Abstract
Cyclooxygenases (COXs) play a significant role in many different viral infections with respect to replication and pathogenesis. Here we investigated the role of COXs in the mouse hepatitis coronavirus (MHV) infection cycle. Blocking COX activity by different inhibitors or by RNA interference affected MHV infection in different cells. The COX inhibitors reduced MHV infection at a post-binding step, but early in the replication cycle. Both viral RNA and viral protein synthesis were affected with subsequent loss of progeny virus production. Thus, COX activity appears to be required for efficient MHV replication, providing a potential target for anti-coronaviral therapy.
Background
Virus infections often cause acute inflammatory responses, which are mediated by several cellular effectors and soluble factors. Although these responses have an important protective role, they may also have deleterious effects on the host. The balance between these protective and deleterious effects may ultimately determine the course of disease after viral infection. Prostaglandins (PGs) are important regulators of this inflammatory reaction. They are synthesized by cyclooxygenases (COXs), converting arachidonic acid into PGH2, which can then be isomerized to generate different biologically active forms of PGs. There are three known isoforms of COXs, with COX-1 and COX-2 being the best characterized. COX-1 is expressed in various cell types and PGs produced by COX-1 are predominantly involved in the regulation of various homeostatic processes [1]. COX-2 is an immediate early response gene, which upon induction generates mainly hyperalgesic and proinflammatory PGs at sites of inflammation [2,3]. PGs from the E series, such as PGE2, also exhibit immunomodulatory activities, preventing hyperactivation of the innate cellular immunity [4]. Furthermore, they can inhibit the secretion of gamma interferon, a cytokine with antiviral activity [5]. A direct role for COXs and PGs in controlling viral replication has been described for a wide range of virus infections, but their actions appear to be dependent on both the virus and cell type [6]. For instance, COXs and/or PGs are required for efficient replication of herpesviruses [7-13], bovine leukemia virus [14], and rotavirus [15]. In case of human cytomegalovirus, human T-lymphotropic virus type 1, and human immunodeficiency virus type-1 PGE2 has been shown to stimulate virus replication by activating viral promotors [16-18]. On the other hand, COXs/PGs negatively affect adenovirus replication, as well as replication of human immunodeficiency virus type 1 in macrophages [19,20]. The mechanisms by which COXs and PGs regulate viral replication are largely unclear.
Coronaviruses (CoVs) constitute a family of enveloped, positive-stranded RNA viruses. They are known pathogens in the veterinary field, causing severe diseases in several domestic species [21]. Recently, their relevance has increased considerably with the discovery of several new human CoVs (HCoVs) such as the severe acute respiratory syndrome (SARS)-CoV [22], HCoV-NL63 [23], and HCoV-HKU1 [24]. The role of COXs during CoV infection and pathogenesis is not well understood. MHV strain 3, which causes fulminant hepatitis, was shown to induce the synthesis of PGE2 in macrophages [25]. However, the exogenous administration of PGE2 could completely prevent the development of hepatic necrosis [26]. More recently, two structural proteins from the SARS-CoV were shown to induce the expression of COX-2 in vitro [27-29], whereas elevated levels of PGE2 were found in the blood of SARS-CoV-infected individuals [30], suggesting a role for COXs and PGs in CoV pathogenesis. However, the requirement for COX activity for CoV replication remains unexplored.
