Abstract
Immunotherapy by using multimerized self-peptides has demonstrated a clear protective effect on experimental models of autoimmune diseases. However, the mechanisms involved remain ill-defined. Here we have evaluated the therapeutic efficacy of multimerized self-peptides at the effector phase of autoimmune diabetes and examined their mechanisms of action. Diabetes was induced in rat insulin promoter-hemagglutinin (HA) mice expressing HA in pancreatic β-cells by adoptive transfer of HA110–119-specific T helper 1 cells. Complete protection was provided by low doses of the HA 4-mer consisting of four covalently linked linear HA107–119 peptides. In vivo, the 4-mer appeared to act directly on the pathogenic HA-specific T helper 1 cells and indirectly by activation/recruitment of lymphocytes with regulatory properties so that mice became resistant to a second transfer of diabetogenic T cells. This effect was associated with a recruitment of Foxp3+ CD4 T cells around islets. Moreover, we show that dominant protection from autoimmunity was transferable by spleen cells, and that development of this regulatory population was crucially dependent on the lymphocytes from treated rat insulin promoter-HA mice. Thus, immunotherapy using multimerized epitopes emerges as a promising strategy in view of the current identification of self-epitopes that are major targets of the pathogenic CD4 T cell response in autoimmune diseases.
Keywords: autoimmunity immune tolerance, immunotherapy, regulatory T cells, Foxp3
The treatment of human autoimmune diseases is particularly challenging. Indeed, diseases such as rheumatoid arthritis, multiple sclerosis, and type 1 diabetes (T1D) are mainly chronic and impact quality of life more than life expectancy. Moreover, treatments are often started long after the onset of the pathogenic process, at a time when the autoimmune recognition has spread to multiple self-antigens (1). In this context, conventional immunosuppression offers only partial benefit at the price of frequent and sometimes serious adverse events. More recently, targeted immunotherapies using antiinflammatory cytokines, antibodies against lymphocyte surface molecules, or neutralization of proinflammatory cytokines or homing receptors have achieved great clinical success. However, by affecting indiscriminately the pathogenic autoimmune process, together with beneficial immune responses, they too can be saddled with serious side effects. An attractive alternative is to use antigen-specific approaches to selectively silence autoreactive T cells (2–5). In animal models, prevention of autoimmune disease can be achieved by i.v., mucosal, or s.c. administration of soluble self-antigen or by vaccination with DNA-encoding self-antigens (3, 5). However, therapeutic results were often inconsistent (2, 3) possibly due to the selective targeting of T cells specific for the injected self-antigen.
Peptides bind MHC class II molecules with both ends extruding from the peptide-binding groove (6). This structural feature provides the opportunity to design linear multimeric peptides that consist of multiple copies of a CD4 T cell epitope connected by spacer sequences (7). This unique structure is designed to form multivalent arrays of peptide:MHC complexes on the surface of the antigen-presenting cells (APCs). The cross-linking of MHC class II molecules and the resulting clustering of peptide:MHC/T cell receptor (TCR) greatly enhance both APC and T cell activation. Therefore, multimerized T cell epitopes increase the immunological potency of soluble peptides and can promote either antigen-specific T cell activation or tolerance through activation-induced cell death in vitro at much lower concentrations than the monomeric peptide (7, 8). Similarly, in vivo immunization with multimerized myelin peptides led to an exacerbated experimental autoimmune disease, whereas the same multimer injected i.v. resulted in inactivation of pathogenic CD4 T cells and dramatic suppression of disease (9, 10). However, despite their great therapeutic potential, little is known about the mechanisms of action of multimerized epitopes in vivo. In the present study, we have evaluated the therapeutic efficacy of multimerized epitopes at the effector phase of a T1D model and assessed their mechanisms of action.
Results
Comparison of the Effects of the 4-Mer and the HA107–119 Peptide on T Cell Recognition.
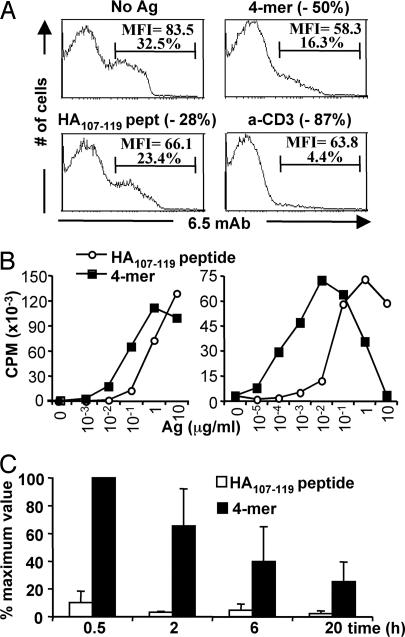
The hemagglutinin (HA) 4-mer used in this study was produced by recombinant techniques and consists of four covalently linked HA107–119 peptides. We first compared the in vitro activity of the 4-mer and the monomeric HA107–119 peptide on HA-specific CD4 T cells from 6.5-TCR transgenic animals. These cells can be specifically labeled with the 6.5 anticlonotypic mAb (11). 6.5+ CD4 T cells exhibited an enhanced TCR down-regulation after in vitro incubation with the 4-mer compared with monomeric HA107–119 peptide (Fig. 1A). Moreover, the half-maximal proliferative response of naïve (Fig. 1B Left) or TH1-differentiated (Fig. 1B Right) 6.5-TCR CD4 T cells was consistently induced by a 10- to 100-fold lower concentration of the 4-mer compared with the monomeric HA107–119 peptide. Furthermore, the 4-mer induced a bell-shaped proliferation curve, suggesting that high concentrations of the multimer favored death and/or anergy of the specific T cells in vitro, as previously documented in other systems (8, 9). An oligomer consisting of 16 copies of the HA107–119 peptide also acted as a superagonist in vitro. Presumably due to aggregation the longer oligomer proved slightly less potent than the 4-mer (data not shown) so that further in vivo investigations were performed by using the 4-mer.
