Abstract
Cones in the vertebrate retina project to horizontal and bipolar cells and the horizontal cells feedback negatively to cones. This organization forms the basis for the center/surround organization of the bipolar cells, a fundamental step in the visual signal processing. Although the surround responses of bipolar cells have been recorded on many occasions, surprisingly, the underlying surround-induced responses in cones are not easily detected. In this paper, the nature of the surround-induced responses in cones is studied. Horizontal cells feed back to cones by shifting the activation function of the calcium current in cones to more negative potentials. This shift increases the calcium influx, which increases the neurotransmitter release of the cone. In this paper, we will show that under certain conditions, in addition to this increase of neurotransmitter release, a calcium-dependent chloride current will be activated, which polarizes the cone membrane potential. The question is, whether the modulation of the calcium current or the polarization of the cone membrane potential is the major determinant for feedback-mediated responses in second-order neurons. Depolarizing light responses of biphasic horizontal cells are generated by feedback from monophasic horizontal cells to cones. It was found that niflumic acid blocks the feedback-induced depolarizing responses in cones, while the shift of the calcium current activation function and the depolarizing biphasic horizontal cell responses remain intact. This shows that horizontal cells can feed back to cones, without inducing major changes in the cone membrane potential. This makes the feedback synapse from horizontal cells to cones a unique synapse. Polarization of the presynaptic (horizontal) cell leads to calcium influx in the postsynaptic cell (cone), but due to the combined activity of the calcium current and the calcium-dependent chloride current, the membrane potential of the postsynaptic cell will be hardly modulated, whereas the output of the postsynaptic cell will be strongly modulated. Since no polarization of the postsynaptic cell is needed for these feedback-mediated responses, this mechanism of synaptic transmission can modulate the neurotransmitter release in single synaptic terminals without affecting the membrane potential of the entire cell.
Keywords: retina, negative feedback, calcium-dependent chloride current, lateral inhibition
INTRODUCTION
The photoreceptors (rods and cones) in the vertebrate retina respond with sustained hyperpolarizing responses to stimulation with small spots of light. They have small receptive fields and feed into a network of horizontal cells (HCs) and bipolar cells (BCs). The HCs are strongly electrically coupled, have large receptive fields (Naka and Rushton 1967; Norton et al. 1968; Baylor et al. 1971) and feed back negatively to the cones (O'Bryan 1973; Verweij et al. 1996; Kraaij et al. 1998). This feedback pathway forms the basis of the spectral coding of HCs (Fuortes and Simon 1974; Stell and Lightfoot 1975; Kamermans et al. 1991) and of the surround responses of BCs (Kaneko 1970; Kaneko and Tachibana 1981; Toyoda and Kujiraoka 1982). Due to the spatial extension of the HC-receptive fields, stimulation in the surround of a cone can induce depolarizing responses in cones (Baylor et al. 1971; O'Bryan 1973; Lasansky 1981). These surround-induced depolarizing responses are mainly carried by Cl− (Lasansky 1981; Thoreson and Burkhardt 1991; Barnes and Deschenes 1992).
On the other hand, it has recently been shown that HCs feedback to cones by modulating the calcium current (ICa) in cones in a γ-aminobutyric acid (GABA)–independent way (Verweij et al. 1996). Unfortunately, the neurotransmitter mediating this feedback signal from HCs to cones is presently unknown. Consistent with this finding is the observation that the surround responses in BCs do not seem to be GABA mediated (Hare and Owen 1996). Hyperpolarization of the HCs shifts the activation function of ICa in cones to more negative potentials, which leads to an increased calcium influx. This finding seems in conflict with the well-established Cl dependence of the surround-induced responses in cones. Several authors (Maricq and Korenbrot 1988; Thoreson and Burkhardt 1991; Barnes and Deschenes 1992) have suggested that the surround-induced depolarizing responses in cones are mediated by a calcium-dependent chloride current (ICl(Ca)), which can be activated by a feedback-induced calcium influx. They suggested that such a pathway could become, under certain conditions, regenerative, leading to the observed feedback-induced polarization of the cone membrane potential.
Depending on the intracellular chloride concentration ([Cl]i) in cones [i.e., different chloride equilibrium potentials (ECl)], surround stimulation would generate either depolarizing or hyperpolarizing responses in cones. This suggests that with values of ECl more positive than the resting membrane potential, feedback from HCs would be negative, whereas with ECl more negative than the resting membrane potential, feedback from HCs would be positive. Estimates of ECl in cones, reported in literature, range from values more positive than −40 mV (Thoreson and Burkhardt 1991) to values more negative than −50 mV (Kaneko and Tachibana 1986). Since the cone dark resting membrane potential is approximately −45 mV, the estimates of ECl, reported in literature, suggest that feedback from HCs can be either positive or negative, which is not consistent with the well-established antagonistic nature of the bipolar cell surround.
The question now arising is whether the modulation of ICa alone is enough to induce a feedback response in second-order neurons or whether depolarization of the cone is essential for transmission of the feedback signal to second-order neurons. In this study, the role of Cl− and ICl(Ca) in the negative feedback pathway from HCs to cones is studied and the relative contribution of the cone depolarization and the modulation of ICa on the feedback-mediated responses in second-order neurons is estimated.
It will be shown that the surround-induced responses are initiated by a feedback-induced calcium influx in the cones, which can activate ICl(Ca). Since the conductance of the Ca channels is relatively small compared with the total conductance of the cell and ICl(Ca) is hardly activated in the dark, the membrane potential of the cone is nearly independent of feedback, whereas the cone neurotransmitter release is strongly modulated by feedback. The consequence is that, as will be shown, HCs can have depolarizing responses even in conditions where cones do not depolarize, which means that the feedback pathway from HCs to cones is electrically almost silent.
MATERIALS AND METHODS
Preparation
Goldfish, Carassius Auratus, (12–16-cm standard body length) were kept at 18°C under a 12-h dark, 12-h light regime. Before the experiment, the fish was kept in the dark for 8 ± 1 min, decapitated, and an eye was enucleated. This eye was hemisected and most of the vitreous was removed with filter paper. The retina was isolated, placed receptor side up in a superfusion chamber and superfused continuously (1.5 ml/min) with oxygenated Ringer's solution (pH 7.8, 18°C). This procedure was done under infrared light illumination (λ = 920 nm) using infrared viewers (Edmund Scientific).
Recording Procedure
Whole cell patch clamp recordings.
The superfusion chamber was mounted on a microscope (Optiphot-X2; Nikon, Inc.). The preparation was illuminated with infrared light (λ > 850 nm, wratten filter 87c; Eastman-Kodak Co.) and viewed with a 40× water immersion objective (numeric aperture, NA = 0.55; Nikon, Inc.), Hoffman modulation contrast optics and a video camera (Philips). Electrodes were mounted on an MP-85 Huxley/Wall-type micro-manipulator (Sutter Instruments Co.) and connected to an integrating patch clamp (3900A; Dagan Corp.). For the data acquisition, control of the patch clamp and optical stimulator (CED 1401 AD/DA convertor; Cambridge Electronic Design Ltd.) and an MS-DOS based computer system was used.