Results
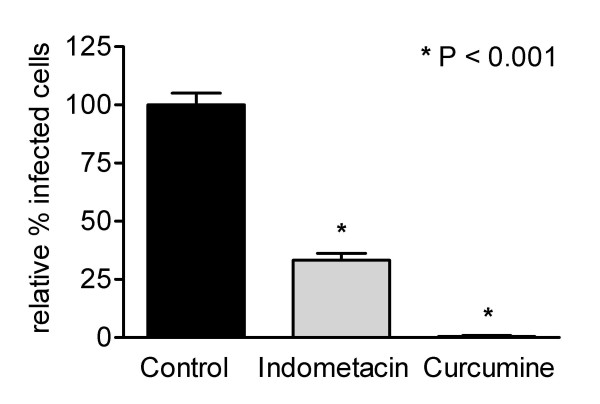
In the present study we investigated the role of COXs in the MHV replication cycle. To this end, Caco-2 cells, stably expressing the MHV receptor glycoprotein (Caco-MHVR) [31], were infected with MHV strain A59 (MHV-A59) at a multiplicity of infection (m.o.i.) of 0.01 in the presence or absence of the COX-1 and COX-2 inhibitors indomethacin and curcumine. The cells were incubated 1 h prior to infection with the inhibitors, and were maintained in the presence of the inhibitors from 30 minutes post infection (p.i.). Cells were fixed at 6 h p.i. with ice-cold methanol, and the number of MHV-infected cells were determined by an indirect immunofluorescence assay (IFA) using anti-MHV antibodies [32]. Possible cytotoxic effects of the inhibitors and their solvents were tested, using cell proliferation reagent WST-1 and lactate dehydrogenase cytotoxicity detection kit (Roche Diagnostics) assays according to the manufacturer's protocol. All inhibitors were used at concentrations that were not toxic to the cells. In the presence of 20 μM indomethacin, MHV infection was reduced by 57%, while curcumine reduced infection by 95% at a concentration of 30 μM (Figure 1). Both drugs affected MHV infection in a concentration-dependent manner (data not shown).
Figure 1.
COX inhibitors are negatively affecting MHV infection. (A) Caco-MHVR cells were incubated with culture medium (containing a concentration of DMSO similar to that present in the inhibitor solutions), 20 μM indomethacin, or 30 μM curcumine 1 h prior to inoculation with MHV-A59 (m.o.i = 0.01). The cells were maintained in the presence of the inhibitors until they were fixed at 6 h p.i. Infected cells were detected by an indirect IFA using an anti-MHV serum and Texas Red conjugated secondary antibodies. Fluorescence was viewed with a Nikon Eclipse E800 microscope. The numbers of MHV-infected cells in the drug-treated cells are presented as a percentage of the average number of infected cells in the mock-treated (control) cell cultures. Data are presented as mean ± standard error of mean (n = 6). For statistical analysis a one-way ANOVA with the Tukey-Kramer test was performed using GraphPad Prism version 3.00 for Windows (GraphPad Software). In all tests, P < 0.05 was considered statistically significant.
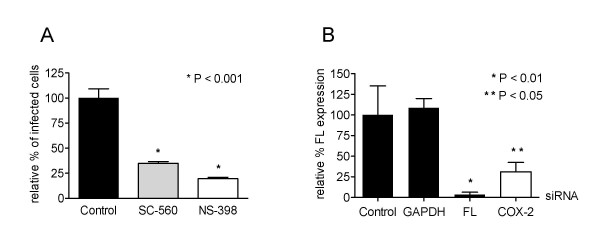
Next, we determined the role of the different COX isoforms. The ability of specific COX-1 and COX-2 inhibitors to reduce MHV infection was determined in a similar way as described above. Both SC-560 and NS-398, which inhibit COX-1 and COX-2, respectively, reduced MHV infection by 65–75% at concentrations that were non-toxic to the cells (1 μM and 0.055 μM respectively) (Figure 2A). Apparently, the activity of both enzymes is required for efficient MHV replication in Caco-MHVR cells. RNA interference technology was applied to confirm the observation that COX-2 activity is important for MHV replication. Parallel cultures of HeLa cells were transfected with siRNAs (purchased from Dharmacon, Inc.) targeting COX-2, firefly luciferase (FL) (positive control) or GAPDH (specificity control) transcripts for degradation. They were infected at 72 h posttransfection with MHV-FLSrec [33], a recombinant MHV expressing the FL reporter gene, the level of which is a reliable measure for MHV replication [34]. Silencing of GAPDH, a cellular housekeeping gene, did not affect FL expression compared to mock-transfected (control) cells (Figure 2B). However, HeLa cells transfected with siRNAs targeting the FL or COX-2 transcripts showed a reduction in FL expression of more than 90% and 65%, respectively. A taqman reverse transcription (RT)-PCR targeting COX-2 mRNA revealed that in cells treated with COX-2 siRNAs, COX-2 mRNA levels were decreased with more than 70% compared to control cells (data not shown). Therefore, these data show the requirement of COX-2 activity for efficient MHV replication.
Figure 2.