Fig. 1.
Influence of epitope multimerization on T cell responses in vitro and on half-life in vivo. (A) Effect on TCR down-regulation. 6.5 expression on gated CD4 cells from 6.5-TCR mice was analyzed by FACS after 12 h of stimulation with medium alone, monomeric HA107–119 peptide (10 μg/ml), 4-mer (10 μg/ml), or anti-CD3ε mAb (0.5 μg/ml). The decrease in 6.5+ CD4+ T cells compared with the unstimulated cells is shown in parentheses. (B) Effect on the proliferative response of HA-specific CD4 T cells (Left) Spleen and lymph nodes of 6.5-TCR mice were incubated with the indicated concentrations of the HA107–119 peptide or 4-mer, and [3H]thymidine incorporation was measured after 72 h of stimulation. Similar results were obtained with shorter (12, 24, and 48 h) stimulation periods (data not shown). (Right) TH1-differentiated 6.5-TCR CD4 T cells were incubated for 24 h with the indicated concentrations of the HA107–119 peptide or 4-mer and irradiated APCs, and [3H]thymidine uptake was evaluated. Results are from one of two similar experiments. (C) In vivo half-life on splenic APCs of the HA107–119 peptide or 4-mer. Splenocytes from BALB/c mice, injected i.v. with 2.5 μg of the HA107–119 peptide or 4-mer, were harvested at the indicated time points to induce proliferation of purified 6.5-TCR CD4 T cells. Maximum [3H]thymidine uptake (62,885 ± 11,655 cpm) was defined as the stimulation induced by splenocytes from BALB/c mice injected 0.5 h earlier with 4-mer. Results from three experiments are presented as the mean percentage (± SEM) of the maximum value.
We next compared the in vivo half-life of the 4-mer and the HA107–119 peptide on APCs. To this end, BALB/c mice received PBS or 2.5 μg of either 4-mer or HA107–119 peptide, and their splenocytes, harvested at different times postinjection, were used to stimulate purified CD4 T cells from 6.5-TCR mice. Splenocytes from mice injected with the peptide induced minimal proliferation, preventing accurate assessment of the in vivo half-life. In contrast, a strong response was elicited by splenocytes from 4-mer-injected mice, revealing an in vivo half-life of the 4-mer of 4.8 h (Fig. 1C).
Injection of 4-Mer Polypeptide Blocks Autoimmune Diabetes Induced by Effector TH1 Cells.
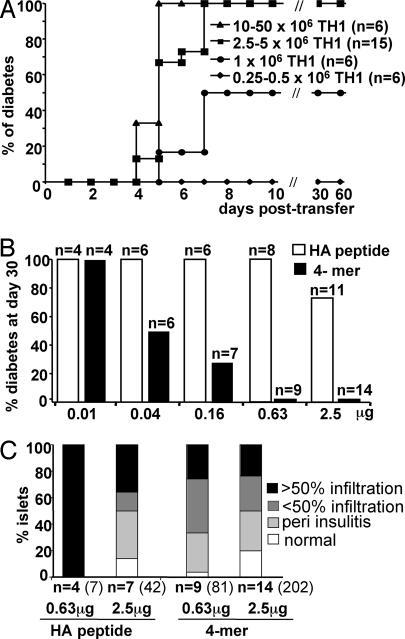
We then tested the ability of the HA 4-mer to induce tolerance to a pancreatic neo-self-antigen in a new T1D model. The model consists of the adoptive transfer of 6.5-TCR TH1 cells into nonirradiated immunocompetent rat insulin promoter (RIP)-HA mice (12). Transfer of 2.5 × 106 TH1 cells or more invariably resulted in diabetes, which proved rapidly fatal (Fig. 2A). To compare the ability of the 4-mer and the HA107–119 peptide to block the effector phase of autoimmune diabetes, increasing doses (0.01–2.5 μg) of either molecule were injected after the adoptive transfer of diabetogenic TH1 cells. Treatment with the HA107–119 peptide resulted in incomplete protection from diabetes only at the highest dose tested (2.5 μg) (P = 0.086). In sharp contrast, treatment with the 4-mer completely protected RIP-HA mice from disease development at the 2.5-μg (P < 0.0001) or 0.63-μg (P < 0.0001) doses (Fig. 2B). Moreover, a partial but significant (P = 0.01) protection from diabetes was observed with as little as 0.04 μg of the 4-mer. The histological analysis of the pancreas showed that the severity of insulitis correlated with the development of disease (Fig. 2C). However, despite full protection from diabetes, pancreatic islets from 4-mer-treated mice still exhibited mononuclear cell infiltration.
Fig. 2.
Injection of 4-mer protects from diabetes induced by transfer of effector 6.5-TCR TH1 cells in RIP-HA mice. (A) Development of the diabetes model. Increasing numbers of 6.5-TCR TH1 cells were injected i.v. into immunocompetent RIP-HA mice, and development of diabetes was plotted against time. (B and C) Protection from diabetes by 4-mer. (B) RIP-HA mice were adoptively transferred with 2.5 × 106 6.5-TCR TH1 cells. The animals were injected with the indicated doses (0.01–2.5 μg) of either the HA107–119 peptide or 4-mer. The frequency of diabetes in each group is shown. All PBS-treated control RIP-HA mice (n = 13) developed diabetes. (C) The extent of insulitis was determined at day 30 as the percentage of normal, periinfiltrated, infiltrated <50%, or infiltrated >50% islets (n = number of mice). Total number of islets is indicated in parentheses. Among the HA107–119 peptide-treated mice, 4 of 8 mice treated with 0.63 μg and 7 of 11 mice treated with 2.5 μg survived and could be analyzed histologically.
Induction of Active Suppression by the 4-Mer Treatment.
