Abstract
Glycogen synthase kinase (GSK)-3 is a protein serine/threonine kinase that regulates differentiation and cell fate in a variety of organisms. This study examined the role of GSK-3 in antigen-specific T cell responses. Using resting T cells from P14 T cell receptor (TCR)-transgenic mice (specific for the lymphocytic choriomeningitis virus and H-2Db), we demonstrated that GSK-3β was inactivated by serine phosphorylation after viral peptide–specific stimulation in vitro. To further investigate the role of GSK-3, we have generated a retroviral vector that expresses a constitutively active form of GSK-3β that has an alanine substitution at the regulatory amino acid, serine 9 (GSK-3βA9). Retroviral transduction of P14 TCR–transgenic bone marrow stem cells, followed by reconstitution, led to the expression of GSK-3βA9 in bone marrow chimeric mice. T cells from chimeric mice demonstrate a reduction in proliferation and interleukin (IL)-2 production. In contrast, in vitro assays done in the presence of the GSK-3 inhibitor lithium led to dramatically prolonged T cell proliferation and increased IL-2 production. Furthermore, in the presence of lithium, we show that nuclear factor of activated T cells (NF-AT)c remains in the nucleus after antigen-specific stimulation of T cells. Together, these data demonstrate that GSK-3 negatively regulates the duration of T cell responses.
Keywords: signaling, NF-AT, T cell activation, cytokines, lymphocytes
Introduction
Understanding the signaling pathways associated with TCR activation may provide insights that clarify the intracellular mechanisms of costimulation in addition to understanding the sensitivity of TCR interactions that range from antagonism to full T cell activation. TCR interactions lead to phosphorylation of CD3 chains and the associated ζ chains by lck and the activation of ZAP-70, followed by the induction of the extracellular signal–regulated kinase–mitogen-activated protein kinase (ERK–MAPK) pathway. In addition, the activation of phospholipase Cγ generates phospholipid second messengers that activate protein kinase C, calcium mobilization, and the downstream calcineurin/nuclear factor of activated T cells (NF-AT) pathway 1 2 3. One kinase that has not been well defined in TCR signaling is glycogen synthase kinase (GSK)-3.
The two members of the protein serine/threonine kinase GSK-3 family, GSK-3α and β, are involved in regulating cell fate and differentiation in a variety of organisms, including Dictyostelium, Xenopus, and Drosophila 4 5. Unlike most kinases, GSK-3 is active in resting cells. Stimulation with mitogens or growth factors leads to the inactivation of GSK-3β by phosphorylation of the regulatory serine residue at position 9 6 7. There is evidence that GSK-3 is downstream of the wingless pathway 8 9 10 as well as the ERK–MAPK pathway 4 8. GSK-3 acts on a wide variety of substrates including glycogen synthase, c-Jun, c-Myc, and eIF-2B 4 5. Beals et al. have shown that GSK-3 enhances nuclear export of NF-AT in brain extracts and transient transfection of COS cells, thereby negatively regulating this pathway 11. Welsh et al. have shown that GSK-3 can be inactivated by treatment with PMA and ionomycin in peripheral human lymphocytes 12. These studies suggest that GSK-3 may play a role in T cell proliferation in vivo.
We have examined the role of GSK-3 in antigen-specific T cell responses. Together, biochemical analysis, bone marrow chimeric mice expressing a constitutively active form of GSK-3β, and studies using inhibitors demonstrate that GSK-3β regulates NF-AT localization, antigen-specific T cell proliferation, and IL-2 production.
Materials and Methods
Mice.
P14 TCR–transgenic mice (327 line) express a Vα2/Vβ8.1 heterodimer specific for lymphocytic choriomeningitis virus (LCMV) glycoprotein peptide (gp) p33 (KAVYNFATM) and H-2Db 13. The P14 receptor is expressed on 70–90% of mature CD8+ T cells. We have also bred the P14 TCR on a recombinase-activating gene (RAG)2−/− background (H-2b). These mice were bred and maintained under specific pathogen–free conditions according to institutional guidelines. C57BL/6 mice were purchased from The Jackson Laboratory.
Western Blot Analysis.
