Abstract
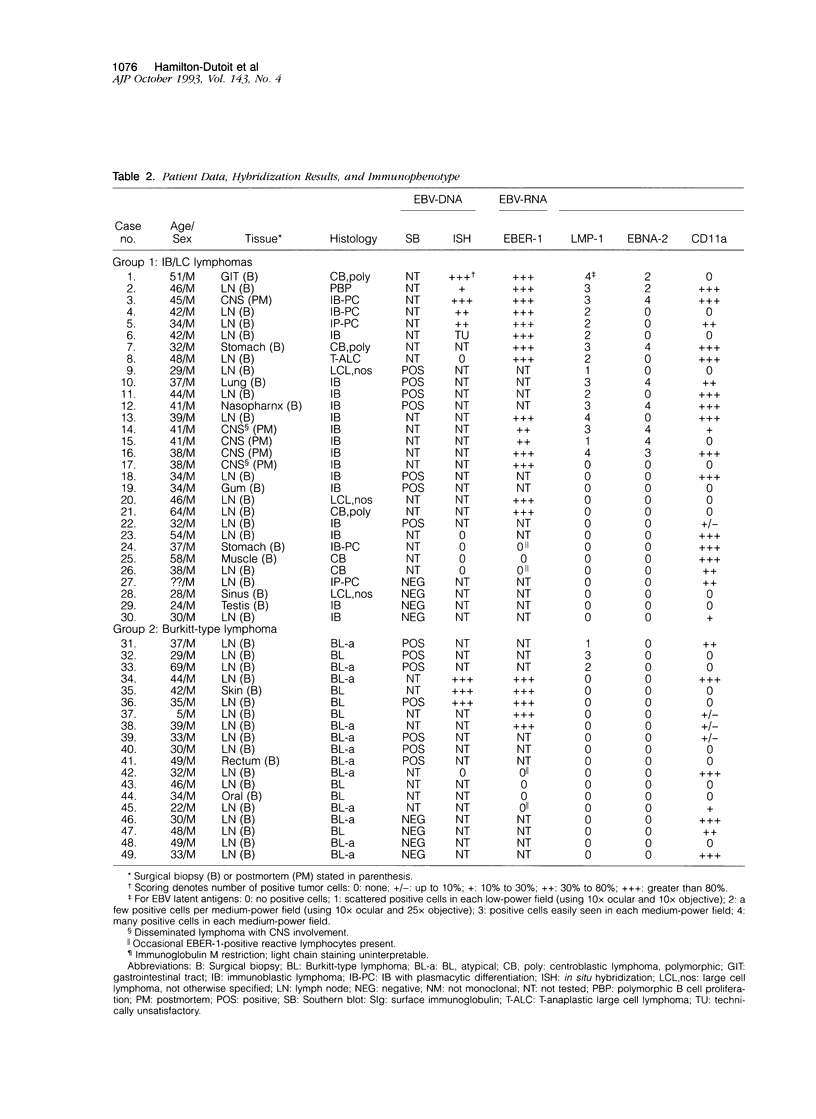
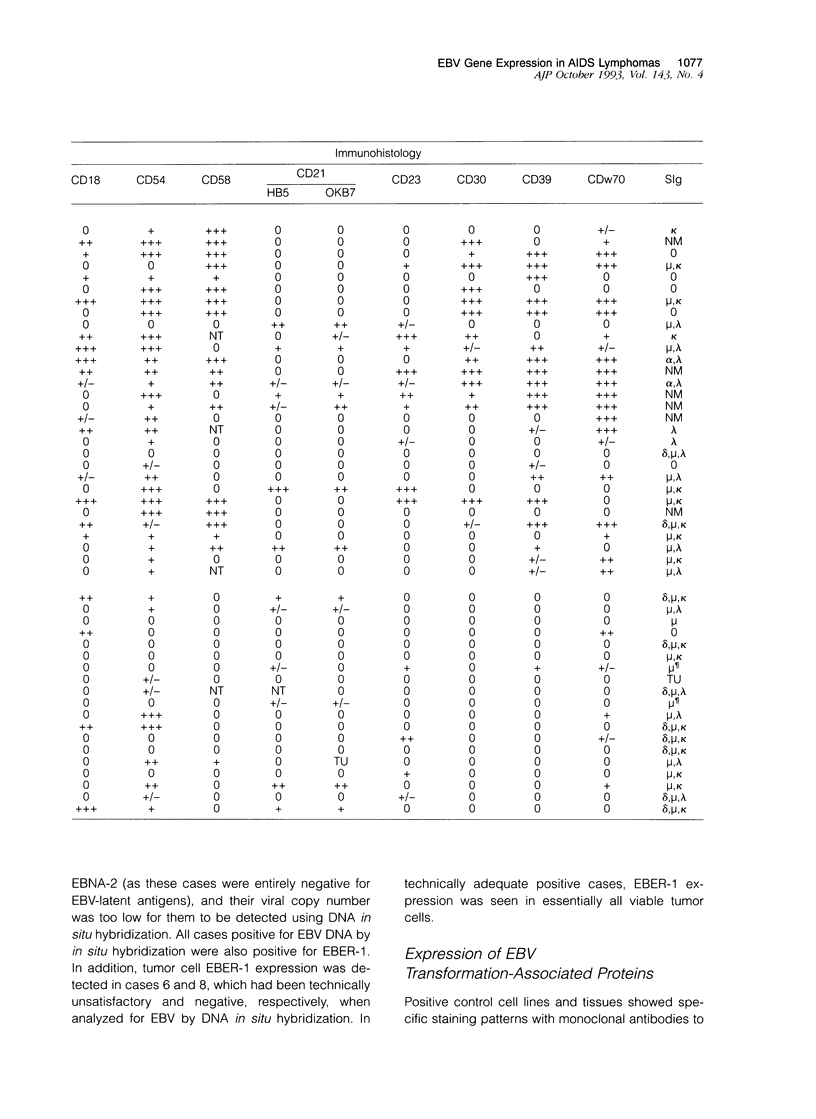
We investigated 49 acquired immunodeficiency syndrome-related lymphomas (ARLs) for Epstein-Barr virus (EBV) by Southern blotting and in situ hybridization and, in positive cases, used cryostat immunohistology to compare EBV-latent gene expression (EBV encoded small RNA-1 [EBER-1], EBV nuclear antigen-2 [EBNA-2], latent membrane protein-1 [LMP-1] and host cell immunophenotype (CD11a, CD18, CD54, CD58, CD21, CD23, CD30, CD39, CDw70, immunoglobulin) patterns with those reported in other EBV infections. EBV+ immunoblast-rich/large cell ARLs (n = 22) showed three patterns of latency: broad (EBER+EBNA-2+/LMP-1+; n = 9), reminiscent of a lymphoblastoid cell line phenotype; restricted (EBER+/EBNA-2-/LMP-1-; n = 6), similar to endemic Burkitt's lymphoma; and intermediate (EBER+/EBNA-2-/LMP-1+; n = 7), a pattern rarely described in vitro but seen in certain EBV-related malignancies. EBNA-2 expression was associated with extranodal lymphomas. EBV+ Burkitt-type ARLs (n = 11) usually showed the restricted latency pattern (n = 8), but some expressed the intermediate form (n = 3). Adhesion (CD54, CD58) and activation (CD30, CD39, CDw70) molecule expression varied with morphology (immunoblast-rich/large cell versus Burkitt-type), but was not independently correlated with EBV-positivity. CD30 and LMP-1 expression were associated. ARLs show heterogeneity regarding both the presence of EBV and latency pattern. Comparison of these phenotypically distinct lymphoma groups with known forms of EBV infection provides clues to their possible pathogenesis.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abbot S. D., Rowe M., Cadwallader K., Ricksten A., Gordon J., Wang F., Rymo L., Rickinson A. B. Epstein-Barr virus nuclear antigen 2 induces expression of the virus-encoded latent membrane protein. J Virol. 1990 May;64(5):2126–2134. doi: 10.1128/jvi.64.5.2126-2134.