Abstract
Tissue factor (TF) is the primary initiator of coagulation, and the TF pathway mediates signaling through protease-activated receptors (PARs). In sepsis, TF is up-regulated as part of the proinflammatory response in lipopolysaccharide (LPS)–stimulated monocytes leading to systemic coagulation activation. Here we demonstrate that TF cytoplasmic domain–deleted (TFΔCT) mice show enhanced and prolonged systemic coagulation activation relative to wild-type upon LPS challenge. However, TFΔCT mice resolve inflammation earlier and are protected from lethality independent of changes in coagulation. Macrophages from LPS-challenged TFΔCT mice or LPS-stimulated, in vitro–differentiated bone marrow–derived macrophages show increased TF mRNA and functional activity relative to wild-type, identifying up-regulation of macrophage TF expression as a possible cause for the increase in coagulation of TFΔCT mice. Increased TF expression of TFΔCT macrophages does not require PAR2 and is specific for toll-like receptor, but not interferon γ receptor, signaling. The presence of the TF cytoplasmic domain suppresses ERK1/2 phosphorylation that is reversed by p38 inhibition leading to enhanced TF expression specifically in wild-type but not TFΔCT mice. The present study demonstrates a new role of the TF cytoplasmic domain in an autoregulatory pathway that controls LPS-induced TF expression in macrophages and procoagulant responses in endotoxemia.
Introduction
Disseminated intravascular coagulation driven by tissue factor (TF) is associated with systemic inflammatory disorders, including viral hemorrhagic fevers and severe sepsis (for reviews, see Riewald and Ruf1 and Ruf2). Inhibition of the TF pathway by potent TF-directed inhibitors reduced inflammatory cytokine levels and increased survival in lethal models of bacterial sepsis in primates3–5 and attenuated acute lung injury.6 Although coagulation blockade is likely to be beneficial in sepsis by reducing microthrombosis, subsequent hypoxic organ dysfunction, and inflammation, blocking thrombin generation per se has poor efficacy in prolonging survival in primate models.7,8 Therefore, it remains of interest to understand how signaling of the TF pathway makes contributions to sepsis mortality. Genetic reduction of TF or VIIa levels in mice increased survival in endotoxemia, but protease activated receptor 1 (PAR1) or PAR2 deficiency alone or in combination did not reduce lethality.9–11 Whether the TF pathway contributes to severe systemic inflammation through signaling thus remains incompletely understood.
In the response to microbial pathogen challenge or lipopolysaccharide (LPS)–induced toll-like receptor 4 (TLR4) activation, cells of the monocyte/macrophage lineage are the primary cell types that up-regulate TF to trigger intravascular coagulation.12–15 However, the subsequent crosstalk between coagulation activation and inflammation is complex. Thrombin and PAR signaling may amplify the coagulant response by further inducing TF in endothelial cells16–18 or attenuate coagulation and inflammation through the activated protein C (PC)/endothelial cell PC receptor (EPCR) pathway that uses PARs as signaling receptors.19–22 In addition, endotoxin challenge of TF cytoplasmic domain–deleted (TFΔCT) mice indicated that TF can regulate leukocyte rolling and recruitment, presumably by altering vessel wall properties.23 The TF cytoplasmic domain also regulates integrin activation and cell migration24,25 and has roles in PAR-2–dependent angiogenesis.26
Here, we identify a novel role of the TF cytoplasmic domain in regulating the procoagulant response induced by TLR signaling in macrophages. LPS activation of macrophages is mediated by a receptor complex of CD14 and TLR4.27 The signaling pathways by which LPS induces TF mRNA expression in monocytes have been studied in detail,28 but maturation of monocytes into macrophages promotes diverse phenotypic changes, including the expression of TF29 as well as the TF-VIIa signaling receptor PAR2.30 We find in macrophages that LPS-mediated TF up-regulation is controlled by the TF cytoplasmic domain through p38-dependent regulation of the ERK1/2 MAP kinase pathway. However, regulation of TF expression is independent of PAR2 signaling and specific for TLR, but not interferon γ, signaling. The data provide new evidence for an autoregulatory role of the TF cytoplasmic domain to control procoagulant responses in endotoxemia.
Materials and methods
Mouse strains
TFΔCT mice and PAR2−/− mice31,32 were backcrossed into C57BL/6. TFΔCT mice were backcrossed for 10 generations and a colony of wild-type animals was derived from this backcross. Survival of wild-type mice derived from the backcrossing did not differ from C57BL/6J mice restocked from Jackson Laboratories (Bar Harbor, ME). PAR2-deficient mice were backcrossed for 9 generations and a homogenous C57BL/6 background was confirmed by genome scanning for 129 markers of founder animals. TFΔCT/PAR2−/− mice were generated by interbreeding after backcrossing was complete. TFΔCT mice were further backcrossed for 3 more generations into C57BL/6J with no change in the observed phenotype. After fostering to pathogen-free mothers and confirmed absence of chronic infections, mice were transferred to and further maintained in a pathogen-free animal facility.
Mouse model of severe endotoxemia
All experiments were carried out in accordance with the National Institutes of Health Animal Protection Guidelines and were approved by The Scripps Research Institute Animal Care and Use Committee. Lipopolysaccharide (LPS, E coli O111:B4; Calbiochem, La Jolla, CA) was dissolved in 37°C normal saline by vortexing for 30 seconds before and after 2 minutes of sonication. Mice between 60 and 80 days of age were intraperitoneally injected under 2% isoflurane with 5 to 9 mg/kg LPS. For each batch of endotoxin, an appropriate dose was established that resulted in 75% to 90% lethality. Because of reported10 sex differences in the dose response to endotoxin, we used male mice only in order to reduce variability. However, the phenotype of TFΔCT mice is not sex specific. All experiments were carried out in a room with alternating 12-hour light and dark cycles under stable conditions of temperature (20°C-22°C) and relative humidity (40%-60%). Mice had constant access to food and water.
Two basic experimental protocols were used. For evaluation of coagulation activation and cytokine levels, mice were challenged with LPS for 4, 6, 12, or 18 hours. Ten minutes prior to blood collection, mice were anticoagulated by intraperitoneal injection of 1000 IU heparin (Elkins-Sinn, Cherry Hill, NJ). Under 3% isoflurane anesthesia, blood was drawn from the inferior vena cava into a 3.2% solution of sodium citrate and mice were killed to terminate the experiment. To establish the survival benefit of knock-out strains, mice were challenged with LPS and tightly monitored over the next 7 days without further manipulations, including blood drawing which may have a negative impact on the recovery of animals. Coagulation inhibitors were applied by intravenous tail vein injection at the time of LPS administration.
TF expression in peritoneal macrophages after LPS stimulation
Mouse peritoneal macrophages were harvested 6 hours after intraperitoneal injection of 5 mg/kg LPS. After killing, peritoneal lavage was performed to harvest resident cells by injecting 3 times 3 to 5 mL sterile cold PBS and recovering the wash through a small abdominal incision. Contaminating red blood cells were lysed with hypotonic buffer and cells were counted. Flow cytometry was used to quantify TF expression on macrophages by gating on CD11b-PerCP and F4/80-PE (Pharmingen, San Diego CA) double-positive cells and detection with fluorescein isothiocyanate (FITC)-conjugated antimouse TF antibody. TF mRNA expression in lungs 6 hours after LPS or sham injection into mice was quantified by TaqMan real-time polymerase chain reaction (PCR) on an ABI PRISM detection system (Applied Biosystems, Foster City, CA).