2–5 × 106 cells were lysed in gentle soft buffer (10 mM NaCl, 20 mM Pipes, pH 7, 0.5% NP-40, 0.05% 2-ME, and inhibitors 0.1 mM PMSF, 100 μM Na3VO4, leupeptin, 50 mM NaF, and 1 mM benzamidine) and run on SDS-PAGE. Western blots were probed with hemagglutinin (HA)-specific antibodies (Upstate Biotechnology) to detect the expression of the retroviral GSK-3βA9, or phospho-specific GSK-3β (serine 9 specific; New England Biolabs, Inc.) or anti-GSK-3 (Upstate Biotechnology). Laser scanning densitometry (Molecular Devices) was used to determine fold increase in phospho-GSK-3β relative to the AV-stimulated control at each time point. Densitometry readings were taken as the sum above background.
Generation of Retroviruses and Bone Marrow Chimeras.
Recombinant retroviruses were packaged using a packaging cell line GP+E and titrated on NIH 3T3 cells as previously described 14. Packaging cell lines producing high-titer viruses (106 CFU/ml) were used to infect bone marrow from 2–4-mo-old P14 TCR–transgenic mice as previously described 15. In brief, cells were cultured at 5 × 105 cells/ml in IMDM supplemented with 50 μM 2-ME, 10% heat-inactivated FCS (Sigma-Aldrich), and IL-3– and IL-6–conditioned media. After 48 h, bone marrow cells were cocultivated with the packaging cell line producing the replication-defective retroviruses for a further 48 h. Selection of bone marrow cells was done with 0.75 mg/ml G418 (GIBCO BRL) for 24 h. Approximately 106 cells were infused into irradiated recipients (900 rads), and animals were reconstituted for 8–12 wk.
Proliferation Assays and IL-2 Production.
Splenocytes (2 × 105) were cocultivated with 105 irradiated C57BL/6 splenocytes as APCs that were previously pulsed with 10−7 M peptides for 1–2 h at 37°C in 96-well flat-bottomed plates. After 48 h, 1 μCi of [3H]thymidine (NEN Life Science Products) was added to the wells and cultured overnight. Cells were harvested and counted on a Matrix 96 direct β-counter (Canberra Packard). The peptides have been characterized to be a strong agonist ligand, p33 (KAVYNFATM); a weaker agonist, A4Y (KAVANFATM); or a nonstimulatory control ligand, AV (SGPSNTPPEI) 16 17. Peptides were generated and purified as previously described 16. In some assays, 10 mM LiCl was added, or as a control, 10 mM KCl.
For IL-2 production, supernatants were removed at the time points indicated in Fig. 3 and Fig. 4. IL-2 activity was assayed by proliferation of the IL-2–dependent CTLL-2 cell line. 5 × 103 CTLL-2 cells were cultured with the supernatant for 24 h, followed by a pulse of 1 μCi of [3H]thymidine overnight.
Figure 3.
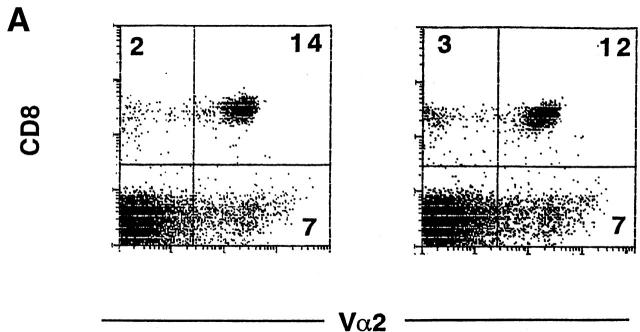
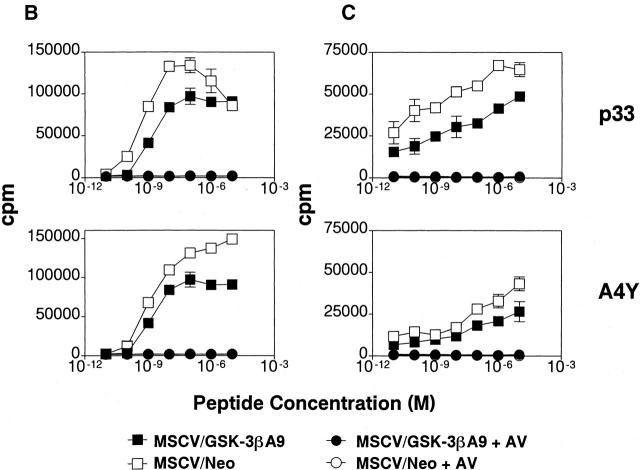
Overexpression of constitutively active GSK-3β inhibits antigen-specific T cell proliferation and IL-2 production. (A) Similar reconstitution of P14 TCR–transgenic T cells in retroviral transduced bone marrow chimeric mice. The spleen cells from chimeric mice, reconstituted with P14 TCR–transgenic bone marrow transduced with MSCV/GSK-3βA9 (left panel) or MSCV/neo (right panel), were stained with antibodies specific for CD8 and Vα2. T cells from bone marrow chimeric mice expressing GSK-3βA9 show decreased proliferation and IL-2 production. (B) Splenocytes from bone marrow chimeric mice transduced with MSCV/GSK-3βA9 show reduced proliferation in response to the strong agonist peptides p33 and weaker agonist A4Y compared with control MSCV-transduced T cells. Proliferative responses using the nonstimulatory AV peptide for the P14 TCR was <500 cpm. Proliferation was measured on day 2. (C) A reduction in IL-2 production was also seen from P14-transgenic T cells from MSCV/GSK-3βA9 chimeric mice. Supernatants from cultures were removed after 24 h, and the amount of IL-2 was quantitated by measuring proliferation of the IL-2–dependent cell line CTLL-2.