Blocking COX-1 or COX-2 activity by specific inhibitors, or by siRNAs targeting COX-2 mRNA reduce MHV infection. (A) Caco-MHVR cells were incubated with COX-1 (SC-560; 1 μM) or COX-2 (NS-398; 0.055 μM) inhibitor 1 h prior to inoculation with MHV-A59 (m.o.i. = 0.01) and were maintained in the presence of the inhibitors until they were fixed. The numbers of MHV-infected cells were determined with an indirect IFA and are presented as described in the legend of Figure 1. (B) HeLa cells were transfected with 10 nM siRNAs, targeting the indicated transcripts, 72 h prior to inoculation with MHV-FLSrec. Cell viability was measured for 30 minutes at 6 h p.i. using a WST-1 assay as described previously [37], after which the intracellular luciferase levels were determined as relative light units (RLU). Luciferase levels in siRNA-transfected cells are expressed as a percentage of the levels in the mock-transfected (control) cells and were corrected for the percentage of viable cells (n = 3).
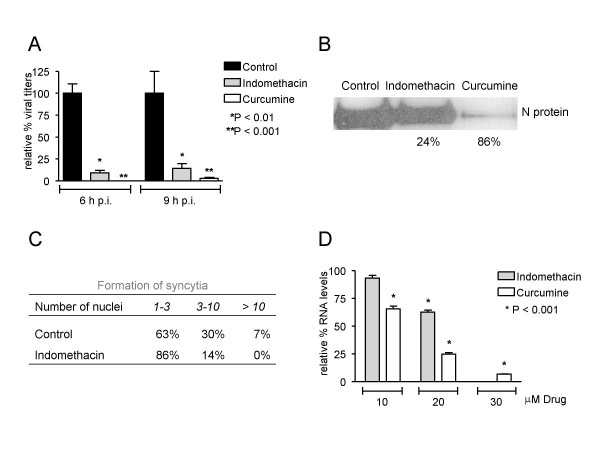
To determine which step of the MHV replication cycle was affected by the COX inhibitors, the production of infectious particles, of viral protein and of viral RNA was analyzed. For this purpose, Caco-MHVR cells were inoculated with MHV-A59 (m.o.i. = 1) in the presence or absence of indomethacin or curcumine. The amount of infectious viral progeny present in cells and culture media was monitored by determining the number of fluorescent focus-forming units (ffu) at different time points p.i. Inhibition of COX activity by curcumine and indomethacin resulted in a significant decrease in the yield of infectious viral progeny by more than 95% and 85%, respectively (Figure 3A). In addition, the amount of N protein present in cell lysates was analyzed by Western blotting using a polyclonal anti-MHV serum. N protein expression levels were markedly reduced by curcumine (86% reduction), and to a lesser extent by indomethacin (24%) (Figure 3B). Consistent with these results, much smaller syncytia were observed after infection of Caco-MHVR cells in the presence of the COX inhibitors (Figure 3C). Reduced expression levels of the MHV S protein, which is responsible for cell-cell fusion in MHV-infected cells [35] are likely to explain the lack of syncytium formation after COXs inhibition. Finally, viral RNA synthesis was analyzed in the presence of COX inhibitors. At 6 h p.i., total RNA was isolated and viral RNA synthesis was monitored by Taqman RT-PCR using a probe and primers that detect the N gene (details in legend Figure 3). Indomethacin and curcumine both inhibited viral RNA synthesis in a dose-dependent manner (Figure 3D). These results indicate that the COX inhibitors interfere with viral RNA and protein synthesis and consequently affect the production of infectious particles. In agreement with our findings, a recent study described the potent antiviral effect of indomethacin on SARS and canine coronavirus (CCoV) replication [36].
Figure 3.
