Abstract
This study examined the effect of attention in young infants on the saccadic localization of peripheral stimuli. Infants ranging in age from 5 to 14 weeks were tested using a peripheral stimulus detection paradigm. The presence of a central fixation stimulus decreased detection probability, particularly if attention was engaged with the central stimulus. Peripheral stimulus localization usually was accomplished with a single eye movement. When localization was accomplished by multiple eye movements, corrective saccades occurred most frequently and fixed-amplitude hypometric saccades occurred less frequently. A decrease in the slope of the linear component of the main sequence was found from 5 to 11 weeks of age, and this decrease was independent of attention.
From birth, human infants have the ability to use their eyes to gather information about the world. For that reason, the visual system in the human infant has been widely studied. One area of this research that has received a good deal of attention in infants is peripheral stimulus localization. Some of this research has been descriptive in nature, focusing on performance characteristics such as percent localization (Aslin & Salapatek, 1975; Butcher, Kalverboer, & Geuze, 2000; Finlay & Ivinskis, 1982, 1984; Goldberg, Maurer, & Lewis, 1997; Harris & MacFarlane, 1974; Hicks & Richards, 1998; Richards, 1997) or latency to look away from a central stimulus to a peripheral target (Braddick, Atkinson, & Hood, 1996; Hicks & Richards, 1998; Hood & Atkinson, 1993; Tronick, 1972). Other studies have evaluated accuracy and quantitative characteristics (i.e., main sequence) of the eye movements used in peripheral stimulus localization (Aslin & Salapatek, 1975; Hainline, Turkel, Abromov, Lemerise, & Harris, 1984; Richards & Hunter, 1997; Salapatek, Aslin, Simonson, & Pulos, 1980). Only a few studies (Hicks & Richards, 1998; Richards, 1997; Richards & Hunter, 1997) have assessed the effect of attention on these characteristics.
One important factor found to affect performance characteristics (i.e., probability or latency) of peripheral stimulus localization is the presence of a central stimulus. Many studies indicate that infants are slower and less likely to detect a peripheral target if a competing central stimulus is present and fixation is directed toward this stimulus. For example, Aslin and Salapatek (1975) reported higher proportions of peripheral stimulus localization when the peripheral stimulus replaced the central stimulus (replacement condition) than when the peripheral stimulus was presented in addition to the central stimulus (addition condition). Also, in the addition condition, the latency of the eye movement toward the peripheral target was increased. Early research in this area interpreted the decrease in localization percentages as a decrease in the size of the effective visual field (Harris & MacFarlane, 1974) in the presence of a competing central stimulus.
Findings from more recent studies (Hicks & Richards, 1998; Richards, 1997; Richards & Hunter, 1997) indicate that it is not just the presence of a central stimulus that is responsible for these effects. Rather, attention must be engaged toward the central stimulus for central stimulus presence to affect peripheral stimulus detection or localization latency. For example, Richards and Hunter (1997) used heart rate (HR) changes in 14-, 20-, and 26-week-old infants as an index of attention to the central stimulus. It has been postulated that phasic changes in HR correspond to different levels of attention engagement (Berg & Richards, 1997; Richards & Casey, 1992; Richards & Hunter, 1998). Attention is thought to be engaged when HR decelerates below prestimulus levels and remains at low levels (sustained attention). Attention is hypothesized to be unengaged when the HR returns to the prestimulus level but fixation continues on the stimulus (attention termination). They found that infants were least likely to localize a peripheral target when the center stimulus remained on and attention was engaged. Theoretical models based on adult research have suggested that attention blocks saccades to peripheral targets when it is engaged by a central stimulus (Fischer, 1986; Fischer & Breitmeyer, 1987). This is true in infants, however, only when attention is engaged by the central stimulus. When attention was not engaged by the central stimulus and a peripheral stimulus was presented, peripheral stimulus localization on the addition trials occurred at probabilities similar to replacement trials (e.g., Richards, 1997).
One goal of this study was to investigate the effect of attention (as indicated by HR phases) to a central stimulus on these performance characteristics in 5- to 14-week-old infants. The definition of attention in this model has been primarily used with infants of 14 weeks and older (e.g., Richards, 1997; Richards & Hunter, 1997). HR was used as an index of attention engagement in only two studies with 8-week-old infants (Hicks & Richards, 1998; Richards, 1989). This study is a first attempt to systematically apply HR-defined attention phases to the age range below 8 weeks. If the visual attention system is functioning in the ages tested in this study, peripheral stimulus localization should occur with the highest probabilities under conditions in which attention is not engaged with the central stimulus (e.g., preattention or attention termination; Richards, 1987, 1997). Attention engagement with the central stimulus, if operating at these ages, should attenuate peripheral stimulus localization. Because sustained attention increases with age, the older infants may show poorer accuracy in peripheral target localization during sustained attention than the younger infants, poorer accuracy compared to their own performance in inattentive conditions, or both.
The quantitative characteristics of eye movements used in peripheral stimulus localization by young infants have been the focus of fewer studies. Infants, like adults, use saccades to localize peripheral targets. These eye movements have been reported to be grossly hypometric in infants. Aslin and Salapatek (1975) reported that 1- and 2-month-old infants often localize targets with a series of steplike, fixed-amplitude, hypometric saccades. Approximately 60% of the saccades were of this type. Other studies have differed from the Aslin and Salapatek study on the existence and proportion of these hypometric saccades (Hainline & Abramov, 1985; Hainline et al., 1984; Richards & Hunter, 1997). Richards and Hunter (1997) found only 21% of the multiple eye movements of 14- to 26-week-old infants to be of the hypometric, fixed-amplitude, steplike variety, compared with the findings of Aslin and Salapatek (1975) that about 60% of 4- and 9-week-old infants’ eye movements were hypometric. Hainline and colleagues (1984) reported less than 5% of the eye movements in their study met this criterion. In addition, Richards and Hunter (1997) reported that the large majority (79%) of multiple saccades sequences consisted of a large first saccade, followed by a small corrective saccade that localized the target. The differences between the Aslin and Salapatek (1975) and the Richards and Hunter (1997) findings may be due to the older participants in the latter study. This study used the procedures from the Richards and Hunter (1997) study with infants ranging in age from 5 to 14 weeks to examine this quantitative characteristic of saccadic eye movements at earlier age ranges.
As is the case with accuracy, the relation between saccade velocity and amplitude (or duration and amplitude), the main sequence, has been the focus of few published studies involving infants (Ashmead, 1984; Hainline et al., 1984; Richards & Hunter, 1997). The main sequence is a lawful relationship between the velocity of a saccade and its amplitude (or the duration of the saccade and its amplitude; Bahill, Clark, & Stark, 1975). The main sequence is represented by a particular function for which the relation between maximum saccade velocity and saccade amplitude is linear up to about 20°, with maximum velocity leveling off near 20° to 25°. It is the direct result of the firing rate and firing duration of brain stem motor neurons connected to the eye muscles (Moschovakis & Highstein, 1994). During the first 2 months of life, saccadic eye movements are thought to be reflexive in nature and the result of this system (with input from the superior colliculus; Johnson, 1990, 1995; Richards & Hunter, 1998). Because these structures are thought to be well-developed at birth, changes in the quantitative parameters relating saccade velocity to saccade amplitude would not be expected. There are some indications, however, that changes do occur both with age and under differing attentional demands. For example, Hainline and colleagues (1984) recorded eye movements with corneal reflection procedures while infants viewed complex visual scenes. They found adultlike main sequence relations in infants during scanning of interesting (i.e., complex) visual scenes, but a decrease in the peak, velocity, and amplitude slope for uninteresting (i.e., simple) scenes. This decreased slope represents slower speeds per degree of saccade eccentricity and was interpreted as a lack of interest. Their study, however, did not specifically investigate the attentional factors involved.