Figure 4.
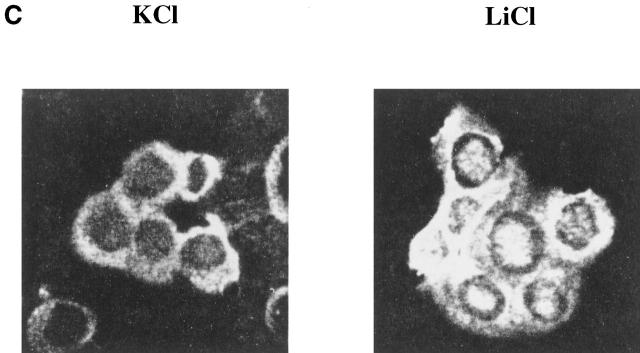
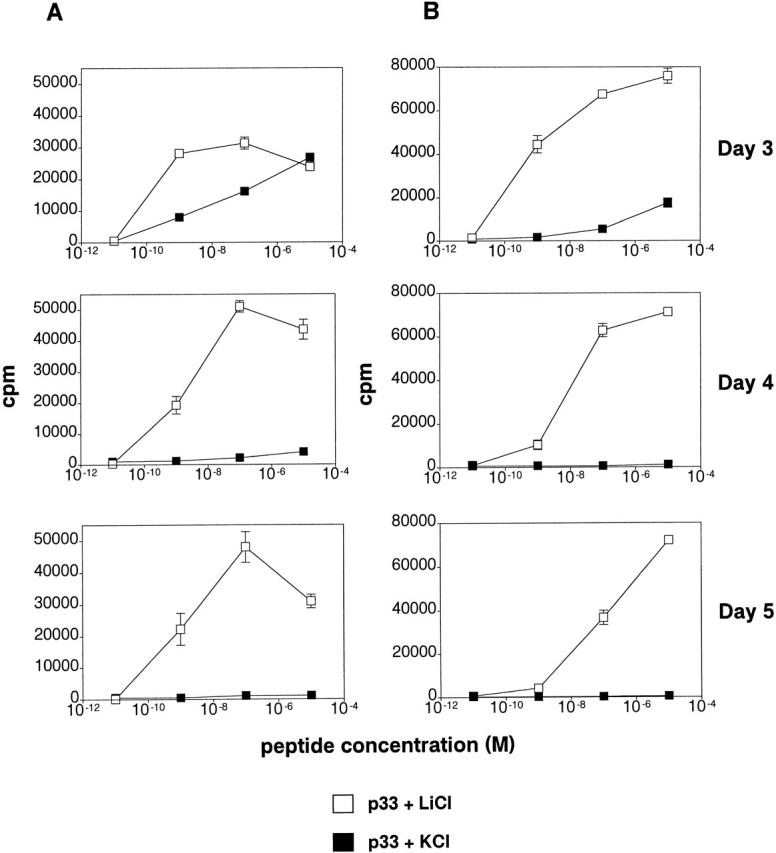
Inhibition of GSK-3 leads to prolonged T cell proliferation and IL-2 production. Splenocytes from P14 TCR–transgenic mice were cocultivated with APCs pulsed with the strong agonist ligand p33 or control AV peptide, in the presence of LiCl or KCl. (A) Proliferation of T cells was measured by thymidine incorporation. Proliferation in response to AV was below 300 cpm. (B) IL-2 production was measured by proliferation of the IL-2–dependent cell line CTLL-2 by the addition of supernatant from cultures harvested from the indicated days. (C) GSK-3 regulates NF-ATc nuclear localization. Purified CD8+ T cells from P14 TCR–transgenic RAG2−/− mice were cocultured with APCs pulsed with p33 for 3 d in the presence of LiCl or KCl. Cells were fixed, permeabilized, and stained using an NF-ATc–specific antibody.