Intracellular recordings.
For the intracellular recordings from HCs, the retina was mounted on an inverted microscope (IMT-2; Olympus Corp.), illuminated with an infra red light emitting diode (LED SFH 484-II; Telefunken), and viewed through a 2× objective of the microscope and a video camera (Philips). The recordings were made with a S7000A microelectrode amplifier with a S7071A electrometer module (World Precision Instruments, Inc.).
Data acquisition, control of the microelectrode amplifier, and of the optical stimulator were done with a CED 1401 AD/DA convertor with a sample frequency of 1.0 kHz (Cambridge Electronic Design Ltd.) and an MS-DOS based computer system.
Optical Stimulator
Whole cell patch clamp recordings.
A 450-W xenon-lamp supplied two beams of light. Both beams were projected through Uniblitz VS14 shutters (Vincent Associates), neutral density filters (NG Schott), interference filters with a bandwidth of 8 ± 3 nm (Ealing Electro-Optics, Inc.), lenses, and apertures. The 65-μm diameter spots were projected through the 40× water immersion objective and the 3,000-μm diameter spots were projected through the condenser (N.A. = 1.25) of the microscope.
Intracellular recordings.
The optical stimulator consisted of two beams from a 450-W xenon light source, projected through a monochromator (Ebert), or interference filters with a bandwidth of 8 ± 3 nm (Ealing Electro-Optics, Inc.) and a pair of circular neutral density filters (Barr & Strout). The full-field light stimuli were projected onto the retina through a 2× objective lens (N.A. = 0.08) of an inverted microscope (IMT-2; Olympus Corp.). The photon flux density of 4.0 ×108 photons μm−2 s−1 corresponds to an intensity of 0 log.
Patch Electrodes and Pipette Medium
Whole cell patch clamp recordings.
The patch pipettes were pulled from borosilicate glass (GC150TF-10; Clark) with a micropipette puller (P-87; Sutter Instruments Co.) and had impedances between 5 and 10 MΩ when filled with standard patch pipette medium and measured in Ringer's solution. The series resistance during the whole cell recording was between 10 and 20 MΩ.
The standard patch pipette medium contained (mM): 20.0 KCL, 70.0 d-gluconic-K, 5.0 KF, 1.0 MgCl2, 0.1 CaCl2, 1.0 EGTA, 5.0 HEPES, 4.0 ATP-Na2, 1.0 GTP-Na3, 0.2 3′:5′-cGMP-Na, 20 phosphocreatine-Na2, 50 U/ml creatine phosphokinase. To change the Cl− equilibrium potential, KCl was exchanged for equimolar d-gluconic-K. The pH of the pipette medium was adjusted to 7.25 with KOH. All chemicals were obtained from Sigma-Aldrich.
Intracellular recordings.
Microelectrodes were pulled on a micropipette puller (P-80-PC; Sutter Instruments Co.) using aluminosilicate glass (o.d. = 1.0 mm, i.d. = 0.5 mm; Clark), and had impedances ranging 100–200 MΩ when filled with 4 M KAc. The intracellular voltages were amplified (WPI S7000A; World Precision Instruments, Inc.), recorded on paper (Graphtec Linearcorder), and sampled using an MS-DOS–based computer system and an AD/DA converter (CED 1401; Cambridge Electronic Design).
Liquid junction potential.
The liquid junction potential was measured with a patch electrode, filled with pipette medium, and positioned in a pipette medium containing bath. The reference electrode was a patch electrode filled with 3 M KCl. After the potential was adjusted to zero, the bath solution was replaced with Ringer's solution. The resulting potential change was considered as the junction potential and all data were corrected accordingly. All results presented in this paper are obtained ∼15 min after whole cell configuration was achieved, unless otherwise indicated.
Classification
Cones were selected visually and the voltage light responses to 500-ms flashes of 65 μm spots with seven different wavelengths and various intensities were recorded. Cells with a maximal response of ∼450, 550, and 650 nm were classified as short, middle, and long wavelength–sensitive cones, respectively. Upon classification, only white stimuli of either 65 or 3000 μm were used.
Horizontal cells were searched by penetrating the retina with a microelectrode, while stimulating the retina with alternating 550- and 650-nm full-field stimuli. Subsequently, the HCs were classified based on their spectral properties formulated by Norton et al. 1968.
Ringer's Solution
The Ringer's solution contained (mM): 102.0 NaCl, 2.6 KCl, 1.0 MgCl2, 1.0 CaCl2, 28.0 NaHCO3, 5.0 glucose, and in some cases 0.1 niflumic acid, and was continuously gassed with ∼2.5% CO2 and 97.5% O2, yielding a pH of 7.8. All chemicals were obtained from Sigma-Aldrich.
RESULTS
Cl Dependence of the Surround-induced Response in Cones
Many of the surround-induced responses in cones reported in literature were measured with intracellular electrodes. Those responses were depolarizing and seemed to depend on the intracellular Cl concentration. Since in those experiments the intracellular Cl concentration could only be controlled to a limited extent, the first set of experiments in this study deals with the Cl dependence of the surround-induced responses in cones.
Feedback-induced voltage responses can be measured when cones are saturated with a bright 65-μm spot and stimulated with a full-field stimulus (3,000 μm). Since feedback is most efficient at potentials between −55 and −45 mV (Verweij et al. 1996) and the bright saturating spot hyperpolarized the cones further than that potential range, a small inward current was injected into the cones such that they depolarized again to their resting membrane potential between −55 and −45 mV. By recording from a large number of cones with different intracellular Cl concentration, the dependence of the surround-induced responses on Cl was determined without interference of the strongly nonlinear behavior and voltage dependence of the feedback-induced currents.
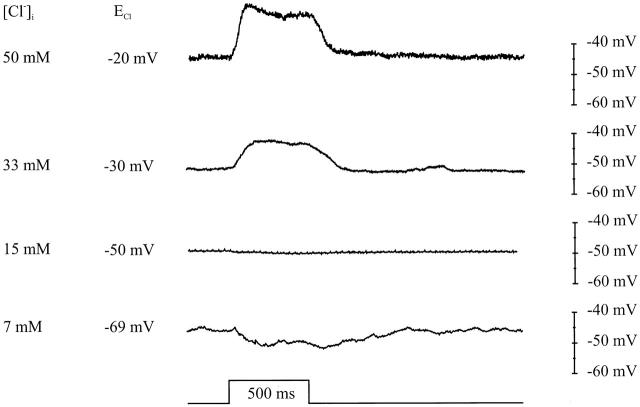
Fig. 1 (top) shows the surround-induced response of a cone with a [Cl]i of 50 mM. In this condition, surround stimulation induces a large depolarizing response. Fig. 1 (bottom) shows the response of a cone with [Cl]i of 7 mM. In that condition, the surround-induced response is a small hyperpolarizing response. If this response was generated by a residual light sensitivity of the cone, the response should become larger with hyperpolarization, but at −60 mV surround-induced responses were absent (not shown), indicating that these hyperpolarizing responses were due to feedback. The two traces in between show the surround-induced responses of a cone with intermediate [Cl]i. This figure illustrates that surround stimulation of cones with high [Cl]i evokes depolarizing responses, whereas hyperpolarizing responses are generated in cones with low [Cl]i. With [Cl]i of 15 mM, surround stimulation induces no polarization. This [Cl]i yields a calculated ECl of −50 mV.
