Abstract
IL-18 is known to induce IFN-γ production, which is enhanced when combined with IL-2. In the present study, we investigated whether the combination of exogenous IL-2 and IL-18 alters airway hyperresponsiveness (AHR) and airway inflammation. Sensitized mice exposed to ovalbumin (OVA) challenge developed AHR, inflammatory cells in the bronchoalveolar lavage (BAL) fluid, and increases in levels of Th2 cytokines and goblet cell numbers. The combination of IL-2 and IL-18, but neither alone, prevented these changes while increasing levels of IL-12 and IFN-γ. The combination of IL-2 and IL-18 was ineffective in IFN-γ–deficient and signal transducer and activator of transcription (STAT)4-deficient mice. Flow cytometry analysis showed significant increases in numbers of IFN-γ–positive natural killer (NK) cells in the lung after treatment with the combination therapy, and transfer of lung NK cells isolated from sensitized and challenged mice treated with the combination significantly suppressed AHR and BAL eosinophilia. These data demonstrate that the combination of IL-2 and IL-18 prevents AHR and airway inflammation, likely through IL-12–mediated induction of IFN-γ production in NK cells.
Keywords: IL-2, IL-18, STAT4, IFN-γ, airway hyperresponsiveness
CLINICAL RELEVANCE
The data support the concept that manipulation of critical cytokines, in this case IL-2 and IL-18, can significantly impact allergic airway inflammation. Thus, the data support the use of these cytokines to interfere with asthma pathophysiology.
The pathogenesis of asthma is complex, and involves a variety of factors, inflammatory cells, cytokines and chemokines. IL-2 was originally identified as a potent T cell growth factor and a regulator of lymphocyte responses (1, 2). Endogenous IL-2, produced by activated T cells, enhances cytokine production and cytolytic activities of T and natural killer (NK) cells (3, 4). Administration of high or intermediate doses of exogenous IL-2 has been used as an anti-tumor treatment in humans, but was limited because of systemic side effects such as vascular leakage, renal dysfunction, fever, and hypotension (5, 6).
IL-18 is a potent immunoregulatory cytokine that induces IFN-γ production (7). IL-18 exhibits many activities including activation of T cells and NK cells leading to cytokine production, proliferation, and cytolytic activity (8–10). The combination of IL-2 and IL-18 has been shown to potently induce cytotoxicity against tumor cells, IFN-γ production, and expansion of NK cells (4, 11, 12). The combination of low doses of IL-2 with IL-18 may reduce toxicity while augmenting NK cell activity (13). To date, a small number of studies have investigated the effects of the combination of IL-2 and IL-18 on T helper cell (Th)2 responses (14–16), while the effects of the combination on relative contributions to Th1 and Th2 levels has not been well characterized.
Bronchial asthma is thought to be a Th2 cytokine–mediated airway disease. Many cell types, including T cells, eosinophils, and mast cells, are recruited to the airways and are activated to secrete Th2 type cytokines and chemokines, resulting in airway inflammation and airway hyperresponsiveness (AHR) (17). The Th1 cytokines IFN-γ and IL-12 are potent negative regulators of Th2 responses. IFN-γ production is enhanced by IL-18 when combined with IL-12 (18), and allergic inflammation and AHR were attenuated (19). However, IL-18 has also been shown to increase IgE levels, Th2 cytokines, and eosinophils (20), and induce allergic inflammation such as atopic dermatitis in mice (21). To date, the consequences of combining IL-2 and IL-18 on allergic airway inflammation has not been defined. Moreover, our focus was to examine normal and not genetically deficient mice or mice treated with “depleting” antibodies, and to focus on the post-sensitization phase of the response to allergen challenge.
In this study, we demonstrate that the combination of IL-18 together with relatively small doses of IL-2 effectively suppressed allergen-induced AHR and airway inflammation. Further, this effect was shown to be dependent on the local production of IFN-γ by NK cells, mediated through an IL-12–STAT4 pathway.
MATERIALS AND METHODS
Animals
Male and female C57BL/6 mice (Jackson Laboratories, Bar Harbor, ME) were maintained on an ovalbumin (OVA)-free diet. Male and female IFN-γ−/− mice (background: C57BL/6 strain) and homozygous STAT4−/− mice (background: B6 129 strain) bred at the National Jewish Medical and Research Center were studied at ages of 8–12 wk. All experimental animals used in this study were under a protocol approved by the Institutional Animal Care and Use Committee of the National Jewish Medical and Research Center.