Immunofluorescence.
105 cells were stained with antibodies specific for CD4, CD8, Vβ8, or Vα2 (PharMingen) in PBS, 1% FCS, and 0.1% sodium azide. Cells were washed once and then fixed in 1% paraformaldehyde.
For intracellular NF-ATc staining, P14 TCR RAG−/− splenocytes were cocultivated with thioglycollate-stimulated macrophages pulsed with the LCMV-gp peptide p33. After 3 d in the presence of 10 mM KCl or LiCl, cells were harvested and spun onto slides. Cells were dipped in acetone for 15 s, followed by methanol for 6 min. Slides were incubated in blocking buffer (PBS, 2% FCS, 5% donkey serum, 5% BSA) for 1 hr, followed by anti–NF-ATc1 (a gift from G. Crabtree, Stanford University, Stanford, CA) for 72 h. After washing, the donkey anti–mouse secondary antibody (The Jackson Laboratory) was added for 1 h, followed by streptavidin–FITC (Sigma-Aldrich).
Results and Discussion
Studies have suggested that GSK-3 may be involved in lymphocyte activation 11 12. To determine whether antigen-specific stimulation of lymphocytes inactivates GSK-3, naive CD8+ splenocytes were sorted from P14 TCR–transgenic mice (specific for LCMV-gp [amino acid 33–41, p33] and H-2Db) and stimulated with macrophages pulsed with the antigenic ligand p33 or a control peptide AV (Fig. 1). Western blot analysis using an antibody specific for inactive GSK-3β (GSK-3 serine phosphorylated at position 9) shows an increase in phosphorylated GSK-3β after peptide-specific stimulation. Densitometry analysis indicates that the amount of phosphorylated GSK-3β has increased one- to twofold. This demonstrates that GSK-3β becomes inactivated after antigen-specific T cell stimulation.
Figure 1.
TCR-specific stimulation leads to inactivation of GSK-3. CD8-purified P14 TCR–transgenic T cells were incubated with macrophages pulsed with the antigenic ligand p33 (P) or nonstimulatory ligand AV (A). After the indicated time periods, cells were lysed and analyzed by Western blot using phospho-serine–specific GSK-3 antibodies. Numbers indicate the densitometry measurements as fold increase relative to AV-treated cells. Total GSK-3 was shown as a control.
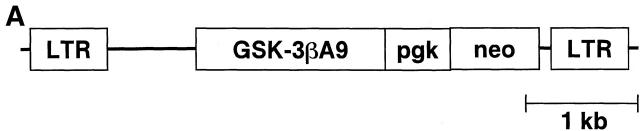
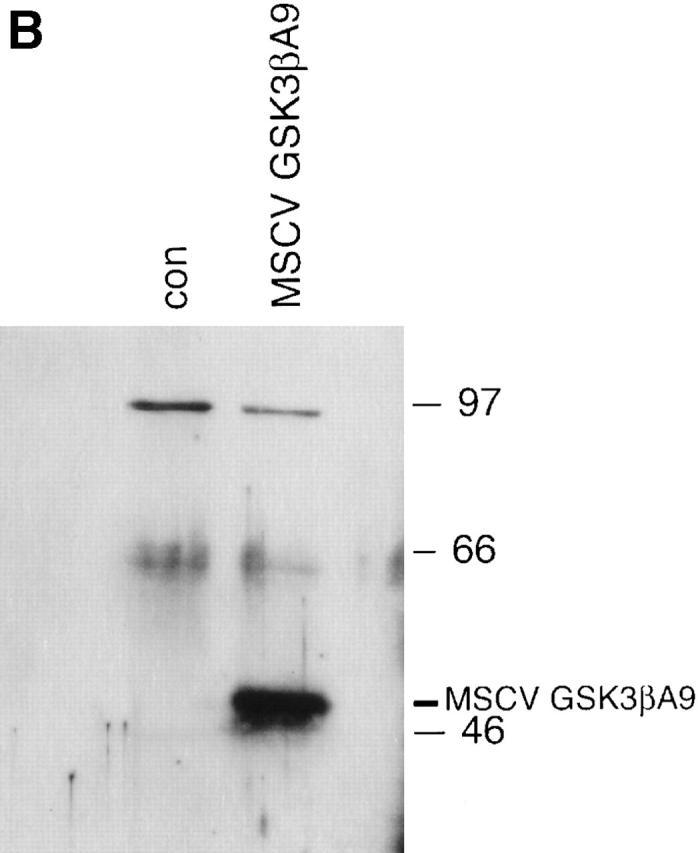
Previous studies have demonstrated that a substitution of alanine for serine at amino acid position 9 of GSK-3β leads to constitutive kinase activity in human cell lines and transgenic mice 7 18. To understand the role of GSK-3 in T cell responses, an HA-tagged constitutively active form of GSK-3β (GSK-3βA9) was cloned into the retroviral vector murine stem cell virus (MSCV; Fig. 2 A; reference 14). Expression was confirmed by transfecting NIH 3T3 cells with MSCV/GSK-3βA9 and examining expression by Western blot analysis using HA-specific mAbs (Fig. 2 B). A high-titer packaging cell line for MSCV/GSK-3βA9 and control MSCV was obtained (106 CFU/ml) and used to infect bone marrow stem cells from P14 TCR–transgenic mice. The frequency of bone marrow precursors that were transduced with the retrovirus was quantitated by selecting cells with neomycin. Routinely, 60–90% of stem cells carried the selectable marker. These cells were used to generate bone marrow chimeric mice with C57BL/6 irradiated host animals.