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Billaud M., Rousset F., Calender A., Cordier M., Aubry J. P., Laisse V., Lenoir G. M. Low expression of lymphocyte function-associated antigen (LFA)-1 and LFA-3 adhesion molecules is a common trait in Burkitt's lymphoma associated with and not associated with Epstein-Barr virus. Blood. 1990 May 1;75(9):1827–1833. [PubMed] [Google Scholar]
- Brooks L., Yao Q. Y., Rickinson A. B., Young L. S. Epstein-Barr virus latent gene transcription in nasopharyngeal carcinoma cells: coexpression of EBNA1, LMP1, and LMP2 transcripts. J Virol. 1992 May;66(5):2689–2697. doi: 10.1128/jvi.66.5.2689-2697.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chappuis B. B., Müller H., Stutte J., Hey M. M., Hübner K., Müller-Hermelink H. K. Identification of EBV-DNA in lymph nodes from patients with lymphadenopathy and lymphomas associated with AIDS. Virchows Arch B Cell Pathol Incl Mol Pathol. 1990;58(3):199–205. doi: 10.1007/BF02890072. [DOI] [PubMed] [Google Scholar]
- Cohen J. I., Wang F., Mannick J., Kieff E. Epstein-Barr virus nuclear protein 2 is a key determinant of lymphocyte transformation. Proc Natl Acad Sci U S A. 1989 Dec;86(23):9558–9562. doi: 10.1073/pnas.86.23.9558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dambaugh T., Wang F., Hennessy K., Woodland E., Rickinson A., Kieff E. Expression of the Epstein-Barr virus nuclear protein 2 in rodent cells. J Virol. 1986 Aug;59(2):453–462. doi: 10.1128/jvi.59.2.453-462.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deacon E. M., Pallesen G., Niedobitek G., Crocker J., Brooks L., Rickinson A. B., Young L. S. Epstein-Barr virus and Hodgkin's disease: transcriptional analysis of virus latency in the malignant cells. J Exp Med. 1993 Feb 1;177(2):339–349. doi: 10.1084/jem.177.2.339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gratama J. W., Zutter M. M., Minarovits J., Oosterveer M. A., Thomas E. D., Klein G., Ernberg I. Expression of Epstein-Barr virus-encoded growth-transformation-associated proteins in lymphoproliferations of bone-marrow transplant recipients. Int J Cancer. 1991 Jan 21;47(2):188–192. doi: 10.1002/ijc.2910470205. [DOI] [PubMed] [Google Scholar]