Figure 1.
Voltage light responses of cones, with various [Cl]i, to 550-nm, 500-ms flashes of 3,000-μm surround fields. Hyperpolarizing responses are found with ECl above −50 mV and depolarizing responses are found with ECl below −50 mV. The scaling, timing, [Cl]i, and calculated ECl are indicated in the figure.
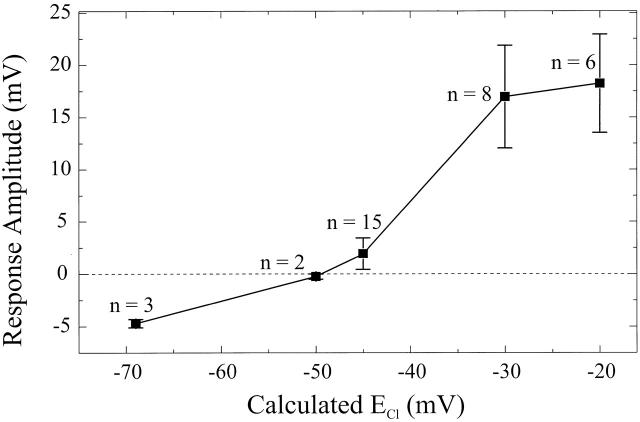
Fig. 2 shows the mean amplitude of the surround responses of 34 cones as function of ECl. As can be seen, the response amplitude becomes zero with ECl around −50 mV, which is slightly more negative than the mean resting membrane potential, which was −46.3 ± 8.3 mV (mean ± SD), under these experimental conditions. At ECl values more negative than −50 mV the surround responses are hyperpolarizing, and at ECl values more positive than −50 mV the surround responses are depolarizing. This shows that the amplitude and polarity of the surround-induced responses varies with ECl in a manner suggesting that these responses are mediated mainly by chloride ions.
Figure 2.
Mean sustained response amplitudes of surround-induced light responses in cones as a function of the calculated ECl. The error bars indicate the SEM. The number of cells used for each data point is indicated near each data point.
On some particular occasions, surround-induced responses can become regenerative. Fig. 3 shows the surround-induced responses of a cone with ECl at −20 mV. Sometimes the cone membrane potential did not hyperpolarize after the surround stimulus was turned off (arrow). In this continued depolarized condition, surround stimulation did not induce a significant further depolarization (star). Similar results are obtained in 14 cones as long as ECl is more positive than −30 mV, indicating that in those conditions surround simulation can trigger a regenerative process, which depolarizes the cones to the reversal potential of the current underlying the surround-induced response; i.e., the Cl current. These spontaneous depolarizing responses were never found immediately after achieving whole cell configuration, indicating that under physiological conditions ECl is more negative than −30 mV. It has been suggested that a possible source of this regenerative behavior of the surround-induced responses is the combined activity of ICa and ICl(Ca) (Thoreson and Burkhardt 1991; Barnes and Deschenes 1992). To test this, the currents induced by surround stimulation were studied.
Figure 3.
Surround-induced depolarizing responses in a cone with [Cl]i = 50 mM. Occasionally the cone depolarized spontaneously or remained depolarized for a long time (arrow). In this depolarized condition, only small surround-induced responses could be measured (*). The scaling and surround stimulus are indicated in the figure.
Feedback-induced Current in Cones
Surround stimulation induces an initial inward current, usually followed by a slowly developing current. The I-V relation of a cone without and with the full-field stimulus is shown in Fig. 4 A. Surround stimulation only induces significant changes in the I-V relation in the range between −20 and −60 mV. Previously it was shown that this change in I-V relation is due to a shift in the activation function of the calcium current to more negative potentials (Verweij et al. 1996). In our experimental conditions, the surround-induced ICa was ∼10 pA at −46 mV, while the light-modulated current was ∼100 pA, showing that, at that potential, the feedback-induced changes in ICa are relatively small. The initial surround-induced current triggers a slowly developing current. The sign of this slowly developing current depends strongly on ECl. This current is outward when ECl is set at −40 mV (Fig. 4 B) and inward when ECl is set at −20 mV (Fig. 4 C). 100 μM niflumic acid, a blocker of the ICl(Ca), blocks the slowly developing current (Fig. 4 D), while the initial inward current remained.
Figure 4.
(A) Cone I-V relations with and without surround stimulation. The cone was held at −77 mV and ramped in 500 ms to −7 mV. The I-V relation depicted with the solid line was determined when the cone was illuminated with a 20 μm saturating white spot and the I-V curve depicted with the dotted line was determined when the cone was illuminated with a 20-μm saturating white spot plus a 3,000 μm white spot stimulating the cone surround. (B) Current traces of a voltage clamped cone, clamped at −58 mV and stepped for 2,000 ms to −43, −38, and −33 mV. During this protocol, the cone was continuously saturated with an intense white spot, while a 3,000-μm full field of 550 nm was illuminating the cone surrounding for 500 ms. In this cone, the [Cl]i was 22 mM, which results in a calculated ECl of −40 mV. Surround stimulation induced an inward current, followed by a secondary slowly developing outward current above −38 mV. (C) Current traces of a voltage-clamped cone, clamped at −58 mV and stepped for 2,000 ms to −43, −38, and −33 mV. During this protocol, the cone was continuously saturated with an intense white spot, while a 3000-μm full field of 550 nm was illuminating the cone surrounding for 500 ms. In this cone, the [Cl]i was 50 mM, which results in a calculated ECl of −20 mV. Surround stimulation induced an inward current, followed by a secondary slowly developing inward current above −40 mV. (D) Current traces of a voltage-clamped cone, clamped at −58 mV and stepped for 2, 000 ms to −43, −38, and −33 mV. During this protocol, the cone was continuously saturated with an intense white spot, while a 3000-μm full field of 550 nm was illuminating the cone surrounding for 500 ms. In this cone, the [Cl]i was 22 mM, which results in a calculated ECl of −40 mV, and 100 μM niflumic acid was added to the Ringers solution. Surround stimulation induced only an initial inward current, without a slowly developing secondary current. The scaling and timing are shown in the figure.