Figure 2.
Retroviral expression of GSK-3βA9. (A) A retroviral vector was generated using MSCV and a constitutively active form of GSK-3β (GSK-3βA9) tagged with HA. GSK-3βA9 is expressed from the retroviral LTR, using the splice donor and splice acceptor sites. The selectable neomycin (G418)-resistant marker is expressed from the phosphoglycerate kinase (pgk) promoter. (B) NIH 3T3 cells were transduced with the retrovirus MSCV/GSK-3βA9, and expression of GSK-3βA9 was examined by Western blot using HA-specific antibodies, compared with control (con) NIH 3T3 cells.
Chimeric mice were compared from P14 TCR–transgenic bone marrow transduced with MSCV/GSK-3βA9 and control MSCV. After reconstitution, flow cytometry analysis using antibodies specific for CD4, CD8, and Vα2 (expressed by the P14 transgenic TCR) showed comparable numbers of P14-transgenic CD8+ T cells in the thymus (data not shown) and spleen (Fig. 3 A). This suggests that positive selection and survival of peripheral T lymphocytes was not dramatically affected by the presence of GSK-3βA9. Mature T cell LCMV peptide-specific responses were assessed using a strong agonist ligand p33, the weaker agonist A4Y, and nonstimulatory control peptide AV. Proliferative responses were consistently reduced in GSK-3βA9–reconstituted animals (Fig. 3 B). IL-2 production from these cultures was also assessed by measuring proliferation of an IL-2–dependent line, CTLL-2. Fig. 3 C shows that IL-2 production was also reduced in T cells from MSCV/GSK-3βA9 chimeric mice. Therefore, inactivation of GSK-3 is required for maximal proliferation and IL-2 production.
T cell responses were also examined in the presence of LiCl, which selectively inhibits GSK-3 activity in a variety of cells, including COS cells, rat PC12 cells, Drosophila, Xenopus, and Dictyostelium 9 19 20. Splenocytes from P14 TCR–transgenic mice were incubated with the strong antigenic peptide p33 and weaker agonist A4Y ligand, in the presence of 10 mM LiCl or 10 mM KCl, and pulsed with [3H]thymidine at several time points. The initial proliferative responses after 24 h were similar (data not shown). However, in the presence of lithium, dramatically increased and prolonged proliferative responses to p33 and A4Y were seen after 3, 4, and 5 d (Fig. 4 A). In addition, IL-2 production was dramatically increased in the presence of lithium, as measured by proliferation of the IL-2–dependent line CTLL-2 (Fig. 4 B). Together, these data suggest that GSK-3 regulates the duration of T lymphocyte responses.
