm h−1 at start of leflunomide showed better leflunomide survival at complete follow-up [HR (95% CI) 1.38 (1.01, 1.88)] compared with patients with ESR ≥35 mm h−1. The attending rheumatologist was associated with leflunomide drug survival with a P < 0.10 and entered in the model for multivariate analysis. The rheumatologist with the median number of included patients was used as the reference rheumatologist for calculating maximal contrasts within the group of attending rheumatologists in this study. That is, the range of hazard ratios for leflunomide survival found for every individual rheumatologist compared with the rheumatologist with the median number of patients attending, gives an estimate of the maximal differences in leflunomide survival.
Table 2.
Univariate analysis for predictors for longer leflunomide drug survival
| Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|
| Factor | P-value | Hazard ratio (95% CI) | P-value | Hazard ratio (95% CI) |
| Concomitant systemic corticosteroids | 0.05 | 0.006 | ||
| No | 1 (reference) | 1 (reference) | ||
| Yes | 1.35 (1.00, 1.83) | 1.58 (1.14, 2.21) | ||
| ESR start (mm h–1) | 0.04 | 0.03 | ||
| ≥ 35 | 1 (reference) | 1 (reference) | ||
| <35 | 1.38 (1.01, 1.88) | 1.42 (1.03, 1.96) | ||
| Rheumatologist | 0.095 | 0.60–2.661 | 0.02 | 0.54–2.301 |
95% CI, 95% confidence interval; ESR start, erythrocyte sedimentation rate at the start of leflunomide therapy.
Maximal contrasts between rheumatologists.
Multivariate analysis showed attending rheumatologist, concomitant systemic corticosteroid use and an ESR <35 mm h−1 at start of leflunomide to be related to leflunomide survival (Table 2).
Withdrawal for adverse drug reactions or ineffectiveness. When analysing the results for the specific reason of withdrawal, i.e. for ineffectiveness or adverse drug reactions, we found the following set of predictive factors.
For withdrawal due to ineffectiveness of leflunomide treatment, a total of 188 patients remain. Univariate analysis shows concomitant systemic corticosteroid use at start of leflunomide (P = 0.068) and attending rheumatologist (P = 0.065) to be predictive with P < 0.10. These factors are entered into multivariate analysis. In multivarate analysis concomitant systemic corticosteroid use remains a significant predictive factor [P = 0.025; HR (95% CI) 1.71 (1.07, 2.72)], but attending rheumatologist does not remain a significant predictor (P = 0.15).
Withdrawal due to adverse drug reactions on leflunomide treatment. A total of 207 patients remain. Univariate analysis shows ESR at start of leflunomide treatment (P = 0.002) and attending rheumatologist predictive for withdrawal due to adverse drug reactions with P < 0.10. These factors are entered into multivariate analysis. In multivariate analysis ESR remains a significant predictive factor [P = 0.016; HR (95% CI) 1.79 (1.17, 2.78)], but attending rheumatologist does not remain a significant predictor (P = 0.15).
In both analyses other factors in univariate analysis were not predictive (gender, age, disease duration, rheumatoid factor status, number of previous DMARDs and CRP).
Discussion
We performed an analysis of predictive factors for continuation of leflunomide for the treatment of RA. Our data show attending rheumatologist, concomitant systemic corticosteroid use and ESR at the start of leflunomide treatment to be predictors for survival of leflunomide in an outpatient population with RA in the setting of care-as-usual.
At the time we started our study no information on the predictors for longer survival of leflunomide was published. Wolfe et al.[7], defining treatment failure as a combination of withdrawal or addition of a second DMARD, recognized adverse effects and clinical status prior to starting methotrexate or leflunomide as predictors for treatment failure. For obvious reasons, adverse events occurring during treatment were not part of our analysis.
We found concomitant use of systemic corticosteroids at the start of leflunomide therapy to be predictive for leflunomide survival. Wolfe et al.[7] did not find differences in survival curves between patients with or without previous prednisone therapy. This difference may be explained by the fact that in our study systemic corticosteroid use at the start of leflunomide was scored while Wolfe et al. studied previous prednisone therapy.
Wolfe et al. report the exclusive use of self-reported data and the absence of laboratory data to predict discontinuation as one of the limitations of their study. In our study we used laboratory data as far as they are part of routine rheumatological care. Lower ESR, in contrast to CRP, was found to be a significant predictor of leflunomide survival.
Siva et al.[9] found age <44 or >75 years, annual family income <$60.000 and the use of a 3-day 100-mg loading dose to be predictive for leflunomide discontinuation. In our study age was not found to be predictive. Since all but three patients in our study started leflunomide using the dosing schedule advised in the Summary of Product Characteristics, the absence of a loading dose could not be recognized as a predictor in our study.
Our data suggest that the individual rheumatologist influences leflunomide survival significantly, the maximal difference between the rheumatologists on leflunomide survival being over fourfold, with hazard ratios differing from 0.54 to 2.30. The influence of the attending rheumatologist seems to be a major contributor to leflunomide survival, compared with the other variables found to be associated with leflunomide survival. Concomitant corticosteroid use [HR 1.58 (1.14, 2.21)] and ESR [HR 1.42 (1.03, 1.96)] at the start of leflunomide show a mean 58% and 42% difference in leflunomide survival depending on concomitant corticosteroid use and ESR category, respectively.
A recent publication on the long-term survival of methotrexate also concluded that attending rheumatologist was a predictor for long-term methotrexate survival [10]. Our findings are in accordance with this result and again suggest that remarkable heterogeneity exists in the treatment strategies of individual rheumatologists. Although the prescription of leflunomide was not prospectively standardized in our study, leflunomide was not considered the option of first choice in the study period. Clinical guidelines aim to reduce variations in practice and to promote uniform and consistent best practice. Whether development of guidelines on the application of leflunomide in RA will lead to more uniform strategies and improvement of treatment outcomes remains to be determined.