- Gregory C. D., Murray R. J., Edwards C. F., Rickinson A. B. Downregulation of cell adhesion molecules LFA-3 and ICAM-1 in Epstein-Barr virus-positive Burkitt's lymphoma underlies tumor cell escape from virus-specific T cell surveillance. J Exp Med. 1988 Jun 1;167(6):1811–1824. doi: 10.1084/jem.167.6.1811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamilton-Dutoit S. J., Delecluse H. J., Raphael M., Lenoir G., Pallesen G. Detection of Epstein-Barr virus genomes in AIDS related lymphomas: sensitivity and specificity of in situ hybridisation compared with Southern blotting. J Clin Pathol. 1991 Aug;44(8):676–680. doi: 10.1136/jcp.44.8.676. [DOI] [PMC free article] [PubMed] [Google Scholar]
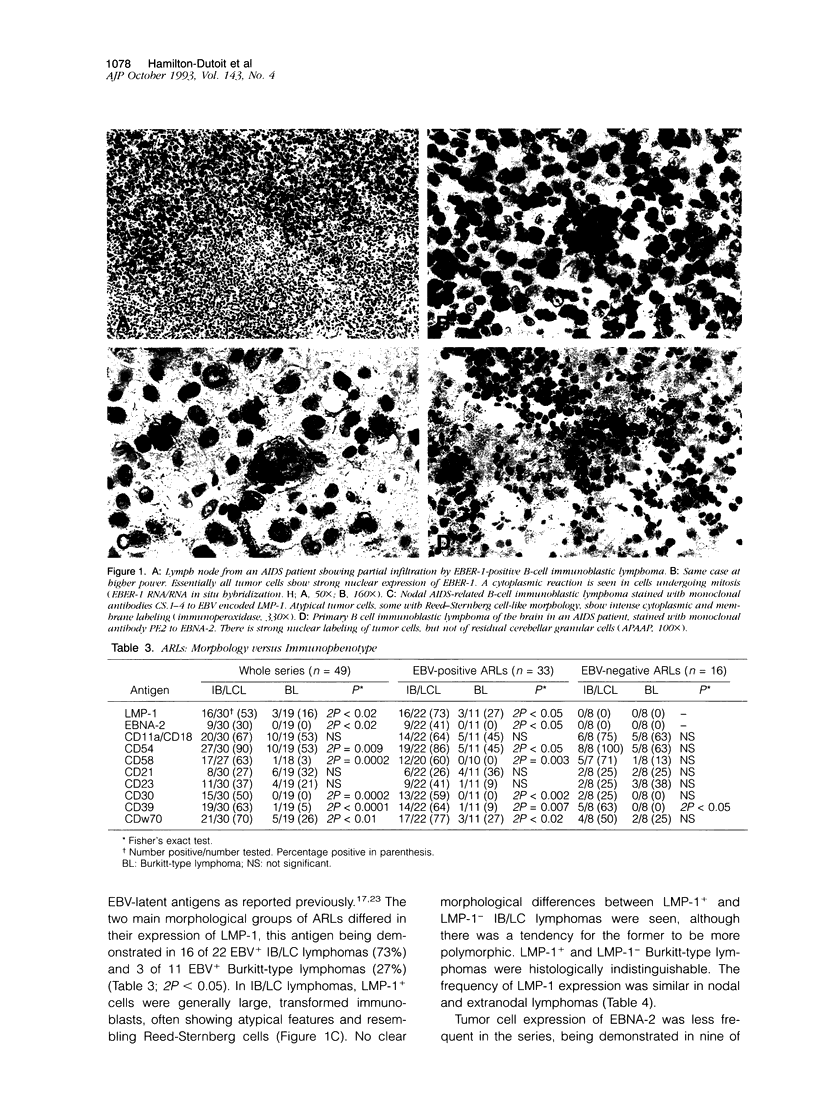
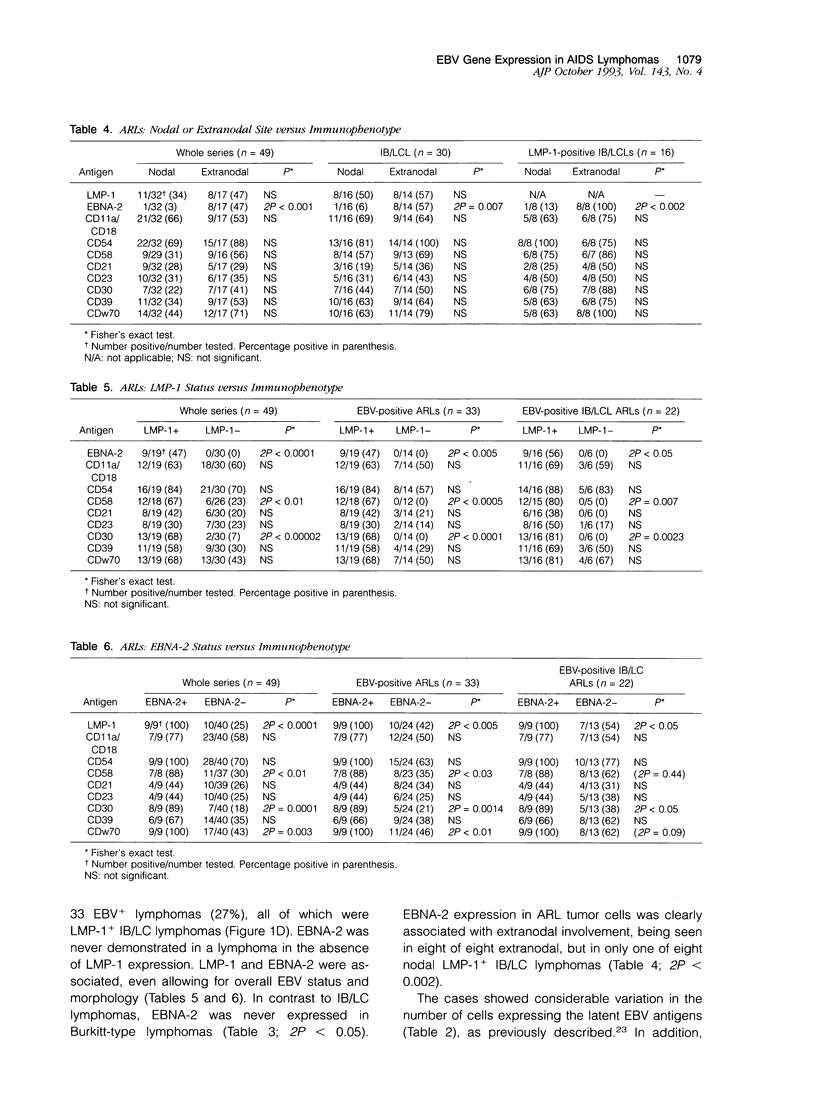
- Hamilton-Dutoit S. J., Pallesen G. A survey of Epstein-Barr virus gene expression in sporadic non-Hodgkin's lymphomas. Detection of Epstein-Barr virus in a subset of peripheral T-cell lymphomas. Am J Pathol. 1992 Jun;140(6):1315–1325. [PMC free article] [PubMed] [Google Scholar]
- Hamilton-Dutoit S. J., Pallesen G., Franzmann M. B., Karkov J., Black F., Skinhøj P., Pedersen C. AIDS-related lymphoma. Histopathology, immunophenotype, and association with Epstein-Barr virus as demonstrated by in situ nucleic acid hybridization. Am J Pathol. 1991 Jan;138(1):149–163. [PMC free article] [PubMed] [Google Scholar]
- Hamilton-Dutoit S. J., Raphael M., Audouin J., Diebold J., Lisse I., Pedersen C., Oksenhendler E., Marelle L., Pallesen G. In situ demonstration of Epstein-Barr virus small RNAs (EBER 1) in acquired immunodeficiency syndrome-related lymphomas: correlation with tumor morphology and primary site. Blood. 1993 Jul 15;82(2):619–624. [PubMed] [Google Scholar]