Previous studies have shown that GSK-3 phosphorylates NF-ATc, which promotes cytoplasmic localization and also export from the nucleus 11. Therefore, the presence of LiCl should inactivate GSK-3β, resulting in prolonged dephosphorylation of NF-ATc and sustained nuclear localization. We examined the localization of NF-ATc 3 d after T cells were stimulated with the antigenic peptide p33 in the presence or absence of LiCl. Fluorescent microscopy showed that NF-ATc was found in the nucleus in the presence of inactive GSK-3 (Fig. 4 C). This demonstrates that GSK-3 plays an important role in antigen-specific T cell activation by regulating NF-ATc localization.
Our studies suggest that TCR signals inactivate GSK-3, which negatively regulates T cell proliferation and IL-2 production by altering the nuclear import/export of NF-AT. Evidence from other systems support this model. Studies using HeLa and BHK cells have shown that Crm1 and Ran are involved in NF-AT export from the nucleus and have suggested that GSK-3 contributes to this process 21 22. In addition, mutation of the serine residue in NF-AT that is phosphorylated by GSK-3 leads to constitutive nuclear localization 23. Astoul et al. 24 have examined the role of GSK-3 in B cell receptor signaling using the B lymphoma cell line A20. Cross-linking the B cell receptor with F(ab′)2 fragments lead to the phosphorylation and inactivation of the GSK-3α isoform. Together, studies from a variety of models support a role for GSK-3 in receptor-mediated signals that shuttle NF-AT from the nucleus.
Surprisingly little is known about the negative regulation of T cell activation. Cell surface interactions through molecules such as CTLA-4 negatively regulate T cell responses by interacting with the costimulatory ligands B7-1 and B7-2 25. Other molecules such as CD5 have been shown to negatively influence TCR-mediated signals 26 27. In addition, signaling molecules such as the tyrosine kinase Csk 28, SHP-1 (motheaten; PTP-1C; references 29 30 31 32), Cabin 1 33, and the adaptor Cbl 34 35 have also been demonstrated to play a role in negatively regulating T cell responses. In this study, we have shown that GSK-3, a molecule that is generally known to be active in resting cells, is inactivated upon TCR stimulation. We show that GSK-3 is involved in the regulation of TCR-mediated proliferation and that one role of GSK-3 is to negatively regulate NF-ATc activity and IL-2 production. However, GSK-3 has a multitude of substrates, and it will be interesting to understand how these different effectors interplay with other pathways to orchestrate resting T cell survival and activation.
Acknowledgments
We would like to thank Dr. Gerald Crabtree for the generous gift of the NF-AT antibodies and Dr. Satoshi Matsuda for critical reading of the manuscript.
This work was supported by the Medical Research Council of Canada. P.S. Ohashi is a Medical Research Council Scientist.
Footnotes
Abbreviations used in this paper: gp, glycoprotein peptide; GSK, glycogen synthase kinase; LCMV, lymphocytic choriomeningitis virus; MSCV, murine stem cell virus; NF-AT, nuclear factor of activated T cells; RAG, recombinase-activating gene.
References
- Qian D., Weiss A. T cell antigen receptor signal transduction. Curr. Opin. Cell Biol. 1997;9:205–212. doi: 10.1016/s0955-0674(97)80064-6. [DOI] [PubMed] [Google Scholar]
- Alberola-Ila J., Takaki S., Kerner J.D., Perlmutter R.M. Differential signalling by lymphocyte antigen receptors. Annu. Rev. Immunol. 1997;15:125–154. doi: 10.1146/annurev.immunol.15.1.125. [DOI] [PubMed] [Google Scholar]
- Rao A., Luo C., Hogan P.G. Transcription factors of the NFAT familyregulation and function. Annu. Rev. Immunol. 1997;15:707–747. doi: 10.1146/annurev.immunol.15.1.707. [DOI] [PubMed] [Google Scholar]
