Abstract
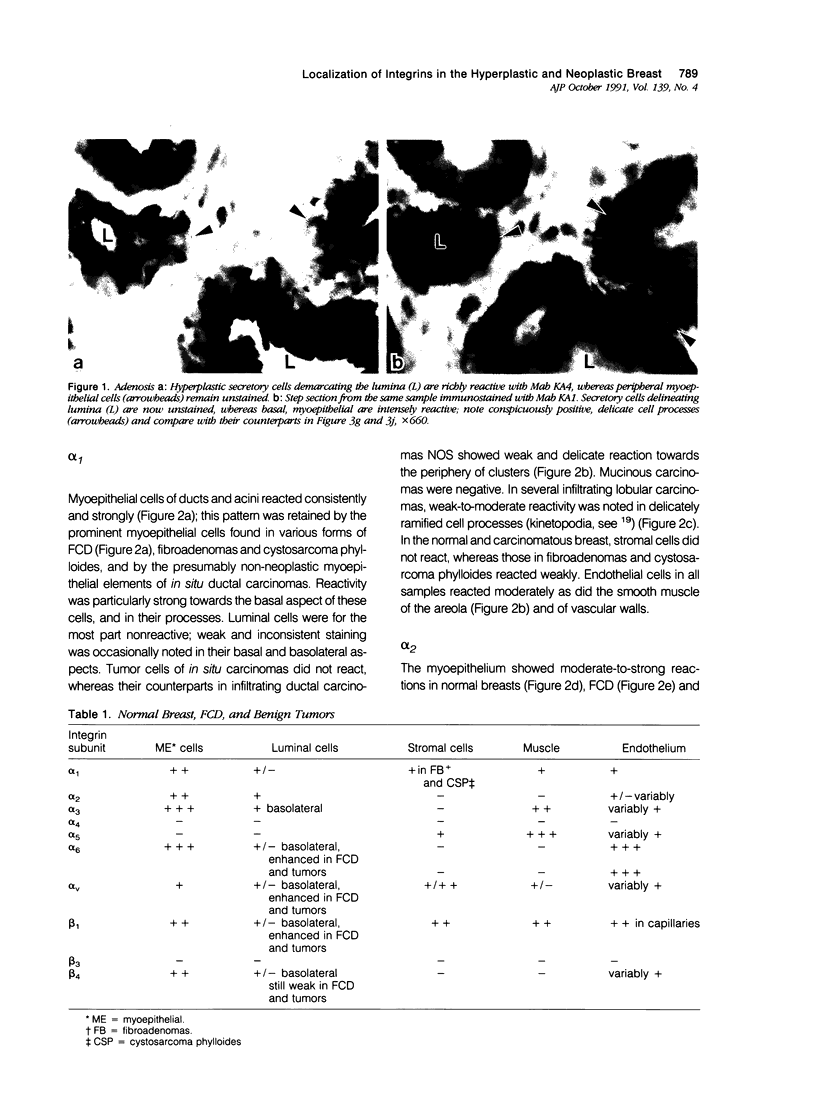
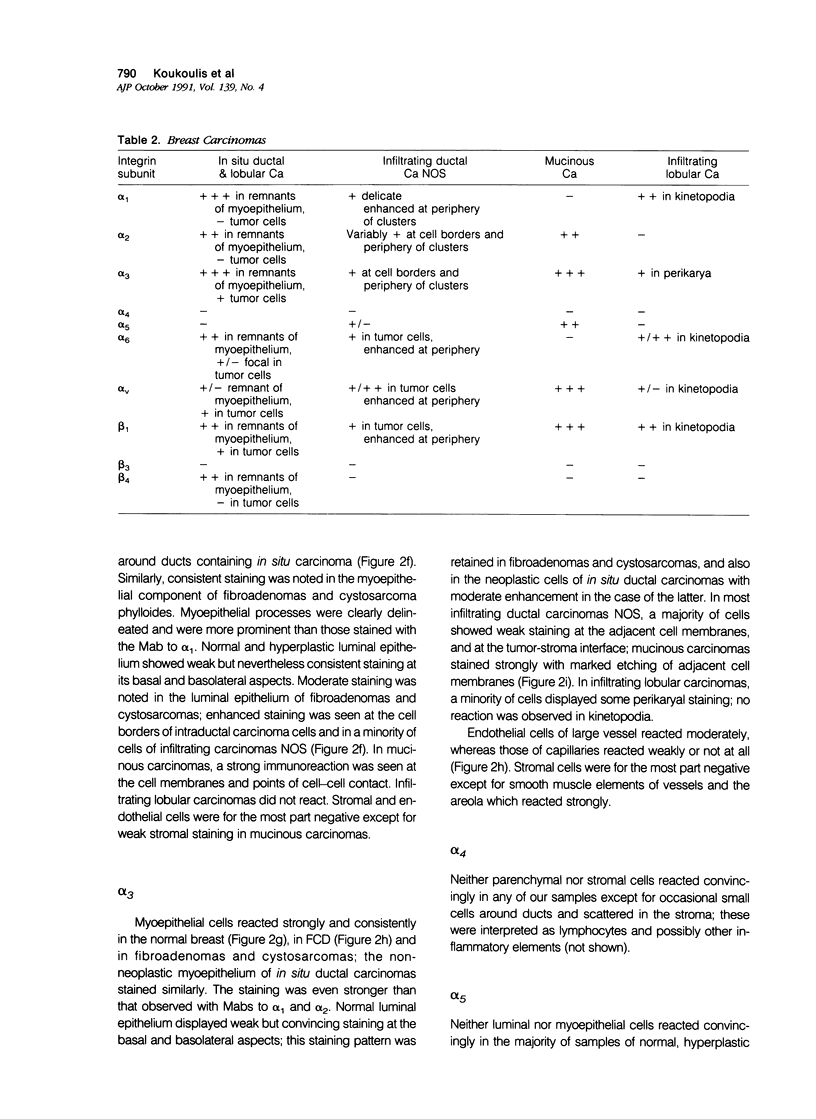
Integrins comprise a family of transmembrane glycoproteins that modulate cell-matrix and cell-cell relationships by acting as receptors to extracellular protein ligands, and also as direct adhesion molecules. The authors studied by immunohistochemistry the distribution of the alpha 1-6,v and the beta 1,3,4 subunits of integrins in samples of normal breast, the spectrum of fibrocystic disease (FCD), and representative benign and malignant neoplasms. Monoclonal antibodies (Mabs) specific for each subunit were applied to cryosections by the avidin-biotin-complex method; selected samples were studied by double immunofluorescence microscopy with the Mabs and a polyclonal antiserum to myosin. The authors found that the alpha 1-3,6,v and the beta 1, integrin subunits were detectable in the normal breast parenchyma; myoepithelial cells were consistently more prominently stained than the basolateral aspect of the luminal cells. This immunoprofile was retained, and in cases enhanced through the spectrum of FCD, in benign tumors and in ductal and lobular carcinomas in situ. In most infiltrating ductal carcinomas, integrin staining tended to decrease except for some cases that reacted strongly for the alpha v subunit. Several mucinous carcinomas reacted strongly for alpha 2,3,6,v and beta 4 subunits, and even more so for the alpha 5 subunit that was not found in the normal breast. Subsets of infiltrating lobular carcinomas stained convincingly for alpha 1,3,6,v and beta 1 subunits in delicate but abundant kinetopodia. Our findings indicate that in hyperplasias and in benign tumors integrin expression patterns parallel those of the normal breast, whereas in carcinomas, variations include decrease, enhancement, and emergence of certain subunits that are not in the normal repertory. Alterations of integrin expression parallel phenotypic changes in breast carcinoma cells; they also reflect their disrupted interaction with the similarly disrupted extracellular matrix. Enhancement of certain integrins in some carcinomas may reflect the selection of subpopulations with increased binding capacity which in turn may impact on their invasive and metastatic properties.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Achstätter T., Moll R., Anderson A., Kuhn C., Pitz S., Schwechheimer K., Franke W. W. Expression of glial filament protein (GFP) in nerve sheaths and non-neural cells re-examined using monoclonal antibodies, with special emphasis on the co-expression of GFP and cytokeratins in epithelial cells of human salivary gland and pleomorphic adenomas. Differentiation. 1986;31(3):206–227. doi: 10.1111/j.1432-0436.1986.tb00401.x. [DOI] [PubMed] [Google Scholar]
- Adams J. C., Watt F. M. Fibronectin inhibits the terminal differentiation of human keratinocytes. Nature. 1989 Jul 27;340(6231):307–309. doi: 10.1038/340307a0. [DOI] [PubMed] [Google Scholar]