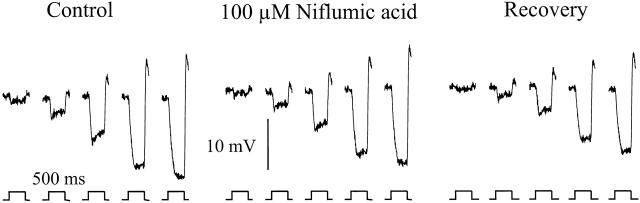
To exclude that niflumic acid might have affected the retinal network by affecting calcium and/or potassium channels in a nonspecific way, we tested whether the synaptic transmission between cones and HCs was still intact. Fig. 5 shows the responses of a monophasic horizontal cell (MHC) in the isolated goldfish retina to full field white light stimuli of various intensities before (left), during (middle), and after (right) application of 100 μM niflumic acid. Fig. 5 shows that, although the surround-induced slowly developing current in cones is blocked by niflumic acid, the MHC resting membrane potential and the MHC light responses are hardly affected (n = 4).
Figure 5.
Monophasic horizontal cell responses to 500 ms lasting full-field white light stimuli of five intensities. (Left) Responses in control Ringers solution before application of niflumic acid. (Middle) Responses are recorded in a 100-μM niflumic acid containing Ringers solution. (Right) Responses again in control Ringers solution after application of niflumic acid.
Summarizing: surround stimulation generates a fast inward current (ICa) and, secondary to this, a slowly developing current whose presence depends on the activation of the calcium current. Furthermore, this secondary current can be blocked by niflumic acid and its sign depends on [Cl]i, features that are characteristic for ICl(Ca). Thus the results presented so far suggest that the surround-induced voltage responses of cones are carried by ICl(Ca). If this is indeed the case, then it should be possible to block the surround-induced voltage responses by niflumic acid. Fig. 6 shows the surround-induced voltage responses of a cone with ECl at −30 mV before (left) during (middle), and after (right) application of 100 μM niflumic acid. Niflumic acid had no large effect on the resting membrane potential of the cones, indicating that ICl(Ca) is only slightly activated at physiological membrane potentials. Before application of niflumic acid the cone depolarized ∼15 mV in response to surround stimulation. This response could be blocked completely by niflumic acid and recovered slightly. Similar results were obtained in all nine cells tested this way.
Figure 6.
Surround-induced light responses of current-clamped cones to 500-ms flashes of a 3,000-μm surround field (ECl = −20 mV). In control conditions (left), surround stimulation induced depolarizing responses, in 100 μM niflumic acid these responses were absent (middle), and after wash the depolarizing responses recovered slightly (right).
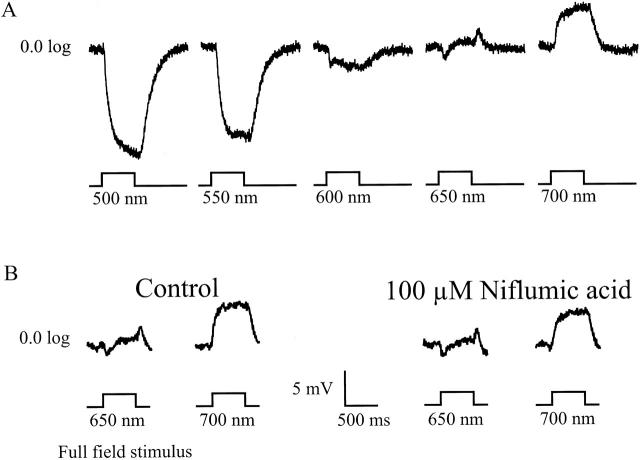
The results presented so far show that niflumic acid can effectively block the surround-induced depolarizations in cones, without affecting the feedback-induced modulation of ICa. The question now arising is whether the cone depolarization or the modulation of ICa is most important for feedback-induced response in second-order neurons, such as the biphasic horizontal cells (BHCs). The depolarizing responses of the BHCs, due to red light stimulation, are thought to be generated by feedback from the MHCs to the middle wavelength–sensitive cones (Fuortes and Simon 1974; Stell et al. 1975; Stell 1976; Kamermans et al. 1991). Fig. 7 A shows the responses of a BHC to flashes of 500 ms with wavelength ranging from 500 to 700 nm in 50-nm steps. The neutral point, the wavelength where the hyperpolarizing response changes into a depolarizing response, is close to 650 nm. If niflumic acid blocks feedback, then the depolarizing response to 700 nm should be blocked and the neutral point should shift to longer wavelength. Fig. 7 B shows the responses to 650 and 700 nm before and during niflumic acid application. As is clear from this figure, niflumic acid does not block the depolarizing light responses and did not shift the neutral point of the BHC. Although minor changes in the response amplitude were seen occasionally, the feedback-induced responses in BHCs were never blocked in all cells tested (n = 5), whereas the feedback-induced depolarizations in cones were always blocked completely. This experiment shows that depolarization of the cones is not essential for the transmission of a feedback response to second-order neurons.
Figure 7.
(A) Light responses of a biphasic horizontal cell, in normal Ringers solution, to 500-ms lasting full-field light stimuli with wavelengths ranging from 500 to 700 nm in 50-nm steps. (B) Biphasic horizontal cell responses to 500-ms lasting full-field light stimuli of 600, 650, and 700 nm. (Left) Depolarizing light responses in control Ringers solution. (Right) Depolarizing light responses recorded in a 100-μM niflumic acid containing Ringers solution.
Estimation of the Cl Equilibrium Potential under Physiological Conditions
So far we have shown: (a) that the size and sign of the surround-induced responses in cones depend on [Cl]i, (b) that these responses are carried by ICl(Ca), and (c) that the feedback-induced depolarizing responses in cones are not essential for depolarizing responses in BHCs. To quantify the role of the ICl(Ca) in this synapse further, it is essential to know the value of ECl under physiological conditions. With ECl more positive than the resting membrane potential, cones will depolarize due to surround stimulation. This depolarization might amplify the effect of the feedback signal received by the cones on the cone output. On the other hand with ECl more negative than the resting membrane potential, cones will hyperpolarize due to surround stimulation, which might lead to a reduction of the effect of the feedback signal received by the cones on the second-order neurons.
In voltage-clamp experiments, the properties of ICl(Ca) can be examined rather easily since ICl(Ca) has three distinct properties: (a) it is a slowly developing current that depends on ECl, (b) it can generate slow tail currents, and (c) it can be blocked by niflumic acid (Barnes and Deschenes 1992). In Fig. 8, the current traces of two voltage-clamped cones are shown. These cones were clamped at −77 mV and stepped for 2,000 ms to −25, −15, and −5 mV. In Fig. 8, left, ECl was −50 mV. Stepping the cone membrane potential to the depolarized potentials yields slowly developing outward currents, preceded by an inward current. Small tail currents are visible when the membrane potential is hyperpolarized back to −77 mV. Similar results were found in 20 cones. With ECl at −20 mV (middle), the properties of the slowly developing current change strongly. Now stepping from −77 to −25 mV yields a slowly developing inward current and large tail current after returning to −77 mV. This was found in all 15 cones tested in this way. This slowly developing current could be blocked by 100 μM niflumic acid (right). Note that in this condition both the slowly developing current and the tail currents are absent except for a small transient tail current in the first 50 ms. All four cells tested this way behaved similarly. These experiments show that goldfish cones have a slowly developing current that depends on ECl and can generate tail currents. This current can be used for the estimation of the physiological value of ECl.