Richards and Hunter (1997) recorded eye movements using electro-oculargram (EOG) technology while monitoring HR as an index of attention. They found a strong relation between peak velocity and amplitude of the localizing saccades during a prestimulus period using a simple, nonpatterned stimulus array. There were no age differences in this main sequence relation, and they assumed that adultlike relations characterized the velocity–amplitude relations under these conditions. However, when attention was maximally engaged (during HR deceleration) to a central stimulus unusual velocity–amplitude relations for the peripheral stimulus localization were found. However, unlike the main sequence relations found by Hainline and colleagues (1984), there was a slower peak velocity when attention was maximally engaged relative to that found during prestimulus periods when attention was not yet engaged. These attention differences were found in the 20- and 26-week-old infants but not in the 14-week-olds.
A second goal of this study was to further examine these quantitative eye movement characteristics of infants from 5 to 14 weeks of age. In this regard we were interested in the proportion and characteristics of multiple saccades used to localize a peripheral stimulus, particularly with respect to fixed amplitude hypometric saccades. This would allow a direct comparison between the effect of attention on this eye movement characteristic and the ages used in Aslin and Salapatek’s (1975) study of hypometric saccades. If attention is a factor in determining the accuracy of these eye movements, then differential effects might be found between the attentive and inattentive conditions. However, this is not likely the case, because Richards and Hunter (1997) found no differences in these conditions in older infants with respect to accuracy. The effects of age and attention on the parameters of the main sequence also were also examined. This extends the study of the main sequence relation between saccade velocity and amplitude down to younger ages than tested previously (e.g., 14- to 26-week-olds; Richards & Hunter, 1997) and may help clarify the role of attention on main sequence relations. These age groups are more in keeping with those infants tested in the Hainline and colleagues (1984) study. If attention engagement differentially affects the main sequence relation, then differences should be found in the attentive and inattentive conditions. Further, as the effect of attention increases with age (as was indicated in Richards & Hunter, 1997), differences in the main sequence relation may also emerge.
Infants were tested at 5, 8, 11, and 14 weeks of age. These ages were chosen to cover a range of ages in which the effect of attention on peripheral stimulus localization has not previously been studied extensively, and to find a range of ages comparable to the ages of the infants in the Aslin and Salapatek (1975) study. An interesting visual stimulus was presented in the infant’s focal visual field. Peripheral stimuli at eccentricities of 10°, 15°, 20° or 25° were presented after a delay. The delays were designed to represent attention engagement to the central stimulus (e.g., 2-sec delay and delay until HR showed a significant deceleration) or inattentive fixation (e.g., return of HR to its prestimulus level after a significant HR deceleration). We also included a condition (prestimulus) for which a blinking dot attracted fixation and the peripheral stimulus was presented without the presentation of a visual stimulus in the focal visual field, and a condition for which the focal and visual stimuli were presented simultaneously (immediate). Infant attention is not engaged by the blinking dot in the prestimulus condition, nor has had a chance to become engaged in the immediate condition. Therefore, these two conditions represent a period of time before attention has been engaged (Hicks & Richards, 1998; Richards, 1997; Richards & Hunter, 1997). The EOG was used to measure saccade amplitude and velocity. The EOG recording provided the opportunity to study the main sequence relation that has not been studied in infants at this age range and quantify the saccade amplitude on multiple saccades to study hypometric and corrective saccades in this age range.
METHODS
Participants
Infants were recruited from the Columbia, South Carolina, newspaper. Participants had to be full term, which was defined as having a birth weight of more than 2,500 g, and a gestational age of at least 38 weeks based on the mother’s report of her last menstrual cycle. No infants had acute or chronic pre- or perinatal medical complications, and all were in good health at the time of the recording session. There were 120 infants sampled cross-sectionally at 5 (n = 27, M = 36.915 days, SD = 2.42), 8 (n = 32, M = 57.97 days, SD = 4.97), 11 (n = 31, M = 78.14 days, SD = 2.83), and 14 (n = 30, M = 100.32 days, SD = 3.73) weeks postnatal age. Five to 7 infants in each age group were randomly assigned to one of five testing conditions—prestimulus, immediate, 2-sec, HR deceleration, and HR acceleration. These testing conditions are explained in greater detail in the “Procedures” section. An additional 5 infants were tested at 8 weeks as a control group. Their data were used to verify there was no effect of infants being tested in a car seat (5-week-olds) versus a parent’s lap. Fewer infants were tested at 5 weeks of age due to the omission of one of the delay testing conditions (HR return to prestimulus level). An additional 74 infants were tested (26 at 5 weeks, 19 at 8 weeks, 10 at 11 weeks, and 14 at 14 weeks). Of these 74 infants, 13 were excluded because of equipment problems and 56 were excluded because of fussiness or inability to remain alert.
Apparatus
Infants who were 5 weeks of age were placed in an infant car seat secured to a chair approximately 51 cm from the center of a black-and-white 49-cm (19-in.) monitor. Infants whose postnatal age was 8, 11, or 14 weeks were held on his or her parent’s lap approximately 51 cm from the center of the monitor. The 5-week-olds had soft pillows on each side of the head to restrain head movement, and the parent holding the older children slightly restrained the infants’ head movement so that large head movements did not occur. The plane of the TV was parallel to the infant’s eyes and subtended a 44° visual angle. The area surrounding the TV and infants was covered in black cloth. A video camera was centered above the TV to allow an observer in an adjacent room to judge infant fixations and allowed for recording of the sessions with a time code to synchronize physiological and experimental information for analysis.
The first fixation stimulus was a 1° blinking square and was presented on the TV monitor within 15° of the center. It was used to attract the infant’s initial fixation to the TV monitor. The second fixation stimulus consisted of 10° square patterns presented centered at the same location as the first fixation stimulus. The central stimulus was one of 15 dynamic computer-generated patterns (e.g., a rotating star and a changing face) that had been used in previous studies and had been found to elicit similar patterns of HR response and fixation duration in infants at similar ages (e.g., 8–26 weeks of age; Hicks & Richards, 1998; Richards, 1987, 1989, 1997; Richards & Hunter, 1997). The peripheral stimulus was a rectangle presented on the same TV as the first and second fixation stimuli. The rectangle had a constantly changing sine wave grating and was 2° horizontal width and 6° vertical width. The peripheral stimulus’ innermost side was at 10°, 15°, 20°, or 25° from the center of the central stimulus.
Measurement and Quantification of Physiological Variables
The electrocardiogram (ECG) was recorded with Ag-AgCl electrodes placed on the infant’s chest. The ECG was digitized online at 1000 Hz (1 msec). The R-wave of the ECG was identified, and interbeat intervals were computed within 1 msec of the R-wave occurrence for the online evaluation of HR changes. A significant HR deceleration was indicated when online evaluation detected five successive beats, with interbeat intervals each longer than the median of the five prestimulus beats (i.e., sustained attention; Richards, 1987, 1997; Richards & Casey, 1991, 1992). The return of HR to its prestimulus level was indicated when online evaluation determined that (a) a significant HR deceleration had occurred and (b) HR returned to its prestimulus level, defined as five beats with interbeat intervals shorter than median of the five prestimulus beats (i.e., attention termination; Richards, 1987, 1997; Richards & Casey, 1991).
The EOG was recorded with 6-mm Ag-AgCl electrodes placed on the outer canthus of each eye using disposable electrode collars. The EOG was digitized online at 1000 Hz, amplified at 2K, and a DC recording was made. Using an algorithm presented in Matsuoka and Ueda (1986; Matsuoka & Harato, 1983; see Richards & Holley, 1999; Richards & Hunter, 1997), saccades were separated from the composite EOG record. This algorithm uses a third-order differential equation to identify saccades. A computer-based editing program was used to confirm the algorithm identification and select the onset and offset of the saccade. The EOG amplitude (μV) at the beginning, middle, and end of the saccades was also recorded.