- Akiyama S. K., Yamada K. M. Biosynthesis and acquisition of biological activity of the fibronectin receptor. J Biol Chem. 1987 Dec 25;262(36):17536–17542. [PubMed] [Google Scholar]
- Albelda S. M., Buck C. A. Integrins and other cell adhesion molecules. FASEB J. 1990 Aug;4(11):2868–2880. [PubMed] [Google Scholar]
- Badley R. A., Lloyd C. W., Woods A., Carruthers L., Allcock C., Rees D. A. Mechanisms of cellular adhesion. III. Preparation and preliminary characterisation of adhesions. Exp Cell Res. 1978 Dec;117(2):231–244. doi: 10.1016/0014-4827(78)90136-2. [DOI] [PubMed] [Google Scholar]
- Bronner-Fraser M. Alterations in neural crest migration by a monoclonal antibody that affects cell adhesion. J Cell Biol. 1985 Aug;101(2):610–617. doi: 10.1083/jcb.101.2.610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bronner-Fraser M. An antibody to a receptor for fibronectin and laminin perturbs cranial neural crest development in vivo. Dev Biol. 1986 Oct;117(2):528–536. doi: 10.1016/0012-1606(86)90320-9. [DOI] [PubMed] [Google Scholar]
- Carter W. G., Kaur P., Gil S. G., Gahr P. J., Wayner E. A. Distinct functions for integrins alpha 3 beta 1 in focal adhesions and alpha 6 beta 4/bullous pemphigoid antigen in a new stable anchoring contact (SAC) of keratinocytes: relation to hemidesmosomes. J Cell Biol. 1990 Dec;111(6 Pt 2):3141–3154. doi: 10.1083/jcb.111.6.3141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carter W. G., Wayner E. A., Bouchard T. S., Kaur P. The role of integrins alpha 2 beta 1 and alpha 3 beta 1 in cell-cell and cell-substrate adhesion of human epidermal cells. J Cell Biol. 1990 Apr;110(4):1387–1404. doi: 10.1083/jcb.110.4.1387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W. T., Chen J. M., Mueller S. C. Coupled expression and colocalization of 140K cell adhesion molecules, fibronectin, and laminin during morphogenesis and cytodifferentiation of chick lung cells. J Cell Biol. 1986 Sep;103(3):1073–1090. doi: 10.1083/jcb.103.3.1073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheresh D. A., Harper J. R. Arg-Gly-Asp recognition by a cell adhesion receptor requires its 130-kDa alpha subunit. J Biol Chem. 1987 Feb 5;262(4):1434–1437. [PubMed] [Google Scholar]
- Couchman J. R., Rees D. A., Green M. R., Smith C. G. Fibronectin has a dual role in locomotion and anchorage of primary chick fibroblasts and can promote entry into the division cycle. J Cell Biol. 1982 May;93(2):402–410. doi: 10.1083/jcb.93.2.402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Luca M., Tamura R. N., Kajiji S., Bondanza S., Rossino P., Cancedda R., Marchisio P. C., Quaranta V. Polarized integrin mediates human keratinocyte adhesion to basal lamina. Proc Natl Acad Sci U S A. 1990 Sep;87(17):6888–6892. doi: 10.1073/pnas.87.17.6888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dedhar S., Saulnier R. Alterations in integrin receptor expression on chemically transformed human cells: specific enhancement of laminin and collagen receptor complexes. J Cell Biol. 1990 Feb;110(2):481–489. doi: 10.1083/jcb.110.2.