Figure 8.
Current traces of two voltage-clamped cones, clamped at −77 mV and stepped for 2,000 ms to −25, −15, and −5 mV. (Left) ECl was −50 mV. Above −25 mV, an outward current is present that generates a tail current. (Middle) ECl was −20 mV. In this condition, at −25 mV, a slowly activating inward current developed. At −15 mV, this current reverses and becomes outward and generates large tail currents. (Right) Current traces of the same cone are presented, but now with 100 μM niflumic acid added to the Ringer's solution. Both the slowly developing current and tail currents are completely blocked with niflumic acid.
The rationale for the next set of experiments is that, immediately after achieving whole cell configuration, the pipette solution will not yet have diffused into the cell and at that moment ECl will still have a value close to the physiological value, whereas after 20 min the cell interior has been perfused with the pipette solution and thus ECl in the cones will have the value of ECl in the pipette. As shown in Fig. 8, the sign of ICl(Ca) depends strongly on the value of ECl, and thus could be used to estimate the physiological ECl. When ICl(Ca) is outward, ECl is more negative than the clamp potential, and when ICl(Ca) is inward, ECl is more positive than the clamp potential. Due to the nature of ICl(Ca), this method is limited to conditions where the intracellular calcium concentration ([Ca]i) is sufficiently high to activate ICl(Ca). This happens at potentials above −40 mV, which means that we can only determine ECl exactly when it is more positive than −40 mV.
Fig. 9 shows the results of two experiments in which the effect of [Cl]i on the sign of ICl(Ca) was studied as a function of time. Since the size of ICl(Ca) varies between the various cones, the cells may not be compared with each other, but changes in ICl(Ca) with time in one individual cell can be used to estimate ECl. Fig. 9 (top) shows the current traces of a cone clamped at various potentials with ECl at −50 mV at 2 and 22 min after achieving whole-cell configuration. In this condition, at both moments in time, a slowly developing outward current can be seen at potentials of −40 mV and above. In both conditions, small tail currents are present. The overall response to the voltage steps does not seem to change dramatically over time. This behavior was observed in all 29 cells tested this way. With ECl at −20 mV, on the other hand, the responses to the same voltage steps strongly change with time (Fig. 9, bottom). At 2 min after achieving whole-cell configuration, only a very small slowly developing outward current can be observed and the tail currents are very small. However, at 21 min, a voltage step to −40 mV induces a large slowly developing inward current and returning back to −75 mV generates a very large tail current. This indicates that ECl has changed strongly during the 20-min whole cell configuration. In all 55 cones tested directly after achieving whole cell configuration, voltage steps to −40 mV never induced a slowly developing inward current and mostly induced a slowly developing outward current. These results indicate that the physiological ECl is about or more negative than −40 mV.
Figure 9.
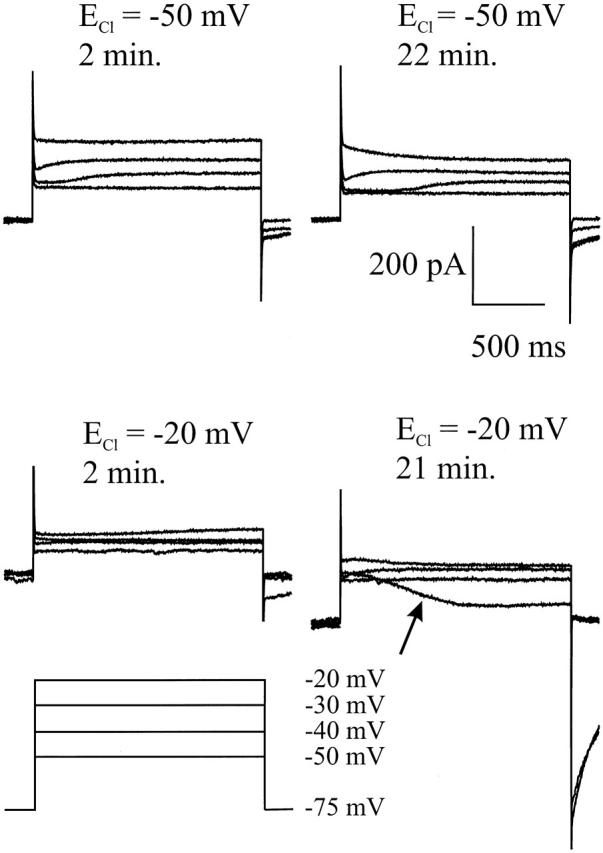
Time dependence of ICl(Ca) in cones with two different [Cl]i. The cones were held at −75 mV and stepped to −50, −40, −30, and −20 mV. For both values of ECl, the current responses are given at 2 min and >20 min after whole-cell configuration was achieved. With ECl at −20 mV, the slowly developing current became inward after an ∼21-min whole-cell configuration (arrow), while it remained outward when ECl was −50 mV.
Since this method to estimate ECl has a limited resolution and works only in the range where ICl(Ca) is activated, a second method for the determination of ECl is needed. Therefore, a second estimate of ECl was made using the tail currents. The experiments of Fig. 8 show that the tail currents can be almost completely blocked by 100 μM niflumic acid, indicating that these currents are carried by ICl(Ca). Fig. 10 shows the current traces of a cone filled with a 33 mM Cl-containing pipette solution 8 min after the whole-cell configuration was achieved. The cone was clamped at −77 mV, stepped to −20 mV for 500 ms and successively clamped for 400 ms to various potentials more negative than −50 mV. The step to −20 mV activates ICa and ICl(Ca), and the subsequent step to negative potentials induces the ICl(Ca)-dependent tail current. For the estimation of the reversal potential of the tail current, no potentials above −50 mV were used because at these potentials the activation of the calcium current interferes with the measurements. To correct for a possible remaining leak current that might be present in the potential range between −100 and −50 mV, the difference between the tail current measured 50 and 300 ms after the step from −20 mV to the negative potentials (gray bars), was used to determine the reversal potential of the tail current. In this way, an estimate of the size of the tail current was obtained for various potentials. In the insert, this estimate is plotted as a function of the potential. The dotted line is a linear curve fitted through the data points using the linear regression algorithm. The intersection with the x axis gives the reversal potential of the tail currents. Since the tail currents are mainly carried by Cl, this value is an estimate of ECl.
Figure 10.

(A) An example of a tail current measurement, 8 min after achieving whole-cell configuration. The cone was recorded with a 33-mM Cl−-containing patch pipette, clamped at −77 mV, stepped to −20 mV for 500 ms, and successively clamped for 400 ms to various potentials. The step to −20 mV activates the ICl(Ca), and the step to the various potentials induces the Ca-dependent tail current. For the estimation of the reversal potential of the tail current, no potentials above −50 mV were used, because at these potentials the activation of the Ca current interferes with the measurements. The difference between the mean current for 50 ms, measured 50 and 300 ms after the step from −20 mV to the various clamp potentials, ranging from −100 up to −50 mV, was used to determine ECl by assuming that the tail current reverses at ECl; i.e., in that condition the difference between the mean current at 50 and 300 ms is zero. (B) Estimation of ECl using the tail-current measurements. The measured ECl's are shown for 11 cells with [Cl] in the pipette (33 mM, •) and for 2 cells with [Cl] in the pipette (7 mM, ○), measured at various moments after achieving the whole-cell configuration. At 2 min, the estimated ECl yields values of approximately ∼55 mV for both values of [Cl]. For the low [Cl], ECl becomes more negative with time, and, for the high [Cl], ECl becomes more positive with time.