Indomethacin and curcumine inhibit MHV replication at the level of RNA synthesis. Caco-MHVR cells were incubated with and maintained in culture medium containing DMSO, 20 μM indomethacin, or 30 μM curcumine as described in figure legend 1. After 1 h, the cells were inoculated with MHV-A59 (m.o.i. = 1). At 6 and 9 h p.i., supernatants were collected and cells were harvested to isolate infectious viral particles, proteins and total RNA. (A) Caco-MHVR cells were inoculated with serial dilutions of combined supernatants and cleared cell homogenates from mock-treated (black bars), indomethacin-treated (grey bars) and curcumine-treated (white bars) cultures collected at 6 and 9 h p.i. The amount of ffu in the samples was determined with an indirect IFA as described in the legend of Figure 1 (n = 3). (B) Protein samples were analyzed on a SDS-15% polyacrylamide gel followed by Western blotting using the polyclonal anti-MHV serum. The N protein levels and the percentage of reduction (normalized for β-tubulin expression (data not shown)) in drug-treated cells compared to mock-treated cells are indicated. (C) The size of the observed syncytia was measured by counting the number of nuclei per syncytium of MHV-infected cells in the absence or presence of 20 μM indomethacin. (D) The expression levels of the N gene of MHV were determined by Taqman RT-PCR using primers 2915 (5'-GCCTCGCCAAAAGAGGACT-3') and 2916 (5'-GGGCCTCTCTTTCCAAAACAC-3') and a dual labeled probe (5'-6-FAM-CAAACAAGCAGTGCCCAGTGCAGC-TAMRA-3'). The relative amount of viral RNA in the drug-treated cells was expressed as a percentage of the average amount of viral RNA in the mock-treated cells.
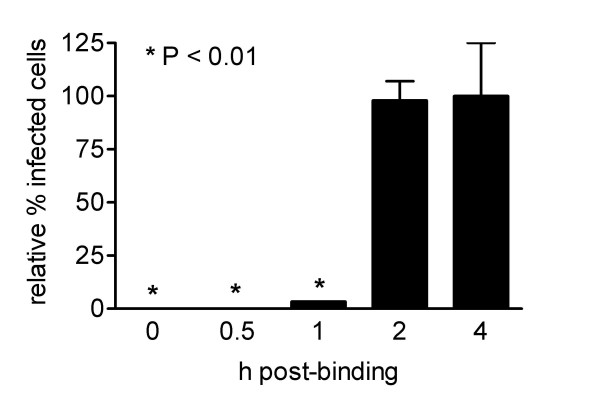
To study the kinetics of inhibition of MHV replication in more detail Caco-MHVR cells were inoculated with MHV-A59 (m.o.i. = 0.01) for 2 h at 4°C to allow binding of the virus to the cells without entry. After removing any unbound viral particles, the cells were placed at 37°C to induce virus entry and 20 μM indomethacin was added at the time points indicated (Figure 4). MHV infection was significantly reduced, as measured by the indirect IFA described above, if indomethacin was added up to 1 h after the cells were placed at 37°C. The maximum inhibitory effect was obtained when indomethacin was added immediately after the cells were placed at 37°C. No significant inhibition of the infection was observed if indomethacin was added 2 h after the cells were placed at 37°C. This result demonstrates that COX activity plays an important role early in the virus infection cycle, at a post-binding step. Thus, COX activity might either be required for efficient entry or for an initial step in RNA replication. Similarly, rotavirus replication was also negatively affected by the addition of COX inhibitors early, but not late in the infection cycle [15]. In conclusion, our results clearly show that COX activity is required for efficient virus replication in vitro early during MHV infection. These findings may offer new possibilities for anti-CoV therapy.
Figure 4.
COX inhibition affects MHV infection at a post-binding step. Caco-MHVR cells were inoculated with MHV-A59 (m.o.i. = 0.01) at 4°C for 2 h. Subsequently, cells were placed at 37°C and 20 μM indomethacin was added to the culture medium immediately (t = 0 h post binding) or at the indicated times. Cells were maintained in culture medium containing indomethacin until they were fixed at 6 h post-binding. The numbers of infected cells are presented as described in the legend of Figure 1 (n = 3).
Competing interests
The author(s) declare that they have no competing interests.
Authors' contributions
MR, LJAT, MvH, JB, JWAR, and RR conducted all the experiments. MR wrote the manuscript. AWCE, HAB, CAMdeH, and JWAR coordinated the research efforts and assisted with writing the manuscript. All authors read and approved the final manuscript.
Acknowledgments
Acknowledgements
This work was supported by grants from the Sophia Foundation for Medical Research, Rotterdam, The Netherlands, and The Netherlands Organization for Scientific Research.
Contributor Information
Matthijs Raaben, Email: m.raaben@vet.uu.nl.
Alexandra WC Einerhand, Email: a.einerhand@erasmusmc.nl.
