Abstract
The molecular mechanism responsible for a decline of stem cell functioning after replicative stress remains unknown. We used mouse embryonic fibroblasts (MEFs) and hematopoietic stem cells (HSCs) to identify genes involved in the process of cellular aging. In proliferating and senescent MEFs one of the most differentially expressed transcripts was Enhancer of zeste homolog 2 (Ezh2), a Polycomb group protein (PcG) involved in histone methylation and deacetylation. Retroviral overexpression of Ezh2 in MEFs resulted in bypassing of the senescence program. More importantly, whereas normal HSCs were rapidly exhausted after serial transplantations, overexpression of Ezh2 completely conserved long-term repopulating potential. Animals that were reconstituted with 3 times serially transplanted control bone marrow cells all died due to hematopoietic failure. In contrast, similarly transplanted Ezh2-overexpressing stem cells restored stem cell quality to normal levels. In a “genetic genomics” screen, we identified novel putative Ezh2 target or partner stem cell genes that are associated with chromatin modification. Our data suggest that stabilization of the chromatin structure preserves HSC potential after replicative stress.
Introduction
Hematopoietic stem cell (HSC) self-renewal is driven by both intrinsic and extrinsic factors, but the molecular mechanism specifying whether developmental potential is lost or retained during asymmetric cell divisions is unknown. Serial transplantation studies have clearly indicated that self-renewal potential of HSCs is impaired after replicative stress.1,2 HSC activity may be irreversibly lost in a single cell division,3,4 indicating that the epigenetic regulation of gene expression, largely dictated by the histone code, may play an important role. Recently, substantial attention has focused on the role of Polycomb group (PcG) proteins in stem cell self-renewal. To date, 2 distinct Polycomb repressive complexes, (PRCs) 1 and 2, have been identified. Mammalian PRC1 contains Cbx, Mph, Ring, Bmi1, and Mel185 and is thought to be important in the maintenance of gene repression. The second complex, PRC2, contains Ezh2, Eed, and Su(z)125 and is thought to be involved in initiation of gene repression. PcG complexes are targeted to cis-regulatory Polycomb response elements (PREs) by DNA-binding transcription factors.
The role of only few PcG proteins in murine hematopoiesis has been established. Melo18 (Pcqf2) negatively regulates self-renewal of HSCs because its loss leads to an increase of HSCs in G0 and to enhanced HSC self-renewal.6 Mph1/Rae28 (Phc1) mutant mice are embryonic lethal because HSC activity in these animals is not sufficient to maintain hematopoiesis during embryonic development.7 Bmi1 (Pcgf4)-/- HSCs derived from fetal liver were not able to contribute to long-term hematopoiesis in competitive repopulation experiments, demonstrating cell autonomous impairment of their self-renewal potential.8 In contrast, overexpression of Bmi-1 extends the replicative lifespan of mouse and human fibroblasts and causes lymphomas in transgenic mice.9 Heterozygosity for a null allele of Eed caused marked myeloproliferative and lymphoproliferative defects, indicating a negative regulation of cell cycle activity of both lymphoid and myeloid progenitor cells.10 Eed, which is contained in the PRC2 complex, seems to have an opposite effect on hematopoiesis than proteins that are present in PRC1.
Deficiency of Ezh2, also present in the PRC2 complex, resulted in lethality at early stages of mouse development. Mutant blastocysts either ceased developing after implantation or failed to complete gastrulation.11 Conditional deletion of Ezh2 in HSCs resulted in impaired T-cell generation after transplantation.12 Expression levels of EZH2 increased with differentiation of bone marrow cells.13 In contrast, EZH2 was down-regulated on differentiation of HL-60 cells to mature granulocytes.14 In humans EZH2 is commonly up-regulated in different cancer types.15-20 Inhibition of Ezh2 levels by antisense oligonucleotides or siRNA experiments led to cell cycle arrest.14,15,21 Recently, it has been shown that EZH2 is down-regulated in senescent human fibroblasts,22 implying a role for EZH2 during cellular senescence.
During development, PcG protein complexes are thought to maintain long-term and heritable gene silencing through local alterations of the chromatin structure.23 Methylation of the N-terminal tail of histone H3 causes stable conformational changes in chromatin that define the status of gene expression.24,25 The Drosophila Enhancer of zeste (E(z)) protein contains histone methyltransferase (HMT) activity in its SET domain, which is specific for lysines 9 and 27 of H3.26 It has been suggested that E(z) plays an important role not only in the establishment of PcG-specific methylation marks but also in their maintenance during replication, as well as after recruiting PRC1.27 E(z)-null Drosophila showed general decondensation of chromatin,28 reflected by increased chromosome breakage and a low mitotic index.29 In mammalian cells, histone deacetylases (HDACs) 1 and 2 are also present in the Eed-Ezh2 complex,30 suggesting that transcriptional repression of this complex is also mediated via histone deacetylation. In addition, PcG genes are known to play an important role in maintaining the silent state of Hox gene expression.31 Overexpression of specific Hox genes resulted in increased HSC pool size.32
Although an important function for several PcG genes in hematopoiesis has been established, the role of Ezh2 has not yet been elucidated. In the current study, Ezh2 was identified as a senescence-preventing gene in a genetic screen using young and senescent mouse embryonic fibroblasts. Moreover, Ezh2 was abundantly expressed in highly purified HSCs and was rapidly down-regulated on differentiation. We established that overexpression of Ezh2 in HSCs fully preserves stem cell potential and prevents HSC exhaustion even after multiple rounds of serial transplantation. Finally, we identified putative novel partner or target genes of Ezh2 in stem cells.
Materials and methods
Animals
Timed pregnant female C57BL/6 mice were used to obtain day 14 postcoitus embryos from which mouse embryonic fibroblasts (MEFs) were isolated. Eight- to 12-week-old female B6.SJL-PtprcaPep3b/BoyJ (CD45.1) mice were used as donors for transplantation and were bred at the Central Animal Facility of the University of Groningen. Eight- to 12-week-old female C57BL/6 (CD45.2) mice were purchased from Harlan (Horst, The Netherlands) and were used as recipients.
MEFs
Day 14 postcoitus embryos were mechanically dissociated into single cells and cultured in DMEM with 10% FCS and penicillin and streptomycin (Invitrogen, Breda, The Netherlands). MEFs were passaged every 3 to 4 days and viable cells were counted by trypan blue exclusion. Population doublings were calculated as per Todaro and Green.33
Gene array analysis
Total RNA was isolated from passage 1 (young p1) and passage 5 (aged p5) MEFs with a RNeasy mini kit (Qiagen, Venlo, The Netherlands). During cDNA synthesis, samples were labeled with [33P]-deoxycytidine triphosphate (dCTP; MP Biochemicals, Irvine, CA). Mouse filter gene arrays (GF400a; Research Genetics, Invitrogen) were hybridized and analysis was performed as described previously.34 Expression patterns were verified by reverse transcription-polymerase chain reaction (RT-PCR).
Retroviral vectors
The MIEV vector (a kind gift of Prof Dr C. Jordan, University of Rochester, Rochester, NY) contains an internal ribosomal entry site (IRES) sequence and the gene for enhanced green fluorescent protein (eGFP). This MIEV vector served as a control and backbone for cloning of an Ezh2 cDNA upstream of the IRES to create MIEV-Ezh2 vector (Figure 1D).
Figure 1.