Measurements of thrombin-antithrombin complex (TAT), cytokine levels, and blood counts
Citrated blood drawn from the vena cava inferior was immediately centrifuged at 2000g for 10 minutes. Plasma was separated, aliquoted, and stored at −80°C for subsequent determination of TAT and cytokine levels. Enzyme-linked immunosorbent assays (ELISAs) were used to determine TAT (Enzygnost; Dade Behring, Marburg, Germany), IL6, IL1β, and MCP1 (Quantikine; R&D Systems, Minneapolis, MN) levels. Cytokine profiles were determined by Luminex multiplex analysis (Invitrogen, Carlsbad, CA). For blood counts, 300 μL blood was drawn into EDTA-containing microtainer tubes (Becton Dickinson, Franklin Lakes, NJ) from the inferior vena cava 6 hours after LPS injection. White blood cells, hemoglobin, hematocrit, and platelets were measured on an ADVIA 120 Hematology Analyzer (Bayer, New York, NY) and compared to established reference ranges for mice.
Statistical analysis
Six to 10 mice per genotype were injected in a block randomized manner and experiments were typically repeated 2 to 4 times. All data are presented as means and standard deviations (mean ± SD), and differences were considered significant at P less than .05. Kaplan-Meier curves were plotted and survival differences were analyzed using a log-rank test. Statistical analysis of continuous data was performed for 2 group comparison by the Mann-Whitney U test.
Culture of bone marrow–derived macrophages (BMMs)
BMMs were in vitro–differentiated using endotoxin-free cell culture reagents according to published protocols.33 Briefly, bone marrow cells were obtained by flushing femurs and tibias with RPMI-1640 medium. Following washing with RBC lysis buffer (Sigma, St Louis, MO), cells were plated into Petri dishes containing RPMI-1640 medium supplemented with 20% to 30% L929 cell–conditioned medium, 10% heat-inactivated FBS, 100 units/mL penicillin, 1000 units/mL streptomycin, and 20 mM l-glutamine and maintained in a 8% CO2 atmosphere. Replacing L929 cell–conditioned medium with recombinant MCSF yielded macrophages with similar properties in the TF-dependent signaling. At day 7, the adherent cellular monolayer was used for experiments. Morphologic and fluorescence-activated cell sorter (FACS) analyses showed that 98% to 100% of cells were macrophages. For analysis of expression of activation markers, BMMs were left unstimulated or stimulated with LPS (1 μg/mL) for 18 hours. Cells were harvested, washed in FACS buffer (PBS, 2% FBS, 2 mM EDTA), and resuspended in FACS staining buffer with Fc-block (Pharmingen) for 10 minutes at 4°C. Cells were then incubated for an additional 30 minutes with fluorochrome-conjugated antibodies at 4°C in the dark. After washing in PBS, cells were analyzed by flow cytometry. Antibodies to CD11b, CD80, CD86, and CD69b were from Pharmingen and anti-TLR4 and F4/80 from eBioscience (San Diego, CA).
Characterization of BMMs
BMMs (2 × 105 cells/mL) were plated after 7 days of differentiation and allowed to adhere overnight in L929 cell–conditioned medium. Controls and cells stimulated with LPS (1 μg/mL) at 37°C for the indicated times were rinsed twice with cold PBS and lysed in SDS sample buffer for Western blotting using antiphospho or panspecific ERK1/2 and p38 antibodies (Cell Signaling Technologies, Beverly MA). FcγR-dependent phagocytosis was evaluated using sheep red blood cells coated with or without subagglutinating concentrations of rabbit antisheep red blood cell antibodies (ICN Biomedicals, Aurora, CA) at 37°C for 60 minutes. After washes, coated red blood cells were added to BMMs on coverslips for uptake in RPMI-1640 medium, 10% FCS. After washes, noningested red blood cells were lysed with hypotonic buffer (Sigma-Aldrich, St Louis, MO), and specific uptake of opsonized cell was determined by counting representative views. For confocal microscopy, BMMs grown on coverslips were stimulated with LPS (1 μg/mL) for 18 hours and permeabilized at 4°C for 15 minutes using 0.05% saponine. Cells were stained with monoclonal phospho-specific ERK1/2 antibody (Cell Signaling Technologies) and rabbit antimouse TF (rabbit 552), followed by appropriate secondary antibodies conjugated to FITC or Texas Red and nuclear staining with ToPro3 dye. Optical sections of 0.2-0.5 μm were obtained with 60× oil immersion objective (numerical aperture 1.3) on a Nikon TE2000-U confocal microscope (Nikon Instruments, Melville, NY). Images were collected using Laser pix software (Bio-Rad, Hercules, CA) and assembled using Photoshop (Adobe, San Jose, CA). Single optical sections are shown. For detection of nitric oxide synthase 2 (NOS2) activation, BMMs were stimulated with LPS (1 μg/mL) for 18 hours and cell lysates were subjected to Western blotting using anti-NOS2 antibody (SantaCruz Biotechnology, Santa Cruz, CA).
TF assays
BMMs were stimulated either with LPS (1 μg/mL) or IFNγ (100 ng/mL) for 6 hours after preincubation of the indicated inhibitors for 30 minutes. After stimulation, BMMs were lysed at 37°C for 15 minutes in 15 mM octyl-d-glucopyranoside. TF coagulant activity was determined by a one-stage clotting assay in mouse plasma using a calibration curve in which a 50-second clotting time was arbitrarily set to 1000 mU. VIIa-dependent Xa generation was determined by 2-stage chromogenic assay using mouse VIIa (kindly provided by Dr Lars Petersen, Novo Nordisk, Malov, Denmark) and human factor X. TF mRNA levels were determined by real-time reverse-transcription (RT)–PCR on an ABI Prism Sequence Detection System (Applied Biosystems) using sequence-specific TaqMan primers designed using Primer Express Version 2 software (Applied Biosystems). The mRNA was extracted with Trizol reagent from directly lysed cultured cells or from tissue homogenates prepared by tissue grinder or cell beater. TaqMan probes and primers were custom designed for mouse TF: the sequence of forward primer, 5′-acctgggcctatgaagcaaa-3′; reverse primer, 5′-ttggtctccgtccccatg-3′; and TaqMan probe, 5′-6-FAM-tcctctctgtcccacggaggaactca-TAMRA-3′. All samples were normalized with mouse GAPDH or β-actin probes and primers obtained from Applied Biosystems.
Results
TF cytoplasmic domain–deleted mice display increased coagulation activation but are protected from lethality independent of altered coagulation
We addressed the role of the TF cytoplasmic domain in endotoxemia in a homogenous C57BL/6 genetic background. We established an appropriate endotoxin dose (5-8 mg/kg, depending on individual LPS batches) to yield 75% to 90% lethality in male mice. With the preparations of LPS used, this target lethality was typically achieved at a dose approximately 1.6-fold higher than an LD50, indicating a steep dose-response curve. Lethality in this model occurred 1 to 3 days after endotoxin challenge and all mice that lived for 7 days were considered long-term survivors. At this time, surviving mice had returned to normal feeding behavior, appeared healthy, and were active.