Antigen Sensitization and Challenge
Mice were sensitized as previously described (22). Briefly, mice received an intraperitoneal injection of 20 μg OVA (Grade V; Sigma Chemical Co., St. Louis, MO) emulsified in 2 mg aluminum hydroxide (AlumImuject; Pierce, Rockford, IL) in a total volume of 100 μl on Days 0 and 14, and then were challenged via the airways, using nebulized OVA (1% in 0.9% saline), delivered with an ultrasonic nebulizer (NE-07; Omron, Kyoto, Japan) for 20 min daily on Days 28, 29, and 30. On Day 32, AHR was assessed, and bronchoalveolar lavage (BAL) fluid, blood, and lung tissue were collected. Human recombinant IL-2 (2,000 IU/mouse; obtained from National Institutes of Health, Bethesda, MD) and mouse recombinant IL-18 (0.6 μg/mouse; Biosource, Camarillo, CA) were administered intraperitoneally in a volume of 5 ml/kg on Day 27 and 10 min before each of the OVA challenges.
Determination of Airway Resistance
Airway resistance (RL) was determined as changes in airway function after aerosolized methacholine (MCh) challenge. Mice were anesthetized with sodium pentobarbital (90 mg/kg), tracheostomized, and mechanically ventilated at a rate of 160 breaths/min with a constant tidal volume of air (0.2 ml). Lung function was assessed as previously described (22). Aerosolized MCh was administered for 10 breaths at a rate of 60 breath/min, tidal volume of 500 μl by a second ventilator (Model 683; Harvard Apparatus, South Natick, MA) in increasing concentrations (12.5, 25, 50, and 100 mg/ml). After each MCh challenge, the data were continuously collected for 1–5 min and maximum values of RL were taken to express changes in these functional parameters.
Determination of Cell Numbers and Cytokine Levels in BAL
Immediately after the assessment of AHR, lungs were lavaged via the tracheal cannula with Hanks' balanced salt solution (HBSS; 1 ml/mouse). Total leukocyte numbers were counted (Coulter Counter; Coulter Corporation, Hialeah, FL). Differential cell counts were made from cytocentrifuged preparations (Cytospin 2; Shandon Ltd., Runcorn, Cheshire, UK), stained with Leukostat (Fisher Diagnostics, Pittsburgh, PA). Cells were identified as macrophages, eosinophils, neutrophils, and lymphocytes by standard hematologic procedures and at least 200 cells counted under ×400 magnification in a blinded manner.
BAL supernatants were collected and kept frozen at −80°C until assayed. The levels of cytokine secreted into the supernatants of BAL fluid samples were determined by enzyme-linked immunosorbent assay. IL-4, IL-5, IL-12, IFN-γ (all from BD Pharmingen, San Diego, CA) and IL-13 (from R&D Systems, Minneapolis, MN) were measured following the manufacturers' directions. The limits of detection were 4 pg/ml for IL-4 and IL-5, 10 pg/ml for IL-12 and IFN-γ, and 1.5 pg/ml for IL-13.
Histopathologic Study
Lungs were fixed after inflation and immersion in 10% formalin. For detection of mucus-containing cells in formalin-fixed airway tissue, sections were stained with periodic acid-Schiff (PAS) and were quantitated as previously described (23, 24).
Lung Cell Isolation and Flow Cytometry Analysis
Lung cells were isolated as previously described using collagenase digestion (25). After lung digestion, cells were resuspended in HBSS, and mononuclear cells were purified by 35% percoll gradient centrifugation. After IL-2 and IL-18 treatment, 3.4 ± 0.6 × 106 NK cells were isolated from each lung. In untreated mice, the number of NK cells isolated were 1.5 ± 0.3 × 106 per lung.
After purification, 1 × 106 cells were incubated with allophycocyanin-conjugated anti-CD3, FITC-conjugated anti-CD4 or anti-CD8, and PE-conjugated NK1.1 antibodies (all from BD Pharmingen), then analyzed by flow cytometry (FACSCalibur; BD Biosystems, San Jose, CA). Intracytoplasmic cytokine staining for IFN-γ was performed as previously described (26). The number of IFN-γ–positive CD4+ T, CD8+ T, natural killer T (NKT) (NK1.1+, CD3+), and NK (NK1.1+, CD3−) cells per lung were derived by multiplying the percentage of stained cells by the total number of lung cells isolated after PMA and ionomycin (PI) stimulation.