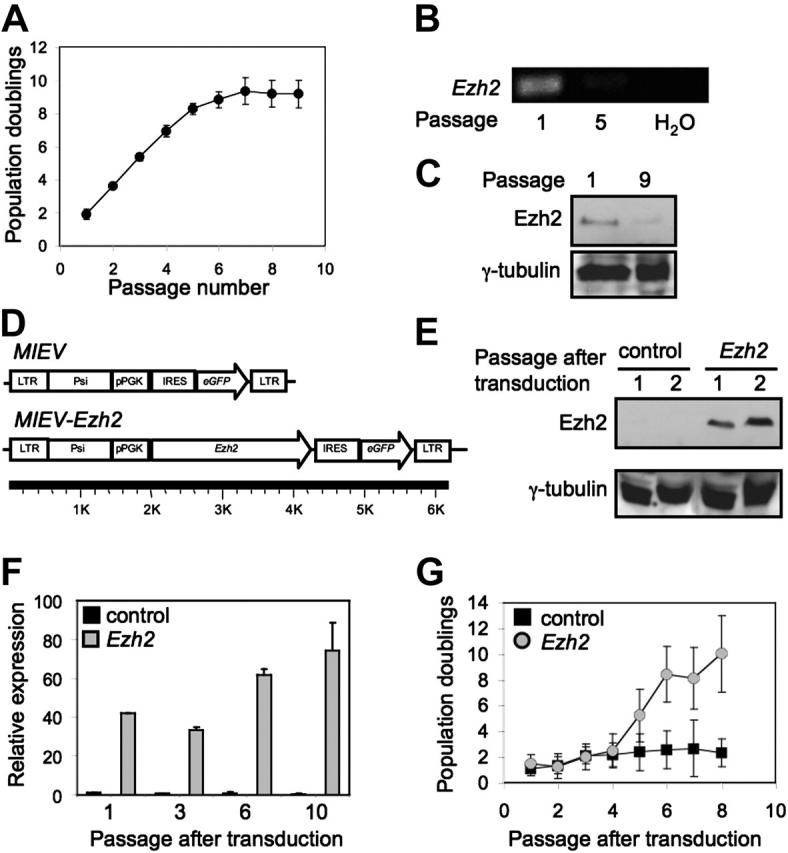
Expression of Ezh2 in MEFs. (A) At first MEFs showed rapid proliferation, but senesced after 8 population doublings. (B) RT-PCR analysis of young (p1) and aged (p5) MEFs. (C) Protein levels of Ezh2 in untreated MEFs at passage 1 and 9. (D) Schematic representation of retroviral vectors used to overexpress Ezh2. (E) Detection of Ezh2 protein in MEFs transduced with control and Ezh2 vector at different passages after transduction. (F) Relative expression of Ezh2 mRNA in MEFs transduced with the control vector (▪) or with the vector containing Ezh2 ( ) at different passages after transduction. Expression levels of Ezh2 were determined by quantitative PCR and calculated relative to MEFs transduced with control at passage 1 after transduction. (G) Growth of MEFs after retroviral transduction (control, ▪; Ezh2,
) at different passages after transduction. Expression levels of Ezh2 were determined by quantitative PCR and calculated relative to MEFs transduced with control at passage 1 after transduction. (G) Growth of MEFs after retroviral transduction (control, ▪; Ezh2,  .
.
Ecotropic Phoenix packaging cells (Nolan Lab, Stanford, CA) were transfected with 2 μg plasmid DNA and Fugene 6 (Roche, Basel, Switzerland), 24 hours after plating 3 × 105 cells/well in a 6-well plate. Virus-containing supernatants from transfected ecotropic Phoenix packaging cells were used to infect MEFs and HSCs.
Retroviral overexpression of Ezh2 in MEFs
MEFs were plated at a density of 105 cells/well in a 6-well plate 24 hours prior to transduction. Viral supernatant was harvested 48 and 72 hours after transfection and added to the MEFs with addition of 8 μg Polybrene (Sigma, St Louis, MO). Three days later GFP+ cells were selected using a MoFlo flow cytometer (DakoCytomation, Fort Collins, CO). The experiment was performed 3 times in triplicate.
Retroviral overexpression of Ezh2 in HSCs
Primary bone marrow (BM) cells were transduced as previously described with some adjustments.35 Briefly, BM cells were extracted from mice (CD45.1) given intraperitoneal injections of 150 mg/kg 5-fluorouracil (5-FU; Pharmachemie Haarlem, Haarlem, The Netherlands) 4 days earlier. Cells were cultured for 48 hours in StemSpan (StemCell Technologies, Vancouver, BC, Canada) supplemented with 10% FCS, 300 ng/mL PEG-rrSCF (Amgen, Thousand Oaks, CA), 20 ng/mL rmIL-11 (R&D Systems, Minneapolis, MN), 10 ng/mL Flt3 ligand (Amgen), penicillin, and streptomycin. Viral supernatant was harvested 24 and 48 hours after transfection of ecotropic Phoenix cells and inoculated in 6-well plates that were coated with 50 μg retronectin (Takara, Kyoto, Japan). Plates containing the viral supernatant were spun for 1 hour at 800 g at room temperature. Four hours later viral supernatant was removed, and 7.5 × 105 cultured BM cells were inoculated per well together with 4 μg Polybrene (Sigma). At the second transduction 2 μg Polybrene was added. Four days later transduction efficiency was determined by flow cytometry (FACSCalibur; Becton Dickinson, Palo Alto, CA).
Transplantation
After transduction, 3 × 106 CD45.1 BM cells were transplanted into lethally irradiated (9.5Gy, IBL 637 137Cs γ source; CIS Biointernational, Gif-sur-Yvette, France) recipients (CD45.2). GFP+ cells were not selected after the transduction protocol, so transplants consisted of a mixture of transduced and nontransduced cells. At several points after transplantation, blood was drawn from the retro-orbital plexus and white blood cell (WBC) numbers were counted using a Coulter Counter (Beckman Coulter, Fullerton, CA). The percentage GFP+CD45.1+ leukocytes was assessed by flow cytometry (FACSCalibur; Becton Dickinson). Two independent experiments were carried out. The total number of primary recipients was 11 controls and 11 for Ezh2.
Serial transplantation
Four months after primary transplantation, recipients of the control and Ezh2 group were killed and BM cells were isolated by flushing 2 hind legs. Unfractionated BM cells were transplanted into lethally irradiated (9.5 Gy) secondary recipients (CD45.2) in limiting dilution with 106 freshly isolated C57BL/6 competitor BM cells (CD45.2). Reconstitution by the transduced HSCs was assessed for each recipient 3 months after serial transplantation by analysis of peripheral blood cell chimerism. Animals showing more than 1% GFP+ engraftment in myeloid and lymphoid lineages were considered to be reconstituted by the transduced HSCs. The competitive repopulating index (CRI) and the frequency of long-term repopulating cells present in primary recipients (LTRA) were calculated. The formula to calculate the CRI was: CRI = [(% CD45.1+GFP+ cells/% CD45.1+GFP- cells)/(ratio of CD45.1+GFP+/CD45.1+GFP- cells transplanted)]. The entire experiment was repeated twice. In total, 35 secondary recipients were given transplants with Ezh2-overexpressing cells and 35 recipients with control cells. In addition, a secondary transplantation without cotransplantation of competitors was performed. A total of 2 × 106 BM cells were transplanted into 10 lethally irradiated (9.5 Gy) recipients.
Four months after secondary transplantation, a tertiary transplantation was performed, identical to the secondary transplantation. For both control and Ezh2-overexpressing cells, a total of 28 recipients was used for competitive repopulation assays and 12 animals underwent transplantation without additional support of competitor BM cells.
Sorting hematopoietic cell populations
Different populations of hematopoietic cells were sorted using flow cytometry as described before.34 The 4 different populations (Lin-Sca-1- c-kit-, Lin-Sca-1+c-kit-, Lin-Sca-1-c-kit+, Lin-Sca-1+c-kit+) were directly sorted in RNA lysis buffer of the RNeasy mini kit (Qiagen), using a MoFlo flow cytometer (DakoCytomation) for gene expression analysis. In addition, Lin-Sca-1+c-kit+ cells were sorted for further culturing.
To assess clonogenic activity in purified stem cell fractions after primary transplantation, the cobblestone area-forming cell (CAFC) assay was performed as described previously.36 Early-appearing CAFCs day 7 correspond to relatively committed progenitor cells, whereas late-appearing CAFCs day 35 reflect more primitive cell subsets.
In vitro differentiation of purified HSCs
Purified Lin-Sca-1+c-kit+ (LSK) cells were cultured in IMDM (Invitrogen) containing 100 ng/mL PEG-rrSCF (Amgen) and 10 ng/mL GM-CSF (Behringwerke, Marburg, Germany) to stimulate rapid in vitro differentiation. Total cell number was assessed using trypan blue exclusion. At different time points during culturing RNA was isolated for gene expression analysis.
Western blotting
Cells were resuspended in PBS, lysed by addition of sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) sample buffer, and sonicated. Proteins were separated by 10% SDS-PAGE and transferred to nitrocellulose. After blocking in 3% skim milk, membranes were probed with a 1:100 dilution of mouse anti-Ezh2 mAb (M18EZH2, kindly provided by Prof Dr A. P. Otte, Swammerdam Institute for Life Science, University of Amsterdam, The Netherlands37,38), washed, and incubated with anti-mouse horseradish peroxidase-conjugated secondary antibody (Amersham Biosciences, Buckinghamshire, United Kingdom). Membranes were developed using enhanced chemiluminescence (ECL) reagents (Amersham Biosciences). Equal loading of membranes was verified with rabbit anti-γ-tubulin (Sigma).