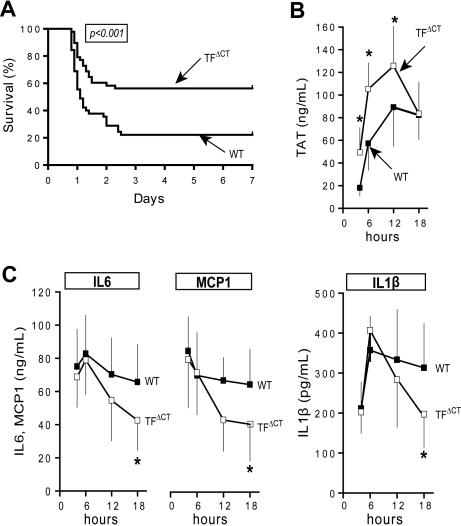
In this model, mice challenged for 6 hours develop an overall leukopenia with increased neutrophil and decreased lymphocyte counts, signs of hemoconcentration, and reduced platelet counts, as also described by Camerer et al.10 No differences in leukocyte or platelet counts were detected between TFΔCT and wild-type mice 6 hours after LPS challenge (Table 1). This represents the time when differences in leukocyte counts were most prominent in the study by Sharma et al23 who studied TFΔCT mice in a mixed genetic background and attributed early reduction in mortality to diminished leukocyte rolling and recruitment to the lung. It is well appreciated that inflammation models are susceptible to strain variations. Nevertheless, homogenous C57BL/6 TFΔCT mice showed significantly increased long-term survival relative to wild-type controls (Figure 1A).
Table 1.
Blood counts of wild-type or TFΔCT mice 6 hours after LPS stimulation
| WBC count 109/L | Neutrophil (% of WBC) | Lymphocyte (% of WBC) | Monocyte (% of WBC) | Eosinophil (% of WBC) | Hematocrit (%) | Platelet count | |
|---|---|---|---|---|---|---|---|
| Normal range | 3.2-12.7 | 7-31 | 60-95 | 0-4 | 0-6 | 36.7-46.8 | 766-1657 |
| Wild type | |||||||
| Mean | 1.3 | 49 | 44 | 3 | 3 | 47 | 658 |
| SD | 0.5 | 17 | 19 | 1 | 2 | 1 | 118 |
| TFΔCT | |||||||
| Mean | 1.3 | 60 | 29 | 2 | 5 | 48 | 647 |
| SD | 0.6 | 5 | 12 | 2 | 6 | 3 | 231 |
Wild-type, n = 6; TFΔCT, n = 9.
WBC indicates white blood cell.
Figure 1.
Long-term survival benefit of TF cytoplasmic domain–deleted mice in severe endotoxemia. (A) Kaplan-Meier survival plot of TFΔCT (n = 45) versus wild-type (n = 48) male mice after intraperitoneal LPS challenge (6 repeat experiments with 7-8 mice/group). TAT levels (B) as well as IL6, IL1β, and MCP1 inflammatory cytokine levels (C) were measured in separate experiments at 4, 6, 12, and 18 hours in wild-type (filled squares) and TFΔCT (open squares) male mice after LPS challenge (TFΔCT 4 hours: n = 9; WT 4 hour: n = 8; TFΔCT 6 hours: n = 6; WT 6 hours: n = 6; TFΔCT 12 hours: n = 6; WT 12 hours: n = 6; TFΔCT 18 hours: n = 14; WT 18 hours: n = 14). *denotes significant difference between groups, P < .05 by Mann-Whitney test.
In order to monitor the inflammatory and coagulant response under our challenge conditions, we obtained plasma samples from separate cohorts by terminal bleeding after 4, 6, 12, or 18 hours for determination of thrombin-antithrombin complex (TAT) levels and of circulating cytokines IL6, IL1β, and MCP1. TFΔCT mice showed significantly increased coagulation activation, measured as TAT levels, until 12 hours after LPS challenge relative to wild-type mice (Figure 1B). The increase in coagulation activation was accompanied by cytokine production that was indistinguishable from wild-type animals at 6 hours, indicating that LPS-induced inflammatory cytokine elaboration was not regulated by the TF cytoplasmic domain during this period (Figure 1C). At 12 hours, inflammation markers of TFΔCT mice began to deviate from wild-type controls. After 18 hours, TAT levels were similar between TFΔCT and wild-type mice. However, levels of all inflammatory cytokines were significantly reduced in TFΔCT mice, indicating that despite similar coagulation activation, TFΔCT mice can more readily resolve the systemic inflammatory response.
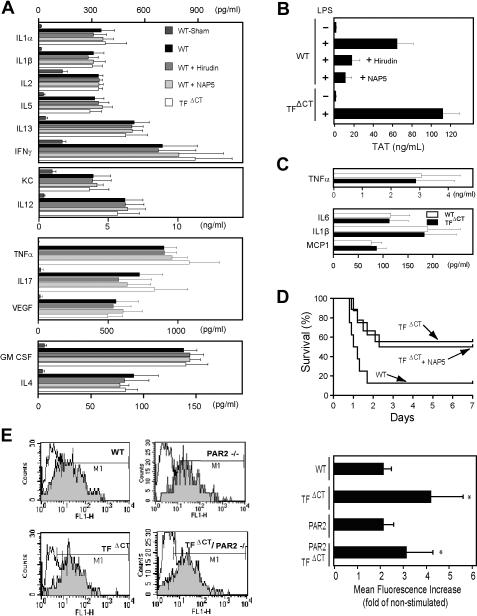
In order to demonstrate that the measured cytokines are representative of the early inflammatory response of TFΔCT mice, we determined a large panel of cytokines by multiplex analysis of 6-hour plasma samples from TFΔCT and wild-type mice. In addition, we blocked coagulation activation in wild-type mice with a single bolus injection of the thrombin inhibitor hirudin or the Xa inhibitor NAP5 to address whether early coagulation activation contributed to the inflammatory responses in this model (Figure 2A-B). None of the inflammatory parameters showed differences between wild-type and TFΔCT mice, confirming at a large scale that early LPS-induced inflammation between these strains is indistinguishable. Blocking coagulation activation had also no effect on inflammatory cytokine levels, further demonstrating that attenuating coagulation has no immediate effect on inflammation. We further confirmed that TFΔCT mice had no defect in the initial phase of inflammation by measuring selected cytokines, including TNFα, after 2 hours of stimulation (Figure 2C). Thus, the changes observed in TFΔCT mice are not in the onset, but rather in the resolution of the systemic inflammatory response.
Figure 2.
TF induction is regulated by its cytoplasmic domain in peritoneal macrophages. (A) Multiplex cytokine array analysis of plasma samples from TFΔCT and WT mice treated with an intravenous, single bolus of the Xa inhibitor NAP5 (2 mg/kg) or the thrombin inhibitor hirudin (40 mg/kg) at the time of LPS challenge. Six hours after LPS stimulation, heparinized blood was drawn (n = 8/group). Sham denotes mice that received saline instead of LPS. (B) TAT levels for the treatment groups shown in panel A. (C) TNFα, IL6, IL1β, and MCP1 levels were measured by ELISA in samples obtained 2 hours after LPS challenge. (D) Survival curve of LPS-challenged WT or TFΔCT mice receiving a single intravenous injection of saline or NAP5 (2 mg/kg) at the time of intraperitoneal LPS administration. (E) TF expression by peritoneal macrophages after LPS (5 mg/kg) stimulation. Representative histograms of TF staining of CD11b and F4/80 double-positive peritoneal cells recovered 6 hours after intraperitoneal injection of saline (open histograms) or LPS (shaded histograms). Changes in mean fluorescence over control-treated animals were calculated for each depicted genotype (*denotes different from wild type, P < .05, n ≥ 3).