Fig. 10 B shows the estimates of ECl for 11 cells with the [Cl] in the pipette being 33 mM (•) and 2 cells with [Cl] in the pipette being 7 mM (○) as function of time. At 2 min, the estimate yields values of −55 ± 7 mV (n = 5). This value is independent of the [Cl] in the pipette. For low [Cl] (○), ECl does not change very much, whereas for high [Cl] (•), ECl becomes −33 ± 4 mV (n = 5) after 30 min and approaches its calculated ECl value of −30 mV. This experiment shows that the physiological value of ECl is approximately −55 mV and that ECl shifts in time with the [Cl] in the pipette. With ECl around −55 mV, surround stimulation will not induce a substantial polarization of the cone membrane potential, as indicated in Fig. 1.
DISCUSSION
In this paper we have shown that: (a) surround-induced polarizations in cones are due to the activation of ICa and ICl(Ca), (b) ECl is approximately −55 mV under physiological conditions, and (c) the effect of negative feedback from HCs to cones on second-order neurons is still present even when the cones do not depolarize during surround stimulation.
The Feedback Pathway from Horizontal Cells to Cones
Surround stimulation hyperpolarizes the HCs, which in turn shifts the ICa activation function in cones to more negative potentials (Verweij et al. 1996). This shift will increase the Ca-influx leading to an increase in [Ca]i and finally to activation of ICl(Ca). Depending on ECl, this will lead to either a hyperpolarizing or depolarizing response of the cones. Since HCs feed back to cones negatively, one could argue that cones should depolarize to surround stimulation. Therefore, ECl should be more positive than the resting membrane potential. Indeed, for turtle cones, some evidence exists that ECl might be more positive than the resting membrane potential (Thoreson and Burkhardt 1991). However, others found in turtle that ECl is more negative than the resting membrane potential (Tachibana and Kaneko 1984; Kaneko and Tachibana 1986). This negative value of ECl was also found in other species (Miller and Dacheux 1983; Wu 1986) and is in complete agreement with the findings presented in this paper. The question addressed in this paper is whether cone polarizations are essential for feedback-induced responses in second-order neurons. We showed that, when the modulation of ICl(Ca) in cones is blocked by niflumic acid, negative feedback-mediated responses in second-order neurons are still present. This means that the polarization of the cone by negative feedback is of lesser importance than the direct modulation of the Ca current.
This makes the feedback synapse from HCs to cones a very unique synapse. Polarization of the presynaptic cell (HC) leads to Ca influx in the postsynaptic cell (cone). Since the Ca conductance is small relative to the total conductance of the cone, modulation of ICa will hardly polarize the cone. Moreover, the depolarizing effect of ICa on the cone membrane potential will be counteracted by the activation of ICl(Ca), which tend to hyperpolarize the cone. As a result, the membrane potential of the postsynaptic cell is hardly modulated, whereas the output of the postsynaptic cell is strongly modulated. This mechanism of synaptic transmission is restricted to one synaptic terminal and does not integrate the input to the whole neuron. It is an open but very intriguing question whether such synapses are also present in the central nervous system.
Consequences for Surround Responses in Bipolar Cells
The consequence of this type of synaptic transmission is that, in the cones, the information about the stimulus within its receptive field is visible in the membrane potential, whereas the information about the stimulus outside the receptive field of the cones, but inside the receptive field of the HCs expresses itself mainly in changes in [Ca]i in the synaptic terminal of the cone, without affecting the cone membrane potential. Consistent with this finding is the work of Hare and Owen 1996, who studied the effect of the GABA analogue d-aminovaleric acid (AVA) on the surround responses of BCs in tiger salamander. They found that AVA blocked the surround responses of BCs without any obvious effect on the cone membrane potential. This result could mean two things: (a) HCs feed directly into BCs via an AVA-sensitive pathway or (b) feedback from HCs to cones does not modulate the cone membrane potential (Hare and Owen 1996). In view of the results presented in this paper, the later interpretation seems more likely.
Taken together, it seems that the polarization of the cone membrane potential is not a good estimate of the output of the cones. Therefore, determining the transfer function of this synapse by describing the postsynaptic potential changes as a function of the presynaptic potential will not yield the function governing the complete signal flow between cones and HCs. This calls for a re-evaluation of the data concerning the transfer functions of cones to HCs and BCs.
Functional Role of the Ca-dependent Cl Current
The results presented in this paper indicate that ICl(Ca) can have a large effect on the cone membrane potential when ECl is more positive than −30 mV. However, under physiological conditions, ECl was estimated to be about −55 mV. As indicated by Fig. 1, with ECl at this potential, surround stimulation hardly modulates the cone membrane potential. Furthermore, the niflumic acid experiments have shown that blocking ICl(Ca) hardly affects the cone and HC resting membrane potential or the cone and HC light responses. This illustrates that in the dark ICl(Ca) is hardly activated. Since light stimulation will hyperpolarize the cones, this suggests that ICl(Ca) will never be activated substantially. This seems counterintuitive because in the dark the Ca influx is large. However, there are conditions in which ICl(Ca) could be activated. First one has to realize that, although the Ca-influx is large, in the dark it is not maximal. Depolarizing a cone further than the dark membrane potential will substantially increase the Ca influx and might activate ICl(Ca). Such depolarizations might occur at light offset when cones can generate depolarizing light-off transients. In that condition, ICl(Ca) might be activated and will yield a hyperpolarizing influence on the cone membrane potential that terminates the depolarizing light-off transients. Therefore, the role of ICl(Ca) seems to be to prevent the cone from large depolarizations.
Another condition in which ICl(Ca) could be activated is when only the surround of a cone is stimulated with, for instance, an annular stimulus. In that condition, the central cone will remain at its dark-membrane potential, but the activation function of ICa will be shifted to more negative potentials. This will induce a larger Ca influx that might become large enough to activate ICl(Ca). For this cone, the increased ICa tends to depolarize the cone, whereas the increased ICl(Ca) has the opposite effect on the cone. The overall result might be that the cone membrane potential in that condition is hardly modulated by feedback, whereas the glutamate release is increased. The functional consequence is that the driving force for the modulation of the cGMP-gated channel in the outer segment is not affected by negative feedback, which makes the gain of the light response of the cones independent of surround stimulation. Similar effects can be found in the spectral domain as well. For instance, short wavelength–sensitive cones when stimulated with a red background do not respond to that background, but will receive a feedback signal from the HCs (Kraaij et al. 1998). Also, in this condition the activation function of ICa in the cones shifts to more negative potentials, leading to an increase in the Ca influx, which tend to depolarize the cone, whereas the activation of ICl(Ca) counteracts this depolarization.