Quantitative PCR
Total RNA was isolated using the RNeasy Mini kit (Qiagen) and standard cDNA synthesis was performed. Quantitative PCRs (qPCRs) were performed in triplicate. PCR amplification, using SYBR Green, was performed in 96-well microtiter plates in an iCycler thermal cycler (Bio-Rad, Hercules, CA). Sample cDNAs were compared with expression of house-keeping genes Gapdh and actin using the relative quantification ΔΔCT technique.39 Relative expression levels in the different LSK populations were estimated by first calculating the number of molecules formed at reaching the CT. By correcting this value for the initial number of cells used, relative expression was determined.
Search for putative targets or partners of Ezh2
To find putative targets or partners of Ezh2, we used The GeneNetwork, an online stem cell database.40 We have recently described the establishment of this database.41 The GNF Hematopoietic Cells U74Av2 (Mar04) RMA database was chosen and information on Ezh2 expression levels in different BXD strains was retrieved. Subsequently, linkage analysis as implemented in GeneNetwork40 was performed to determine by which locus differences in expression levels of Ezh2 were regulated. Furthermore, it is feasible to identify which stem cell transcripts show similar or opposite expression profiles as Ezh2 across the 30 BXD strains. These transcripts are potential partners or targets of Ezh2. The top 100 best correlating transcripts were selected and imported in WebGestalt (http://genereg.ornl.gov/webgestalt/, a web-based Gene Set Analysis Toolkit implemented in GeneNetwork40).
Statistical analysis
Statistical differences between means were assessed using the 2-tailed t test assuming unequal variances and ANOVA with 2 factors. Kaplan-Meier survival analysis was performed for mice that received a tertiary transplant of control or Ezh2-transduced HSCs. Differences in survival were tested for significance using a log-rank test. Statistical analysis was performed using the SPSS (Chicago, IL) statistical package.
Results
Ezh2 and cellular senescence in MEFs
An established in vitro model to study cellular senescence involves the serial passaging of MEFs.42 MEFs were cultured from 14-day-old C57BL/6 embryos. As expected, MEFs were able to undergo a limited number (8) of population doublings before they senesced (Figure 1A). To screen for genes that were differently expressed in young versus senescent MEFs, expression-profiling experiments were conducted. One of the most differentially expressed transcripts was Enhancer of zeste homolog 2 (Ezh2) (see Table S1 for all differentially expressed genes; see the Supplemental Table link at the top of the online article, at the Blood website). Differences in expression levels of Ezh2 in young and senescent MEFs were confirmed by RT-PCR (Figure 1B). Protein levels of Ezh2 in MEFs correlated with RNA levels; Ezh2 was more abundant at early passage (p1) compared to senescent MEFs (p9; Figure 1C). We next established the growth kinetics of MEFs in which Ezh2 was ectopically overexpressed using retroviral transductions. The retroviral vectors used to induce overexpression of Ezh2 are shown schematically in Figure 1D. As control we used a vector that only expresses eGFP (MIEV). The MIEV vector (Ezh2) contains Ezh2 and eGFP sequences, separated by an IRES. Western blotting failed to detect expression of Ezh2 in MEFs transduced with the control vector, whereas protein levels were clearly increased in cells transduced with the Ezh2 vector (Figure 1E). At different passages after retroviral transduction expression levels of Ezh2 were assessed by qPCR (Figure 1F). We confirmed that Ezh2 was readily overexpressed in MEFs transduced with the Ezh2 vector relative to cells transduced with the control vector. Levels of overexpression gradually increased from 40-fold to 70-fold at different time points after transduction. MEFs transduced with the control vector senesced rapidly after transduction (Figure 1G). In contrast, MEFs in which Ezh2 was overexpressed were able to escape senescence and showed continuous growth several passages after transduction (Figure 1G).
Ezh2 expression in HSCs
Different members of the PcG gene family have recently been implicated as having a role in HSC self-renewal.6-8 In contrast, the potential function of Ezh2 in long-term repopulating HSCs has not been investigated. To quantify Ezh2 expression in different hematopoietic cell populations, BM cells were isolated and stained with a cocktail of lineage (Lin)-specific antibodies and for the stem cell markers Sca-1 and c-kit. The 5% most lineage-negative fraction (Figure 2A) was further subfractionated in 4 different populations based on Sca-1 and c-kit expression (Figure 2B), and levels of Ezh2 were determined for all 4 fractions by qPCR (Figure 2C). In 2 separate experiments, highest expression levels were found in the Lin-Sca-1-c-kit+ population, which is known to be enriched for progenitors.43 The Lin-Sca-1+c-kit+ (LSK) population is highly enriched for the most primitive HSCs.44-47
Figure 2.
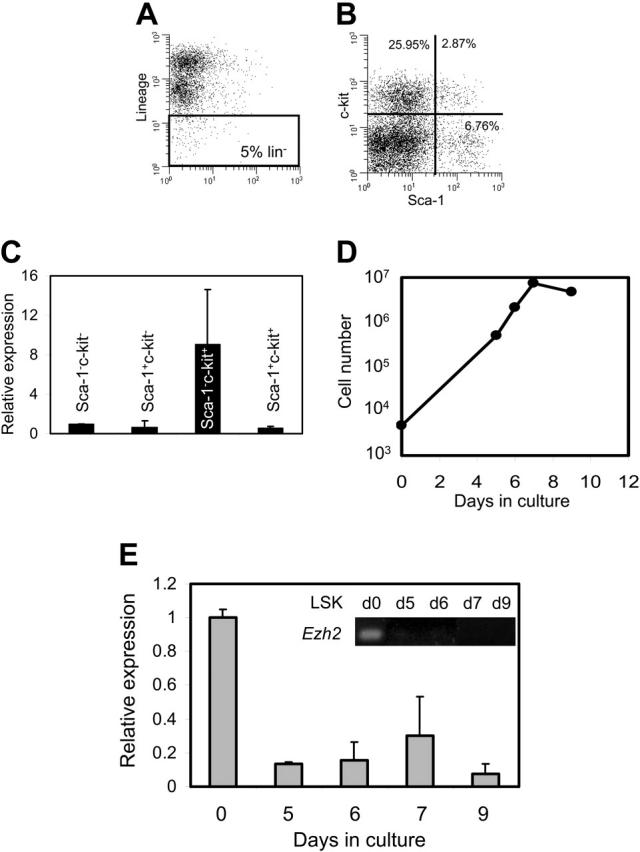
Expression of Ezh2 in hematopoietic cells. (A) To isolate the different lineage-negative (Lin-) populations from BM, the 5% most Lin- cells were selected. (B) Cells in the Lin- population were sorted based on Sca-1 and c-kit surface markers. (C) Expression of Ezh2 as measured by qPCR in the different Sca-1 and c-kit populations relative to Sca-1-c-kit- BM cells. (D) Growth of Lin-Sca-1+c-kit+ cells cultured in the presence of GM-CSF and SCF. (E) Relative expression of Ezh2 was monitored by qPCR at different time points after initiation of differentiation with GM-CSF and SCF. Day 0 was set at 1. Insert shows gel analysis of amplified RT-PCR products.
Next, LSK cells were purified and differentiation was induced by culture in the presence of GM-CSF and SCF. As expected, LSK cells showed rapid growth during the first few days in culture and stopped proliferating at day 7 (Figure 2D) with accompanying changes in morphology (data not shown). At several time points after initiation of differentiation the relative expression of Ezh2 was assessed by quantitative PCR. Ezh2 expression was rapidly down-regulated on differentiation (Figure 2E).
Overexpression of Ezh2 in HSCs
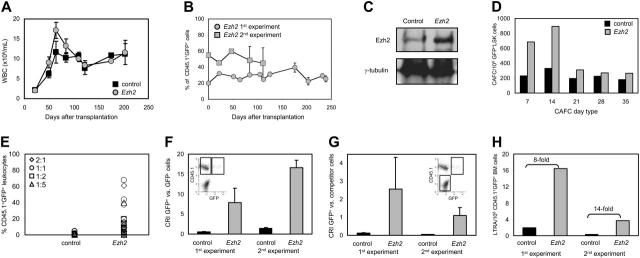
Primary transplantation. Because Ezh2 was expressed in freshly isolated HSCs, but down-regulated during differentiation-inducing cultures, we wished to assess the functional consequences of enforced overexpression of Ezh2 in HSCs. The 5-FU-treated BM cells (CD45.1) were transduced with the control or Ezh2 vector and 3 × 106 cells were transplanted without sorting for GFP+ cells, in lethally irradiated CD45.2 recipients. In this way, the relative contribution of Ezh2-overexpressing (CD45.1+GFP+) stem cells compared to identically treated, but nontransduced stem cells (CD45.1+GFP-) could be determined. Shortly after transplantation, peripheral WBC counts were somewhat higher (P = .15) in mice given Ezh2 BM cells, but returned to normal levels and remained similar to control values as from 100 days after transplantation (Figure 3A). After transplantation, chimerism was assessed by quantifying the percentage of GFP+ cells within the donor CD45.1+ cell fraction (Figure 3B). No significant skewing in contribution of transduced (CD45.1+GFP+) cells was observed.