Figure 2B documents that the attenuation of coagulation activation by both inhibitors was efficient for at least 6 hours. Consistent with the study by Camerer et al,10 blocking early coagulation activation by different inhibitors had no effect on the long-term survival of wild-type mice in our model (data not shown). Increased coagulation activation in TFΔCT mice may have induced antiapoptotic or barrier protective pathways19,22,34,35 without changes in inflammatory parameters. In order to address whether early coagulation activation protected TFΔCT mice, we administered the potent Xa inhibitor NAP5 at the time of LPS challenge and monitored the survival of these animals versus wild-type and TFΔCT controls (Figure 2D). Attenuation of early coagulation activation had no effect on the survival of TFΔCT mice, suggesting that these mice are protected by alterations in direct TF-dependent signaling, rather than changes in coagulation activation.
Peritoneal TFΔCT macrophages of endotoxin-stimulated mice have increased TF expression independent of PAR2
Circulating monocytes are critical in the initial inflammatory response to endotoxin. Because monocytes express TF only after LPS induction, we reasoned that the TF cytoplasmic domain cannot play a regulatory role in monocytes. Consistently, all early inflammatory markers were found to be unchanged and TFΔCT mice develop a full-blown inflammatory response (Figure 2A,C). The observed increased coagulation activation in TFΔCT mice may result from changes in the cell surface procoagulant activity or alternatively from increased TF expression. We detected approximately 2-fold increased TF mRNA levels in the lung of TFΔCT relative to wild-type mice 6 hours after LPS challenge (wild-type 1.9 ± 1.4-fold versus TFΔCT 4.2 ± 1.8-fold up-regulation over sham-treated control, P < .02, n = 8), but the magnitude of the difference was considered to be too low for further analysis of lungs by immunohistochemistry.
Maturation of monocytes into macrophages results in TF expression, but macrophages remain sensitive to LPS stimulation by further up-regulation of TF.29 To test whether macrophages potentially contribute to systemic coagulation activation, we designed an in vivo experiment where resident macrophages were stimulated with LPS for 6 hours and TF expression was evaluated on CD11b and F4/80 double-positive cells isolated from the peritoneal cavity. LPS stimulation increased TF expression on peritoneal macrophages from basal levels by approximately 2-fold in wild-type mice. TFΔCT macrophages from LPS-stimulated mice expressed approximately 2-fold higher TF levels relative to wild-type (Figure 2E). The TF cytoplasmic domain is linked to PAR2 by signaling crosstalks.26,36 To address whether PAR2 plays a role in increased TF induction in peritoneal macrophages, we determined macrophage TF expression levels after stimulation of PAR2−/− and TFΔCT/PAR2−/− mice. PAR2−/− macrophages expressed similar TF levels as wild-type mice, but TF expression was significantly elevated in TFΔCT/PAR2−/− macrophages, similar to that in TFΔCT mice. Thus, increased TF expression in TFΔCT macrophages is independent of PAR2 expression.
TFΔCT, PAR2−/−, TFΔCT/PAR2−/− bone marrow progenitor cells develop normally into macrophages
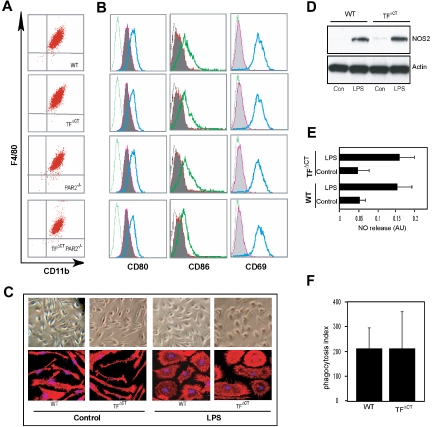
In order to distinguish between direct effects of the TF cytoplasmic domain in macrophages versus indirect effects of TF cytoplasmic domain signaling originating from other cell types in vivo, we further characterized TF induction in bone marrow–derived macrophages. Bone marrow from wild-type, TFΔCT, PAR2−/−, and TFΔCT/PAR2−/− mice yielded similar amounts of pure populations of differentiated macrophages after 7 days in culture, as shown by similar double immunostaining with CD11b and F4/80 markers (Figure 3A). In addition, overnight stimulation with LPS induced surface expression of the costimulatory molecules CD80 and CD86, and the activation marker CD69b, demonstrating that the TF cytoplasmic domain is not required for differentiation and activation of macrophages by endotoxin (Figure 3B). Overnight stimulation of macrophages with LPS-induced similar morphologic changes and cytoskeletal reorganization visualized by phalloidin staining of wild-type or TFΔCT mice (Figure 3C). In addition, NOS2 induction and nitric oxide production upon LPS stimulation, as well as Fc-mediated phagocytosis of IgG-coated red blood cells were similar (Figure 3D-F), indicating that the TF cytoplasmic domain has no major role to modulate macrophage effector functions.
Figure 3.
Characterization of bone marrow–derived macrophages (BMMs). (A) BMMs obtained by culture of bone marrow cells from 7- to 10-week-old wild-type, TFΔCT, PAR2−/−, or TFΔCT/PAR2−/− mice were analyzed by flow cytometry for the macrophage surface markers F4/80 and CD11b. (B) Expression of costimulatory molecules on BMMs after overnight stimulation with LPS (1 μg/mL). Isotype-matched control antibody staining (dotted histogram) or specific staining of non–LPS-stimulated (shaded area) or LPS-stimulated (solid line) BMMs are shown; an example of at least 3 independent experiments is given. (C) Morphology of wild-type and TFΔCT BMMs with and without overnight LPS stimulation by phase-contrast microscopy (upper panel) or after phalloidin staining (red) by confocal microscopy; nucleus is stained with ToPro3 (blue) (lower panel).[AU34} (D-F) Effector functions of wild-type or TFΔCT BMMs. Overnight LPS (1 μg/mL) stimulation leads to similar up-regulation of NOS2 by Western blotting (D) and nitric oxide (NO) production (E) determined using the Griess reagent37 (n = 3). (F) FcγR-mediated phagocytosis of IgG-opsonized sheep red blood cells by wild-type and TFΔCT BMMs.