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duband J. L., Rocher S., Chen W. T., Yamada K. M., Thiery J. P. Cell adhesion and migration in the early vertebrate embryo: location and possible role of the putative fibronectin receptor complex. J Cell Biol. 1986 Jan;102(1):160–178. doi: 10.1083/jcb.102.1.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ekblom P. Developmentally regulated conversion of mesenchyme to epithelium. FASEB J. 1989 Aug;3(10):2141–2150. doi: 10.1096/fasebj.3.10.2666230. [DOI] [PubMed] [Google Scholar]
- Fradet Y., Cordon-Cardo C., Thomson T., Daly M. E., Whitmore W. F., Jr, Lloyd K. O., Melamed M. R., Old L. J. Cell surface antigens of human bladder cancer defined by mouse monoclonal antibodies. Proc Natl Acad Sci U S A. 1984 Jan;81(1):224–228. doi: 10.1073/pnas.81.1.224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giancotti F. G., Ruoslahti E. Elevated levels of the alpha 5 beta 1 fibronectin receptor suppress the transformed phenotype of Chinese hamster ovary cells. Cell. 1990 Mar 9;60(5):849–859. doi: 10.1016/0092-8674(90)90098-y. [DOI] [PubMed] [Google Scholar]
- Giltay J. C., Brinkman H. J., Modderman P. W., von dem Borne A. E., van Mourik J. A. Human vascular endothelial cells express a membrane protein complex immunochemically indistinguishable from the platelet VLA-2 (glycoprotein Ia-IIa) complex. Blood. 1989 Apr;73(5):1235–1241. [PubMed] [Google Scholar]
- Gould V. E., Jao W., Battifora H. Ultrastructural analysis in the differential diagnosis of breast tumors. The significance of myoepithelial cells, basal lamina, intracytoplasmic lumina and secretory granules. Pathol Res Pract. 1980 May;167(1):45–70. doi: 10.1016/S0344-0338(80)80181-6. [DOI] [PubMed] [Google Scholar]
- Gould V. E., Koukoulis G. K., Jansson D. S., Nagle R. B., Franke W. W., Moll R. Coexpression patterns of vimentin and glial filament protein with cytokeratins in the normal, hyperplastic, and neoplastic breast. Am J Pathol. 1990 Nov;137(5):1143–1155. [PMC free article] [PubMed] [Google Scholar]
- Gould V. E., Koukoulis G. K., Virtanen I. Extracellular matrix proteins and their receptors in the normal, hyperplastic and neoplastic breast. Cell Differ Dev. 1990 Dec 2;32(3):409–416. doi: 10.1016/0922-3371(90)90057-4. [DOI] [PubMed] [Google Scholar]
- Gould V. W., Snyder R. W. Ultrastructural features of papillomatosis and carcinoma of nipple ducts. The significance of myoepithelial cells and basal lamina in benign, "questionable," and malignant lesions. Pathol Annu. 1974;9(0):441–469. [PubMed] [Google Scholar]
- Guirguis R., Margulies I., Taraboletti G., Schiffmann E., Liotta L. Cytokine-induced pseudopodial protrusion is coupled to tumour cell migration. Nature. 1987 Sep 17;329(6136):261–263. doi: 10.1038/329261a0. [DOI] [PubMed] [Google Scholar]