Figure 3.
Overexpression of Ezh2 in HSCs. (A) WBC counts of mice given transplants with BM cells transduced with control (▪) or Ezh2 ( ) vectors (n = 11 recipients per group, 2 independent experiments). Mean values plus or minus standard error of the mean (SEM) are shown. (B) Chimerism at different time points after transplantation. The percentage of donor-derived transduced CD45.1+GFP+ WBCs is shown. Average values ± 1 SEM of 2 independent experiments are shown. (C) Ezh2 protein expression in the spleen about 120 days after primary transplantation of control or Ezh2 CD45.1 BM cells. (D) CAFC frequencies of sorted LSK GFP+ cells 120 days after primary transplantation (control, ▪; Ezh2,
) vectors (n = 11 recipients per group, 2 independent experiments). Mean values plus or minus standard error of the mean (SEM) are shown. (B) Chimerism at different time points after transplantation. The percentage of donor-derived transduced CD45.1+GFP+ WBCs is shown. Average values ± 1 SEM of 2 independent experiments are shown. (C) Ezh2 protein expression in the spleen about 120 days after primary transplantation of control or Ezh2 CD45.1 BM cells. (D) CAFC frequencies of sorted LSK GFP+ cells 120 days after primary transplantation (control, ▪; Ezh2,  ). (E) Chimerism levels of secondary recipients, competitively transplanted with various ratios (2:1, ⋄; 1:1, ○; 1:2, □; 1:5, ▵) of transduced/nontransduced and freshly isolated BM cells, analyzed 3 months after transplantation. Values show data from individual recipients in 2 independent experiments (n = 35/group). (F) CRI calculated for CD45.1+GFP+ (transduced) versus CD45.1+GFP- (nontransduced) cells (see insert). Values (+ 1 SEM) are averages 3 months after secondary transplantation, based on 11 and 24 individual mice in the first and second experiment, respectively (control, ▪; Ezh2,
). (E) Chimerism levels of secondary recipients, competitively transplanted with various ratios (2:1, ⋄; 1:1, ○; 1:2, □; 1:5, ▵) of transduced/nontransduced and freshly isolated BM cells, analyzed 3 months after transplantation. Values show data from individual recipients in 2 independent experiments (n = 35/group). (F) CRI calculated for CD45.1+GFP+ (transduced) versus CD45.1+GFP- (nontransduced) cells (see insert). Values (+ 1 SEM) are averages 3 months after secondary transplantation, based on 11 and 24 individual mice in the first and second experiment, respectively (control, ▪; Ezh2,  ). (G) CRI calculated for transduced cells (CD45.1+GFP+) compared to freshly isolated BM cells (CD45.2+; see insert). Averages values (+ 1 SEM) of 11 and 24 individual mice of the first and second experiment, respectively, are shown (control, ▪; Ezh2,
). (G) CRI calculated for transduced cells (CD45.1+GFP+) compared to freshly isolated BM cells (CD45.2+; see insert). Averages values (+ 1 SEM) of 11 and 24 individual mice of the first and second experiment, respectively, are shown (control, ▪; Ezh2,  ). (H) LTRA frequencies in CD45.1+GFP+ (transduced) cell fractions calculated from limiting dilution analyses 3 months after secondary transplantation in 2 independent experiments (control, ▪; Ezh2,
). (H) LTRA frequencies in CD45.1+GFP+ (transduced) cell fractions calculated from limiting dilution analyses 3 months after secondary transplantation in 2 independent experiments (control, ▪; Ezh2,  ).
).
Secondary transplantation. Primary recipients were killed 4 months after transplantation (at a time point when no skewing of CD45.1+GFP+ contribution was observed). At this time point protein levels of Ezh2 were analyzed in the spleen. As expected, Ezh2 was present at a higher level in mice given transplants with Ezh2-overexpressing cells compared to controls (Figure 3C). LSK GFP+ cells were isolated from primary recipients and assayed for CAFC activity. Overexpression of Ezh2 resulted in a marked increase in CAFC day 7 and 14 frequencies and a more subtle effect for later appearing subsets (Figure 3D).
Secondary recipients were given transplants with harvested donor BM cells alone or in competitive repopulation with different ratios of freshly isolated CD45.2+ BM cells. At multiple time points after transplantation the recipients were analyzed for presence of CD45.1+GFP+ cells. In the 2 independent experiments serially passaged retrovirally transduced control cells (CD45.1+GFP+) showed little to no contribution to the peripheral blood in these secondary recipients, whereas Ezh2-overexpressing stem cells contributed to significantly higher (P < .001) levels (Figure 3E). The CRI, comparing stem cell activity of transduced cells (CD45.1+GFP+) with nontransduced cells (CD45.1+GFP-), was calculated 3 months after secondary transplantation. The CRI is a measurement of the quality of reconstitution of the transplanted cells, where a CRI of 1 by definition indicates equal stem cell potential of the 2 populations. The CRI was about 12-fold higher (P < .02) in Ezh2-overexpressing stem cells, when these were compared with serially passaged, but nontransduced (CD45.1+GFP-) cells (Figure 3F). The CRI was also calculated for transduced cells (CD45.1+GFP+) compared to freshly isolated CD45.2+ BM cells (Figure 3G). A 10-fold decrease of stem cell quality (ie, a CRI of 0.1) can usually be expected after a single serial transplantation1 and was indeed observed for control cells. Strikingly, overexpression of Ezh2 in HSCs completely prevented loss of stem cell quality after serial transplantation, as the CRI levels were maintained at a value of 1 (Figure 3G). CRI measurements do not necessarily correlate with actual stem cell frequency. To quantify stem cell numbers, we used limiting dilution analysis of chimerism data of all secondary recipients. Animals that showed more than 1% donor cell contribution in both myeloid and lymphoid compartments were considered engrafted. The BM of secondary recipients given transplants with Ezh2-overexpressing stem cells contained an 8- to 14-fold higher number of stem cells with long-term repopulating ability, showing that overexpression of Ezh2 results in an increase of the stem cell pool (Figure 3H).
Tertiary transplantation. Four months after secondary transplantation, recipients were killed and BM cells were once more serially transplanted in a third cohort of lethally irradiated recipients with addition of different ratios of freshly isolated CD45.2+ competitor BM cells. Figure 4A shows that virtually all myeloid cells in the peripheral blood were derived from Ezh2-overexpressing HSCs, whereas approximately half of the lymphoid lineage was donor-derived. Similar to primary and secondary recipients, peripheral blood cell values were normal in tertiary recipients of Ezh2-overexpressing stem cells (data not shown). The chimerism levels of all individual recipients of control and Ezh2-overexpressing stem cells 3 months after transplantation are shown in Figure 4B. Virtually no donor contribution was detected in recipients receiving control cells, whereas very high chimerism levels originated from Ezh2-overexpressing stem cells. In agreement, recipients that received serially transplanted control cells without competitor cells all died within 40 days after transplantation, whereas Ezh2-overexpressing stem cells provided radioprotection and long-term repopulation to tertiary recipients (Figure 4C). Similarly as after the secondary transplantation, the CRI was calculated for CD45.1+GFP+ cells over CD45.1+GFP- cells. Figure 4D shows these values for both secondary and tertiary recipients and documents that stem cells transduced with Ezh2 show a 10- and 25-fold increase in stem cell quality, respectively. Remarkably, when the quality of stem cells that were transduced with Ezh2 was compared with freshly isolated BM cells, no loss of stem cell quality could be demonstrated (Figure 4E).
Figure 4.