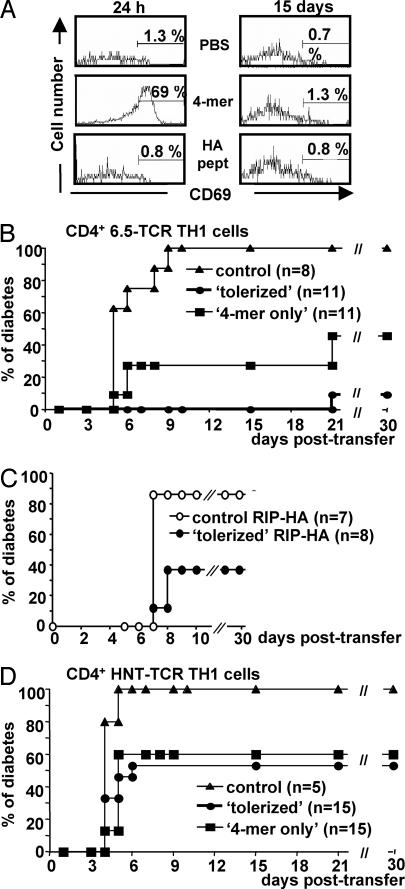
To investigate the mode of action of the 4-mer treatment, we evaluated the duration of 4-mer presentation by host APCs in vivo. RIP-HA mice were first injected with 2.5 μg of 4-mer or HA107–119 peptide, or PBS alone. One or 15 days later, they received CFSE-labeled splenocytes from 6.5-TCR RAG2−/− mice, and CD69 induction was assessed on CFSE+ HA-specific CD4 T cells as an indicator of in vivo priming. Expression of CD69 on HA-specific CD4 T cells was only increased in mice that received the 4-mer 24 h before the cell transfer (Fig. 3A). These results establish that the 4-mer is no longer presented 15 days after its injection.
Fig. 3.
The 4-mer treatment induces dominant tolerance. (A) Duration of 4-mer presentation by APCs in vivo. RIP-HA mice were treated with PBS or with 2.5 μg of HA107–119 peptide or 4-mer. One day (Left) or 15 days (Right) later, mice were injected with 30 × 106 CFSE-labeled 6.5-TCR RAG2−/− splenocytes. The proportion of HA-specific CD4+ CFSE+ T cells expressing CD69 was determined 24 h later in the spleen of recipient mice. (B–D) Dominant tolerance induced by 4-mer. RIP-HA mice received 5 × 106 6.5-TCR TH1 cells and were treated with 2.5 μg of 4-mer. These tolerized mice (filled circles) were challenged 15 days later by a second transfer of 5 × 106 6.5-TCR TH1 cells (HA107–119-specific) (B) or 25 × 106 HNT-TCR TH1 cells (HA126–138-specific) (D). In parallel, adoptive transfer was carried out in 4-mer-only mice that had received only the 4-mer treatment (filled squares). Control RIP-HA recipient mice were left untreated to confirm the diabetogenic potential of each TH1 preparation (filled triangles). (C) RIP-HA mice were transferred with 5 × 107 splenocytes from control unmanipulated RIP-HA mice (empty circles) or tolerized RIP-HA mice (filled circles). The next day, these recipient mice received 5 × 106 6.5-TCR TH1 cells, and the development of diabetes was assessed.
We then challenged protected RIP-HA mice with a second transfer of 6.5-TCR TH1 cells 15 days after the first cell transfer without further injection of the multimer. Ten of 11 tolerized mice were able to control this second wave of diabetogenic TH1 cells (Fig. 3B; P < 0.0001). Moreover, a protective effect was also observed in mice, referred to as the “4-mer-only” group, which initially received only the 4-mer treatment (but no diabetogenic T cells) and were injected 15 days later with 6.5-TCR TH1 cells (Fig. 3B; P = 0.0007). These results suggest that multimer injection can induce dominant tolerance.
To investigate whether protection from diabetes could be transferred to naive animals, splenocytes from unmanipulated or tolerized RIP-HA mice were transferred to a second group of RIP-HA mice. The next day, these recipient mice received 6.5-TCR TH1 diabetogenic cells. Splenocytes from tolerized RIP-HA mice significantly (P = 0.022) protected the recipients from diabetes induced by TH1 cells (Fig. 3C). Collectively, these results indicate that a regulatory cell population capable of transferring protection from diabetes is induced and/or expanded after 4-mer injection.
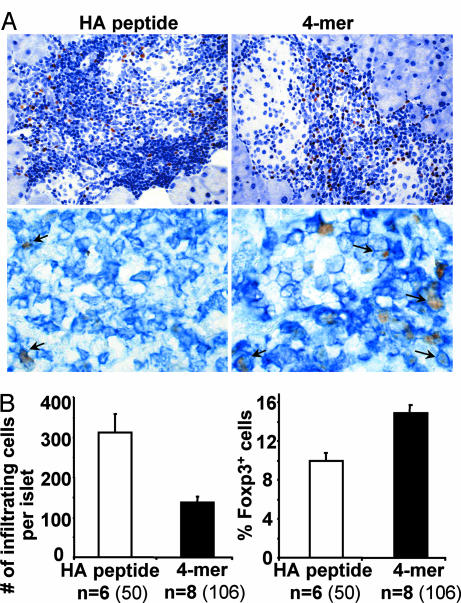
This conclusion led us to evaluate the proportion of infiltrating mononuclear cells expressing Foxp3, a major specification factor for regulatory T cells (13, 14), in the islets of 4-mer versus HA peptide-treated mice by immunohistochemistry (Fig. 4). The insulitis of 4-mer-treated animals was characterized by a significant decrease in mononuclear cells (P = 7 × 10−4) and an ≈50% increase in the proportion of Foxp3+ cells (P = 2 × 10−6) (Fig. 4B), and >90% of these cells were also stained with an anti-CD4 mAb (Fig. 4A Lower) These Foxp3+ CD4 T cells usually displayed a periinsular distribution, with few cells penetrating within the islets (Fig. 4A Upper) Collectively, these data show that the 4-mer-treated mice exhibit a nondestructive insulitis enriched in Foxp3+ CD4 T cells.
Fig. 4.