- Henderson S., Rowe M., Gregory C., Croom-Carter D., Wang F., Longnecker R., Kieff E., Rickinson A. Induction of bcl-2 expression by Epstein-Barr virus latent membrane protein 1 protects infected B cells from programmed cell death. Cell. 1991 Jun 28;65(7):1107–1115. doi: 10.1016/0092-8674(91)90007-l. [DOI] [PubMed] [Google Scholar]
- Jat P., Arrand J. R. In vitro transcription of two Epstein-Barr virus specified small RNA molecules. Nucleic Acids Res. 1982 Jun 11;10(11):3407–3425. doi: 10.1093/nar/10.11.3407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerr B. M., Lear A. L., Rowe M., Croom-Carter D., Young L. S., Rookes S. M., Gallimore P. H., Rickinson A. B. Three transcriptionally distinct forms of Epstein-Barr virus latency in somatic cell hybrids: cell phenotype dependence of virus promoter usage. Virology. 1992 Mar;187(1):189–201. doi: 10.1016/0042-6822(92)90307-b. [DOI] [PubMed] [Google Scholar]
- MacMahon E. M., Glass J. D., Hayward S. D., Mann R. B., Becker P. S., Charache P., McArthur J. C., Ambinder R. F. Epstein-Barr virus in AIDS-related primary central nervous system lymphoma. Lancet. 1991 Oct 19;338(8773):969–973. doi: 10.1016/0140-6736(91)91837-k. [DOI] [PubMed] [Google Scholar]
- Magrath I. The pathogenesis of Burkitt's lymphoma. Adv Cancer Res. 1990;55:133–270. doi: 10.1016/s0065-230x(08)60470-4. [DOI] [PubMed] [Google Scholar]
- Moss D. J., Misko I. S., Burrows S. R., Burman K., McCarthy R., Sculley T. B. Cytotoxic T-cell clones discriminate between A- and B-type Epstein-Barr virus transformants. Nature. 1988 Feb 25;331(6158):719–721. doi: 10.1038/331719a0. [DOI] [PubMed] [Google Scholar]
- Murray R. J., Kurilla M. G., Brooks J. M., Thomas W. A., Rowe M., Kieff E., Rickinson A. B. Identification of target antigens for the human cytotoxic T cell response to Epstein-Barr virus (EBV): implications for the immune control of EBV-positive malignancies. J Exp Med. 1992 Jul 1;176(1):157–168. doi: 10.1084/jem.176.1.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray R. J., Wang D., Young L. S., Wang F., Rowe M., Kieff E., Rickinson A. B. Epstein-Barr virus-specific cytotoxic T-cell recognition of transfectants expressing the virus-coded latent membrane protein LMP. J Virol. 