TFΔCT BMMs show increased LPS-induced TF expression
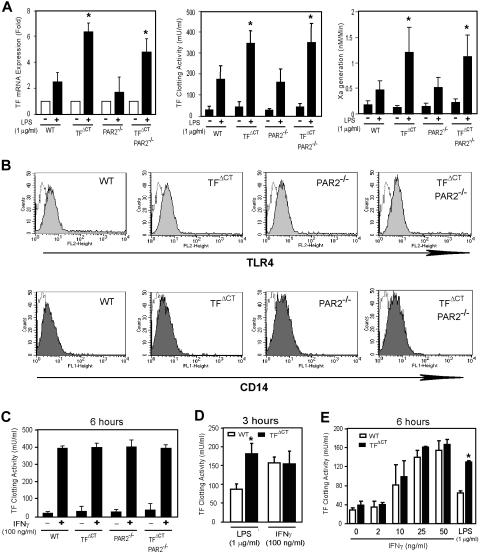
TF expression was analyzed after 6 hours of LPS stimulation by measuring TF clotting activity and VIIa-dependent Xa generation, as well as by determining TF mRNA levels by quantitative real-time PCR. Baseline TF activity was similar in all genotypes, but BMMs from TFΔCT and TFΔCT/PAR2−/− mice showed approximately 2-fold increased LPS-induced TF expression relative to wild-type or PAR2−/− macrophages (Figure 4A). Flow cytometry showed that BMMs from TFΔCT and wild-type mice expressed similar levels of TLR4 and CD14, excluding that changes in expression levels of the LPS signaling receptors accounted for the difference in LPS responsiveness (Figure 4B). Interferon γ (IFNγ) induces macrophage TF expression by a pathway synergistic with LPS, and this pathway therefore is presumably independently regulated.38 Stimulation of BMMs with IFNγ induced TF indistinguishable among all genotypes tested (Figure 4C). Analysis of TF expression after 3 hours of stimulation further demonstrated that differential expression of LPS versus IFNγ was not caused by different kinetics of TF induction (Figure 4D). Dose-response curves for IFNγ-stimulated wild-type and TFΔCT macrophages also did not uncover a difference at suboptimal IFNγ dosage. Thus, the TF cytoplasmic domain specifically regulates LPS-mediated, but not IFNγ-mediated, TF expression.
Figure 4.
TF cytoplasmic domain negatively regulates LPS-induced TF expression in BMMs. (A) TF mRNA expression, TF clotting activity, and TF-VIIa–dependent Xa generation of unstimulated or 6-hour LPS (1 μg/mL)–stimulated BMMs from the indicated genotypes. TF mRNA induction was quantified by real-time PCR (Applied Biosystems); TF procoagulant activity and Xa generation were determined after standard octyl-glucoside cell lysates. (B) TLR4 and CD14 expression in unstimulated BMMs from the indicated genotypes by flow cytometry. Dotted histograms represent isotype-matched control antibody and the filled histograms represent specific surface marker staining. (C) TF levels in BMMs from the indicated genotypes that were stimulated with and without IFNγ for 6 hours. (D) TF clotting activity measured after 3 hours of stimulation. (E) Dose response of IFNγ-induced TF expression after 6 hours of stimulation in comparison to LPS-treated controls. TF activity was determined in standard octyl-glucoside cell lysates. *denotes different from wild-type, P < .05, t test.
The TF cytoplasmic domain suppresses TLR4-mediated ERK1/2 activation dependent on p38 activation
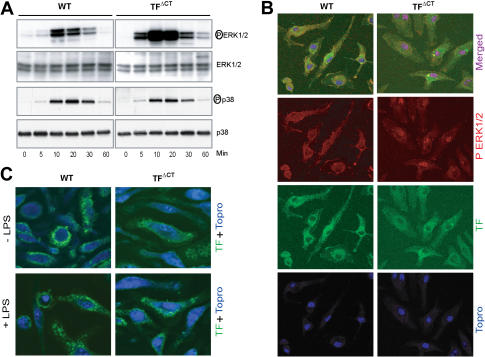
Mitogen-activated protein kinase (MAPK) pathways downstream of TLR4 signaling are required for TF induction.39 We analyzed ERK1/2 and p38 phosphorylation in LPS-stimulated BMMs from wild-type and TFΔCT mice (Figure 5A). Although p38 phosphorylation was similar between the genotypes, TFΔCT displayed greatly enhanced ERK1/2 phosphorylation throughout the time course. In order to induce transcriptional responses, phosphorylated ERK1/2 translocates into the nucleus. Twenty-minute LPS stimulation caused enhanced accumulation of nuclear phospho-ERK1/2 visualized by the merged purple pseudocolor in TFΔCT relative to wild-type mice (Figure 5B). Because it was not clear from the staining of permeabilized cells whether cytoplasmic domain deletion altered TF cell surface expression, we stained nonpermeabilized cells without and with 10-minute LPS stimulation. No differences in staining patterns were apparent between wild-type and TFΔCT macrophages (Figure 5C). Thus, ERK1/2 phosphorylation and nuclear translocation is enhanced in BMMs upon TF cytoplasmic domain deletion independent of changes in TF cell surface localization.
Figure 5.
TF cytoplasmic domain deletion enhances ERK1/2 phosphorylation in macrophages. (A) Time course shows enhanced ERK1/2, but similar p38, phosphorylation after stimulation with LPS (1 μg/mL) in TFΔCT compared to wild-type BMMs. Control experiments with confirmed TLR4-selective LPS gave similar results. (B) Nuclear translocation of phosphorylated ERK1/2 was analyzed in BMMs stimulated with LPS (1 μg/mL) for 20 minutes by triple staining of permeabilized cells with anti-phospho ERK1/2 (red), antimouse TF (green), and nuclear staining with ToPro3 (blue). (C) TF staining (green) in nonpermeabilized BMMs with and without LPS stimulation for 10 minutes.
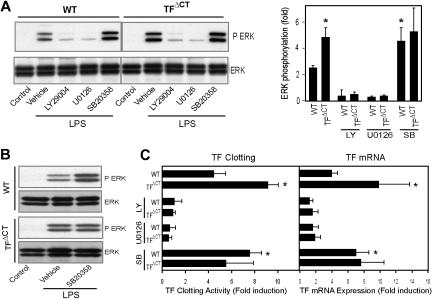
Pharmacological inhibition of ERK1/2 activation or PI3 kinase by U0126 or LY292004, respectively, prevented TF induction in BMMs of both wild-type and TFΔCT mice (Figure 6B). The PI3 kinase inhibitor LY292004 also blocked LPS-induced ERK1/2 phosphorylation (Figure 6A). Thus, TF induction is ERK1/2 dependent and PI3 kinase is upstream of ERK1/2 signaling. Of interest, blocking p38 with SB203580 increased ERK1/2 phosphorylation (Figure 6A) in wild-type mice, but had no effect on ERK1/2 phosphorylation in TFΔCT mice, indicating that the TF cytoplasmic domain may act as a scaffold to promote p38-dependent suppression of ERK1/2 phosphorylation.
Figure 6.
TF cytoplasmic domain regulates TLR4-induced ERK1/2 phosphorylation and TF expression via modulation of the p38 MAPK pathway. (A) LPS-induced ERK1/2 phosphorylation in BMMs is dependent on PI3-kinase and negatively regulated by p38 in wild-type, but not TFΔCT, BMMs. Cells were pretreated for 30 minutes with the p38 inhibitor SB203580 (10 μM), the PI-3 kinase inhibitor LY292004 (30 μM), or the MEK inhibitor U0126 (10 μM) followed by stimulation with LPS (1 μg/mL) for 10 minutes. A representative blot (left panel) and densitometric analysis of 4 experiments (right panel) are shown. (B) BMMs obtained by culturing of bone marrow cells in medium containing (20 ng/mL) recombinant mouse M-CSF. Cells were pretreated for 30 minutes with the p38 inhibitor SB203580 (10 μM) or DMSO (vehicle) followed by stimulation with LPS (1 μg/mL) for 10 minutes; phosphorylated ERK1/2 was determined by Western blotting. (C) LPS-induced TF induction in BMMs is negatively regulated by p38 in wild-type, but not TFΔCT, BMMs. BMMs were stimulated for 6 hours in the presence or absence of the indicated inhibitors at concentrations as described for panel A. TF mRNA induction was quantified by real-time PCR and TF activity was quantified by one stage clotting assay. *denotes statistically significant differences relative to wild-type control.