Because the EOG measures the eye position relative to the head, the rotation of the eyes in the orbit is proportional to the EOG signal. The degree of the eye rotation in the orbit necessary to fixate the blinking dot or peripheral stimulus was calculated, along with the difference in EOG μV values for these fixations. The amplitude of saccades was calibrated by using the fixation on the blinking dot (1° horizontal) and the peripheral stimulus (3° horizontal) as fixation points instead of a separate calibration procedure (see Richards & Holley, 1999; Richards & Hunter, 1997). The degree of difference between fixation on the blinking dot and peripheral stimulus was calculated on each peripheral stimulus trial. A more detailed discussion of this calibration procedure may be found in Richards and Holley (1999) or Richards and Hunter (1997).
Procedure
The experimental trials consisted of presentation of the blinking dot, followed by presentation of the central stimulus, alone or with the peripheral stimulus. There were two within-subjects factors. The addition–replacement factor had two levels. Addition trials consisted of the presentation of the peripheral stimulus in addition to the central stimulus. Replacement trials consisted of presentation of the peripheral stimulus replacing the central stimulus. There were an equal number of replacement and addition trials. Peripheral stimulus eccentricity was the second within-subjects factor. Each participant received peripheral stimulus presentations at nominal eccentricities of 10°, 15°, 20°, and 25°. A fifth level of this condition was a control trial in which no peripheral stimulus was presented but the central stimulus remained on (addition) or was turned off (replacement). The five levels of the eccentricity factor were each done as addition and replacement trials, resulting in 10 trial types. These trial types were presented in 10-trial blocks. Each infant received a minimum of two blocks of trials and a maximum of three blocks of trials.
There was a between-subject factor in this experiment. The peripheral stimulus was presented to each participant in only one of five different conditions defined by predetermined delays between the onset of the central stimulus and the onset of the peripheral stimulus (Table 1). In the prestimulus condition, the initial blinking dot was turned off (replacement) or left on (addition) and a peripheral stimulus was presented at in the periphery for 2 sec. For the other conditions, a central stimulus was presented in place of the blinking dot, and the peripheral stimulus was presented for 2 sec at the predetermined delays. The immediate condition consisted of a central stimulus with the peripheral stimulus (addition), or the central stimulus came on briefly (<100 msec) and was replaced by the peripheral stimulus (replacement). The 2-sec condition consisted of a 2-sec delay between the onset of the central stimulus and the replacement of or addition to the central stimulus with the peripheral stimulus. The HR deceleration condition consisted of the central stimulus remaining on until HR deceleration and then the replacement or addition of the central stimulus with the peripheral stimulus. The HR return to prestimulus level condition required that a HR deceleration occur and then HR returned to its prestimulus level. If the infant looked toward the peripheral stimulus while it was on (or within 1 sec of its being turned off), the central stimulus was turned off and the peripheral stimulus remained on for 3 sec. Otherwise, the central stimulus remained on for 2 sec after the offset of the peripheral stimulus. A minimum of 5 sec interstimulus interval occurred between each trial.
TABLE 1.
Experimental Trial Type Conditions
| Trial Type | Peripheral Stimulus Delay |
|---|---|
| Prestimulus | Peripheral stimulus only |
| Immediate | Central and peripheral stimulus presented simultaneously |
| 2-sec | Central stimulus on, peripheral stimulus delayed for 2 sec |
| HR deceleration + 2 sec | Central stimulus on, peripheral stimulus delayed until HR deceleration + 2 sec |
| HR return to prestimulus level | Central stimulus on, HR deceleration, peripheral stimulus delayed until HR returns to prestimulus level |
Note. HR = heart rate.
Testing was done only if the participants maintained an alert, awake state during the entire procedure (eyes open, no fussing or crying, responding to protocol). The HR-defined delay trials were restarted if no HR deceleration occurred within 10 sec following fixation onset. Trials were also restarted if the infant looked away when the peripheral stimulus was to be presented. After every four experimental trials, a recording of a Sesame Street movie was shown for approximately 10 sec. These presentations were included to prevent the association of temporal sequencing between the central and peripheral stimuli and to allow time to center the EOG recording due to DC shifts. They were not included in any of the analyses.
Peripheral Stimulus Localization Judgments
A single observer judged the infant’s fixation during the experiment in an adjacent room on a TV monitor to control the experimental protocol. Each session was also judged offline. A time code recorded on the videotapes allowed the judgment to have msec accuracy. The observer judged the looks during trials as looking at the stimulus on the right side of the TV, looking at the stimulus on the left side of the TV, or looking away. The time code on the videotape was used by the computer to determine what part of the experimental protocol was occurring (interstimulus interval, blinking square, central stimulus, or peripheral stimulus). The offline judgments were used to assess the integrity of the experimental protocol and if a peripheral stimulus localization had occurred. The judgment of the localization gave the approximate time of the localization that aided in the identification of saccades used to localize the peripheral stimulus.
Localizations were based on the observer’s fixation judgments along with the existence of saccades in the EOG. For any trial period to be included in the analysis, the observer had to determine that the infant was looking toward the TV at the central stimulus at the time of the peripheral stimulus onset or looking at the central stimulus at the onset of the periods defined by the delay conditions. A look was considered a localization when the observer judged that the infant’s eyes moved from the location of the fixation stimuli to the area of the peripheral target and a saccade occurred in the EOG recording in the appropriate direction and with the appropriate amplitude. The appropriate amplitude of the saccade was determined by first plotting the summed magnitude of all saccades for a particular trial. Using the amplitude from a trial on which the observer had indicated a localization had occurred and the EOG record indicated a single saccade for localization a b weight was developed for that participant. That b weight was then used on a trial-by-trial basis to determine if localization had occurred and if multiple eye movements were used in accomplishing localization. Localizations were also defined in the absence of a judgment by the observer that the infant shifted fixation if (a) there was a saccade of appropriate amplitude and direction and (b) the observer judged that the infant was looking at the TV at the time of peripheral stimulus presentation. Any trial in which the observer judged that the infant was not looking at the TV during the saccade (i.e., the saccade was a look away from the central stimulus) was excluded. Nonlocalizations were said to have occurred if the observer’s judgment indicated that fixation remained on the central stimulus and there was no saccade (of appropriate amplitude) in the EOG recording.
RESULTS
Peripheral Stimulus Localization Percentage
The probability of peripheral stimulus localization was examined in relation to the experimental factors with an Age (4) × Experimental Delay (5) × Eccentricity (4) × Addition–Replacement (2) design. Linear categorical modeling was used rather than calculating percentages of individual participation localizations and using analysis of variance (ANOVA)-based analyses. This was done because of the prevalence of empty or zero probability cells in the crossing of experimental and participant factors. There was an overall increase in localization percentage across the four testing ages, χ2(3, N = 1,647) = 89.97, p < .0001. Localization percentages across the four testing ages were 34.81%, 40.38%, 59.17%, and 63.02% for the infants at 5, 8, 11, and 14 weeks of age, respectively. The largest difference in the localization percentages was the change from 8 to 11 weeks. There was also a main effect of eccentricity, χ2(3, N = 1,647) = 9.20, p = .0267. Post hoc comparisons showed that localization percentage decreased as eccentricity increased for the first three eccentricities, 10° (57.66%, N = 362), 15° (51.35%, N = 416), 20° (45.30, N = 458), but did not change from 20° to 25° (45.70, N = 411). As expected, the replacement trials resulted in a larger localization percentage (60.46%, N = 822) than the addition trials (39.64%, N = 825), χ2(1, N = 1647) = 38.09, p < .0001.