Lucas JA Taminiau, Email: taminiau42@hotmail.com.
Michel van Houdt, Email: Michel.Vanhoudt@hu.nl.
Janneke Bouma, Email: j.bouma@erasmusmc.nl.
Rolien H Raatgeep, Email: h.raatgreep@erasmusmc.nl.
Hans A Büller, Email: h.buller@erasmusmc.nl.
Cornelis AM de Haan, Email: c.a.m.dehaan@vet.uu.nl.
John WA Rossen, Email: j.rossen@elisabeth.nl.
References
- Spencer AG, Woods JW, Arakawa T, Singer. Smith WL. Subcellular localization of prostaglandin endoperoxide H synthases-1 and -2 by immunoelectron microscopy. J Biol Chem. 1998;273:9886–9893. doi: 10.1074/jbc.273.16.9886. [DOI] [PubMed] [Google Scholar]
- Newton R, Kuitert LM, Bergmann M, Adcock IM, Barnes PJ. Evidence for involvement of NF-kappaB in the transcriptional control of COX-2 gene expression by IL-1beta. Biochem Biophys Res Commun. 1997;237:28–32. doi: 10.1006/bbrc.1997.7064. [DOI] [PubMed] [Google Scholar]
- Newton R, Stevens DA, Hart LA, Lindsay M, Adcock IM, Barnes PJ. Superinduction of COX-2 mRNA by cycloheximide and interleukin-1beta involves increased transcription and correlates with increased NF-kappaB and JNK activation. FEBS Lett. 1997;418:135–138. doi: 10.1016/S0014-5793(97)01362-8. [DOI] [PubMed] [Google Scholar]
- Betz M, Fox BS. Prostaglandin E2 inhibits production of Th1 lymphokines but not of Th2 lymphokines. J Immunol. 1991;146:108–113. [PubMed] [Google Scholar]
- Hasler F, Bluestein HG, Zvaifler NJ, Epstein LB. Analysis of the defects responsible for the impaired regulation of EBV-induced B cell proliferation by rheumatoid arthritis lymphocytes. II. Role of monocytes and the increased sensitivity of rheumatoid arthritis lymphocytes to prostaglandin E. J Immunol. 1983;131:768–772. [PubMed] [Google Scholar]
- Steer SA, Corbett JA. The role and regulation of COX-2 during viral infection. Viral Immunol. 2003;16:447–460. doi: 10.1089/088282403771926283. [DOI] [PubMed] [Google Scholar]
- Baker DA, Thomas J, Epstein J, Possilico D, Stone ML. The effect of prostaglandins on the multiplication and cell-to-cell spread of herpes simplex virus type 2 in vitro. Am J Obstet Gynecol. 1982;144:346–349. doi: 10.1016/0002-9378(82)90589-0. [DOI] [PubMed] [Google Scholar]
- Janelle ME, Gravel A, Gosselin J, Tremblay MJ, Flamand L. Activation of monocyte cyclooxygenase-2 gene expression by human herpesvirus 6. Role for cyclic AMP-responsive element-binding protein and activator protein-1. J Biol Chem. 2002;277:30665–30674. doi: 10.1074/jbc.M203041200. [DOI] [PubMed] [Google Scholar]
- Symensma TL, Martinez-Guzman D, Jia Q, Bortz E, Wu TT, Rudra-Ganguly N, Cole S, Herschman H, Sun R. COX-2 induction during murine gammaherpesvirus 68 infection leads to enhancement of viral gene expression. J Virol. 2003;77:12753–12763. doi: 10.1128/JVI.77.23.12753-12763.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thiry E, Mignon B, Thalasso F, Pastoret PP. Effect of prostaglandins PGE2 and PGF alpha 2 on the mean plaque size of bovine herpesvirus 1. Ann Rech Vet. 1988;19:291–293. [PubMed] [Google Scholar]
- Zhu H, Cong JP, Yu D, Bresnahan WA, Shenk TE. Inhibition of cyclooxygenase 2 blocks human cytomegalovirus replication. Proc Natl Acad Sci U S A. 2002;99:3932–3937. doi: 10.1073/pnas.052713799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ray N, Bisher ME, Enquist LW. Cyclooxygenase-1 and -2 are required for production of infectious pseudorabies virus. J Virol. 2004;78:12964–12974. doi: 10.1128/JVI.78.23.12964-12974.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rott D, Zhu J, Burnett MS, Zhou YF, Zalles-Ganley A, Ogunmakinwa J, Epstein SE. Effects of MF-tricyclic, a selective cyclooxygenase-2 inhibitor, on atherosclerosis progression and susceptibility to cytomegalovirus replication in apolipoprotein-E knockout mice. J Am Coll Cardiol. 2003;41:1812–1819. doi: 10.1016/S0735-1097(03)00304-8. [DOI] [PubMed] [Google Scholar]