Macrophage differentiation was carried out in conditioned medium from L929 cells that may contain factors that were responsible for the observed phenotypic changes of TFΔCT macrophages. To exclude this possibility, we generated BMMs using recombinant macrophage colony-stimulating factor. Figure 6B shown that the p38-dependent, differential ERK1/2 phosphorylation between wild-type and TFΔCT macrophages was independent of an unidentified factor in the L929 cell–conditioned medium. Inhibition of p38 also increased TF induction in wild-type BMMs, but TF expression in TFΔCT BMMs was similar in the presence or absence of p38 inhibitor (Figure 6C). Taken together, these experiments identify p38-dependent regulation of ERK1/2 as the underlying mechanism by which the TF cytoplasmic domain specifically controls the expression levels of TF in LPS-stimulated macrophages.
Discussion
These experiments provide new insight into functions of the TF cytoplasmic domain in the procoagulant response during endotoxemia in vivo. TLR4 engagement activates various members of the mitogen-activated protein kinase (MAPK) family, including ERK1/2, c-Jun amino terminal kinases (JNKs), and p38 in monocytes leading to TLR4-mediated induction of inflammatory mediators and TF. TF gene induction in monocytes has been extensively studied and shown to involve both NF-κB and MAP kinase pathways.28 Macrophages, unlike endothelial cells and monocytes, up-regulate TF during maturation and constitutively express TF.29 Although the TF cytoplasmic domain per se does not regulate the basal TF expression, it imposes regulatory control over TLR4-mediated, but not interferon γ-mediated, TF induction in macrophages.
ERK1/2 phosphorylation induced by TLR4 was found to be entirely dependent on PI3 kinase activation in macrophages. In monocytes and endothelial cells, TF expression in response to endotoxin or angiogenic growth factors is known to be negatively regulated by PI3 kinase signaling.40,41 Our data do not exclude that PI3 kinase activation also regulates TF induction in macrophages. However, the requirement for PI3 kinase to activate ERK1/2 precludes further experiments to address this question, because in macrophages TF up-regulation is entirely dependent on ERK1/2 similar to other cell types. The presented results identify a regulatory pathway in which the TF cytoplasmic domain suppresses ERK1/2 phosphorylation through a p38 pathway. Negative regulation of ERK1/2 activation by p38 has also been documented for growth factor,42 G-protein–coupled receptor,43,44 cytokine receptor,45 and innate immune46 signaling.
Our data indicate that the TF cytoplasmic domain may act as a scaffold to orchestrate a crosstalk between p38 and ERK1/2 signaling. Ott et al have shown that ligation of TF can stimulate the small GTPase rac and p38 in a TF cytoplasmic domain–dependent manner in cancer cells.25 The observed migratory response in their experimental system may reflect cytosolic sequestration of ERK1/2 following simultaneous activation of p38 and ERK1/2. Consistently, TFΔCT smooth muscle cells also show reduced cell migration in vitro.47 However, the TF cytoplasmic domain can negatively regulate migration dependent on certain integrins, and cell migration is stimulated when TF cytoplasmic domain phosphorylation is induced, for example, by TF-VIIa–mediated PAR2 signaling.24 The present study shows that signaling crosstalk of the TF cytoplasmic domain is not restricted to regulation of cell migration, but that the TF cytoplasmic domain can control transcriptional responses as well.
Maturation of monocytes into macrophages induces diverse phenotypic changes, including the up-regulation of PAR2.30 Deletion of PAR2 showed that the TF cytoplasmic domain influences TLR4 signaling independent of PAR2, providing evidence for an autonomous signaling function of TF to suppress in a p38-dependent manner ERK1/2 activation and TF up-regulation. Negative regulatory roles of the TF cytoplasmic domain were also demonstrated in angiogenesis. Because diabetic neoangiogenesis showed colocalization of phosphorylated TF with PAR2,26 PAR2 signaling may serve as an upstream signal to turn off the regulatory control of TF through phosphorylation. Our data indicate that one immediate downstream effect of such loss of regulatory control in cells exposed to LPS is the up-regulation of TF, which conversely could amplify TF-dependent PAR2 signaling responses.
Loss of the TF cytoplasmic domain leads to an enhanced coagulopathy in a model of severe endotoxemia. Peritoneal macrophages from LPS-stimulated mice showed enhanced TF expression independent of PAR2 expression, validating in vivo findings with in vitro–differentiated, bone marrow–derived macrophages. Although coagulation activation was exacerbated in TFΔCT mice, inflammatory cytokine production was not increased in early stages of the endotoxin challenge model. In addition, attenuation of coagulation had no effect on the initial cytokine responses in this model. Taken together, these data show that the TLR-induced cytokine storm is not dependent on coagulation or TF. Rather, our results indicate that the TF pathway plays roles in regulating the subsequent inflammatory response and contributes to the resolution and rebalancing of severe systemic inflammation.
Robust coagulation activation improves sepsis outcome in heterozygous factor V Leiden carriers,21 but not thrombomodulin mutant mice,48 implicating the protein C pathway. Although enhanced activation of the protein C pathway and resulting barrier and cytoprotective signaling may have contributed to the improved survival of TFΔCT mice, our data show that survival was not changed when coagulation was attenuated pharmacologically to levels well below wild-type mice during the critical period of enhanced coagulation activation in this strain. These data indicate that the elevated levels of TF did not protect through increased coagulation, but rather led to enhanced PAR2 signaling by currently unresolved pathways. Consistent with this concept, our preliminary data show that the survival benefit of TFΔCT mice is abolished by PAR2 deficiency.
The presented in vitro and in vivo data identify macrophages as a critical cell type in which the TF cytoplasmic domain plays a regulatory role in TLR4-dependent TF induction. Whether macrophages are the sole cell type that causes the enhanced circulating TAT level in this model remains to be addressed. In particular, in late-stage endotoxemia, endothelial cells and other myeloid inflammatory cells in the spleen express TF,49 and these cells may contribute to the sustained coagulation activation at later stages when the cytokine profiles indicate that activation of the innate immune system is attenuated in TFΔCT mice. This study elucidates a regulatory pathway by which the TF cytoplasmic domain differentially controls intravascular coagulation and inflammation during endotoxemia.
Acknowledgments
This study was supported by NIH grants HL77753 and HL78614 (W.R.), and stipend NI 540/2-1 from the Deutsche Forschungsgemeinschaft (F.N.).
We thank Cindi Biazak, Pablito Tejada, Jennifer Royce, and Rachael Deleers for their excellent technical assistance, and Drs Peter Carmeliet and Patricia Andrade-Gordon for providing TFΔCT and PAR2−/− mice, respectively.