Accumulation of Foxp3+ mononuclear cells in the infiltrated islets of 4-mer-treated mice. (A) Pancreatic sections from 4-mer- or HA107–119 peptide-treated mice described in Fig. 2B were stained for nuclear Foxp3 (brown, counterstained by H&E). (Upper) Islet-infiltrating mononuclear cells and Foxp3+ cells were counted. (Lower) Double staining was performed on frozen sections to reveal Foxp3 (brown) and CD4 (blue). (B) The number of Foxp3+ and Foxp3− mononuclear cells was counted for each infiltrated islets for a total of 30,096 cells. The mean number (± SEM) of infiltrating cells (Left) and the proportion of Foxp3+ cells (Right) are indicated (n = number of mice). The number of islets is indicated in parentheses.
In contrast to the islets, no significant difference in the proportion of CD25− or CD25+ Foxp3+ CD4 T cells was observed in the spleen and pancreatic lymph nodes of 4-mer- versus HA peptide-treated mice [see supporting information (SI) Fig. 6]. These findings suggest that in lymphoid organs the relevant regulatory T cells are diluted among polyclonal regulatory T cells and/or that these cells are recruited selectively to the islets. Study of cytokine production by splenocytes and pancreatic lymph node cells ex vivo indicated that the production of IL-6, IL-10, TNF-α, and IFN-γ in response to HA peptide was reduced or even inhibited in 4-mer-treated mice (see SI Fig. 7). No increase in basal or HA-induced TGF-β production was observed in 4-mer-treated animals, and in vivo neutralization of IL-4, IL-10, and TGF-β either alone or in combination did not impair the ability of the 4-mer to protect from diabetes (data not shown).
Control of Pathogenic Effector T Cells with Different HA Specificity.
To assess whether the regulatory cell population acts on pathogenic T cells bearing different antigenic specificity (bystander suppression), diabetogenic HNT-TCR TH1 cells, specific for the I-Ad:HA126–138 complex, were used. As described above, RIP-HA mice were tolerized by transfer of 6.5-TCR TH1 cells, followed by injection of the 4-mer or treated with the 4-mer only. Fifteen days later, mice were challenged with a dose of HNT-TCR TH1 cells that induces fully penetrant diabetes in RIP-HA mice (Fig. 3D). Nearly half of the tolerized RIP-HA mice were able to control a subsequent transfer of HNT-TCR TH1 cells (8 of 15 mice became diabetic; P = 0.02). Similar results were obtained with animals treated with the 4-mer alone (9 of 15 mice became diabetic; P = 0.005). These results indicate that treatment with the 4-mer induces a regulatory cell population able to confer protection from T1D induced by diabetogenic CD4 T cells with a different specificity.
Development of Dominant Tolerance Requires an Endogenous Lymphocyte Population.
Because treatment with the 4-mer alone was almost as effective as the combined tolerization with 4-mer and adoptive transfer of TH1 cells, conversion of the transferred T cells into regulatory cells does not appear to be a major mechanism of protection. To determine the contribution of recipients' lymphocytes in the regulation, adoptive transfer experiments were carried out in T and B cell-deficient RAG2−/− RIP-HA recipients. These RAG2−/− RIP-HA mice received 6.5-TCR TH1 cells and were injected with 2.5 μg of the 4-mer or PBS. PBS-treated mice developed T1D within 12 days of transfer, whereas mice treated with the 4-mer presented a significantly delayed diabetes onset (Fig. 5; P = 0.014). This result indicates that endogenous lymphocytes were not responsible for the early control of the diabetogenic TH1 cells. This initial control was associated with a rapid but partial deletion of the pathogenic T cells. Indeed, 48 h after treatment, the number of HA-specific T cells was decreased by 88% in the spleen and 55% in pancreatic lymph nodes in 4-mer-treated compared with PBS-treated mice (data not shown).
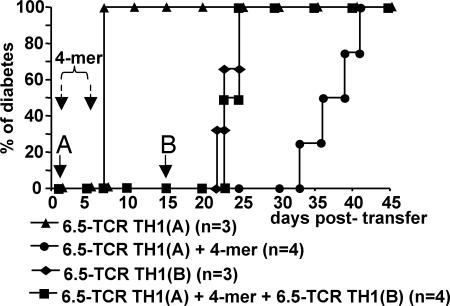
Fig. 5.
The 4-mer-induced dominant tolerance depends on endogenous lymphocytes. RAG2−/− RIP-HA mice received a first injection of 5 × 106 6.5-TCR TH1 cells, called here TH1(A), and were injected i.v. with 2.5 μg of 4-mer (filled circles) or PBS only (filled triangles). A group of mice were tolerized by the combined treatment with 4-mer and 6.5-TCR TH1 cells (filled squares), and a group of control untreated mice (filled diamonds) received on day 15 a second transfer of diabetogenic T cells, called here TH1(B). Adoptive T cell transfers are indicated by solid arrows and i.v. injections of 4-mer by dashed arrows. All PBS-treated mice (6 of 6) eventually developed fatal diabetes within 12 days of TH1 cell transfer.
To determine the influence of endogenous T or B cells on the subsequent induction of dominant tolerance, a group of RAG2−/− RIP-HA mice that had been tolerized by the combined application of 6.5-TCR TH1 cells and 4-mer received a second transfer of diabetogenic 6.5-TCR TH1 cells 15 days later. In contrast to immunocompetent RIP-HA mice (Fig. 3B), the tolerized RAG2−/− RIP-HA mice developed diabetes with the same kinetics as untreated mice (Fig. 5). Thus, in the absence of endogenous T and B cells, the 4-mer is able to delay the onset of disease by directly targeting the transferred TH1 cells, but cannot establish long-lasting dominant tolerance.