Adoptive Transfer of NK Cells
Mononuclear cells were isolated from lungs of sensitized and challenged C57BL/6 mice treated with or without IL-2 and IL-18. Lungs were digested and cells were isolated as described above. Cells were then washed and isolated by HISTOPAQUE-1083 (Sigma) gradient centrifugation at 2,000 rpm for 20 min. NK cells were positively selected using magnetic beads labeled with anti-DX5 antibody (Miltenyi Biotec, Gladbach, Germany) according to the manufacturer's direction. These isolated CD3−, NK1.1+ NK cells (> 95% purity) were intravenously transferred (2 × 106 cells/mouse) to sensitized C57BL/6 mice just before the first of three OVA challenges. Forty-eight hours after the last OVA challenge, all assays were performed.
Data Analysis
Comparisons for all pairs were performed using the Student's t test, and P values for significance were set at 0.05. All data were expressed as the mean ± SEM.
RESULTS
Effects of IL-2 and IL-18 on Allergen-Induced AHR and Airway Inflammation
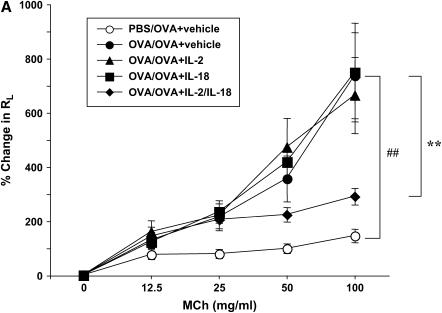
To assess the effects of the combination of IL-2 and IL-18 on airway inflammation, we monitored OVA-induced AHR and accumulation of inflammatory cells in the BAL fluid. Airway responsiveness to MCh was significantly (P < 0.01) increased after three consecutive OVA challenges in sensitized mice compared with nonsensitized mice (Figure 1A). Based on preliminary experiments, we defined the minimum effective concentrations of the combination of IL-2 (2,000 IU/mouse) and IL-18 (0.6 μg/mouse) which were effective. The combination of IL-2 and IL-18 significantly inhibited the development of AHR, whereas neither IL-2 nor IL-18 alone showed any effect. Total cell and eosinophil numbers in BAL fluid were significantly increased in the OVA-sensitized and -challenged group (Figure 1B), while administration of IL-2 and IL-18 significantly reduced the increases in total cells and eosinophils; IL-2 or IL-18 alone did not affect these responses (Figure 1B). No significant changes were observed in numbers of lymphocytes or neutrophils.
Figure 1.
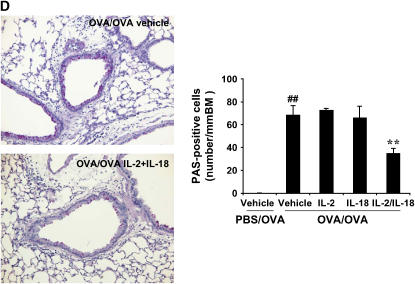
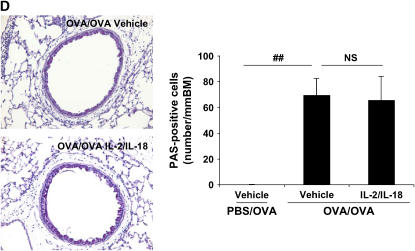
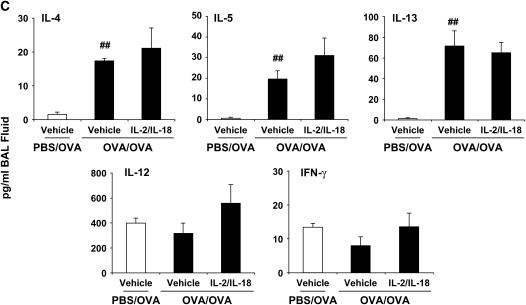
Synergistic effects of exogenous IL-2 and IL-18 on allergic AHR and airway inflammation in C57BL/6 mice. (A) Allergen-induced AHR. Sensitized mice received intraperitoneal injections of IL-2, IL-18, the combination, or vehicle beginning 1 d before the first OVA challenge to the day of the last OVA challenge. Results are expressed as the percentage of change in RL after MCh inhalation. (B) BAL cell composition. (C) Cytokine levels. (D) Goblet cell metaplasia. Goblet cell metaplasia was detected by PAS staining 48 h after the last OVA challenge (original magnification ×100). Results are expressed as the number of PAS-positive cells per millimeter of basement membrane (BM). PBS/OVA: nonsensitized and challenged; OVA/OVA: sensitized and challenged. Results represent mean ± SEM from three separate experiments (n = 12). #P < 0.05, ##P < 0.01 comparing sensitized and challenged to challenged alone. *P < 0.05, **P < 0.01 comparing vehicle-treated to combination of IL-2– and IL-18–treated sensitized and challenged mice.