Footnotes
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: J.A. designed and performed experiments, analyzed and interpreted data, and wrote the paper; F.N. performed research and analyzed data; T.K., Y.K.L., and G.B. performed research; J.H.M. provided vital reagents; W.R. supervised the project and wrote the paper. J.A. and F.N. contributed equally to this study.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Wolfram Ruf, Department of Immunology, SP258, The Scripps Research Institute, 10550 North Torrey Pines Road, La Jolla, CA 92037; e-mail: ruf@scripps.edu.
References
- 1.Riewald M, Ruf W. Science review: role of coagulation protease cascades in sepsis. Crit Care. 2003;7:123–129. doi: 10.1186/cc1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ruf W. Emerging roles of tissue factor in viral hemorrhagic fever. Trends Immunol. 2004;25:461–464. doi: 10.1016/j.it.2004.07.002. [DOI] [PubMed] [Google Scholar]
- 3.Creasey AA, Chang ACK, Feigen L, et al. Tissue factor pathway inhibitor (TFPI) reduces mortality from E. coli septic shock. J Clin Invest. 1993;91:2850–2860. doi: 10.1172/JCI116529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Taylor FB, Jr, Chang ACK, Peer G, et al. Active site inhibited factor VIIa (DEGR VIIa) attenuates the coagulant and interleukin-6 and -8, but not tumor necrosis factor, responses of the baboon to LD100 Escherichia coli. Blood. 1998;91:1609–1615. [PubMed] [Google Scholar]
- 5.Taylor FB, Jr, Chang A, Ruf W, et al. Lethal E. coli septic shock is prevented by blocking tissue factor with monoclonal antibody. Circ Shock. 1991;33:127–134. [PubMed] [Google Scholar]
- 6.Welty-Wolf KE, Carraway MS, Ortel TL, et al. Blockade of tissue factor-factor X binding attenuates sepsis-induced respiratory and renal failure. Am J Physiol Lung Cell Mol Physiol. 2006;290:L21–L31. doi: 10.1152/ajplung.00155.2005. [DOI] [PubMed] [Google Scholar]
- 7.Taylor FB, Jr, Chang ACK, Peer GT, et al. DEGR-factor Xa blocks disseminated intravascular coagulation initiated by Escherichia coli without preventing shock or organ damage. Blood. 1991;78:364–368. [PubMed] [Google Scholar]
- 8.Randolph MM, White GL, Kosanke SD, et al. Attenuation of tissue thrombosis and hemorrhage by ala-TFPI does not account for its protection against E. coli: a comparative study of treated and untreated non-surviving baboons challenged with LD100 E. coli. Thromb Haemost. 1998;79:1048–1053. [PubMed] [Google Scholar]
- 9.Pawlinski R, Pedersen B, Schabbauer G, et al. Role of tissue factor and protease activated receptors in a mouse model of endotoxemia. Blood. 2003;103:1342–1347. doi: 10.1182/blood-2003-09-3051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Camerer E, Cornelissen I, Kataoka H, et al. Roles of protease-activated receptors in a mouse model of endotoxemia. Blood. 2006;107:3912–3921. doi: 10.1182/blood-2005-08-3130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xu H, Ploplis VA, Castellino FJ. A coagulation factor VII deficiency protects against acute inflammatory responses in mice. J Pathol. 2006;210:488–496. doi: 10.1002/path.2073. [DOI] [PubMed] [Google Scholar]
- 12.Franco RF, De Jonge E, Dekkers PEP, et al. The in vivo kinetics of tissue factor messenger RNA expression during human endotoxemia: relationship with activation of coagulation. Blood. 2000;96:554–559. [PubMed] [Google Scholar]
- 13.Schoenmakers SH, Groot AP, Florquin S, Reitsma PH, Spek CA. Blood cell-derived tissue factor influences host response during murine endotoxemia. Blood Cells Mol Dis. 2004;32:325–333. doi: 10.1016/j.bcmd.2003.12.002. [DOI] [PubMed] [Google Scholar]
- 14.Drake TA, Morrissey JH, Edgington TS. Selective cellular expression of tissue factor in human tissues. Am J Pathol. 1989;134:1087–1097. [PMC free article] [PubMed] [Google Scholar]
- 15.Gregory SA, Morrissey JH, Edgington TS. Regulation of tissue factor gene expression in the monocyte procoagulant response to endotoxin. Mol Cell Biol. 1989;9:2752–2755. doi: 10.1128/mcb.9.6.2752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Alm A-K, Norström E, Sundelin J, Nystedt S. Stimulation of proteinase activated receptor-2 causes endothelial cells to promote blood coagulation in vitro. Thromb Haemost. 1999;81:984–988. [PubMed] [Google Scholar]
- 17.Minami T, Sugiyama A, Wu SQ, et al. Thrombin and phenotypic modulation of the endothelium. Arterioscler Thromb Vasc Biol. 2004;24:41–53. doi: 10.1161/01.ATV.0000099880.09014.7D. [DOI] [PubMed] [Google Scholar]
- 18.Takeya H, Gabazza EC, Aoki S, Ueno H, Suzuki K. Synergistic effect of sphingosine 1-phosphate on thrombin-induced tissue factor expression in endothelial cells. Blood. 2003;102:1693–1700. doi: 10.1182/blood-2002-11-3607. [DOI] [PubMed] [Google Scholar]
- 19.Riewald M, Petrovan RJ, Donner A, Mueller BM, Ruf W. Activation of endothelial cell protease activated receptor 1 by the protein C pathway. Science. 2002;296:1880–1882. doi: 10.1126/science.1071699. [DOI] [PubMed] [Google Scholar]
- 20.Esmon CT. Interactions between the innate immune and blood coagulation systems. Trends Immunol. 2004;25:536–542. doi: 10.1016/j.it.2004.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kerlin BA, Yan SC, Isermann BH, et al. Survival advantage associated with heterozygous Factor V Leiden mutation in patients with severe sepsis and in mouse endotoxemia. Blood. 2003;102:3085–3092. doi: 10.1182/blood-2003-06-1789. [DOI] [PubMed] [Google Scholar]
- 22.Joyce DE, Gelbert L, Ciaccia A, DeHoff B, Grinnell BW. Gene expression profile of antithrombotic protein C defines new mechanisms modulating inflammation and apoptosis. J Biol Chem. 2001;276:11199–11203. doi: 10.1074/jbc.C100017200. [DOI] [PubMed] [Google Scholar]
- 23.Sharma L, Melis E, Hickey MJ, et al. The cytoplasmic domain of tissue factor contributes to leukocyte recruitment and death in endotoxemia. Am J Pathol. 2004;165:331–340. doi: 10.1016/S0002-9440(10)63300-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dorfleutner A, Hintermann E, Tarui T, Takada Y, Ruf W. Crosstalk of integrin α3β1 and tissue factor in cell migration. Mol Biol Cell. 2004;15:4416–4425. doi: 10.1091/mbc.E03-09-0640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ott I, Weigand B, Michl R, et al. Tissue factor cytoplasmic domain stimulates migration by activation of the GTPase Rac1 and the mitogen-activated protein kinase p38. Circulation. 2005;111:349–355. doi: 10.1161/01.CIR.0000153333.52294.42. [DOI] [PubMed] [Google Scholar]