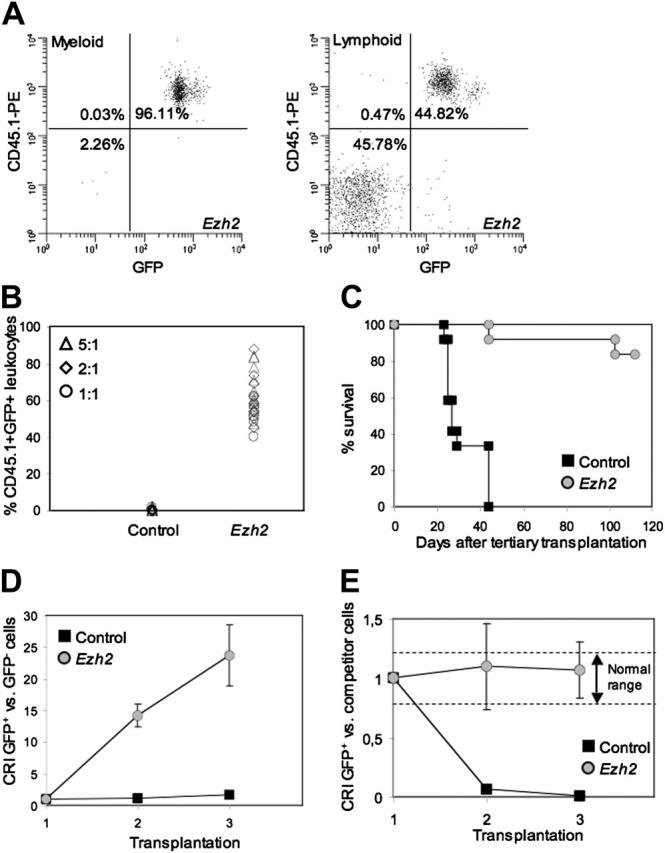
Effects of overexpression of Ezh2 in HSCs in tertiary recipients. (A) Representative fluorescence-activated cell sorting (FACS) plot showing donor contribution in a recipient transplanted with 2.5 × 106 twice serially transplanted Ezh2-overexpressing cells in competition with 5 × 105 freshly isolated CD45.2+ BM cells. Left panel shows contribution when gated on myeloid cells; right panel shows donor contribution for lymphoid cells. (B) The percentage of CD45.1+GFP+ cells in the peripheral blood in all recipients (n = 28) 3 months after tertiary transplantation. Cells were cotransplanted in different ratios (5:1, ▵; 2:1, ⋄; 1:1, ○) with freshly isolated BM cells. (C) Survival curve of tertiary recipients that received transplantations of serially transplanted BM cells without cotransplantation of freshly isolated BM cells (control, ▪; Ezh2,  ). (D) CRI comparing transduced CD45.1+GFP+ stem cells versus nontransduced CD45.1+GFP- stem cells in primary, secondary, and tertiary recipients (control, ▪; Ezh2,
). (D) CRI comparing transduced CD45.1+GFP+ stem cells versus nontransduced CD45.1+GFP- stem cells in primary, secondary, and tertiary recipients (control, ▪; Ezh2,  ). (E) CRI comparing transduced CD45.1+GFP+ stem cells with freshly isolated BM cells after 1, 2, and 3 serial transplantations (control, ▪; Ezh2,
). (E) CRI comparing transduced CD45.1+GFP+ stem cells with freshly isolated BM cells after 1, 2, and 3 serial transplantations (control, ▪; Ezh2,  ).
).
Stem cell gene networks involving Ezh2
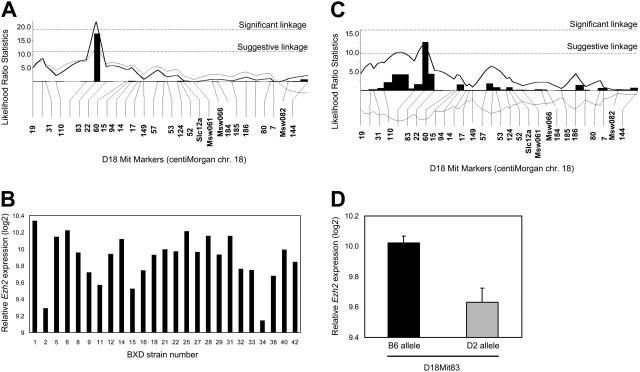
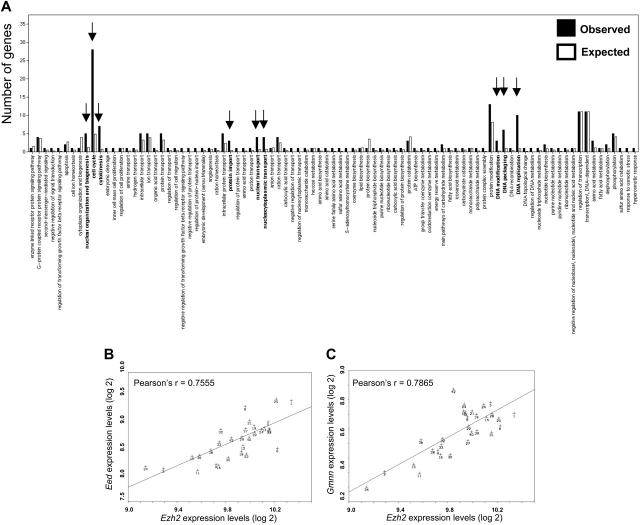
We have previously shown that different inbred strains of mice vary substantially with respect to numerous stem cell traits, including variation in stem cell frequency.48 In a panel of 30 BXD recombinant inbred strains, offspring of DBA/2J mice (containing high numbers of stem cells) and C57BL/6 mice (containing low numbers of stem cells), genomic loci that contribute to these traits were mapped. We identified a quantitative trait locus (QTL) on chromosome 18 that was associated with variation in stem cell pool size (Figure 5A).48 Recently, we have also measured genome-wide variation in gene expression levels in HSCs isolated from these BXD strains.41 This genetic genomic analysis has enabled us to identify clusters of genes that are transcriptionally controlled by a common locus. These data have been made accessible at GeneNetwork,40 an online genetic database that allows the query of coregulated transcripts for stem cell genes of interest. In the present study we determined that Ezh2 was highly, but differentially, expressed in HSCs of the various BXD strains (Figure 5B). Strikingly, variation in Ezh2 levels was largely accounted for by the exact chromosome 18 locus that we had previously associated with variation in stem cell frequency (Figure 5C). The gene encoding Ezh2 is located on chromosome 6 in mice, so variation in Ezh2 expression is regulated in trans by an as yet unidentified gene on chromosome 18. Further analysis showed that mice that had inherited a C57BL/6 allele at D18Mit83 (the marker most strongly associated with variation in Ezh2 levels) had significantly higher expression of Ezh2 (P < .003) than mice carrying a DBA/2J allele (Figure 5D). The limited genetic resolution precludes direct identification of the chromosome 18 gene that is responsible for the variation in Ezh2 gene expression. However, Ezh2 coregulated transcripts in stem cells can easily be retrieved from GeneNetwork.40 The 100 genes of which the transcript levels correlated best with variation in Ezh2 expression were identified. These genes were classified according to their functional annotation using WebGestalt (http://genereg.ornl.gov/webgestalt). We identified 3 functional categories for which transcripts correlating with variation in Ezh2 levels were significantly enriched (Figure 6A). A high number of coregulated stem cell genes was involved in DNA modification, DNA packaging, and DNA replication (Table 1). Two other categories of transcripts that were significantly more often observed than expected were associated with cell cycle/cytokinesis and, surprisingly, nucleocytoplasmic transport (Table 1).
Figure 5.
Variation in CAFC day 35 frequency and Ezh2 expression levels in BXD strains. (A) Variation in HSC frequency (CAFC d35/105 BM cells) in BXD recombinant inbred mice is associated with a QTL mapping to chromosome 18. Data can be retrieved from www.genenetwork.org.41,48 (B) Ezh2 transcript levels were measured in LSK cells isolated from the BM of 30 BXD strains, using Affymetrix gene chips. (C) Variation in Ezh2 expression in LSK cells isolated from BXD mice is regulated by a QTL mapping to chromosome 18. (D). Average Ezh2 expression levels (+ 1 SEM) in BXD recombinant inbred mice segregated according to the presence of the B6 (▪) or D2 ( ) allele for marker D18Mit83.
) allele for marker D18Mit83.
Figure 6.