The percentage of localizations for the five delay conditions are shown in Table 2. The effect of peripheral stimulus delay condition on peripheral stimulus localization percentage was not statistically significant. However, based on the findings of Richards and Hunter (1997; cf. Hicks & Richards, 1998; Richards, 1997) experimental conditions were combined that differed on the type of attention engagement. The HR return to prestimulus level condition represents inattentiveness in the presence of the central stimulus after attention to the central stimulus has waned. The 2-sec and HR deceleration conditions were combined because they represent periods in which attention was engaged. The prestimulus and immediate condition were combined because attention has not yet been engaged in these conditions. Figure 1 shows the localization percentage for these three categories as a function of the addition–replacement and eccentricity–control factors. Two effects may be seen in this figure. First, the addition–replacement factor affected localization percentage independent of the three attention phases (addition–replacement effect in prior analysis). Infants were less likely to localize peripheral targets if the central stimulus remained on the screen (addition trials) than if it was turned off on peripheral target presentation (replacement trials). Second, the eccentricity of the stimulus affected localization percentage primarily at the beginning of stimulus presentation (immediate condition) or when attention was engaged (2-sec and HR deceleration), χ2(3, N = 744) = 15.03, p < .0018. There was a steady decrease in localization percentage as eccentricity increased only when attention to the central stimulus was engaged and the central stimulus remained on (addition trials). This decrease in localization percentage with increase in eccentricity was larger on the addition trials (e.g., 50% at 10° to 27% at 25°) than on the replacement trials (e.g., 66% at 10° to 54% at 25°). Stimulus eccentricity did not significantly affect localization percentage when attention was unengaged (ps > .20 for the preattentive and inattentive results in Figure 1).
TABLE 2.
Localizations on Peripheral Stimulus Present Trials as a Function of Delay
| Prestimulus | Immediate | 2-Sec | HR Declaration | HR Return to Normal | All Types | |
|---|---|---|---|---|---|---|
| Number of trials | 337 | 357 | 411 | 333 | 209 | 1,647 |
| % localization | 53 | 46 | 49 | 52 | 41 | 48 |
Note. HR = heart rate.
FIGURE 1.
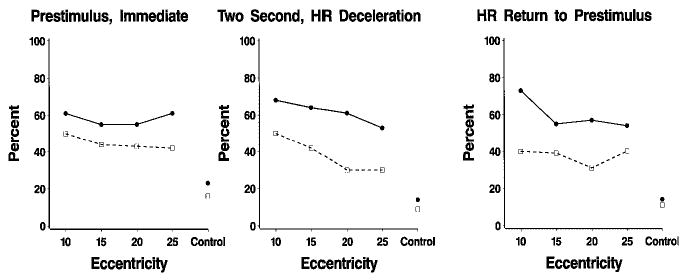
Percentage of stimulus localization as a function of addition (dashed lines and open squares) and replacement trials (solid line and filled circles), experimental conditions, and stimulus eccentricity. The conditions represent periods with no attention engaged (prestimulus), attention engaged (immediate, 2-sec, HR deceleration), and after attention had waned (return of HR to prestimulus level).
Characteristics of Localization Saccades: Multiple Saccades
Infants localized the peripheral targets with either a single saccade or multiple saccades. Table 3a shows the proportion of trials on which there was peripheral stimulus localization for single and multiple eye movements for each age group. For every age group, a majority of the localizations were accomplished with a single eye movement. No more than 5% of the localizations for any of the age groups required more than two eye movements.
TABLE 3a.
Localization Percentages for Single Saccades and Multiple Saccades on Peripheral Stimulus-Present Trials as a Function of Testing Age
| 5 Weeks | 8 Weeks | 11 Weeks | 14 Weeks | All Participants | |
|---|---|---|---|---|---|
| Single saccade | 68 | 74 | 63 | 76 | 70 |
| Two saccades | 26 | 23 | 33 | 20 | 26 |
| Three saccades | 5 | 3 | 4 | 4 | 4 |
It has been reported (Aslin & Salapatek, 1975; Salapatek et al., 1980) that infant saccades in 1- and 2-month-olds can be characterized as being of fixed amplitude and hypometric. We examined that relationship in the multiple saccades found in these data. We defined hypometric saccades as those multiple eye movements for which the saccade amplitudes of the multiple saccades were within 20% of each other (following Aslin & Salapatek, 1975). Saccades not meeting this criterion were defined as corrective. These saccades represented an initial attempt to localize the peripheral stimulus followed by recomputation of stimulus position and a corrective saccade. Table 3b contains the number of saccades that were classified as hypometric or corrective at each testing age. Most of the multiple saccades were corrective (18% of all saccades, n = 157) rather than hypometric (12% of all saccades, n = 104). The proportion of hypometric or corrective saccades did not change over the four testing ages. Table 3c contains the number of saccades classified as hypometric or corrective for the four testing eccentricities. There was an increase in the number of corrective saccades that occurred with increases in eccentricity, whereas the number of saccades that were classified as hypometric stayed relatively constant over the four testing eccentricities. The number of multiple saccades and the proportion of hypometric or corrective saccades was approximately equal across the five delay conditions.
TABLE 3b.
Multiple Saccades: Hypometric and Corrective Saccades as a Function of Testing Age (n/% of Age for Multiple Saccades/% of Age for All Saccades)
| 5 Weeks | 8 Weeks | 11 Weeks | 14 Weeks | All Participants | |
|---|---|---|---|---|---|
| Hypometric | 15/36/11 | 22/47/12 | 43/40/14 | 24/37/8 | 104/40/12 |
| Corrective | 27/64/20 | 25/53/13 | 64/60/22 | 41/63/15 | 157/60/18 |
TABLE 3c.
Multiple Saccades: Hypometric and Corrective Saccades as a Function of Stimulus Eccentricity (n/% of Saccade Type of Multiple Saccades/% of Age for All Saccades)
| 10° | 15° | 20° | 25° | All Participants | |
|---|---|---|---|---|---|
| Hypometric | 26/25/8 | 23/22/5 | 29/28/10 | 26/25/6 | 104/100/12 |
| Corrective | 27/17/5 | 39/25/6 | 38/24/8 | 53/34/8 | 157/100/18 |
The multiple saccades were further examined by plotting a distribution of the proportion of the distance the first saccade traveled, separately for the four testing ages (Figure 2). There appeared to be separate distributions for eye movements. The middle distribution represents the hypometric saccades (n = 104, 40% of multiple saccades, 12% of all saccades). The first saccade traversed approximately half the distance to the saccade target. The second distribution represents those eye movements that can be characterized as corrective where there was a large first saccade that traversed most of the distance followed by a small corrective eye movement (n = 109, 41% of multiple saccades, 12% of all saccades). These two plots appear to have a peak resembling the normal distribution (Figure 2). Some saccades (n = 48, 18% of multiple saccades, 5% of all saccades) consisted of a small first saccade followed by a large corrective saccade.
FIGURE 2.
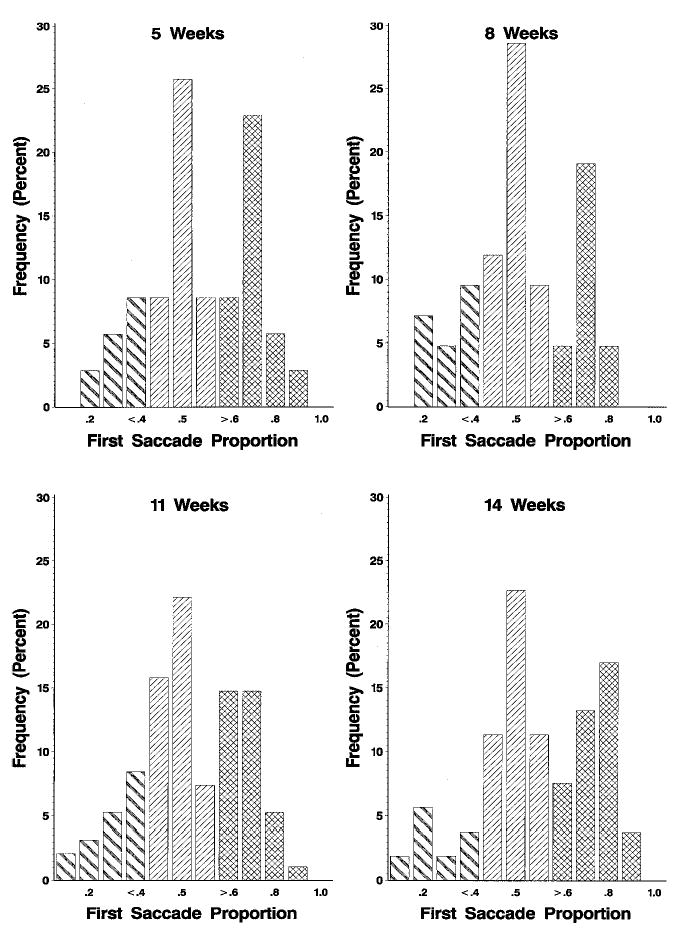
The frequency of the proportion of the first saccade to the total eccentricity of the multiple saccades as a function of testing age. In each case the frequency distributions were separated at .4 and .6, representing hypometric saccades (.4–.6, light slanted lines), corrective saccades with a large first saccade (>.6, cross-hatched lines), and corrective saccades with a small first saccade (<.4, dark slanted lines).