- Pyeon D, Diaz FJ, Splitter GA. Prostaglandin E(2) increases bovine leukemia virus tax and pol mRNA levels via cyclooxygenase 2: regulation by interleukin-2, interleukin-10, and bovine leukemia virus. J Virol. 2000;74:5740–5745. doi: 10.1128/JVI.74.12.5740-5745.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossen JW, Bouma J, Raatgeep RH, Buller HA, Einerhand AW. Inhibition of cyclooxygenase activity reduces rotavirus infection at a postbinding step. J Virol. 2004;78:9721–9730. doi: 10.1128/JVI.78.18.9721-9730.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kline JN, Hunninghake GM, He B, Monick MM, Hunninghake GW. Synergistic activation of the human cytomegalovirus major immediate early promoter by prostaglandin E2 and cytokines. Exp Lung Res. 1998;24:3–14. doi: 10.3109/01902149809046050. [DOI] [PubMed] [Google Scholar]
- Moriuchi M, Inoue H, Moriuchi H. Reciprocal interactions between human T-lymphotropic virus type 1 and prostaglandins: implications for viral transmission. J Virol. 2001;75:192–198. doi: 10.1128/JVI.75.1.192-198.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dumais N, Barbeau B, Olivier M, Tremblay MJ. Prostaglandin E2 Up-regulates HIV-1 long terminal repeat-driven gene activity in T cells via NF-kappaB-dependent and -independent signaling pathways. J Biol Chem. 1998;273:27306–27314. doi: 10.1074/jbc.273.42.27306. [DOI] [PubMed] [Google Scholar]
- Hayes MM, Lane BR, King SR, Markovitz DM, Coffey MJ. Prostaglandin E(2) inhibits replication of HIV-1 in macrophages through activation of protein kinase A. Cell Immunol. 2002;215:61–71. doi: 10.1016/S0008-8749(02)00017-5. [DOI] [PubMed] [Google Scholar]
- Ongradi J, Telekes A, Farkas J, Nasz I, Bendinelli M. The effect of prostaglandins on the replication of adenovirus wild types and temperature-sensitive mutants. Acta Microbiol Immunol Hung. 1994;41:173–188. [PubMed] [Google Scholar]
- Weiss SR, Navas-Martin S. Coronavirus pathogenesis and the emerging pathogen severe acute respiratory syndrome coronavirus. Microbiol Mol Biol Rev. 2005;69:635–664. doi: 10.1128/MMBR.69.4.635-664.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drosten C, Gunther S, Preiser W, van der Werf S, Brodt HR, Becker S, Rabenau H, Panning M, Kolesnikova L, Fouchier RA, Berger A, Burguiere AM, Cinatl J, Eickmann M, Escriou N, Grywna K, Kramme S, Manuguerra JC, Muller S, Rickerts V, Sturmer M, Vieth S, Klenk HD, Osterhaus AD, Schmitz H, Doerr HW. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003;348:1967–1976. doi: 10.1056/NEJMoa030747. [DOI] [PubMed] [Google Scholar]
- van der Hoek L, Pyrc K, Jebbink MF, Vermeulen-Oost W, Berkhout RJ, Wolthers KC, Wertheim-van Dillen PM, Kaandorp J, Spaargaren J, Berkhout B. Identification of a new human coronavirus. Nat Med. 2004;10:368–373. doi: 10.1038/nm1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo PC, Lau SK, Chu CM, Chan KH, Tsoi HW, Huang Y, Wong BH, Poon RW, Cai JJ, Luk WK, Poon LL, Wong SS, Guan Y, Peiris JS, Yuen KY. Characterization and complete genome sequence of a novel coronavirus, coronavirus HKU1, from patients with pneumonia. J Virol. 2005;79:884–895. doi: 10.1128/JVI.79.2.884-895.