- Gustafsson H., Virtanen I., Thornell L. E. Glial fibrillary acidic protein and desmin in salivary neoplasms. Expression of four different types of intermediate filament proteins within the same cell type. Virchows Arch B Cell Pathol Incl Mol Pathol. 1989;57(5):303–313. doi: 10.1007/BF02899095. [DOI] [PubMed] [Google Scholar]
- Hall D. E., Neugebauer K. M., Reichardt L. F. Embryonic neural retinal cell response to extracellular matrix proteins: developmental changes and effects of the cell substratum attachment antibody (CSAT). J Cell Biol. 1987 Mar;104(3):623–634. doi: 10.1083/jcb.104.3.623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hay E. D. Extracellular matrix, cell skeletons, and embryonic development. Am J Med Genet. 1989 Sep;34(1):14–29. doi: 10.1002/ajmg.1320340107. [DOI] [PubMed] [Google Scholar]
- Hedin U., Bottger B. A., Luthman J., Johansson S., Thyberg J. A substrate of the cell-attachment sequence of fibronectin (Arg-Gly-Asp-Ser) is sufficient to promote transition of arterial smooth muscle cells from a contractile to a synthetic phenotype. Dev Biol. 1989 Jun;133(2):489–501. doi: 10.1016/0012-1606(89)90052-3. [DOI] [PubMed] [Google Scholar]
- Hemler M. E., Crouse C., Sonnenberg A. Association of the VLA alpha 6 subunit with a novel protein. A possible alternative to the common VLA beta 1 subunit on certain cell lines. J Biol Chem. 1989 Apr 15;264(11):6529–6535. [PubMed] [Google Scholar]
- Hemler M. E., Sanchez-Madrid F., Flotte T. J., Krensky A. M., Burakoff S. J., Bhan A. K., Springer T. A., Strominger J. L. Glycoproteins of 210,000 and 130,000 m.w. on activated T cells: cell distribution and antigenic relation to components on resting cells and T cell lines. J Immunol. 1984 Jun;132(6):3011–3018. [PubMed] [Google Scholar]
- Horwitz A., Duggan K., Buck C., Beckerle M. C., Burridge K. Interaction of plasma membrane fibronectin receptor with talin--a transmembrane linkage. Nature. 1986 Apr 10;320(6062):531–533. doi: 10.1038/320531a0. [DOI] [PubMed] [Google Scholar]
- Howeedy A. A., Virtanen I., Laitinen L., Gould N. S., Koukoulis G. K., Gould V. E. Differential distribution of tenascin in the normal, hyperplastic, and neoplastic breast. Lab Invest. 1990 Dec;63(6):798–806. [PubMed] [Google Scholar]
- Hsu S. M., Raine L., Fanger H. Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem. 1981 Apr;29(4):577–580. doi: 10.1177/29.4.6166661. [DOI] [PubMed] [Google Scholar]
- Humphries M. J. The molecular basis and specificity of integrin-ligand interactions. J Cell Sci. 1990 Dec;97(Pt 4):585–592. doi: 10.1242/jcs.97.4.585. [DOI] [PubMed] [Google Scholar]
- Hynes R. O. Integrins: a family of cell surface receptors. Cell. 1987 Feb 27;48(4):549–554. doi: 10.1016/0092-8674(87)90233-9. [DOI] [PubMed] [Google Scholar]
- Korhonen M., Ylänne J., Laitinen L., Virtanen I. Distribution of beta 1 and beta 3 integrins in human fetal and adult kidney. Lab Invest. 1990 May;62(5):616–625. [PubMed] [Google Scholar]