Characteristics of Localization Saccades: Main Sequence
The main sequence (Bahill et al., 1975) is a lawful relation between maximum saccade velocity and saccade amplitude, or saccade duration and saccade amplitude. A polynomial regression model was developed on saccades found in the prestimulus period. This model was then tested for its fit to saccades occurring in parts of the experimental trials. This was done to compare the saccades in the inattentive prestimulus period to the saccades when attention may have been directed to the central stimulus. A summary of the findings is that the regression parameters developed for the prestimulus saccades did not provide a satisfactory account for the saccades found in the experimental trials, and there was a decrease in the linear relation between saccade velocity and amplitude from 8 to 11 weeks.
The saccades identified in the prestimulus recording period were selected and a polynomial regression equation was estimated. Table 4 has the coefficients for the polynomial regression for the prestimulus recording period. The coefficients for the linear, quadratic, and cubic terms were statistically significant (ps < .001) but any terms beyond the cubic term were not statistically significant (e.g., quartic term, p = .1412). Figure 3 (top panel) shows the scatterplot for the maximum velocity–total degrees relation and the predicted line for the linear and polynomial regression equation. Table 4 also contains the b weights and R2 comparing the prestimulus saccade model with the models from the experimental conditions. For each condition (all localizations, preattentive, attentive, and inattentive) there was an improvement in the regression model by estimating the b weights from the actual data, and this primarily occurred in the quadratic and cubic polynomial trends (Table 4). Figure 3 (bottom panel) shows the scatterplot for the saccades in the experimental conditions and the predicted regression line for the preattentive, attentive, and inattentive experimental conditions. There was a clear difference in the main sequence relation in the saccades coming from the experimental conditions and saccades coming from the prestimulus period. The saccades coming from the three experimental conditions did not appear to differ in their relation (Figure 3, bottom panel), and tests of the b weights among the preattentive, attentive, and inattentive experimental conditions showed they were not significantly different.
TABLE 4.
Main Sequence Relation Multiple Regression Coefficients for Third-Order Polynomial Equations, for the Different Components of the Experimental Procedure
| Multiple R2 |
Polynomial Coefficients
|
|||||
|---|---|---|---|---|---|---|
| Experimental Procedure | N | R2 From Prestimulus b Weights | R2 From Estimated b Weights | blinear | bquadratic | bcubic |
| Preexperimental periods | 1,388 | .7773 | — | 0.0268 | −0.1660 | 0.0053 |
| All prestimulus localizations | 1,228 | .5500 | .6184** | 0.0554** | −0.1201** | 0.0924** |
| Preattentive (prestimulus, immediate) | 524 | .4972 | .5586** | 0.0525* | −0.1154** | 0.0910** |
| Attentive (2-sec, HR deceleration) | 546 | .5719 | .6406** | 0.0593 | −0.1283** | 0.0984** |
| Inattentive (return to prestimulus) | 156 | .6866 | .7650** | 0.0511 | −0.1071** | 0.0799** |
Note. Significance test for R2 represents a significant improvement in R2 for the model estimated with the empirical data relative to the model based on the b weights from the pre-experimental trial saccades. Significance tests for the polynomial terms represent the significance of a Wald test for estimating the b weights on the empirical data, relative to using the b weights from the pre-experimental trial saccades. The b weights of all three polynomial terms contribute significantly to the regression equation. HR = heart rate.
p < .05.
p < .01.
FIGURE 3.
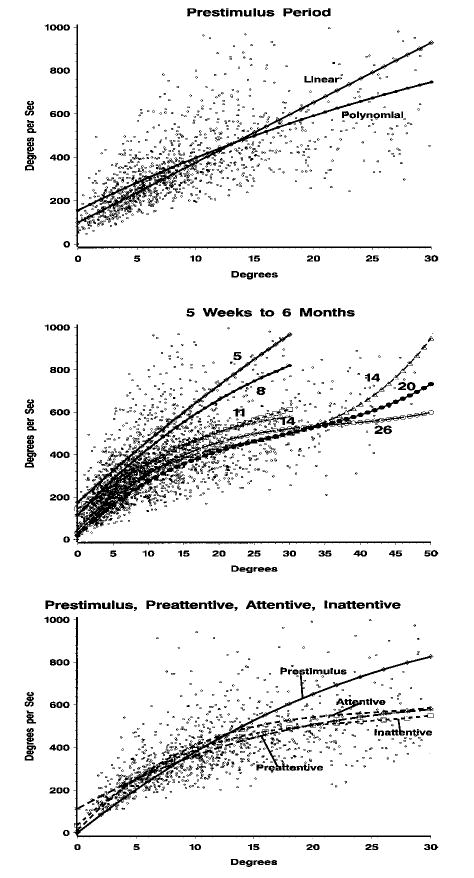
Main sequence relation between maximum saccade velocity and total amplitude of saccade, with regression lines estimated from the best fitting polynomial equations. Top panel: prestimulus saccades, with regression lines from the linear regression and the best fitting polynomial equation. Middle panel: prestimulus saccades, with the regression lines separately for 5, 8, 11, and 14 weeks from this study (lines marked 1, 2, 3, and 4, respectively), and lines for 14, 20, and 26 weeks (3, 4.5, and 6 months) from Richards and Hunter (1997; lines marked 5, 6, and 7, respectively). The regression lines are shown for the infants in this study up to 30°, and for the infants from Richards and Hunter (1997), up to 50°, which are the maximum amplitude of saccades possible for the stimuli from these studies. Bottom panel: saccades from the prestimulus period (diamond) and experimental conditions, with separate regression lines for the preattentive conditions (P), attentive conditions (A), and inattentive condition (I).
The main sequence relation between maximum saccade velocity and saccade amplitude for the prestimulus saccades was examined for the four testing ages. We found an interesting difference over the four testing ages. Figure 3 (middle panel) shows the scatterplot for the prestimulus saccades and the predicted line for the polynomial equation separately for the four testing ages. In addition, we included in Figure 3 the comparable data for 14-, 20-, and 26-week-old infants (3, 4.5, and 6 months) from Richards and Hunter (1997). There was a clear decrease in the linear trend from the 5- to 8- to 11-week-old infants. The linear trend for the 11-, 14-, 20-, and 26-week-olds was similar. The b weights confirmed this decrease in the linear trend, and the b weight for the linear trend was 0.0422 at 5 weeks, 0.0195 at 8 weeks, and around 0.013 for the infants at the other testing ages. This change in the linear trend over the four testing ages looked similiar in all of the experimental conditions (Figure 4).
FIGURE 4.