In turtle, on the other hand, it has been suggested that ECl is more positive than the dark resting membrane potential. Although this a controversial point that is not settled at all, next the effect of a positive value of ECl on the feedback pathway will be discussed. When ECl is positive, activation of ICl(Ca) will depolarize the cones, which in turn activates the voltage-dependent Ca channels and amplify the feedback signal in the cones. This makes [Cl]i, at least in these species, an effective tool to modulate the efficiency of the feedback signal. In this respect, it would be very interesting to see whether ECl in goldfish cones can vary with, for instance, the adaptational state of the retina.
Possible Other Cl-dependent Processes in the Cone Terminal Involved in Feedback
Niflumic acid is known to be a rather nonspecific ICl(Ca) blocker. Could this nonspecific action of niflumic acid have interfered with our analysis? A direct action of niflumic acid on ICa can be ruled out, because the synaptic transmission between cones and HCs remains intact during niflumic acid application, as shown in Fig. 7. Various reports have shown that GABA-gated Cl channels are located in the cones (Tachibana and Kaneko 1984; Kaneko and Tachibana 1986; Yazulla et al. 1989). Can our results be explained by the assumption that niflumic acid blocks the GABA-gated Cl channels in cones? It has been shown that surround stimulation did not modulate the GABA-gated chloride channels in cones (Verweij et al. 1996). Since GABA is not involved in the generation of the surround-induced depolarizing responses in cones, a possible block of the GABA-gated channels by niflumic acid cannot account for the present results.
Another possible mechanism by which feedback could activate a chloride current is by activating the glutamate transporter, which is associated with a chloride conductance (Eliasof and Werblin 1993; Picaud et al. 1995; Arriza et al. 1997). Because glutamate is released in a Ca-dependent manner, an increase of ICa in cones would increase the release of glutamate. Picaud et al. 1995 showed that the glutamate released by the cones can increase the Cl conductance of the glutamate transporters in that cone. If the surround-induced Cl current is mediated by the glutamate transporter, then niflumic acid should block the glutamate transporter. This is not the case because niflumic acid did not affect the HC responses significantly, whereas blocking the transporter with dihydrokainate or dl-threo-β-hydroxyaspartate induces a considerable change in HC response dynamics (Eliasof and Werblin 1993; Vandenbranden et al. 1996).
In summary, we conclude that the underlying current for the surround-induced depolarizing responses is most likely ICl(Ca). We based this conclusion on mainly four findings: (a) the surround-induced currents depend strongly on ECl, (b) these Cl currents only activate after sufficient activation of the Ca current, (c) these currents generate slow tail currents, and (d) the slowly developing surround-induced currents in cones can be blocked by niflumic acid. Finally, in the literature, a number of papers appeared that show that in various species cones have an ICl(Ca) with similar properties as shown in this paper (Maricq and Korenbrot 1988; Barnes and Hille 1989; Barnes and Deschenes 1992; Yagi and MacLeish 1994).
Depolarizing Cone Responses Due to Full-Field Stimulation
Sometimes, cones also depolarize during full-field stimulation (Baylor and Fuortes 1970; Lasansky 1981; Burkhardt et al. 1988; Piccolino 1995). The responses were mostly recorded with KCl- or KAc-filled microelectrodes and are identified as responses generated via feedback from HCs to cones. These results are confusing because full-field stimulation will hyperpolarize all cones, and consequently all HCs should hyperpolarize. The depolarizing responses of cones during full-field stimulation would imply that the effect of the feedback signal on the cone membrane potential has become stronger than the initial cone response. This could not be true for all the cones, because under such a condition the HCs would not remain hyperpolarized. To account for these results, one has to consider the effect of Cl− on the various response components. The depolarizing responses of cones due to full-field stimulation were mostly recorded with Cl-containing electrodes. In this condition, the feedback responses in the cones are enhanced due to the positive value of ECl. Furthermore, we found that the direct light responses of the cones reduce over time with ECl more positive than −30 mV, suggesting that high Cl− in the cones inhibits the phototransduction pathway. Similar observations have been made by Lasansky 1981 with KCl-filled electrodes and by Attwell et al. 1984 with KAc-filled electrodes. In conclusion, the depolarizing responses of cones due to full-field stimulation seems to be due to artificially high anion concentrations in the cones recorded from, which enhances the feedback-induced membrane potential changes and reduces the direct-light responses.
Conclusion
The sequence of events during surround stimulation can be summarized as follows. Surround stimulation leads to hyperpolarization of HCs, which shifts the Ca current-activation function of the cones to more negative potentials. This induces a Ca influx, which leads to an increase in glutamate release. Since the Ca conductance is relatively small compared with the total membrane conductance of the cone, this Ca influx will only slightly depolarize the cone. Under physiological conditions, this modulation of the Ca influx will be too small to activate ICl(Ca) substantially. The result is that the cone membrane potential is hardly modulated by feedback, while the cone's glutamate release is strongly modulated by feedback. Thus, feedback-mediated responses such as the depolarizing responses in the BHC, due to red light stimulation and the surround responses of BCs, remain present in the absence of depolarizing cone responses.
Acknowledgments
This work was supported by the Human Frontier Science Program (Private Investigator, H. Spekreijse) and the Netherlands Organization for Scientific Research (NWO).
Footnotes
Abbreviations used in this paper: BC, bipolar cell; BHC, biphasic horizontal cell; ECl, chloride equilibrium potential; GABA, γ-aminobutyric acid; HC, horizontal cell; MHC, monophasic horizontal cell.