Putative new stem cell targets or partners of Ezh2. (A) The top 100 stem cell genes whose expression correlated with Ezh2 expression in BXD mice were classified according to function by WebGestalt (http://genereg.ornl.gov/webgestalt/). □, the expected number of genes with a certain function to be found if 100 transcripts were randomly selected; ▪, the actually retrieved numbers. Detailed information on genes present in the 3 significantly (P < .01) enriched clusters (arrows) is provided in Table 1. (B) Correlation of relative expression of Ezh2 and Eed in the 30 BXD strains. Each data point in the figure refers to an individual BXD strain. (C) Correlation of relative expression of Ezh2 and Geminin (Gmnn) in the 30 BXD strains. Each data point in the figure refers to an individual BXD strain.
Table 1.
Putative targets or partners of Ezh2 in HSCs
| Symbol | Name | QTL chromosome | Mb, if on chromosome 18 | Correlation |
|---|---|---|---|---|
| DNA modification, packaging, or replication | ||||
| Mcm4 | Minichromosome maintenance deficient 4 homolog (Saccharomyces cerevisiae) | 14, 18 | 20-23 | 0.7545 |
| Mcm3 | Minichromosome maintenance deficient 3 (S cerevisiae) | 9, 18 | 20-23 | 0.7284 |
| Pcna | Proliferating cell nuclear antigen | 18 | 20-23 | 0.8141 |
| Prim1 | DNA primase, p49 subunit | 18 | 12-14, 20-25 | 0.7986 |
| Hat1 | Histone aminotransferase 1 | 11 | — | 0.7973 |
| Recc1 | Replication factor C 1 | 2 | — | 0.7911 |
| Eed | Embryonic ectoderm development | 18 | 47-50 | 0.7555 |
| Pole2 | Polymerase (DNA directed), ε 2 (p59 subunit) | 18 | 20-23 | 0.751 |
| Mcm2 | Minichromosome maintenance deficient 2 mitotin (S cerevisiae) | 14, 18 | 20-23 | 0.739 |
| Rrm1 | Ribonucleotide reductase M1 | 4 | — | 0.7359 |
| Nap1/1 | Nucleosome assembly protein 1-like 1 | 18 | 47-50 | 0.7294 |
| Rpa3 | Replication protein A3 | 18 | 17-20, 35-38 | 0.7239 |
| H2afz | H2A histone family, member Z | 18 | 12-14 | 0.7115 |
| Setdb1 | SET domain, bifurcated 1 | 6, 17 | — | 0.7102 |
| Ahcy | S-adenosylhomocysteine hydrolase | 11 | — | 0.6855 |
| Dntt | Deoxynucleotidyltransferase, terminal | 15 | — | 0.6851 |
| Aurkb | Aurora kinase B | — | — | 0.718 |
| Nuclear organization and biogenesis, cell cycle, and cytokinesis | ||||
| Top2a | Topoisomerase (DNA) II α | 18 | 20-23 | 0.8733 |
| Smc2/1 | SMC2 structural maintenance of chromosomes 2-like 1 (yeast) | — | — | 0.8507 |
| Topbp1 | Topoisomerase (DNA) II β-binding protein | 18 | 20-25 | 0.8497 |
| Mki67 | Antigen identified by monoclonal antibody Ki 67 | 2, 18 | 20-23 | 0.837 |
| Tfdp1 | Transcription factor Dp 1 | 7 | — | 0.8299 |
| Pole2 | Polymerase (DNA directed), ε 2 (p59 subunit) | 18 | 20-23 | 0.7986 |
| Ccnb2 | Cyclin B2 | 13 | — | 0.7949 |
| Recc1 | Replication factor C 1 | 2 | — | 0.7911 |
| Ect2 | Ect2 oncogene | 4 | — | 0.7879 |
| Gmnn | Geminin | 18 | 14-23 | 0.7865 |
| Cdca5 | Cell division cycle associated 5 | 2 | — | 0.764 |
| Prc1 | Protein regulator of cytokinesis 1 | 5, 7 | — | 0.7584 |
| Nusap1 | Nucleolar and spindle-associated protein 1 | 14, 18 | 35-38, 71-73 | 0.7584 |
| Mcm4 | Minichromosome maintenance deficient 4 homolog (S cerevisiae) | 14, 18 | 20-23 | 0.7545 |
| Ppp1cc | Protein phosphatase 1, catalytic subunit, γ isoform | 18 | 50-53 | 0.751 |
| Nek2 | NIMA (never in mitosis gene a)-related expressed kinase 2 | 7 | — | 0.751 |
| Mad2/1 | MAD2 (mitotic arrest deficient, homolog)-like 1 (yeast) | 18 | 12-13, 35-38 | 0.7428 |
| Mcm2 | Minichromosome maintenance deficient 2 mitotin (S cerevisiae) | 14, 18 | 20-23 | 0.739 |
| Rrm1 | Ribonucleotide reductase M1 | 4 | — | 0.7359 |
| Tacc3 | Transforming, acidic coiled-coil-containing protein 3 | 18 | 12-14 | 0.7335 |
| Prim1 | DNA primase, p49 subunit | 18 | 12-14, 20-25 | 0.7321 |
| Pcna | Proliferating cell nuclear antigen | 18 | 20-23 | 0.7321 |
| Nap1/1 | Nucleosome assembly protein 1-like 1 | 18 | 47-50 | 0.7294 |
| Mcm3 | Minichromosome maintenance deficient 3 (S cerevisiae) | 9, 18 | 20-23 | 0.7284 |
| Rbl1 | Retinoblastoma-like 1 (p107) | 14 | — | 0.7268 |
| Rpa3 | Replication protein A3 | 18 | 17-20, 35-38 | 0.7239 |
| Aurkb | Aurora kinase B | — | — | 0.718 |
| H2afz | H2A histone family, member Z | 18 | 12-14 | 0.7115 |
| Setdb1 | SET domain, bifurcated 1 | 6, 17 | — | 0.7102 |
| Stk6 | Serine/threonine kinase 6 (Aurka) | 9 | — | 0.7062 |
| Dntt | Deoxynucleotidyltransferase, terminal | 15 | — | 0.6851 |
| Cdca7 | Cell division cycle-associated 7 | 9, 18 | 14-23 | 0.6843 |
| Cul2 | Cullin 2 | 18 | 50-54 | 0.682 |
| Smc4/1 | SMC4 structural maintenance of chromosomes 4-like 1 (yeast) | 3, 18 | 12-14, 25-27 | — |
| AA545217 | Expressed sequence AA545217 | 16 | — | — |
| Protein import, nuclear transport, and nucleocytoplasmic transport | ||||
| Kpna2 | Karyopherin (importin) α 2 | 18 | 51-53 | 0.78 |
| Tacc3 | Transforming, acidic coiled-coil-containing protein 3 | 18 | 12-14 | 0.7335 |
| Kpnb3 | Karyopherin (importin) β 3 | 5, 8, 13 | — | 0.7277 |
| Xpo1 | Exportin 1, CRM1 homolog (yeast) | 14 | — | 0.7581 |
Ezh2 coregulated transcripts in stem cells were retrieved from www.genenetwork.org. The 100 genes of which the transcript levels correlated best with variation in Ezh2 expression were identified. These genes were classified according to their functional annotation using Webgestalt (http://genereg.oml.gov/webgestalt). Three functional categories were identified. — indicates not applicable. Correlation values refer to Pearson correlation coefficients between variation in the gene of interest and Ezh2 levels (Figure 6B-C).
Interestingly, one of the stem cell transcripts whose levels correlated with variable Ezh2 expression was Eed, its well-known partner (Figure 6B). Other interesting novel putative Ezh2 partners or targets included Mcm2, Mcm3, and Mcm4; Aurka and Aurkb; Gmnn (Figure 6C); Prim1; topoisomerases; and polymerases. A substantial number of these transcripts was also controlled by the chromosome 18 locus (Table 1).
Discussion
In this study we show that Ezh2 is down-regulated on serial passaging of MEFs and during differentiation of primitive, highly proliferative HSCs to committed myeloid cells. These 2 complementary models suggest a general suppressive role for Ezh2 during cellular differentiation and senescence. Indeed, we show that overexpression of Ezh2 leads to bypassing of the senescence program in MEFs. More strikingly, enforced expression of Ezh2 in HSCs completely prevents stem cell exhaustion and stabilizes normal stem cell functioning, even after 3 serial transplantations.