- Woodgett J.R. Regulation and functions of the glycogen synthase kinase-3 subfamily. Cancer Biol. 1994;5:269–275. [PubMed] [Google Scholar]
- Welsh G.I., Wilson C., Proud C.G. GSK3a SHAGGY frog story. Cell Biol. 1996;6:274–279. doi: 10.1016/0962-8924(96)10023-4. [DOI] [PubMed] [Google Scholar]
- Sutherland C., Leighton I.A., Cohen P. Inactivation of glycogen synthase kinase-3 β by phosphorylationnew kinase connections in insulin and growth-factor signalling. Biochem. J. 1993;296:15–19. doi: 10.1042/bj2960015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stambolic V., Woodgett J.R. Mitogen inactivation of glycogen synthase kinase-3β in intact cells via serine 9 phosphorylation. Biochem. J. 1994;303:701–704. doi: 10.1042/bj3030701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook D., Fry M.J., Hughes K., Sumathipala R., Woodgett J.R., Dale T.C. Wingless inactivates glycogen synthase kinase-3 via an intracellular signalling pathway which involves a protein kinase C. EMBO (Eur. Mol. Biol. Organ.) J. 1996;15:4526–4536. [PMC free article] [PubMed] [Google Scholar]
- Stambolic V., Ruel L., Woodgett J.R. Lithium inhibits glycogen synthase kinase-3 activity and mimics Wingless signalling in intact cells. Curr. Biol. 1996;6:1664–1668. doi: 10.1016/s0960-9822(02)70790-2. [DOI] [PubMed] [Google Scholar]
- Moon R.T., Brown J.D., Yang-Snyder J.A., Miller J.R. Structurally related receptors and antagonists compete for secreted Wnt ligands. Cell. 1997;88:725–728. doi: 10.1016/s0092-8674(00)81915-7. [DOI] [PubMed] [Google Scholar]
- Beals C.R., Sheridan C.M., Turck C.W., Gardner P., Crabtree G.R. Nuclear export of NF-ATc enhanced by glycogen synthase kinase-3. Science. 1997;275:1930–1933. doi: 10.1126/science.275.5308.1930. [DOI] [PubMed] [Google Scholar]
- Welsh G.I., Miyamoto S., Price N.T., Safer B., Proud C.G. T-cell activation leads to rapid stimulation of translation initiation factor eIF2B and inactivation of glycogen synthase kinase-3. J. Biol. Chem. 1996;271:11410–11413. doi: 10.1074/jbc.271.19.11410. [DOI] [PubMed] [Google Scholar]
- Pircher H., Bürki K., Lang R., Hengartner H., Zinkernagel R. Tolerance induction in double specific T-cell receptor transgenic mice varies with antigen. Nature. 1989;342:559–561. doi: 10.1038/342559a0. [DOI] [PubMed] [Google Scholar]
- Hawley R.G., Lieu F.H.L., Fong A.Z.C., Hawley T.S. Versatile retroviral vectors for potential use in gene therapy. Gene Ther. 1994;1:136–138. [PubMed] [Google Scholar]
- Ally B.A., Hawley T.S., MacKall-Faienza K., Kündig T.M., Oehen S.U., Pircher H., Hawley R.G., Ohashi P.S. Prevention of autoimmune disease by retroviral-mediated gene therapy. J. Immunol. 1995;155:5404–5408. [PubMed] [Google Scholar]
- Sebzda E., Kündig T.M., Thomson C.T., Aoki K., Mak S.Y., Mayer J., Zamborelli T.M., Nathenson S., Ohashi P.S. Mature T cell reactivity altered by a peptide agonist that induces positive selection. J. Exp. Med. 1996;183:1093–1104. doi: 10.1084/jem.183.3.1093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mariathasan S., Bachmann M.F., Bouchard D., Ohteki T., Ohashi P.S. Degree of TCR internalization and Ca2+ flux correlates with thymocyte selection. J. Immunol. 1998;161:6030–6037. [PubMed] [Google Scholar]
- Brownlees J., Irving N.G., Brion J.-P., Gibb B.J., Wagner U., Woodgett J., Miller C.C. Tau phosphorylation in transgenic mice expressing glycogen synthase kinase-3beta transgenes. Neuroreport. 1997;8:3251–3255. doi: 10.1097/00001756-199710200-00013. [DOI] [PubMed] [Google Scholar]
- Klein P.S., Melton D.A. A molecular mechanism for the effect of lithium on development. Proc. Natl. Acad. Sci. USA. 1996;93:8455–8459. doi: 10.1073/pnas.93.16.8455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedgepeth C.M., Conrad L.J., Zhang J., Huang H.C., Lee V.M., Klein P.S. Activation of the Wnt signaling pathwaya molecular mechanism for lithium action. Dev. Biol. 1997;185:82–91. doi: 10.1006/dbio.1997.8552. [DOI] [PubMed] [Google Scholar]