Why ESR and concomitant steroid use are predictive for leflunomide survival can not be concluded from the current study. One possible explanation is that patients with a lower ESR or without concomitant sytemic corticosteroid use represent a class of patients with specific disease characteristics, e.g. less agressive disease, that are predictive for better leflunomide effectiveness or a decreased incidence of adverse drug reactions with an associated difference in leflunomide survival. However, other disease characteristics such as the number of previous DMARDs and IgM factor rheumatoid status were not found to be predictive for leflunomide survival. In accordance with the principle that observational studies create hypothesis rather than test a hypothesis, our study was not designed to detect the explanation for withdrawing from leflunomide.
In this study a limited dataset was collected. Therefore, some predictors for leflunomide survival may have been missed in the current analysis. Retrospective data collection may have introduced a bias and therefore may have influenced results. However, no patients were excluded from the study, the dataset was complete and the data collected were not subject to observer bias. This suggests that any bias, if present, may have had only a limited influence.
At the time we conducted this study, results from studies with leflunomide in combination with other DMARDs were scarce. Information on the combination of leflunomide with methotrexate showed an increased incidence of hepatotoxicity, an adverse drug reaction that had broad attention of the registration authorities in 2000. This attention led to adaptations in the Summary of Product Characteristics concerning the monitoring of serum liver enzyme (transaminase) activities. The combination of the virtual absence of study results on combination therapy with leflunomide at the time of this study and the ‘Dear doctor letter’ on hepatotoxicity of leflunomide led to a reserve considering leflunomide combination therapy. Whether introduction of combination therapy may lead to differences in leflunomide survival remains to be studied.
Conclusion
Concomitant systemic corticosteroid use, ESR at the start of treatment and attending rheumatologist were found to be predictive for leflunomide survival. Whether specific interventions based on this information, for example by more frequent follow-up of patients with a higher risk of treatment withdrawal, will lead to improved treatment outcomes remains to be studied.
Acknowledgments
Competing interests: None declared.
References
- 1.Black N. Why we need observational studies to evaluate the effectiveness of health care. BMJ. 1996;312:1215–8. doi: 10.1136/bmj.312.7040.1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Benson K, Hartz AJ. A comparison of observational studies and randomized, controlled trials. N Engl J Med. 2000;342:1878–86. doi: 10.1056/NEJM200006223422506. [DOI] [PubMed] [Google Scholar]
- 3.Smolen JS, Kalden JR, Scott DL, Rozman B, Kvien TK, Larsen A, Loew-Friedrich I, Oed C, Rosenburg R. Efficacy and safety of leflunomide compared with placebo and sulphasalazine in active rheumatoid arthritis: a double-blind, randomised, multicentre trial. Lancet. 1999;353:259–66. doi: 10.1016/s0140-6736(98)09403-3. [DOI] [PubMed] [Google Scholar]
- 4.Strand V, Cohen S, Schiff M, Weaver A, Fleischmann R, Cannon G, Fox R, Moreland L, Olsen N, Furst D, Caldwell J, Kaine J, Sharp J, Hurley F, Loew-Friedrich I. Treatment of active rheumatoid arthritis with leflunomide compared with placebo and methotrexate. Arch Intern Med. 1999;159:2542–50. doi: 10.1001/archinte.159.21.2542. [DOI] [PubMed] [Google Scholar]
- 5.Emery P, Breedveld FC, Lemmel EM, Kaltwasser JP, Dawes PT, Gomer B, Van Den Bosch F, Nordstrom D, Bjorneboe O, Dahl R, Horslev-Petersen K, Rodriguez De La Serna A, Molloy M, Tikly M, Oed C, Rosenburg R, Loew-Friedrich I. A comparison of the efficacy and safety of leflunomide and methotrexate for the treatment of rheumatoid arthritis. Rheumatol. 2000;39:655–65. doi: 10.1093/rheumatology/39.6.655. [DOI] [PubMed] [Google Scholar]
- 6.Van Roon EN, Jansen TLThA, Mourad L, Houtman PM, Bruyn GA, Griep EN, Wilffert B, Tobi H, Brouwers JR. Leflunomide in active rheumatoid arthritis: a prospective study in daily practice. Br J Clin Pharmacol. 2004;57:790–7. doi: 10.1111/j.1365-2125.2004.02075.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wolfe F, Michaud K, Stephenson B, Doyle J. Toward a definition and method of assessment of treatment failure and treatment effectiveness: the case of leflunomide versus methotrexate. J Rheumatol. 2003;30:1725–32. [PubMed] [Google Scholar]
- 8.Geborek P, Crnkic M, Petersson IF, Saxne T South Swedish Arthritis Treatment Group. Etanercept, infliximab, and leflunomide in established rheumatoid arthritis: clinical experience using a structured follow-up programme in southern Sweden. Ann Rheum Dis. 2002;61:793–8. doi: 10.1136/ard.61.9.793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Siva C, Eisen SA, Shepherd R, Cunningham F, Fang MA, Finch W, Salisbury D, Singh JA, Stern R, Zarabadi SA. Leflunomide use during the first 33 months after food and drug administration approval: experience with a national cohort of 3,325 patients. Arthritis Rheum. 2003;49:745–51. doi: 10.1002/art.11452. [DOI] [PubMed] [Google Scholar]
- 10.Hoekstra M, van de Laar MA, Bernelot Moens HJ, Kruijsen MW, Haagsma CJ. Longterm observational study of methotrexate use in a Dutch cohort of 1022 patients with rheumatoid arthritis. J Rheumatol. 2003;30:2325–9. [PubMed] [Google Scholar]