References
- Arriza J.L., Eliasof S., Kavanaugh M.P., Amara S.G. Excitatory amino acid transporter 5, a retinal glutamate transporter coupled to a chloride conductance. Proc. Natl. Acad. Sci. USA. 1997;94:4155–4160. doi: 10.1073/pnas.94.8.4155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Attwell D., Wilson M., Wu S.M. A quantitative analysis of interactions between photoreceptors in the salamander (Ambystoma) retina. J. Physiol. 1984;352:703–737. doi: 10.1113/jphysiol.1984.sp015318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes S., Deschenes M.C. Contribution of Ca and Ca-activated Cl channels to regenerative depolarization and membrane bistability of cone photoreceptors. J. Neurophysiol. 1992;68:745–755. doi: 10.1152/jn.1992.68.3.745. [DOI] [PubMed] [Google Scholar]
- Barnes S., Hille B. Ionic channels of the inner segment of tiger salamander cone photoreceptors. J. Gen. Physiol. 1989;94:719–743. doi: 10.1085/jgp.94.4.719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baylor D.A., Fuortes M.G.F. Electrical responses of single cones in the retina of turtle. J. Physiol. 1970;207:77–92. doi: 10.1113/jphysiol.1970.sp009049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baylor D.A., Fuortes M.G.F., O'Bryan P.M. Receptive fields of cones in the retina of the turtle. J. Physiol. 1971;214:265–294. doi: 10.1113/jphysiol.1971.sp009432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burkhardt D.A., Gottesman J., Thoreson W.B. Prolonged depolarization in turtle cones evoked by current injection and stimulation of the receptive field surround. J. Physiol. 1988;407:329–348. doi: 10.1113/jphysiol.1988.sp017418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eliasof S., Werblin F.S. Characterization of the glutamate transport in the retinal cones of the tiger salamander. J. Neurosci. 1993;13:402–411. doi: 10.1523/JNEUROSCI.13-01-00402.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuortes M.G.F., Simon E.J. Interactions leading to horizontal cell responses in the turtle retina. J. Physiol. 1974;240:177–198. doi: 10.1113/jphysiol.1974.sp010606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hare W.A., Owen W.G. Receptive field of the retinal bipolar cella pharmacological study in the tiger salamander. J. Neurophysiol. 1996;76:2005–2019. doi: 10.1152/jn.1996.76.3.2005. [DOI] [PubMed] [Google Scholar]
- Kamermans M., Van Dijk B.W., Spekreijse H. Color opponency in cone-driven horizontal cells in carp retina. Aspecific pathways between cones and horizontal cells. J. Gen. Physiol. 1991;97:819–843. doi: 10.1085/jgp.97.4.819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaneko A. Physiological and morphological identification of horizontal, bipolar and amacrine cells in goldfish retina. J. Physiol. 1970;207:623–633. doi: 10.1113/jphysiol.1970.sp009084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaneko A., Tachibana M. Retinal bipolar cells with double colour-opponent receptive fields. Nature. 1981;293:220–222. doi: 10.1038/293220a0. [DOI] [PubMed] [Google Scholar]
- Kaneko A., Tachibana M. Effects of gamma-aminobutyric acid on isolated cone photoreceptors of the turtle retina. J. Physiol. 1986;373:443–461. doi: 10.1113/jphysiol.1986.sp016057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kraaij D.A., Kamermans M., Spekreijse H. Spectral sensitivity of the feedback signal from horizontal cells to cones in goldfish retina. Vis. Neurosci. 1998;15:799–808. doi: 10.1017/s0952523898154184. [DOI] [PubMed] [Google Scholar]
- Lasansky A. Synaptic action mediating cone responses to annular illumination in the retina of the larval tiger salamander. J. Physiol. 1981;310:205–214. doi: 10.1113/jphysiol.1981.sp013544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maricq A.V., Korenbrot J.I. Calcium and calcium-dependent chloride currents generate action potentials in solitary cone photoreceptors. Neuron. 1988;1:503–515. doi: 10.1016/0896-6273(88)90181-x. [DOI] [PubMed] [Google Scholar]
- Miller R.F., Dacheux R.F. Intracellular chloride in retinal neuronsmeasurement and meaning. Vision Res. 1983;23:399–411. doi: 10.1016/0042-6989(83)90087-1. [DOI] [PubMed] [Google Scholar]
- Naka K.I., Rushton W.A.H. The generation and spread of S-potentials in fish (cyprinidae) J. Physiol. 1967;192:437–461. doi: 10.1113/jphysiol.1967.sp008308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norton A.L., Spekreijse H., Wolbarsht M.L., Wagner H.G. Receptive field organization of the S-potential. Science. 1968;160:1021–1022. doi: 10.1126/science.160.3831.1021. [DOI] [PubMed] [Google Scholar]
- O'Bryan P.M. Properties of the depolarizing synaptic potential evoked by peripheral illumination in cones of the turtle retina. J. Physiol. 1973;235:207–223. doi: 10.1113/jphysiol.1973.sp010385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picaud S.A., Larsson H.P., Wellis D.P., Lecar H., Werblin F.S. Cone photoreceptors respond to their own glutamate release in the tiger salamander. Proc. Natl. Acad. Sci. USA. 1995;92:9417–9421. doi: 10.1073/pnas.92.20.9417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piccolino M. The feedback synapse from horizontal cells to cone photoreceptors in the vertebrate retina. Prog. Retin. Eye Res. 1995;14:141–196. [Google Scholar]
- Stell W.K. Functional polarization of horizontal cell dendrites in goldfish retina. Invest. Ophthalmol. 1976;15:895–908. [Google Scholar]
- Stell W.K., Lightfoot D.O. Colour-specific interconnections of cones and horizontal cells in the retina of the goldfish. J. Comp. Neurol. 1975;159:473–502. doi: 10.1002/cne.901590404. [DOI] [PubMed] [Google Scholar]
- Stell W.K., Lightfoot D.O., Wheeler T.G., Leeper H.F. Goldfish retinafunctional polarization of cone horizontal cell dendrites and synapses. Science. 1975;190:989–990. doi: 10.1126/science.1188380. [DOI] [PubMed] [Google Scholar]
- Tachibana M., Kaneko A. Gamma-aminobutyric acid acts at axon terminals of turtle photoreceptorsdifference in sensitivity among cell types. Proc. Natl. Acad. Sci. USA. 1984;81:7961–7964. doi: 10.1073/pnas.81.24.7961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thoreson W.B., Burkhardt D.A. Ionic influences on the prolonged depolarization of turtle cones in situ. J. Neurophysiol. 1991;65:96–110. doi: 10.1152/jn.1991.65.1.96. [DOI] [PubMed] [Google Scholar]
- Toyoda J.I., Kujiraoka T. Analyses of bipolar cell responses elicited by polarization of horizontal cells. J. Gen. Physiol. 1982;79:131–145. doi: 10.1085/jgp.79.1.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandenbranden C.A.V., Verweij J., Kamermans M., Muller L.J., Ruijter J.M., Vrensen C.F.J.M., Spekreijse H. Diffusion of neurotransmitters in the cone synaptic terminals of goldfish retina. Vision Res. 1996;36:3859–3875. doi: 10.1016/s0042-6989(96)00134-4. [DOI] [PubMed] [Google Scholar]
- Verweij J., Kamermans M., Spekreijse H. Horizontal cells feed back to cones by shifting the cone calcium-current activation range. Vision Res. 1996;36:3943–3953. doi: 10.1016/s0042-6989(96)00142-3. [DOI] [PubMed] [Google Scholar]
- Wu S.M. Effects of gamma-aminobutyric acid on cones and bipolar cells of the tiger salamander retina. Brain Res. 1986;365:70–77. doi: 10.1016/0006-8993(86)90723-7. [DOI] [PubMed] [Google Scholar]
- Yagi T., MacLeish P.R. Ionic conductances of monkey solitary cone inner segments. J. Neurophysiol. 1994;71:656–665. doi: 10.1152/jn.1994.71.2.656. [DOI] [PubMed] [Google Scholar]
- Yazulla S., Studholme K.M., Vitorica J., De Blas A.L. Immunocytochemical localization of GABA a receptors in goldfish and chicken retinas. J. Comp. Neurol. 1989;280:15–26. doi: 10.1002/cne.902800103. [DOI] [PubMed] [Google Scholar]