Discussion
In the present study, we characterized the cellular mechanisms underlying the therapeutic effect of the injection of small doses of a multimerized epitope in a mouse model of T1D. In this model, the 4-mer appeared to act through distinct mechanisms, leading to a shift in the balance between pathogenic and regulatory cells Its first effect was to induce a partial deletion of diabetogenic TH1 cells. However, development of diabetes was only delayed in 4-mer-treated RAG−/− RIP-HA mice. In a second phase, we observed an induction and/or activation of cells with regulatory properties. These regulatory cells developed in immunocompetent RIP-HA mice treated with 4-mer, but not in RAG−/− recipients, suggesting that they originate from the recipients' lymphocytes, rather than from conversion of the transferred pathogenic TH1 cells. In addition, preliminary experiments suggest that the 4-mer treatment does not convert naive conventional HA-specific CD4 T cells into Foxp3+ CD25+ regulatory cells (L.T.M., S.B., and R.S.L., unpublished data). These regulatory cells were able to control diabetogenic TH1 cells injected at a time when presentation of the 4-mer was no longer detectable in the recipients. The dominant regulatory capacity of these cells was further evidenced by their ability to block diabetes when transferred in conjunction with pathogenic T cells (Fig. 3C). In addition, islets from 4-mer-treated mice were significantly enriched in Foxp3+ CD4 T cells, suggesting that tissue-infiltrating regulatory T cells were contributing to the tolerant phenotype, as previously described in other diabetes models (15–18). Very few regulatory T cells are needed to inhibit autoimmunity, provided that they express a relevant specificity. Thus, even if only some of the islet-infiltrating Foxp3+ CD4 T cells from 4-mer-treated mice were HA-specific, it could certainly explain their major clinical effect. However, at this stage, we have no tools to directly assess their antigenic specificity. Although dominant tolerance in vivo has been attributed to antiinflammatory cytokines in several experimental situations (19–21), in our model, administration of mAbs neutralizing IL-4, IL-10, and TGF-β did not impair the diabetes-protective effect of the 4-mer.
Probably the most interesting aspect of the 4-mer effect is its ability to promote suppression of pathogenic T cells that are not specific for the injected multimerized epitope. This type of bystander suppression induced by multimer treatment has previously been documented in the experimental autoimmune encephalomyelitis setting (9). In a likely scenario, regulatory cells directly inactivate effector T cells attracted to their immediate vicinity by a shared source of antigen. Alternatively, the ability of the 4-mer to induce regulatory lymphocytes could be related to a tolerogenic effect on the APCs in the absence of a “danger” signal. These tolerogenic APCs can be further educated by tolerant T cells (22). Consequently, the same tolerogenic APC could be simultaneously presenting different HA epitopes, modulating the diabetogenicity of the two different HA-specific T cell populations analyzed here. One possible implication of our findings is that specifically designed multimers may inhibit T cell responses to autoantigens secondarily recognized during the course of a destructive autoimmune process.
Several antigen-specific strategies have been devised to promote deletion or anergy of pathogenic autoreactive T cells, or to convert them into innocuous or even regulatory cells (2–5, 23). The observation that full protection from experimental autoimmune disease was achieved with very low doses of multimerized self-antigen is potentially of therapeutic interest and represents a distinct advantage over earlier approaches (2, 3). However, antigen-specific immune suppression at very low-antigen doses is not unprecedented. Indeed, similar to the multimer used here, soluble dimeric HA110–120:I-Ed-Fc chimeric molecules display potent stimulatory properties on 6.5-TCR T cells and have a long half-life in vivo (24, 25). Treatment of (6.5-TCR × RIP-HA)F1 double transgenic mice with 2 μg of these chimeric molecules prevented and even reversed diabetes through the induction of T cell anergy in the spleen and stimulation of IL-10-secreting Tr1 cells in the pancreas (25). However, due to the transient nature of the tolerance induced, a chronic treatment was required to maintain the diabetes-protective effect. An analogous approach using peptide:I-Ag7-Fc chimeric molecules was shown to prevent diabetes induced by the adoptive transfer of BDC2.5 transgenic T cells into TCRα−/− nonobese diabetic mice. This treatment induced clonal anergy and antiinflammatory cytokines, but here again diabetes reappeared after suspension of the treatment (26).
To date, the only product approved by the Food and Drug Administration for autoimmune diseases acting at the level of the peptide:MHC/TCR complex is glatiramer acetate (GA) for the treatment of multiple sclerosis (27). GA is made of synthetic amino acid copolymers that promiscuously bind to MHC class II molecules. Notably, GA has been shown to directly impair the capacity of dendritic cells to secrete the TH1-polarizing cytokine, IL-12p70, and to promote the induction of IL-4 and IL-10-secreting T cells (27–30). Moreover, modified amino acid copolymers ameliorate experimental autoimmune encephalomyelitis through mechanisms similar to those described here for the 4-mer (31, 32). Similarities are also found in the mechanisms of action of anti-CD3 Abs, which afford long-term remission from T1D in nonobese diabetic mice by first inducing the depletion or inactivation of T cells and then promoting active tolerance involving TGF-β-producing regulatory CD4 T cells (33). However, in contrast to GA and anti-CD3 mAbs, the multimers specifically target the deleterious autoreactive T cells. In addition, the sequence of T cell epitope multimers can, in principle, be adapted for the treatment of various autoimmune diseases by representing crucial target autoantigens (9, 10).
A major concern regarding immunotherapy using the systemic administration of self-antigens is safety. Indeed, hypersensitivity or anaphylactic reactions can develop in mouse and man (34–37). Moreover, tolerance induction may be preceded by a transient T cell activation phase, which would pose a risk of exacerbating the autoimmune disease (2, 38, 39). Defining the correct dose is also essential to ensure efficacy and avoid side effects. Several additional steps are therefore needed before implementing a multimerized self-peptide therapy in humans (40). These include the early detection of a potentially harmful immune response in vivo in transgenic animals expressing human MHC molecules. A further prerequisite is the ability to accurately monitor the number and function of antigen-specific CD4 T cells in humans (41, 42). In any event, immunomodulation by multimerized epitopes emerges as a promising alternative for organ-specific autoimmune diseases. The ongoing identification of major self-peptides recognized by pathogenic T cells in human diseases, such as diabetes (43) and multiple sclerosis (39), should help the design of therapeutically relevant multimerized epitopes.