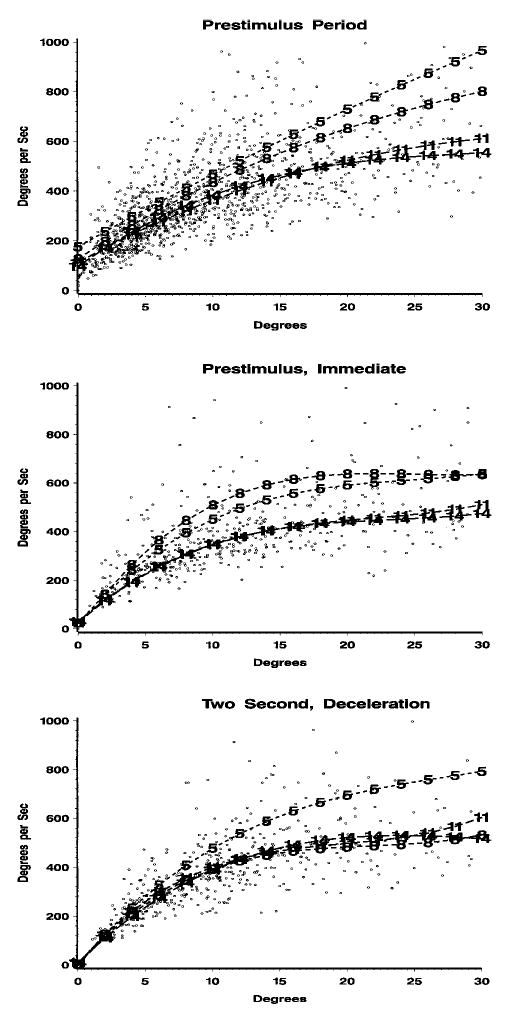
Main sequence relation between maximum saccade velocity and total amplitude of saccade as a function of testing age. Top panel: prestimulus period. Middle panel: preattentive conditions (prestimulus, immediate). Bottom panel: attentive conditions (2 sec, heart rate deceleration). The regression lines are plotted separately for 5, 8, 11, and 14 weeks (marked 1, 2, 3, and 4, respectively).
DISCUSSION
The goals of this study centered on the effect attention has on characteristics of saccadic eye movements used by infants in peripheral stimulus localization. The first goal of this study was to investigate the effect of attention to a central stimulus on the performance characteristics of peripheral stimulus localization in 5- to 14-week-old infants. There was an overall increase in localization percentage over the testing ages, replicating several studies (Hicks & Richards, 1998; Richards, 1997; Richards & Hunter, 1997). As expected, localization percentage decreased as eccentricity of the peripheral stimulus increased. The presence of the central fixation stimulus interfered with peripheral stimulus localization regardless of attention delay condition. The effect of peripheral stimulus eccentricity was the greatest when attention to the central stimulus was engaged (Figure 1).
The second goal of this study was to examine the quantitative eye movement characteristics of very young infants. As with a recent study of older infants (Richards & Hunter, 1997), most localizations were accomplished with a single eye movement (Table 3). Further, for those instances when multiple eye movements were used in peripheral stimulus localization, about 40% were classified as same-size, hypometric; about 40% consisted of a large initial saccade followed by a smaller corrective saccade; and about 20% consisted of a small initial saccade followed by a large corrective saccade (Figure 2). These percentages were not affected by attention phase. There were also changes in the main sequence relation between saccade velocity and amplitude over the four testing ages and with attention phase.
The general pattern found in the analyses of performance characteristics replicates many of the findings present in the existing literature. First, there was an increase in localization percentages with age. The localization percentages of the 5- and 8-week-old infants (29% and 34%, respectively) were similar to those reported for 8-week-old infants (25%) in a similar study (Hicks & Richards, 1998). The localization percentages of the 5-week-old infants were much lower than those found in some other studies (compared to about 80% for Aslin & Salapatek, 1975; and 75% for Goldberg et al., 1997, for similar ages and eccentricities). This difference in localization percentages likely was due to differing experimental manipulations in these studies. For example, in this study and in Hicks and Richards (1998) the peripheral stimulus remained on for 2 sec, and there was only a 3-sec window in which the infant could respond. The level of localization probability and the increase across these testing ages was similar to that found by Butcher and colleagues (2000), who used a 5-sec window for the peripheral stimulus presentation. Alternatively, in Goldberg et al. (1997) the peripheral stimulus remained on until the experimenter judged the infant made an eye movement (in either direction) allowing for an unlimited response time for the infants. In addition, mean latency to respond for 1-month-old infants in Goldberg et al. was greater than 3 sec. Thus, the 75% proportion was for the response to the correct side under unlimited exposures, rather than for detection percentage in under 3 sec. Similarly, in Aslin and Salapatek (1975), the peripheral stimulus remained on for 10 sec and a number of eye movements had latencies greater than 3 sec (Figures 3 & 4; Aslin & Salapatek, 1975). The difference between these studies implies that the absolute peripheral sensitivity may not change dramatically over this age range (e.g., Aslin & Salapatek, 1975; Goldberg et al., 1997) but that the speed of making a localizing response increases over this age range (e.g., this study; Butcher et al., 2000; Hicks & Richards, 1998). There also were differences in the current study in the types of stimuli used from Goldberg et al. (1997), Aslin and Salapatek (1975), and Butcher and colleagues (2000) that may account for some of these differences.
We were most interested in the effect of attention on peripheral stimulus localization. There were two important findings in this respect. First, the presence of a central stimulus affected peripheral stimulus localization percentages independent of attention engagement. The peripheral stimulus was localized more frequently when it replaced the central stimulus than when it was presented in addition to the central stimulus, in the two inattentive conditions (Figure 1, prestimulus, return of HR to prestimulus level) and in the attentive conditions (Figure 1, immediate, 2-sec, HR deceleration). This finding was markedly different from what has been found with older infants. Richards and Hunter (1997) found with infants from 14 to 26 weeks of age that the difference between the addition and replacement trials occurred in the attentive conditions and did not occur in the inattentive conditions. Similar findings using only addition conditions have been reported by Hicks and Richards (1998), and Richards (1987, 1997). Second, there was a significant effect of stimulus eccentricity only for the conditions on which attention was hypothesized to be engaged. Alternatively, for the prestimulus condition in which no central stimulus was present or the return of HR to prestimulus condition in which attention had waned, stimulus eccentricity did not affect peripheral stimulus localization percentages (Figure 1). This finding was similar to that of Richards and Hunter (1997) with older infants (14–26 weeks of age) and stimuli with larger eccentricities (25°, 35°, or 45°). These two findings suggest that attention engagement affects stimulus localization in these very young infants only partially. Infants at the earliest ages may engage in “sticky fixation” or “obligatory fixation” (Butcher et al., 2000; Goldberg et al., 1997; Richards, 1997) based on factors other than attention engagement, such as fixation-based inhibition of saccadic eye movement centers in the brain or the relation between characteristics of the central and peripheral stimulus (Goldberg et al., 1997; Hicks & Richards, 1998). The change from these very young ages (5–14 weeks) to older ages (14–26 weeks) may be the increasingly pervasive effects of visual attention on eye movements and the decline in the effect of stimulus or perceptual factors on eye movements.
The second focus of this study was quantitative characteristics of eye movements used in localizing peripheral stimuli. Aslin and Salapatek (1975) reported that infants often localized peripheral stimuli with a series of fixed-amplitude hypometric saccades. This study and Richards and Hunter (1997) differed from the Aslin and Salapatek (1975) findings in several respects. First, in this study 70% of localizations were accomplished with a single eye movement (Table 3), and only 30% were accomplished with multiple eye movements. Aslin and Salapatek (1975) reported that about 50% of saccades of 1- and 2-month-old infants were accomplished with a single eye movement. Richards and Hunter (1997), with older infants (14 to 26 weeks old), and larger eccentricities (25°, 35°, or 45°) found that 55% of the localizations were accomplished with a single eye movement. Second, when we examined instances in which two or more saccades were used in peripheral stimulus localization we also did not find a large number of same-size, steplike saccades. Only 12% of the saccades in this study could be identified as hypometric according to the criteria defined by Aslin and Salapatek (1975). The majority (60% of multiple saccades and 18% of all saccades) of the multiple saccades were corrective (Table 3b; Figure 2). Similarly, in Richards and Hunter (1997), in older infants the large majority of the multiple saccades were of the localizing-corrective type (78% of multiple saccaees). However, Aslin and Salapatek reported that about 65% of the multiple eye movements were fixed-amplitude hypometric saccades (Note 9, Aslin & Salapatek, 1975).