- Kehlenbach R.H., Dickmanns A., Gerace L. Nucleocytoplasmic shuttling factors including Ran and CRM1 mediate nuclear export of NFAT in vitro. J. Cell Biol. 1998;141:863–874. doi: 10.1083/jcb.141.4.863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu J., McKeon F. NF-AT activation requires suppression of Crm1-dependent export by calcineurin. Nature. 1999;398:256–260. doi: 10.1038/18473. [DOI] [PubMed] [Google Scholar]
- Beals C.R., Clipstone N.A., Ho S.N., Crabtree G.R. Nuclear localization of NF-ATc by a calcineurin-dependent, cyclosporin-sensitive intramolecular interaction. Genes Dev. 1997;11:824–834. doi: 10.1101/gad.11.7.824. [DOI] [PubMed] [Google Scholar]
- Astoul E., Watton S., Cantrell D. The dynamics of protein kinase B regulation during B cell antigen receptor engagement. J. Cell Biol. 1999;145:1511–1520. doi: 10.1083/jcb.145.7.1511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenschow D.J., Walunas T.L., Bluestone J.A. CD28/B7 system of T cell costimulation. Annu. Rev. Immunol. 1996;14:233–258. doi: 10.1146/annurev.immunol.14.1.233. [DOI] [PubMed] [Google Scholar]
- Tarakhovsky A., Kanner S.B., Hombach J., Ledbetter J.A., Müller W., Killeen N., Rajewsky K. A role for CD5 in TCR-mediated signal transduction and thymocyte selection. Science. 1995;269:535–537. doi: 10.1126/science.7542801. [DOI] [PubMed] [Google Scholar]
- Azzam H.S., Grinberg A., Lui K., Shen H., Shores E.W., Love P.E. CD5 expression is developmentally regulated by T cell receptor (TCR) signals and TCR avidity. J. Exp. Med. 1998;188:2301–2311. doi: 10.1084/jem.188.12.2301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmedt C., Saijo K., Niidome T., Kühn R., Aizawa S., Tarakhovsky A. Csk controls antigen receptor-mediated development and selection of T-lineage cells. Nature. 1998;394:901–904. doi: 10.1038/29802. [DOI] [PubMed] [Google Scholar]
- Plas D.R., Johnson R., Pingel J.T., Matthews R.J., Dalton M., Roy G., Chan A.C., Thomas M.L. Direct regulation of ZAP-70 by SHP-1 in T cell antigen receptor signaling. Science. 1996;272:1173–1176. doi: 10.1126/science.272.5265.1173. [DOI] [PubMed] [Google Scholar]
- Pani G., Fischer K.-D., Mlinaric-Rascan I., Siminovitch K.A. Signaling capacity of the T cell antigen receptor is negatively regulated by the PTP1C tyrosine phosphatase. J. Exp. Med. 1996;184:839–852. doi: 10.1084/jem.184.3.839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorenz U., Ravichandran K.S., Burakoff S.J., Neel B.G. Lack of SHPTP1 results in src-family kinase hyperactivation and thymocyte hyperresponsiveness. Proc. Natl. Acad. Sci. USA. 1996;93:9624–9629. doi: 10.1073/pnas.93.18.9624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carter J.D., Neel B.G., Lorenz U. The tyrosine phosphatase SHP-1 influences thymocyte selection by setting TCR signaling thresholds. Int. Immunol. 1999;11:1999–2014. doi: 10.1093/intimm/11.12.1999. [DOI] [PubMed] [Google Scholar]
- Sun L., Youn H.D., Loh C., Stolow M., He W., Liu J.O. Cabin 1, a negative regulator for calcineurin signaling in T lymphocytes. Immunity. 1998;8:703–711. doi: 10.1016/s1074-7613(00)80575-0. [DOI] [PubMed] [Google Scholar]
- Murphy M.A., Schnall R.G., Venter D.J., Barnett L., Bertoncello I., Thien C.B.F., Langdon W.Y., Bowtell D.D.L. Tissue hyperplasia and enhanced T-cell signalling via ZAP-70 in c-Cbl-deficient mice. Mol. Cell. Biol. 1998;18:4872–4882. doi: 10.1128/mcb.18.8.4872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naramura M., Kole H.K., Hu R.-J., Gu H. Altered thymic positive selection and intracellular signals in Cbl-deficient mice. Proc. Natl. Acad. Sci. USA. 1998;95:15447–15552. doi: 10.1073/pnas.95.26.15547. [DOI] [PMC free article] [PubMed] [Google Scholar]