Materials and Methods
Antigens.
The monomeric HA110–119 and HA107–119 peptides were synthesized by NeoMPS (Strasbourg, France). The 4-mer consists of four covalently linked linear HA107–119 peptides separated by 12 aa-long spacer sequences [-(GGPG)3-]. It was produced as previously described (7) and was purified on a reverse-phase C4-HPLC column (Vydac, Hesperia, CA). Absence of endotoxin was confirmed by a standard limulus amoebocyte lysate assay (Cambrex, East Rutherford, NJ).
Mice.
RIP-HA (12), 6.5-TCR (11), and HNT-TCR (44) transgenic mice were backcrossed at least 10 times onto the BALB/c background. Where indicated, RAG2−/− 6.5-TCR and RAG2−/− RIP-HA mice were used. All animal experiments were performed under specific pathogen-free conditions in accordance with the European Union guidelines and had local committee approval.
Flow Cytometry.
The following biotin-, FITC-, and PE-conjugated monoclonal antibodies (mAb) were used: anti-mouse CD4 (clone CT-CD4), CD8 (clones CT-CD8a and 5H10), CD25 (clone 7D4), CD69 (clone H1.2F3), CD62L (clone MEL-14), anti-rat IgG2b-PE (clone 2b10A8), anti-rat IgG2b biotin (clone RG7/11.1), and the 6.5 anti-clonotypic mAb (11). Analysis was performed by using a FACScan flow cytometer, and data were analyzed by using CellQuest software (Becton Dickinson, Mountain View, CA).
In Vitro TH1 Cell Differentiation.
To generate HA-specific TH1 cells, 107 spleen and lymph node cells from 6.5-TCR mice were stimulated with 10 μg/ml HA110–119 peptide in RPMI medium 1640 supplemented with 10% FCS, 1 ng/ml IL-2, and 20 ng/ml IL-12 (R&D Systems, Minneapolis, MN). At day 6, CD4 cells were purified by magnetic sorting by using a rat anti-mouse CD4 mAb (3 μg/ml, clone CT-CD4), goat anti-rat IgG-coated magnetic beads, and separation columns (Miltenyi Biotech, Bergish Gladbach, Germany). Positively selected CD4 T cells (106 per well) were restimulated with HA peptide-pulsed irradiated syngeneic spleen cells (107 per well) in complete medium. At day 9, living cells were collected by Ficoll density separation and used in adoptive transfer experiments. TH1 cells were generated from HNT-TCR mice under the same conditions.
Proliferation Assays.
Single-cell suspensions from spleens and lymph nodes of 6.5-TCR mice (106 per well) or purified TH1 cells were stimulated with anti-CD3ε mAb (0.5 μg/ml; clone 145–2C11; PharMingen, San Diego, CA) or with increasing concentrations of the HA110–119 peptide (10−5 to 10 μg/ml) in the presence of irradiated BALB/c spleen cells (106 per well). Proliferation was assessed by the incorporation of [3H]thymidine (Amersham, Arlington Heights, IL) added for the last 16 h of culture. To measure the in vivo half-life of the 4-mer on APCs, splenocytes (106 cells per well) from BALB/c mice injected i.v. with 2.5 μg of the 4-mer or HA107–119 peptide, or with PBS, were irradiated and used to induce proliferation of purified CD4 T cells from unmanipulated 6.5-TCR mice (APC:T cell ratio of 5:1), without exogenously added antigen (45).
In Vivo Activation of CD4 T Cells.
Splenocytes from 6.5-TCR RAG2−/− mice (106 per ml) were labeled for 10 min at 37°C with 5 μM 5,6-carboxyfluorescein diacetate succinimidyl ester (CFSE; Molecular Probes, Eugene, OR). Thirty million CFSE-labeled cells were adoptively transferred into RIP-HA mice injected with PBS or 2.5 μg of either 4-mer or HA107–119 peptide 1 or 15 days earlier. The percentage of activated (CD69+) CD4+ CFSE+ cells was determined by FACS 24 h after the transfer.
Adoptive Transfer of Diabetes, Prevention of Diabetes by Injection of the Peptides, and in Vivo Neutralization of IL-4, IL-10, and TGF-β.
T1D was induced in RAG2−/− or immunocompetent RIP-HA adult mice by i.v. injection of the indicated number of 6.5-TCR TH1 cells. In some experiments, mice received a second i.v. injection of 5 × 106 6.5-TCR TH1 cells or 25 × 106 HNT-TCR TH1 cells. In cotransfer experiments, RIP-HA mice were first injected i.v. with 5 × 107 total splenocytes from either control RIP-HA mice or RIP-HA mice injected 15 days earlier with 5 × 106 6.5-TCR TH1 cells and the 4-mer. The next day, recipient mice received 5 × 106 diabetogenic 6.5-TCR TH1.
Mice were treated with two identical i.v. injections of the 4-mer or the HA107–119 peptide in 200 μl PBS 3 h and 3 days after TH1 cell transfer. Control animals were injected i.v. with PBS. In some experiments, mice also received i.p. control rat IgG (Sigma–Aldrich, St. Louis, MO) or neutralizing mAbs against IL-10 (clone JES5–2A5, 0.5 mg at days 0 and 3), IL-4 (clone 11B11, 0.5 mg at days 0 and 3), and TGF-β (clone 2G7, 0.5 mg at days −2, 0, 2, 4, 6, and 8). This mAb dose has been previously shown to neutralize cytokine effects in vivo (46, 47).
Glycosuria was assessed by using test strips (Glukotest; Roche-Diagnostic, Mannheim, Germany). Mice were considered diabetic if glycosuria was above 1 g/liter on two consecutive tests. Cumulative diabetes frequencies were compared by using the log-rank test.
Histology.