The differences in multiple saccade characteristics in this study and in the Aslin and Salapatek (1975) study are not easy to resolve. The ages in this study (5, 8, 11, and 14) were similar to the ages in the Aslin and Salapatek (1975) study. The eccentricities of the peripheral stimuli in this study (10°, 15°, 20°, and 25°) were similar to those used by Aslin and Salapatek (10°, 20°, and 30°). We would expect that if the fixed-amplitude hypometric saccades were a robust behavioral type used in this age range for stimuli at these eccentricities that our results would have been more similar. Instead, the proportions of multiple saccades compared to single saccades and the relative proportions of hypometric and corrective saccades were more similar to the study of Richards and Hunter (1997), with older infants (14, 20, and 26 weeks) and larger eccentricities (25°, 35°, and 45°).
The differences in these studies may come from analysis differences or experimental variables. One analysis difference was in the treatment of averages in the Aslin and Salapatek (1975) study. They took the saccades for each infant and averaged them then compared differences in the first and subsequent saccades (Figure 8, Table 1, in Aslin & Salapatek, 1975). This averaging procedure may have obscured the distribution of saccade amplitudes for the multiple saccades. This was evident in this study in which it appears that there were (at least) two distributions of saccades. One type of multiple saccade was hypometric and had a normal distribution of first saccade amplitudes around 50% (Figure 2). A second type of multiple saccade was corrective and had a normal distribution of first saccade amplitudes that nearly accomplished the total localization, followed by a small second saccade for the final localization (Figure 2, right distribution). Experimental conditions in the two studies also were slightly different. The infants in the Aslin and Salapatek (1975) study had head movements slightly restrained (though they report head movements occurred) and were supine, whereas infants in this study and in Richards and Hunter (1997) did not have head movements restrained and were sitting rather than supine. The characteristics of the stimuli used in the studies may have affected the types and proportion of multiple saccades that were found.
In this study and in Richards and Hunter (1997), most of the multiple saccades were similar to corrective saccades that have been described for adult participants (Becker, 1976; Prablanc & Jennerod, 1975; Prablanc, Masse, & Echallier, 1978). These multiple saccades consisted of a large saccade that traversed the majority of the distance to the peripheral target that was followed by a small corrective saccade for final target localization. This study and Richards and Hunter (1997) provide a picture of young infants as making directionally appropriate saccades that typically reach their intended target in a single saccade (70%), making corrective saccades if the target is not reached (18%) and occasionally making hypometric saccades of fixed amplitude (12%). We see no need to hypothesize a separate fixed-amplitude hypometric saccadic system or fixed-amplitude saccadic programming for infants in this age range (e.g., Aslin & Salapatek, 1975; Salapatek et al., 1980). The higher proportion of multiple saccades in infants relative to adults may be due to an immature targeting system, inattentiveness, visual field increases, or other factors that change in a quantitative fashion over age. For example, some of the multiple saccades may have consisted of an eye movement away from the central stimulus that was inadvertently in the appropriate direction even though the peripheral target was not seen. This inadvertent saccade was then followed by a second saccade that was in the appropriate direction toward a perceived peripheral target. This surely is true for many of the saccades in which a small first saccade was followed by a large corrective saccade (Figure 2, left distribution). This may also be true of some of the hypometric saccades with fixed amplitude (Figure 2, middle distribution).
This study systematically examined the main sequence relation in young infants. The main sequence is a lawful relation between maximum saccade velocity and saccade amplitude (Bahill et al., 1975). The general form for the main sequence shows a near linear relation between these two variables up to about 20°, with a decreasing exponential curve after that point. This general shape of the velocity–amplitude relation was found in this study in nearly all respects (see Figure 3 in particular) and was similar to that found with adult participants (e.g., Lebedev, van Gelder, & Tsui, 1996). However, the parameters of the regression function relating maximum saccade velocity and saccade amplitude differed over these testing ages. There was a decrease in the magnitude of the linear regression weight from about 5 to 11 weeks, and this linear relation did not change from 11 weeks to 26 weeks (6 months) of age (Figure 3). This age change occurred in the prestimulus period in which attention was unengaged (Figures 3 and 4) and occurred in the conditions in which a stimulus occurred and attention was engaged (Figure 4). Thus, this age change was independent of any changes in attention that may have been occurring over this age range.
The age change in the linear component of the main sequence relation may represent a change in the low-level eye movement systems of the young infant. The main sequence is the direct result of low-level neural systems that control saccadic eye movements (Moschovakis & Highstein, 1994; Schall, 1995; Schiller, 1998). The muscles that control eye movement are innervated by cranial nerves (oculomotor, trochler, and abducent) that are controlled by eye movement centers in the brain stem and medulla. These eye movement centers have burst neurons that initiate saccades and fire for a duration that is proportional to the expected size of the saccade, and pause neurons that terminate the saccade. The velocity and amplitude components of the main sequence are a product of the activity of these burst and pause cells and their relation to the rotational characteristics of the eye orbit. We believe that the age changes in the main sequence relation found from 5 to 11 weeks of age in this study represent developments occurring in these low-level eye movement centers. Alternatively, at older ages (e.g., 14–26 weeks, Richards & Hunter, 1997; also see Hainline et al., 1984) there may be a significant disruption of the main sequence regression parameters under conditions of attention to the central stimulus. We believe that the effect of attention on the main sequence relation at older ages is due to an increasing influence of cortical areas on eye movement control. Attention-directed eye movements show rapid development from the ages of 3 months through 6 months of age (Johnson, 1990, 1995; Richards & Holley, 1999; Richards & Hunter, 1997, 1998). The two age changes found in the main sequence relation in this study and in Richards and Hunter (1997) suggest that there are changes in the low-level control of eye movements in the first couple of months of infancy (e.g., through 11 weeks; this study) that are followed by changes in the cortical control of eye movements from 3 through 6 months of age (Richards & Hunter, 1997, 1998).
The change in the parameters of the main sequence relation reflect an immaturity of eye movement control in young infants and the increasing integration of eye movement control into other psychological processes. A description of the change in the linear relation was that the youngest age infants (5 and 8 weeks) must achieve a much higher velocity in the saccade to traverse the same distance as the older age infants (11 and 14 weeks). This suggests a difficulty in the youngest ages in making the most basic eye movements to stimuli in their environment and more efficient eye movement control at 11 or 14 weeks of age. The early difficulty in eye movement control may be the cause of the findings in this and other studies that attention engagement is not as effective in visual behavior in 5- and 8-week-old infants as it is in older infants. “Sticky fixation” (Butcher et al., 2000; Goldberg et al., 1997; Richards, 1997), or fixation independent of attention (this study; Hicks & Richards, 1998) may be due to inadequate voluntary control of the eyes and head in the presence of visual stimuli. The smaller percentage of localization for the youngest infants under time-limited presentations (this study; Butcher et al., 2000; Hicks & Richards, 1998) than for infants under unlimited stimulus presentations (Aslin & Salapatek, 1975; Goldberg et al., 1997) may reflect the increasing efficiency with which infants investigate their visual environment. With the increasing efficiency of eye movement control comes the possibility that more complexly controlled psychological processes such as visual attention may have a more pervasive effect on visual behavior.
Acknowledgments
This research was supported by grants from the National Institute of Child Health and Human Development, No. R01-HD18942.