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pope M, Rotstein O, Cole E, Sinclair S, Parr R, Cruz B, Fingerote R, Chung S, Gorczynski R, Fung L, et al. Pattern of disease after murine hepatitis virus strain 3 infection correlates with macrophage activation and not viral replication. J Virol. 1995;69:5252–5260. doi: 10.1128/jvi.69.9.5252-5260.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abecassis M, Falk J, Dindzans V, Lopatin W, Makowka L, Levy G, Falk R. Prostaglandin E2 (PGE2) alters the pathogenesis of MHV-3 infection in susceptible BALB/cJ mice. Adv Exp Med Biol. 1987;218:465–466. doi: 10.1007/978-1-4684-1280-2_58. [DOI] [PubMed] [Google Scholar]
- Liu M, Gu C, Wu J, Zhu Y. Amino acids 1 to 422 of the spike protein of SARS associated coronavirus are required for induction of cyclooxygenase-2. Virus Genes. 2006;33:309–317. doi: 10.1007/s11262-005-0070-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan X, Hao Q, Mu Y, Timani KA, Ye L, Zhu Y, Wu J. Nucleocapsid protein of SARS-CoV activates the expression of cyclooxygenase-2 by binding directly to regulatory elements for nuclear factor-kappa B and CCAAT/enhancer binding protein. Int J Biochem Cell Biol. 2006;38:1417–1428. doi: 10.1016/j.biocel.2006.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Liu M, Yang Y, Gu C, Yue Y, Wu KK, Wu J, Zhu Y. Spike protein of SARS-CoV stimulates cyclooxygenase-2 expression via both calcium-dependent and calcium-independent protein kinase C pathways. Faseb J. 2007. [DOI] [PubMed]
- Lee CH, Chen RF, Liu JW, Yeh WT, Chang JC, Liu PM, Eng HL, Lin MC, Yang KD. Altered p38 mitogen-activated protein kinase expression in different leukocytes with increment of immunosuppressive mediators in patients with severe acute respiratory syndrome. J Immunol. 2004;172:7841–7847. doi: 10.4049/jimmunol.172.12.7841. [DOI] [PubMed] [Google Scholar]
- Rossen JW, Strous GJ, Horzinek MC, Rottier PJ. Mouse hepatitis virus strain A59 is released from opposite sides of different epithelial cell types. J Gen Virol. 1997;78 ( Pt 1):61–69. doi: 10.1099/0022-1317-78-1-61. [DOI] [PubMed] [Google Scholar]
- Rottier PJ, Horzinek MC, van der Zeijst BA. Viral protein synthesis in mouse hepatitis virus strain A59-infected cells: effect of tunicamycin. J Virol. 1981;40:350–357. doi: 10.1128/jvi.40.2.350-357.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Haan CA, Li Z, te Lintelo E, Bosch BJ, Haijema BJ, Rottier PJ. Murine coronavirus with an extended host range uses heparan sulfate as an entry receptor. J Virol. 2005;79:14451–14456. doi: 10.1128/JVI.79.22.14451-14456.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Haan CA, van Genne L, Stoop JN, Volders H, Rottier PJ. Coronaviruses as vectors: position dependence of foreign gene expression. J Virol. 2003;77:11312–11323. doi: 10.1128/JVI.77.21.11312-11323.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vennema H, Heijnen L, Zijderveld A, Horzinek MC, Spaan WJ. Intracellular transport of recombinant coronavirus spike proteins: implications for virus assembly. J Virol. 1990;64:339–346. doi: 10.1128/jvi.64.1.339-346.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amici C, Di Coro A, Ciucci A, Chiappa L, Castilletti C, Martella V, Decaro N, Buonavoglia C, Capobianchi MR, Santoro MG. Indomethacin has a potent antiviral activity against SARS coronavirus. Antivir Ther. 2006;11:1021–1030. [PubMed] [Google Scholar]
- Verheije MH, Wurdinger T, van Beusechem VW, de Haan CA, Gerritsen WR, Rottier PJ. Redirecting coronavirus to a nonnative receptor through a virus-encoded targeting adapter. J Virol. 2006;80:1250–1260. doi: 10.1128/JVI.80.3.1250-1260.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]