1988 Oct;62(10):3747–3755. doi: 10.1128/jvi.62.10.3747-3755.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niedobitek G., Young L. S., Sam C. K., Brooks L., Prasad U., Rickinson A. B. Expression of Epstein-Barr virus genes and of lymphocyte activation molecules in undifferentiated nasopharyngeal carcinomas. Am J Pathol. 1992 Apr;140(4):879–887. [PMC free article] [PubMed] [Google Scholar]
- Pallesen G., Hamilton-Dutoit S. J., Rowe M., Lisse I., Ralfkiaer E., Sandvej K., Young L. S. Expression of Epstein-Barr virus replicative proteins in AIDS-related non-Hodgkin's lymphoma cells. J Pathol. 1991 Dec;165(4):289–299. doi: 10.1002/path.1711650404. [DOI] [PubMed] [Google Scholar]
- Pedersen C., Gerstoft J., Lundgren J. D., Skinhøj P., Bøttzauw J., Geisler C., Hamilton-Dutoit S. J., Thorsen S., Lisse I., Ralfkiaer E. HIV-associated lymphoma: histopathology and association with Epstein-Barr virus genome related to clinical, immunological and prognostic features. Eur J Cancer. 1991;27(11):1416–1423. doi: 10.1016/0277-5379(91)90023-7. [DOI] [PubMed] [Google Scholar]
- Raphael M., Gentilhomme O., Tulliez M., Byron P. A., Diebold J. Histopathologic features of high-grade non-Hodgkin's lymphomas in acquired immunodeficiency syndrome. The French Study Group of Pathology for Human Immunodeficiency Virus-Associated Tumors. Arch Pathol Lab Med. 1991 Jan;115(1):15–20. [PubMed] [Google Scholar]
- Rowe M., Evans H. S., Young L. S., Hennessy K., Kieff E., Rickinson A. B. Monoclonal antibodies to the latent membrane protein of Epstein-Barr virus reveal heterogeneity of the protein and inducible expression in virus-transformed cells. J Gen Virol. 1987 Jun;68(Pt 6):1575–1586. doi: 10.1099/0022-1317-68-6-1575. [DOI] [PubMed] [Google Scholar]