- 26.Belting M, Dorrell MI, Sandgren S, et al. Regulation of angiogenesis by tissue factor cytoplasmic domain signaling. Nat Med. 2004;10:502–509. doi: 10.1038/nm1037. [DOI] [PubMed] [Google Scholar]
- 27.Aderem A, Ulevitch RJ. Toll-like receptors in the induction of the innate immune response. Nature. 2000;406:782–787. doi: 10.1038/35021228. [DOI] [PubMed] [Google Scholar]
- 28.Guha M, Mackman N. LPS induction of gene expression in human monocytes. Cell Signal. 2001;13:85–94. doi: 10.1016/s0898-6568(00)00149-2. [DOI] [PubMed] [Google Scholar]
- 29.van den Eijnden MM, Steenhauer SI, Reitsma PH, Bertina RM. Tissue factor expression during monocyte-macrophage differentiation. Thromb Haemost. 1997;77:1129–1136. [PubMed] [Google Scholar]
- 30.Colognato R, Slupsky JR, Jendrach M, et al. Differential expression and regulation of protease-activated receptors in human peripheral monocytes and monocyte-derived antigen-presenting cells. Blood. 2003;102:2645–2652. doi: 10.1182/blood-2002-08-2497. [DOI] [PubMed] [Google Scholar]
- 31.Damiano BP, Cheung WM, Santulli RJ, et al. Cardiovascular responses mediated by protease-activated receptor- 2 (PAR-2) and thrombin receptor (PAR-1) are distinguished in mice deficient in PAR-2 or PAR-1. J Pharmacol Exp Ther. 1999;288:671–678. [PubMed] [Google Scholar]
- 32.Melis E, Moons L, De Mol M, et al. Targeted deletion of the cytosolic domain of tissue factor in mice does not affect development. Biochem Biophys Res Commun. 2001;286:580–586. doi: 10.1006/bbrc.2001.5425. [DOI] [PubMed] [Google Scholar]
- 33.Fleit SA, Fleit HB, Zolla-Pazner S. Culture and recovery of macrophages and cell lines from tissue culture-treated and -untreated plastic dishes. J Immunol Methods. 1984;68:119–129. doi: 10.1016/0022-1759(84)90142-x. [DOI] [PubMed] [Google Scholar]
- 34.Mosnier LO, Griffin JH. Inhibition of staurosporine-induced apoptosis of endothelial cells by activated protein C requires protease-activated receptor-1 and endothelial cell protein C receptor. Biochem J. 2003;373:65–70. doi: 10.1042/BJ20030341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Feistritzer C, Riewald M. Endothelial barrier protection by activated protein C through PAR1-dependent sphingosine 1-phosphate receptor-1 crossactivation. Blood. 2005;105:3178–3184. doi: 10.1182/blood-2004-10-3985. [DOI] [PubMed] [Google Scholar]
- 36.Ahamed J, Ruf W. Protease-activated receptor 2-dependent phosphorylation of the tissue factor cytoplasmic domain. J Biol Chem. 2004;279:23038–23044. doi: 10.1074/jbc.M401376200. [DOI] [PubMed] [Google Scholar]
- 37.MacMicking J, Xie QW, Nathan C. Nitric oxide and macrophage function. Annu Rev Immunol. 1997;15:323–350. doi: 10.1146/annurev.immunol.15.1.323. [DOI] [PubMed] [Google Scholar]
- 38.Moon DK, Geczy CL. Recombinant IFN-gamma synergizes with lipopolysaccharide to induce macrophage membrane procoagulants. J Immunol. 1988;141:1536–1542. [PubMed] [Google Scholar]
- 39.Guha M, O'Connell MA, Pawlinski R, et al. Lipopolysaccharide activation of the MEK/ERK1/2 pathway in human monocytic cells mediates tissue factor and tumor necrosis factor α expression by inducing Elk-1 phosphorylation and Egr-1 expression. Blood. 2001;98:1429–1439. doi: 10.1182/blood.v98.5.1429. [DOI] [PubMed] [Google Scholar]
- 40.Guha M, Mackman N. The PI3K-Akt pathway limits LPS activation of signaling pathways and expression of inflammatory mediators in human monocytic cells. J Biol Chem. 2002;277:32124–32132. doi: 10.1074/jbc.M203298200. [DOI] [PubMed] [Google Scholar]
- 41.Blum S, Issbruker K, Willuweit A, et al. An inhibitory role of the phosphatidylinositol 3-kinase-signaling pathway in vascular endothelial growth factor-induced tissue factor expression. J Biol Chem. 2001;276:33428–33434. doi: 10.1074/jbc.M105474200. [DOI] [PubMed] [Google Scholar]
- 42.Rice AB, Ingram JL, Bonner JC. p38 mitogen-activated protein kinase regulates growth factor-induced mitogenesis of rat pulmonary myofibroblasts. Am J Respir Cell Mol Biol. 2002;27:759–765. doi: 10.1165/rcmb.2002-0070OC. [DOI] [PubMed] [Google Scholar]
- 43.Kintscher U, Bruemmer D, Blaschke F, Unger T, Law RE. p38 MAP kinase negatively regulates angiotensin II-mediated effects on cell cycle molecules in human coronary smooth muscle cells. Biochem Biophys Res Commun. 2003;305:552–556. doi: 10.1016/s0006-291x(03)00802-7. [DOI] [PubMed] [Google Scholar]
- 44.Kusuhara M, Takahashi E, Peterson TE, et al. p38 Kinase is a negative regulator of angiotensin II signal transduction in vascular smooth muscle cells: effects on Na+/H+ exchange and ERK1/2. Circ Res. 1998;83:824–831. doi: 10.1161/01.res.83.8.824. [DOI] [PubMed] [Google Scholar]
- 45.Boing I, Stross C, Radtke S, et al. Oncostatin M-induced activation of stress-activated MAP kinases depends on tyrosine 861 in the OSM receptor and requires Jak1 but not Src kinases. Cell Signal. 2006;18:50–61. doi: 10.1016/j.cellsig.2005.03.015. [DOI] [PubMed] [Google Scholar]
- 46.Bhattacharyya A, Pathak S, Kundu M, Basu J. Mitogen-activated protein kinases regulate Mycobacterium avium-induced tumor necrosis factor-alpha release from macrophages. FEMS Immunol Med Microbiol. 2002;34:73–80. doi: 10.1111/j.1574-695X.2002.tb00605.x. [DOI] [PubMed] [Google Scholar]
- 47.Ott I, Michaelis C, Schuermann M, et al. Vascular remodeling in mice lacking the cytoplasmic domain of tissue factor. Circ Res. 2005;97:293–298. doi: 10.1161/01.RES.0000177533.48483.12. [DOI] [PubMed] [Google Scholar]
- 48.Weiler H, Lindner V, Kerlin B, et al. Characterization of a mouse model for thrombomodulin deficiency. Arterioscler Thromb Vasc Biol. 2001;21:1531–1537. doi: 10.1161/hq0901.094496. [DOI] [PubMed] [Google Scholar]
- 49.Drake TA, Cheng J, Chang A, Taylor FB., Jr Expression of tissue factor, thrombomodulin, and E-selectin in baboons with lethal E.coli sepsis. Am J Pathol. 1993;142:1458–1470. [PMC free article] [PubMed] [Google Scholar]