Pancreata were fixed in formol solution and processed for paraffin embedding. Sections (5-μm thick) were stained with H&E, and the degree of insulitis was evaluated microscopically. For detection of nuclear Foxp3, sections were first treated with S2368 buffer (pH = 9) (DakoCytomation, Glostrup, Denmark) for epitope retrieval and then incubated with purified anti-mouse Foxp3 mAb (clone FJK-16s; e-Bioscience, San Diego, CA). After washes, a secondary biotinylated rabbit anti-rat Ig was added, and a peroxidase-based ABC detection system (DakoCytomation) using DAB as the chromogenic substrat was used. Slides were counterstained with hematoxylin. Photographs were taken for each infiltrated islet, and the number of Foxp3− and Foxp3+ in each islet was counted by an investigator unaware of the treatment received by the animals.
For double staining, pancreata were snap-frozen, cryosections (5-μm thick) were acetone-fixed, and endogenous peroxydase activity and biotin groups were blocked. Staining was first performed by using anti-mouse Foxp3 mAb as described above. The CD4 staining (L3T4; BD Biosciences) was then performed and revealed by using Vector Blue (Vector Laboratories, Burlingame, CA).
For additional information, see SI Methods.
Supplementary Material
Acknowledgments
We thank F. Capilla for assistance with immunohistochemistry, A. Estival for multiplex cytokine analyses, and Drs. A. Saoudi and D. Gonzalez-Dunia for helpful comments on the manuscript. This work was supported by grants from the Juvenile Diabetes Research Foundation International, the French Institute for Medical Research, the Fondation pour la Recherche Médicale, the Midi-Pyrénées Region (to R.S.L.), and Deutsche Forschungsgemeinschaft Grant SFB650 (to O.R. and K.F.).
Abbreviations
- APC
antigen-presenting cell
- GA
glatiramer acetate
- RIP
rat insulin promotor
- T1D
type 1 diabetes
- TCR
T cell receptor
- TH1
T helper 1.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/cgi/content/full/0610423104/DC1.
References
- 1.Vanderlugt CL, Miller SD. Nat Rev Immunol. 2002;2:85–95. doi: 10.1038/nri724. [DOI] [PubMed] [Google Scholar]
- 2.Liblau R, Tisch R, Bercovici N, McDevitt HO. Immunol Today. 1997;18:599–604. doi: 10.1016/s0167-5699(97)01171-7. [DOI] [PubMed] [Google Scholar]
- 3.Harrison LC, Hafler DA. Curr Opin Immunol. 2000;12:704–711. doi: 10.1016/s0952-7915(00)00166-7. [DOI] [PubMed] [Google Scholar]
- 4.McDevitt H. Proc Natl Acad Sci USA. 2004;101(Suppl 2):14627–14630. doi: 10.1073/pnas.0405235101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Feldmann M, Steinman L. Nature. 2005;435:612–619. doi: 10.1038/nature03727. [DOI] [PubMed] [Google Scholar]
- 6.Stern LJ, Wiley DC. Structure (London) 1994;2:245–251. doi: 10.1016/s0969-2126(00)00026-5. [DOI] [PubMed] [Google Scholar]
- 7.Rotzschke O, Falk K, Strominger JL. Proc Natl Acad Sci USA. 1997;94:14642–14647. doi: 10.1073/pnas.94.26.14642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Falk K, Rotzschke O, Strominger JL. Eur J Immunol. 2000;30:3012–3020. doi: 10.1002/1521-4141(200010)30:10<3012::AID-IMMU3012>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 9.Falk K, Rotzschke O, Santambrogio L, Dorf ME, Brosnan C, Strominger JL. J Exp Med. 2000;191:717–730. doi: 10.1084/jem.191.4.717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stienekemeier M, Falk K, Rotzschke O, Weishaupt A, Schneider C, Toyka KV, Gold R, Strominger JL. Proc Natl Acad Sci USA. 2001;98:13872–13877. doi: 10.1073/pnas.241504598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kirberg J, Baron A, Jakob S, Rolink A, Karjalainen K, von Boehmer H. J Exp Med. 1994;180:25–34. doi: 10.1084/jem.180.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lo D, Freedman J, Hesse S, Palmiter RD, Brinster RL, Sherman LA. Eur J Immunol. 1992;22:1013–1022. doi: 10.1002/eji.1830220421. [DOI] [PubMed] [Google Scholar]
- 13.Hori S, Nomura T, Sakaguchi S. Science. 2003;299:1057–1061. [PubMed] [Google Scholar]
- 14.Fontenot JD, Gavin MA, Rudensky AY. Nat Immunol. 2003;4:330–336. [PubMed] [Google Scholar]
- 15.Pop SM, Wong CP, Culton DA, Clarke SH, Tisch R. J Exp Med. 2005;201:1333–1346. doi: 10.1084/jem.20042398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chen Z, Herman AE, Matos M, Mathis D, Benoist C. J Exp Med. 2005;202:1387–1397. doi: 10.1084/jem.20051409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Green EA, Choi Y, Flavell RA. Immunity. 2002;16:183–191. doi: 10.1016/s1074-7613(02)00279-0. [DOI] [PubMed] [Google Scholar]
- 18.Battaglia M, Stabilini A, Draghici E, Migliavacca B, Gregori S, Bonifacio E, Roncarolo MG. Diabetes. 2006;55:1571–1580. doi: 10.2337/db05-1576. [DOI] [PubMed] [Google Scholar]
- 19.Homann D, Holz A, Bot A, Coon B, Wolfe T, Petersen J, Dyrberg TP, Grusby MJ, von Herrath MG. Immunity. 1999;11:463–472. doi: 10.1016/s1074-7613(00)80121-1. [DOI] [PubMed] [Google Scholar]