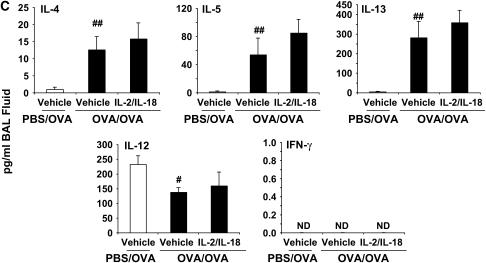
The relative levels of Th1 and Th2 cytokines have been proposed to play an important role in the development of allergic airway inflammation. After OVA challenge of sensitized mice, IL-4, IL-5, and IL-13 levels in BAL were significantly increased (Figure 1C). The combination of IL-2 and IL-18 significantly reduced the levels of IL-5 and IL-13 (the low levels of IL-4 were not altered), whereas neither IL-2 nor IL-18 alone altered the levels of these cytokines. BAL IL-12 levels were significantly decreased after OVA challenge, but in mice that received the combination of IL-2 and IL-18, both IL-12 and IFN-γ levels in BAL were significantly increased (Figure 1C).
Effects of IL-2 and IL-18 on Allergen-Induced Goblet Cell Metaplasia
Goblet cell metaplasia is a major indicator of an allergic response in the lung. To assess goblet cell metaplasia, lung tissue sections were stained with PAS. After OVA challenge, nonsensitized mice showed no PAS-positive cells (0 PAS-positive cells/mm BM), whereas sensitized and challenged mice showed a marked increase in numbers of PAS-positive cells (70.4 ± 8.7 PAS-positive cells/mm BM) (Figure 1D). However, sensitized and challenged mice treated with the combination of IL-2 and IL-18 had far fewer PAS-positive cells (34.6 ± 4.3 PAS-positive cells/mm BM). Neither IL-2 nor IL-18 alone altered the number of the cells (72.7 ± 1.3 and 65.9 ± 10.1 PAS-positive cells/mm BM, respectively) (Figure 1D).
Effects of the Combination of IL-2 and IL-18 on Allergen-Induced AHR and Airway Inflammation in IFN-γ−/− Mice
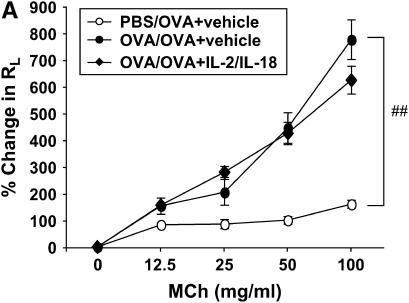
Because the inhibitory effects of the combination of IL-2 and IL-18 on AHR and allergic airway inflammation were associated with elevated levels of IFN-γ in BAL fluid, IFN-γ−/− mice were assessed to address the role of IFN-γ in mediating the suppressive effects of combination therapy. In IFN-γ−/− mice, the development of AHR and eosinophilia in BAL was similar to that in wild-type mice (Figure 2A). However, the combination of IL-2 and IL-18 did not show any suppressive effects on AHR (Figure 2A) or airway eosinophilia (Figure 2B).
Figure 2.