Contributor Information
Sharon K. Hunter, Department of Psychiatry, University of Colorado Health Sciences Center
John E. Richards, Department of Psychology, University of South Carolina
References
- Ashmead D. Parameters of infant saccadic eye movements. Infant Behavior and Development. 1984;7:16. [Google Scholar]
- Aslin RN, Salapatek P. Saccadic localization of visual targets by the very young infant. Perception and Psychophysics. 1975;17:293–302. [Google Scholar]
- Bahill AT, Clark MR, Stark L. The main sequence: A tool for studying human eye movements. Mathematical Bioscience. 1975;24:191–302. [Google Scholar]
- Becker W. Do correction saccades depend exclusively on retinal feedback? A note on the possible role of non-retinal feedback. Vision Research. 1976;16:425–427. doi: 10.1016/0042-6989(76)90209-1. [DOI] [PubMed] [Google Scholar]
- Berg WK, Richards JE. Attention across time in infant development. In: Lang PJ, Simons RF, Balaban MT, editors. Attention and orienting: Sensory and motivational processes. Mahwah, NJ: Lawrence Erlbaum Associates, Inc; 1997. pp. 347–368. [Google Scholar]
- Braddick O, Atkinson J, Hood B. Striate cortex, extrastriate cortex, and colliculus: Some new approaches. In: Vital-Durand F, Braddick O, Atkinson J, editors. Infant vision. Oxford, England: Oxford University Press; 1996. pp. 203–220. [Google Scholar]
- Butcher PR, Kalverboer AF, Geuze RH. Infants’ shifts of gaze from a central to a peripheral stimulus: A longitudinal study of development between 6 and 26 weeks. Infant Behavior and Development. 2000;23:3–21. [Google Scholar]
- Finlay D, Ivinskis A. Cardiac and visual responses to stimuli presented both foveally and peripherally as a function of speed of moving stimuli. Developmental Psychology. 1982;18:692–698. [Google Scholar]
- Finlay D, Ivinskis A. Cardiac and visual responses to moving stimuli presented either successively or simultaneously to the central and peripheral visual fields in 4-month-old infants. Developmental Psychology. 1984;20:29–36. [Google Scholar]
- Fischer B. The role of attention in the preparation of visually guided eye movements in monkey and man. Psychological Research. 1986;48:251–257. doi: 10.1007/BF00309089. [DOI] [PubMed] [Google Scholar]
- Fischer B, Breitmeyer B. Mechanisms of visual attention revealed by saccadic eye movements. Neuropsychologia. 1987;25:73–83. doi: 10.1016/0028-3932(87)90044-3. [DOI] [PubMed] [Google Scholar]
- Goldberg MC, Maurer D, Lewis TL. Influence of a central stimulus on infants’ visual fields. Infant Behavior and Development. 1997;35:856–867. [Google Scholar]
- Hainline L, Abramov I. Saccades and small-field optokinetic nystagmus in infants. Journal of the American Optometric Association. 1985;56:620–626. [PubMed] [Google Scholar]
- Hainline L, Turkel J, Abromov I, Lemerise E, Harris C. Characteristics of saccades in human infants. Vision Research. 1984;24:1771–1780. doi: 10.1016/0042-6989(84)90008-7. [DOI] [PubMed] [Google Scholar]
- Harris P, MacFarlane A. The growth of the effective visual field from birth to seven weeks. Journal of Experimental Child Psychology. 1974;18:340–348. doi: 10.1016/0022-0965(74)90113-1. [DOI] [PubMed] [Google Scholar]
- Hicks JM, Richards JE. The effects of stimulus movement and attention on peripheral stimulus localization by 8- to 26-week-old infants. Infant Behavior and Development. 1998;21:571–589. [Google Scholar]
- Hood B, Atkinson J. Disengaging visual attention in the infant and adult. Infant Behavior and Development. 1993;16:405–422. [Google Scholar]
- Johnson MH. Cortical maturation and the development of visual attention in early infancy. Journal of Cognitive Neuroscience. 1990;2:81–95. doi: 10.1162/jocn.1990.2.2.81. [DOI] [PubMed] [Google Scholar]
- Johnson MH. The development of visual attention: A cognitive neuroscience perspective. In: Gazzaniga MS, editor. The cognitive neurosciences. Cambridge, MA: MIT Press; 1995. pp. 735–747. [Google Scholar]
- Lebedev S, van Gelder P, Tsui WH. Square-root relations between main saccadic parameters. Investigative Ophthalmology & Visual Science. 1996;37:2750–2758. [PubMed] [Google Scholar]
- Matsuoka K, Harato H. Detection of rapid phases of eye movements using third order derivatives. Japanese Journal of Ergonomics. 1983;19:147–153. [Google Scholar]
- Matsuoka K, Ueda Y. Frequency characteristics of the smooth pursuit component in tracking eye movements. Ergonomics. 1986;29:197–214. doi: 10.1080/00140138608968260. [DOI] [PubMed] [Google Scholar]
- Moschovakis AK, Highstein SM. The anatomy and physiology of primate neurons that control rapid eye movements. Annual Review of Neuroscience. 1994;17:465–488. doi: 10.1146/annurev.ne.17.030194.002341. [DOI] [PubMed] [Google Scholar]
- Prablanc C, Jennerod M. Corrective saccades: Dependence on retinal reafferent signals. Vision Research. 1975;15:465–469. doi: 10.1016/0042-6989(75)90022-x. [DOI] [PubMed] [Google Scholar]
- Prablanc C, Masse D, Echallier JF. Error-correcting mechanisms in large saccades. Vision Research. 1978;18:557–560. doi: 10.1016/0042-6989(78)90202-x. [DOI] [PubMed] [Google Scholar]
- Richards JE. Infant visual sustained attention and respiratory sinus arrhythmia. Child Development. 1987;58:488–496. [PubMed] [Google Scholar]
- Richards JE. Sustained visual attention in 8-week-old infants. Infant Behavior and Development. 1989;12:425–436. [Google Scholar]
- Richards JE. Peripheral stimulus localization by infants: Attention, age and individual differences in heart rate variability. Journal of Experimental Psychology: Human Perception and Performance. 1997;23:667–680. doi: 10.1037//0096-1523.23.3.667. [DOI] [PubMed] [Google Scholar]
- Richards JE, Casey BJ. Heart rate defined phases of infant visual information processing. Psychophysiology. 1991;28:43–53. doi: 10.1111/j.1469-8986.1991.tb03385.x. [DOI] [PubMed] [Google Scholar]
- Richards JE, Casey BJ. Development of sustained visual attention in the human infant. In: Campbell BA, Hayne H, Richardson R, editors. Attention and information processing in infants and adults. Mahwah, NJ: Lawrence Erlbaum Associates, Inc; 1992. pp. 30–60. [Google Scholar]
- Richards JE, Holley FB. Infant attention and the development of smooth pursuit tracking. Developmental Psychology. 1999 doi: 10.1037//0012-1649.35.3.856. [DOI] [PubMed] [Google Scholar]
- Richards JE, Hunter SK. Peripheral stimulus localization by infants with eye and head movements during visual attention. Vision Research. 1997;37:3021–3035. doi: 10.1016/s0042-6989(97)00082-5. [DOI] [PubMed] [Google Scholar]
- Richards JE, Hunter SK. Attention and eye movement in young infants: Neural control and development. In: Richards JE, editor. Cognitive neuroscience of attention: A developmental perspective. Mahwah, NJ: Lawrence Erlbaum Associates, Inc; 1998. pp. 131–162. [Google Scholar]
- Salapatek P, Aslin RN, Simonson J, Pulos E. Infant saccadic eye movements to visible and previously visible targets. Child Development. 1980;51:1090–1094. [PubMed] [Google Scholar]
- Schall JD. Neural basis of saccade target selection. Reviews in the Neurosciences. 1995;6:63–85. doi: 10.1515/revneuro.1995.6.1.63. [DOI] [PubMed] [Google Scholar]
- Schiller PH. The neural control of visually guided eye movements. In: Richards JE, editor. Cognitive neuroscience of attention: A developmental perspective. Mahwah, NJ: Lawrence Erlbaum Associates, Inc; 1998. pp. 3–50. [Google Scholar]
- Tronick E. Stimulus control and the growth of the infants’ effective visual field. Perception and Psychophysics. 1972;11:373–376. [Google Scholar]