- Korhonen M., Ylänne J., Laitinen L., Virtanen I. The alpha 1-alpha 6 subunits of integrins are characteristically expressed in distinct segments of developing and adult human nephron. J Cell Biol. 1990 Sep;111(3):1245–1254. doi: 10.1083/jcb.111.3.1245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koukoulis G. K., Gould V. E., Bhattacharyya A., Gould J. E., Howeedy A. A., Virtanen I. Tenascin in normal, reactive, hyperplastic, and neoplastic tissues: biologic and pathologic implications. Hum Pathol. 1991 Jul;22(7):636–643. doi: 10.1016/0046-8177(91)90285-w. [DOI] [PubMed] [Google Scholar]
- Koukoulis G. K., Shin S. S., Gould V. E., Jao W., Gooch G. T., Manderino G. L., Rittenhouse H. G., Tomita J. T. Immunocytochemical evaluation of neoplastic and non-neoplastic breast diseases with Mab A-80. Pathol Res Pract. 1990 Aug;186(4):439–449. doi: 10.1016/s0344-0338(11)80461-9. [DOI] [PubMed] [Google Scholar]
- Krotoski D. M., Domingo C., Bronner-Fraser M. Distribution of a putative cell surface receptor for fibronectin and laminin in the avian embryo. J Cell Biol. 1986 Sep;103(3):1061–1071. doi: 10.1083/jcb.103.3.1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larjava H., Peltonen J., Akiyama S. K., Yamada S. S., Gralnick H. R., Uitto J., Yamada K. M. Novel function for beta 1 integrins in keratinocyte cell-cell interactions. J Cell Biol. 1990 Mar;110(3):803–815. doi: 10.1083/jcb.110.3.803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liotta L. A. Tumor invasion and metastases: role of the basement membrane. Warner-Lambert Parke-Davis Award lecture. Am J Pathol. 1984 Dec;117(3):339–348. [PMC free article] [PubMed] [Google Scholar]
- Lotz M. M., Korzelius C. A., Mercurio A. M. Human colon carcinoma cells use multiple receptors to adhere to laminin: involvement of alpha 6 beta 4 and alpha 2 beta 1 integrins. Cell Regul. 1990 Feb;1(3):249–257. doi: 10.1091/mbc.1.3.249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez-Hernandez A. The extracellular matrix and neoplasia. Lab Invest. 1988 Jun;58(6):609–612. [PubMed] [Google Scholar]
- Menko A. S., Boettiger D. Occupation of the extracellular matrix receptor, integrin, is a control point for myogenic differentiation. Cell. 1987 Oct 9;51(1):51–57. doi: 10.1016/0092-8674(87)90009-2. [DOI] [PubMed] [Google Scholar]
- Mueller S. C., Kelly T., Dai M. Z., Dai H. N., Chen W. T. Dynamic cytoskeleton-integrin associations induced by cell binding to immobilized fibronectin. J Cell Biol. 1989 Dec;109(6 Pt 2):3455–3464. doi: 10.1083/jcb.109.6.3455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagle R. B., Böcker W., Davis J. R., Heid H. W., Kaufmann M., Lucas D. O., Jarasch E. D. Characterization of breast carcinomas by two monoclonal antibodies distinguishing myoepithelial from luminal epithelial cells. J Histochem Cytochem. 1986 Jul;34(7):869–881. doi: 10.1177/34.7.2423579. [DOI] [PubMed] [Google Scholar]