It has been previously shown by others and us that exhaustion of the HSC pool is apparent following serial transplantation,2,49,50 chemotherapy,51,52 and also during normal aging.36,49 The aging of HSCs is genetically regulated, because different inbred mouse strains show distinct patterns,36,53 which are maintained when HSCs of different mouse strains are present in the same microenvironment.54,55 However, the underlying molecular mechanism responsible for a decline of HSC functioning during aging and serial transplantation remains to be elucidated. Several groups have identified genes that directly influence HSC self-renewal potential and their differentiation program, such as Hoxb4,35 Notch1,56 and Bmi-1.8 Our present data document that Ezh2 is able to completely preserve stem cell functioning after repeated serial transplantation, notably without resulting in malignant transformation. Interestingly, a recent gene profiling study in which expression patterns in young and aged HSCs were compared, documented that Ezh2 levels decrease during normal aging.57
The natural variation in Ezh2 transcript levels that can be observed in stem cells of recombinant inbred mouse strains (BXDs), allows 2 approaches.41 First, the genomic locus that regulates Ezh2 expression can be mapped and, second, coregulated transcripts can be identified. Using the first approach we show that variation in expression of Ezh2 is regulated by a locus on chromosome 18. Strikingly, exactly the same locus is also associated with HSC frequency.48 Because many transcripts are located in the critical interval, the Ezh2-regulating gene on chromosome 18 remains unknown at present. Using the second approach, genetic networks associated with variation in Ezh2 expression can be elucidated. We found that Eed expression levels were positively correlated with Ezh2 gene expression. Because Eed is a well-known partner of Ezh2,31 this finding proves the potential power of this approach. Furthermore, expression levels of Gmnn, a DNA replication inhibitor with which PRC1 members are associated,58 were also positively correlated with Ezh2 expression. We identified many Ezh2 coregulated stem cell genes that qualify as chromatin remodeling factors and DNA binding proteins. Interestingly, the interval on chromosome 18 that was associated with variation in Ezh2 expression levels also regulated the expression levels of 12 of 42 coregulated genes. Because this locus is involved in regulating HSC pool size,48 this strongly suggests that chromatin remodeling gene networks affecting epigenetic transcription programs are critically involved in stem cell self-renewal.
Ezh2 can act as a transcriptional repressor as a result of its HMT activity, which ultimately results in DNA condensation and a more compact chromatin structure. Overexpression of Ezh2 is therefore expected to change global chromatin structure and consequently alter gene expression profiles. Moreover, because methylation is considered to be relatively irreversible, these epigenetic changes of gene expression are stable. In stem cell homeostasis a delicate balance exists between self-renewal and terminal differentiation. It is likely that during replicative stress (resulting from serial transplantation, but also from normal aging) this balance weighs in favor of terminal differentiation, resulting in exhaustion of the HSC pool. Emerging evidence suggests that this balance is partly regulated by the histone code, which specifies gene transcription and provides cellular memory. It has been suggested that during normal aging methylation patterns are less strictly maintained, resulting in loss of heterochromatin and deregulated gene transcription.59 It is becoming evident that PcG genes play an important role here because aberrant expression of different PcG genes has been shown to change the program of stem cell self-renewal.
In this study we show that stem cell exhaustion is readily induced but can be prevented in HSCs by overexpressing Ezh2. These data suggest that after replicative stress of HSCs chromatin alterations occur, which impede stem cell functioning. In contrast, in HSCs that overexpress Ezh2 the chromatin structure is stabilized, which provides maintenance of HSC quality. Chemical modifiers of higher order chromatin structure may have a functional role in clinical ex vivo stem cell expansion protocols.
Supplementary Material
Acknowledgments
The authors would like to thank G. Harms and A. van den Berg for their assistance with the mouse filter gene arrays; C. Jordan for providing us with the MIEV vector; A. Otte for providing us with the Ezh2 antibody; G. Mesander and H. Moes for flow cytometry support; and R. van Os for critically reading the manuscript and for advice on CRI calculations.
Prepublished online as Blood First Edition Paper, November 17, 2005; DOI 10.1182/blood-2005-09-3585.
Supported by a grant from the Dutch Organization of Scientific Research (NWO; grant 901-08-339) and the National Institutes of Health (NIH; grant RO1 HL073710).
L.M.K. designed and performed research, analyzed data, and wrote the paper; L.V.B. performed research, contributed vital new analytical tools, analyzed data, and wrote the paper; A.d.B., S.H., J.D., and E.W. performed research and analyzed data; B.D. performed research; and G.d.H. designed research, contributed vital new analytical tools, and wrote the paper.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
- 1.Harrison DE, Astle CM. Loss of stem cell repopulating ability upon transplantation. Effects of donor age, cell number, and transplantation procedure. J Exp Med. 1982;156: 1767-1779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mauch P, Hellman S. Loss of hematopoietic stem cell self-renewal after bone marrow transplantation. Blood. 1989;74: 872-875. [PubMed] [Google Scholar]
- 3.Nakauchi H, Sudo K, Ema H. Quantitative assessment of the stem cell self-renewal capacity. Ann N Y Acad Sci. 2001;938: 18-24. [DOI] [PubMed] [Google Scholar]
- 4.Takano H, Ema H, Sudo K, Nakauchi H. Asymmetric division and lineage commitment at the level of hematopoietic stem cells: inference from differentiation in daughter cell and granddaughter cell pairs. J Exp Med. 2004;199: 295-302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lund AH, van Lohuizen M. Polycomb complexes and silencing mechanisms. Curr Opin Cell Biol. 2004;16: 239-246. [DOI] [PubMed] [Google Scholar]
- 6.Kajiume T, Ninomiya Y, Ishihara H, Kanno R, Kanno M. Polycomb group gene mel-18 modulates the self-renewal activity and cell cycle status of hematopoietic stem cells. Exp Hematol. 2004; 32: 571-578. [DOI] [PubMed] [Google Scholar]
- 7.Ohta H, Sawada A, Kim JY, et al. Polycomb group gene rae28 is required for sustaining activity of hematopoietic stem cells. J Exp Med. 2002;195: 759-770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Park IK, Qian D, Kiel M, et al. Bmi-1 is required for maintenance of adult self-renewing haematopoietic stem cells. Nature. 2003;423: 302-305. [DOI] [PubMed] [Google Scholar]
- 9.Jacobs JJ, Kieboom K, Marino S, DePinho RA, van Lohuizen M. The oncogene and Polycomb-group gene bmi-1 regulates cell proliferation and senescence through the ink4a locus. Nature. 1999;397: 164-168. [DOI] [PubMed] [Google Scholar]
- 10.Lessard J, Schumacher A, Thorsteinsdottir U, et al. Functional antagonism of the Polycomb-group genes eed and Bmi1 in hemopoietic cell proliferation. Genes Dev. 1999;13: 2691-2703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.O'Carroll D, Erhardt S, Pagani M, et al. The polycomb-group gene Ezh2 is required for early mouse development. Mol Cell Biol. 2001;21: 4330-4336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Su IH, Dobenecker MW, Dickinson E, et al. Polycomb group protein ezh2 controls actin polymerization and cell signaling. Cell. 2005;121: 425-436. [DOI] [PubMed] [Google Scholar]
- 13.Lessard J, Baban S, Sauvageau G. Stage-specific expression of polycomb group genes in human bone marrow cells. Blood. 1998;91: 1216-1224. [PubMed] [Google Scholar]
- 14.Fukuyama T, Otsuka T, Shigematsu H, et al. Proliferative involvement of ENX-1, a putative human polycomb group gene, in haematopoietic cells. Br J Haematol. 2000;108: 842-847. [DOI] [PubMed] [Google Scholar]
- 15.Varambally S, Dhanasekaran SM, Zhou M, et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature. 2002; 419: 624-629. [DOI] [PubMed] [Google Scholar]