- Rowe M., Lear A. L., Croom-Carter D., Davies A. H., Rickinson A. B. Three pathways of Epstein-Barr virus gene activation from EBNA1-positive latency in B lymphocytes. J Virol. 1992 Jan;66(1):122–131. doi: 10.1128/jvi.66.1.122-131.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowe M., Rooney C. M., Rickinson A. B., Lenoir G. M., Rupani H., Moss D. J., Stein H., Epstein M. A. Distinctions between endemic and sporadic forms of Epstein-Barr virus-positive Burkitt's lymphoma. Int J Cancer. 1985 Apr 15;35(4):435–441. doi: 10.1002/ijc.2910350404. [DOI] [PubMed] [Google Scholar]
- Rowe M., Rowe D. T., Gregory C. D., Young L. S., Farrell P. J., Rupani H., Rickinson A. B. Differences in B cell growth phenotype reflect novel patterns of Epstein-Barr virus latent gene expression in Burkitt's lymphoma cells. EMBO J. 1987 Sep;6(9):2743–2751. doi: 10.1002/j.1460-2075.1987.tb02568.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowe M., Young L. S., Crocker J., Stokes H., Henderson S., Rickinson A. B. Epstein-Barr virus (EBV)-associated lymphoproliferative disease in the SCID mouse model: implications for the pathogenesis of EBV-positive lymphomas in man. J Exp Med. 1991 Jan 1;173(1):147–158. doi: 10.1084/jem.173.1.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sample J., Brooks L., Sample C., Young L., Rowe M., Gregory C., Rickinson A., Kieff E. Restricted Epstein-Barr virus protein expression in Burkitt lymphoma is due to a different Epstein-Barr nuclear antigen 1 transcriptional initiation site. Proc Natl Acad Sci U S A. 1991 Jul 15;88(14):6343–6347. doi: 10.1073/pnas.88.14.6343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas J. A., Crawford D. H. Epstein-Barr virus associated B-cell lymphomas in AIDS and after organ transplantation. Lancet. 1989 May 13;1(8646):1075–1076. doi: 10.1016/s0140-6736(89)92469-0. [DOI] [PubMed] [Google Scholar]
- Thomas J. A., Hotchin N. A., Allday M. J., Amlot P., Rose M., Yacoub M., Crawford D. H. Immunohistology of Epstein-Barr virus-associated antigens in B cell disorders from immunocompromised individuals. Transplantation. 1990 May;49(5):944–953. doi: 10.1097/00007890-199005000-00022. [DOI] [PubMed] [Google Scholar]