- 20.Maloy KJ, Powrie F. Nat Immunol. 2001;2:816–822. doi: 10.1038/ni0901-816. [DOI] [PubMed] [Google Scholar]
- 21.Green EA, Gorelik L, McGregor CM, Tran EH, Flavell RA. Proc Natl Acad Sci USA. 2003;100:10878–10883. doi: 10.1073/pnas.1834400100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Alpan O, Bachelder E, Isil E, Arnheiter H, Matzinger P. Nat Immunol. 2004;5:615–622. doi: 10.1038/ni1077. [DOI] [PubMed] [Google Scholar]
- 23.Apostolou I, von Boehmer H. J Exp Med. 2004;199:1401–1408. doi: 10.1084/jem.20040249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Casares S, Zong CS, Radu DL, Miller A, Bona CA, Brumeanu TD. J Exp Med. 1999;190:543–553. doi: 10.1084/jem.190.4.543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Casares S, Hurtado A, McEvoy RC, Sarukhan A, von Boehmer H, Brumeanu TD. Nat Immunol. 2002;3:383–391. doi: 10.1038/ni770. [DOI] [PubMed] [Google Scholar]
- 26.Masteller EL, Warner MR, Ferlin W, Judkowski V, Wilson D, Glaichenhaus N, Bluestone JA. J Immunol. 2003;171:5587–5595. doi: 10.4049/jimmunol.171.10.5587. [DOI] [PubMed] [Google Scholar]
- 27.Sela M, Mozes E. Proc Natl Acad Sci USA. 2004;101(Suppl 2):14586–14592. doi: 10.1073/pnas.0404826101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Duda PW, Schmied MC, Cook SL, Krieger JI, Hafler DA. J Clin Invest. 2000;105:967–976. doi: 10.1172/JCI8970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Neuhaus O, Farina C, Wekerle H, Hohlfeld R. Neurology. 2001;56:702–708. doi: 10.1212/wnl.56.6.702. [DOI] [PubMed] [Google Scholar]
- 30.Vieira PL, Heystek HC, Wormmeester J, Wierenga EA, Kapsenberg ML. J Immunol. 2003;170:4483–4488. doi: 10.4049/jimmunol.170.9.4483. [DOI] [PubMed] [Google Scholar]
- 31.Stern JN, Illes Z, Reddy J, Keskin DB, Sheu E, Fridkis-Hareli M, Nishimura H, Brosnan CF, Santambrogio L, Kuchroo VK, Strominger JL. Proc Natl Acad Sci USA. 2004;101:11743–11748. doi: 10.1073/pnas.0403832101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Illes Z, Stern JN, Reddy J, Waldner H, Mycko MP, Brosnan CF, Ellmerich S, Altmann DM, Santambrogio L, Strominger JL, Kuchroo VK. Proc Natl Acad Sci USA. 2004;101:11749–11754. doi: 10.1073/pnas.0403833101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Belghith M, Bluestone JA, Barriot S, Megret J, Bach JF, Chatenoud L. Nat Med. 2003;9:1202–1208. doi: 10.1038/nm924. [DOI] [PubMed] [Google Scholar]
- 34.Kappos L, Comi G, Panitch H, Oger J, Antel J, Conlon P, Steinman L. Nat Med. 2000;6:1176–1182. doi: 10.1038/80525. [DOI] [PubMed] [Google Scholar]
- 35.Pedotti R, Mitchell D, Wedemeyer J, Karpuj M, Chabas D, Hattab EM, Tsai M, Galli SJ, Steinman L. Nat Immunol. 2001;2:216–222. doi: 10.1038/85266. [DOI] [PubMed] [Google Scholar]
- 36.Liu E, Moriyama H, Abiru N, Miao D, Yu L, Taylor RM, Finkelman FD, Eisenbarth GS. J Clin Invest. 2002;110:1021–1027. doi: 10.1172/JCI15488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Smith CE, Eagar TN, Strominger JL, Miller SD. Proc Natl Acad Sci USA. 2005;102:9595–9600. doi: 10.1073/pnas.0504131102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lanoue A, Bona C, von Boehmer H, Sarukhan A. J Exp Med. 1997;185:405–414. doi: 10.1084/jem.185.3.405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bielekova B, Goodwin B, Richert N, Cortese I, Kondo T, Afshar G, Gran B, Eaton J, Antel J, Frank JA, et al. Nat Med. 2000;6:1167–1175. doi: 10.1038/80516. [DOI] [PubMed] [Google Scholar]
- 40.von Herrath MG, Nepom GT. J Exp Med. 2005;202:1159–1162. doi: 10.1084/jem.20051224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Arif S, Tree TI, Astill TP, Tremble JM, Bishop AJ, Dayan CM, Roep BO, Peakman M. J Clin Invest. 2004;113:451–463. doi: 10.1172/JCI19585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mallone R, Nepom GT. Clin Immunol. 2004;110:232–242. doi: 10.1016/j.clim.2003.11.004. [DOI] [PubMed] [Google Scholar]
- 43.Kent SC, Chen Y, Bregoli L, Clemmings SM, Kenyon NS, Ricordi C, Hering BJ, Hafler DA. Nature. 2005;435:224–228. doi: 10.1038/nature03625. [DOI] [PubMed] [Google Scholar]
- 44.Scott B, Liblau R, Degermann S, Marconi LA, Ogata L, Caton AJ, McDevitt HO, Lo D. Immunity. 1994;1:73–83. doi: 10.1016/1074-7613(94)90011-6. [DOI] [PubMed] [Google Scholar]
- 45.Bercovici N, Heurtier A, Vizler C, Pardigon N, Cambouris C, Desreumaux P, Liblau R. J Immunol. 2000;165:202–210. doi: 10.4049/jimmunol.165.1.202. [DOI] [PubMed] [Google Scholar]
- 46.Cautain B, Damoiseaux J, Bernard I, van Straaten H, van Breda Vriesman P, Boneu B, Druet P, Saoudi A. Eur J Immunol. 2001;31:1132–1140. [PubMed] [Google Scholar]
- 47.Chiba A, Oki S, Miyamoto K, Hashimoto H, Yamamura T, Miyake S. Arthritis Rheum. 2004;50:305–313. doi: 10.1002/art.11489. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.