- 16.Kleer C, Cao Q, Varambally S, et al. EZH2 is a marker of aggressive breast cancer and promotes neoplastic transformation of breast epithelial cells. Proc Natl Acad Sci USA. 2003;100: 11606-11611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Visser HP, Gunster MJ, Kluin-Nelemans HC, et al. The Polycomb group protein EZH2 is upregulated in proliferating, cultured human mantle cell lymphoma. Br J Haematol. 2001;112: 950-958. [DOI] [PubMed] [Google Scholar]
- 18.Raaphorst FM, van Kemenade FJ, Blokzijl T, et al. Coexpression of BMI-1 and EZH2 polycomb group genes in Reed-Sternberg cells of Hodgkin's disease. Am J Pathol. 2000;157: 709-715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.van Kemenade FJ, Raaphorst FM, Blokzijl T, et al. Coexpression of BMI-1 and EZH2 polycomb-group proteins is associated with cycling cells and degree of malignancy in B-cell non-Hodgkin lymphoma. Blood. 2001;97: 3896-3901. [DOI] [PubMed] [Google Scholar]
- 20.Rhodes DR, Yu J, Shanker K, et al. Large-scale meta-analysis of cancer microarray data identifies common transcriptional profiles of neoplastic transformation and progression. Proc Natl Acad Sci U S A. 2004;101: 9309-9314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jacobs JJ, van Lohuizen M. Polycomb repression: from cellular memory to cellular proliferation and cancer. Biochim Biophys Acta. 2002;1602: 151-161. [DOI] [PubMed] [Google Scholar]
- 22.Tang X, Milyavsky M, Shats I, et al. Activated p53 suppresses the histone methyltransferase EZH2 gene. Oncogene. 2004;23: 5759-5769. [DOI] [PubMed] [Google Scholar]
- 23.Pirrotta V. Chromatin complexes regulating gene expression in Drosophila. Curr Opin Genet Dev. 1995;5: 466-472. [DOI] [PubMed] [Google Scholar]
- 24.Strahl BD, Allis CD. The language of covalent histone modifications. Nature. 2000;403: 41-45. [DOI] [PubMed] [Google Scholar]
- 25.Kouzarides T. Histone methylation in transcriptional control. Curr Opin Genet Dev. 2002;12: 198-209. [DOI] [PubMed] [Google Scholar]
- 26.Cao R, Wang L, Wang H, et al. Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science. 2002;298: 1039-1043. [DOI] [PubMed] [Google Scholar]
- 27.Breiling A, O'Neill LP, D'Eliseo D, Turner BM, Orlando V. Epigenome changes in active and inactive polycomb-group-controlled regions. EMBO Rep. 2004;5: 976-982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rastelli L, Chan CS, Pirrotta V. Related chromosome binding sites for zeste, suppressors of zeste and Polycomb group proteins in Drosophila and their dependence on Enhancer of zeste function. EMBO J. 1993;12: 1513-1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gatti M, Baker BS. Genes controlling essential cell-cycle functions in Drosophila melanogaster. Genes Dev. 1989;3: 438-453. [DOI] [PubMed] [Google Scholar]
- 30.van der Vlag J, Otte AP. Transcriptional repression mediated by the human polycomb-group protein EED involves histone deacetylation. Nat Genet. 1999;23: 474-478. [DOI] [PubMed] [Google Scholar]
- 31.Cao R, Zhang Y. The functions of E(Z)/EZH2-mediated methylation of lysine 27 in histone H3. Curr Opin Genet Dev. 2004;14: 155-164. [DOI] [PubMed] [Google Scholar]
- 32.Owens BM, Hawley RG. HOX and non-HOX homeobox genes in leukemic hematopoiesis. Stem Cells. 2002;20: 364-379. [DOI] [PubMed] [Google Scholar]
- 33.Todaro GJ, Green H. Quantitative studies of the growth of mouse embryo cells in culture and their development into established lines. J Cell Biol. 1963;17: 299-313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.de Haan G, Bystrykh LV, Weersing E, et al. A genetic and genomic analysis identifies a cluster of genes associated with hematopoietic cell turnover. Blood. 2002;100: 2056-2062. [DOI] [PubMed] [Google Scholar]
- 35.Sauvageau G, Thorsteinsdottir U, Eaves CJ, et al. Overexpression of HOXB4 in hematopoietic cells causes the selective expansion of more primitive populations in vitro and in vivo. Genes Dev. 1995;9: 1753-1765. [DOI] [PubMed] [Google Scholar]
- 36.de Haan G, Nijhof W, Van Zant G. Mouse strain-dependent changes in frequency and proliferation of hematopoietic stem cells during aging: Correlation between lifespan and cycling activity. Blood. 1997;89: 1543-1550. [PubMed] [Google Scholar]
- 37.Hamer K, Sewalt R, den Blaauwen J, et al. A panel of monoclonal antibodies against human polycomb group proteins. Hybrid Hybridomics. 2002;21: 245-252. [DOI] [PubMed] [Google Scholar]
- 38.Sewalt RG, van der Vlag J, Gunster MJ, et al. Characterization of interactions between the mammalian polycomb-group proteins Enx1/EZH2 and EED suggests the existence of different mammalian polycomb-group protein complexes. Mol Cell Biol. 1998;18: 3586-3595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25: 402-408. [DOI] [PubMed] [Google Scholar]
- 40.University of Tennessee. The GeneNetwork. http://www.genenetwork.org. Accessed on March 11, 2005.
- 41.Bystrykh L, Weersing E, Dontje B, et al. Uncovering regulatory pathways that affect hematopoietic stem cell function using `genetical genomics'. Nat Genet. 2005;37: 225-232. [DOI] [PubMed] [Google Scholar]
- 42.Hayflick L. The limited in vitro lifetime of human diploid cell strains. Exp Cell Res. 1965;37: 614-636. [DOI] [PubMed] [Google Scholar]
- 43.Ogawa M, Matsuzaki Y, Nishikawa S, et al. Expression and function of c-kit in hematopoietic progenitor cells. J Exp Med. 1991;174: 63-71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Morrison SJ, Uchida N, Weissman IL. The biology of hematopoietic stem cells. Annu Rev Cell Dev Biol. 1995;11: 35-71. [DOI] [PubMed] [Google Scholar]
- 45.Spangrude GJ, Heimfeld S, Weissman IL. Purification and characterization of mouse hematopoietic stem cells. Science. 1988;241: 58-62. [DOI] [PubMed] [Google Scholar]
- 46.Trevisan M, Iscove NN. Phenotypic analysis of murine long-term hemopoietic reconstituting cells quantitated competitively in vivo and comparison with more advanced colony-forming progeny. J Exp Med. 1995;181: 93-103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Okada S, Nakauchi H, Nagayoshi K et al. Enrichment and characterization of murine hematopoietic stem cells that express c-kit molecule. Blood. 1991;78: 1706-1712. [PubMed] [Google Scholar]
- 48.de Haan G, Van Zant G. Intrinsic and extrinsic control of hemopoietic stem cell numbers: mapping of a stem cell gene. J Exp Med. 1997;186: 529-536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kamminga LM, van Os R, Ausema A, et al. Impaired hematopoietic stem cell functioning after serial transplantation and during normal aging. Stem Cells. 2005;23: 82-92. [DOI] [PubMed] [Google Scholar]
- 50.Morrison SJ, Wandycz AM, Akashi K, Globerson A, Weissman IL. The aging of hematopoietic stem cells. Nat Med. 1996;2: 1011-1016. [DOI] [PubMed] [Google Scholar]
- 51.Gardner RV, Astle CM, Harrison DE. Hematopoietic precursor cell exhaustion is a cause of proliferative defect in primitive hematopoietic stem cells (PHSC) after chemotherapy. Exp Hematol. 1997;25: 495-501. [PubMed] [Google Scholar]
- 52.Van Zant G, Liang Y. The role of stem cells in aging. Exp Hematol. 2003;31: 659-672. [DOI] [PubMed] [Google Scholar]
- 53.Chen JC, Astle CM, Harrison DE. Genetic regulation of primitive hematopoietic stem cell senescence. Exp Hematol. 2000;28: 442-450. [DOI] [PubMed] [Google Scholar]
- 54.Kamminga LM, Akkerman I, Weersing E, et al. Autonomous behavior of hematopoietic stem cells. Exp Hematol. 2000;28: 1451-1459. [DOI] [PubMed] [Google Scholar]
- 55.Van Zant G, Scott-Micus K, Thompson BP, Fleischman RA, Perkins S. Stem cell quiescence/activation is reversible by serial transplantation and is independent of stromal cell genotype in mouse aggregation chimeras. Exp Hematol. 1992;20: 470-475. [PubMed] [Google Scholar]
- 56.Stier S, Cheng T, Dombkowski D, Carlesso N, Scadden DT. Notch1 activation increases hematopoietic stem cell self-renewal in vivo and favors lymphoid over myeloid lineage outcome. Blood. 2002;99: 2369-2378. [DOI] [PubMed] [Google Scholar]
- 57.Rossi DJ, Bryder D, Zahn JM, et al. Cell intrinsic alterations underlie hematopoietic stem cell aging. Proc Natl Acad Sci U S A. 2005;102: 9194-9199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Luo L, Yang X, Takihara Y, Knoetgen H, Kessel M. The cell-cycle regulator geminin inhibits Hox function through direct and polycomb-mediated interactions. Nature. 2004;427: 749-753. [DOI] [PubMed] [Google Scholar]
- 59.Villeponteau B. The heterochromatin loss model of aging. Exp Gerontol. 1997;32: 383-394. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.