- Wang D., Liebowitz D., Kieff E. An EBV membrane protein expressed in immortalized lymphocytes transforms established rodent cells. Cell. 1985 Dec;43(3 Pt 2):831–840. doi: 10.1016/0092-8674(85)90256-9. [DOI] [PubMed] [Google Scholar]
- Wang F., Gregory C., Sample C., Rowe M., Liebowitz D., Murray R., Rickinson A., Kieff E. Epstein-Barr virus latent membrane protein (LMP1) and nuclear proteins 2 and 3C are effectors of phenotypic changes in B lymphocytes: EBNA-2 and LMP1 cooperatively induce CD23. J Virol. 1990 May;64(5):2309–2318. doi: 10.1128/jvi.64.5.2309-2318.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang F., Tsang S. F., Kurilla M. G., Cohen J. I., Kieff E. Epstein-Barr virus nuclear antigen 2 transactivates latent membrane protein LMP1. J Virol. 1990 Jul;64(7):3407–3416. doi: 10.1128/jvi.64.7.3407-3416.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu T. C., Mann R. B., Charache P., Hayward S. D., Staal S., Lambe B. C., Ambinder R. F. Detection of EBV gene expression in Reed-Sternberg cells of Hodgkin's disease. Int J Cancer. 1990 Nov 15;46(5):801–804. doi: 10.1002/ijc.2910460509. [DOI] [PubMed] [Google Scholar]
- Young L. S., Dawson C. W., Clark D., Rupani H., Busson P., Tursz T., Johnson A., Rickinson A. B. Epstein-Barr virus gene expression in nasopharyngeal carcinoma. J Gen Virol. 1988 May;69(Pt 5):1051–1065. doi: 10.1099/0022-1317-69-5-1051. [DOI] [PubMed] [Google Scholar]
- Young L. S., Finerty S., Brooks L., Scullion F., Rickinson A. B., Morgan A. J. Epstein-Barr virus gene expression in malignant lymphomas induced by experimental virus infection of cottontop tamarins. J Virol. 1989 May;63(5):1967–1974. doi: 10.1128/jvi.63.5.1967-1974.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young L., Alfieri C., Hennessy K., Evans H., O'Hara C., Anderson K. C., Ritz J., Shapiro R. S., Rickinson A., Kieff E. Expression of Epstein-Barr virus transformation-associated genes in tissues of patients with EBV lymphoproliferative disease. N Engl J Med. 1989 Oct 19;321(16):1080–1085. doi: 10.1056/NEJM198910193211604. [DOI] [PubMed] [Google Scholar]