- Otey C. A., Pavalko F. M., Burridge K. An interaction between alpha-actinin and the beta 1 integrin subunit in vitro. J Cell Biol. 1990 Aug;111(2):721–729. doi: 10.1083/jcb.111.2.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruoslahti E. Fibronectin and its receptors. Annu Rev Biochem. 1988;57:375–413. doi: 10.1146/annurev.bi.57.070188.002111. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E., Giancotti F. G. Integrins and tumor cell dissemination. Cancer Cells. 1989 Dec;1(4):119–126. [PubMed] [Google Scholar]
- Ruoslahti E. Integrins. J Clin Invest. 1991 Jan;87(1):1–5. doi: 10.1172/JCI114957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonnenberg A., Daams H., Van der Valk M. A., Hilkens J., Hilgers J. Development of mouse mammary gland: identification of stages in differentiation of luminal and myoepithelial cells using monoclonal antibodies and polyvalent antiserum against keratin. J Histochem Cytochem. 1986 Aug;34(8):1037–1046. doi: 10.1177/34.8.2426332. [DOI] [PubMed] [Google Scholar]
- Sonnenberg A., Janssen H., Hogervorst F., Calafat J., Hilgers J. A complex of platelet glycoproteins Ic and IIa identified by a rat monoclonal antibody. J Biol Chem. 1987 Jul 25;262(21):10376–10383. [PubMed] [Google Scholar]
- Sonnenberg A., Modderman P. W., Hogervorst F. Laminin receptor on platelets is the integrin VLA-6. Nature. 1988 Dec 1;336(6198):487–489. doi: 10.1038/336487a0. [DOI] [PubMed] [Google Scholar]
- Sorokin L., Sonnenberg A., Aumailley M., Timpl R., Ekblom P. Recognition of the laminin E8 cell-binding site by an integrin possessing the alpha 6 subunit is essential for epithelial polarization in developing kidney tubules. J Cell Biol. 1990 Sep;111(3):1265–1273. doi: 10.1083/jcb.111.3.1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamura R. N., Rozzo C., Starr L., Chambers J., Reichardt L. F., Cooper H. M., Quaranta V. Epithelial integrin alpha 6 beta 4: complete primary structure of alpha 6 and variant forms of beta 4. J Cell Biol. 1990 Oct;111(4):1593–1604. doi: 10.1083/jcb.111.4.1593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Virtanen I., Korhonen M., Kariniemi A. L., Gould V. E., Laitinen L., Ylänne J. Integrins in human cells and tumors. Cell Differ Dev. 1990 Dec 2;32(3):215–227. doi: 10.1016/0922-3371(90)90034-t. [DOI] [PubMed] [Google Scholar]
- Werb Z., Tremble P. M., Behrendtsen O., Crowley E., Damsky C. H. Signal transduction through the fibronectin receptor induces collagenase and stromelysin gene expression. J Cell Biol. 1989 Aug;109(2):877–889. doi: 10.1083/jcb.109.2.877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ylänne J., Virtanen I. The Mr 140,000 fibronectin receptor complex in normal and virus-transformed human fibroblasts and in fibrosarcoma cells: identical localization and function. Int J Cancer. 1989 Jun 15;43(6):1126–1136. doi: 10.1002/ijc.2910430628. [DOI] [PubMed] [Google Scholar]
- Zutter M. M., Santoro S. A. Widespread histologic distribution of the alpha 2 beta 1 integrin cell-surface collagen receptor. Am J Pathol. 1990 Jul;137(1):113–120. [PMC free article] [PubMed] [Google Scholar]