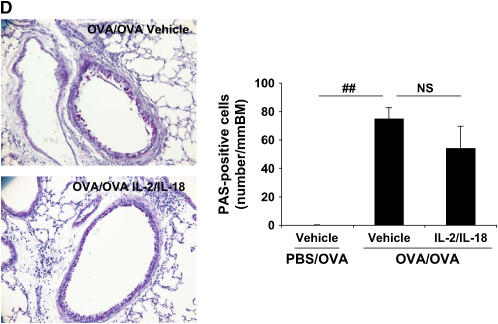
Combination of IL-2 and IL-18 does not suppress allergic AHR and airway inflammation in IFN-γ−/− mice. (A) Allergen-induced AHR. (B) BAL cell composition (C) Cytokine levels in BAL fluid. (D) Goblet cell metaplasia. PBS/OVA: nonsensitized and challenged; OVA/OVA: sensitized and challenged. Results represent mean ± SEM from two separate experiments (n = 8). #P < 0.05 and ##P < 0.01 comparing sensitized and challenged to challenged alone. NS: not significant.
The combination of IL-2 and IL-18 did not show any effects on the reduced levels of IL-12 in sensitized and challenged mice or the increased levels of IL-5 or IL-13 in the IFN-γ−/− mice (Figure 2C). In parallel, the combination of IL-2 and IL-18 was ineffective in reducing the numbers of PAS-positive cells in sensitized and challenged IFN-γ−/− mice (Figure 2D).
Effects of IL-2 and IL-18 on Allergen-Induced AHR and Airway Inflammation in STAT4−/− Mice
Based on the studies in IFN-γ−/− mice, IFN-γ appeared necessary for expression of the suppressive effects of IL-2 and IL-18 on allergic AHR and airway inflammation. In addition, the combination of IL-2 and IL-18 enhanced IL-12 levels in the BAL fluid. As the interplay between these two cytokines was likely involved in the regulation of these lung allergic responses, we investigated the role of STAT4 using deficient mice to determine if interfering with the IL-12 signaling pathway alters the effect of treatment with IL-2 and IL-18. In sensitized STAT4−/− mice, development of AHR and airway eosinophila developed normally after OVA challenge of sensitized mice (Figures 3A and 3B). However, the combination of IL-2 and IL-18 failed to show any effect on AHR (Figure 3A), airway eosinophilia (Figure 3B), cytokine levels (Figure 3C), or numbers of PAS+ cells (Figure 3D) in STAT4−/− mice, similar to IFN-γ−/− mice.
Figure 3.
Combination of IL-2 and IL-18 does not suppress allergic AHR and airway inflammation in STAT4−/− mice. (A) Allergen-induced AHR. (B) BAL cell composition. (C) Cytokine levels in BAL fluids. (D) Goblet cell metaplasia. PBS/OVA: nonsensitized and challenged; OVA/OVA: sensitized and challenged. Results represent mean ± SEM from two separate experiments (n = 8). #P < 0.05, ##P < 0.01 comparing sensitized and challenged to challenged alone, or comparing vehicle-treated to combination of IL-2– and IL-18–treated sensitized and challenged mice. NS: not significant.
Combination of IL-2 and IL-18 Increases Numbers of IFN-γ+ NK Cells
To investigate the influence of IL-2 and IL-18 on cell subpopulations, the proportion of NK, NKT, CD4+, and CD8+ cells was determined. As shown in Table 1, only NK cells were increased after IL-2 and IL-18 treatment. The numbers of NKT, CD4+, and CD8+ cells were not altered after the treatment. To determine the source of IFN-γ under these experimental conditions, we examined intracellular IFN-γ staining of isolated lung cells after PI stimulation. CD4+, CD8+, NK (NK1.1+, CD3−), and NKT (NK1.1+, CD3+) cells were identified as potential sources of IFN-γ. Administration of IL-2 and IL-18 to sensitized and challenged mice significantly increased the numbers of IFN-γ–positive lung NK cells (Figure 4), without altering the numbers of IFN-γ-positive CD4+, CD8+, or NKT cells (Figure 4).
TABLE 1.
CELL DISTRIBUTION OF LUNG MONONUCLEAR CELLS FROM MICE AFTER TREATMENT WITH IL-2 AND IL-18
| Percentage of Lung Mononuclear Cells
|
||||
|---|---|---|---|---|
| NK | NKT | CD4+ | CD8+ | |
| OVA/OVA + vehicle | 12.7 ± 0.7 | 1.3 ± 0.2 | 17.3 ± 1.1 | 11.7 ± 0.2 |
| OVA/OVA + IL-2/IL-18 | 22.5 ± 3.7* | 1.1 ± 0.1 | 14.4 ± 0.6 | 10.6 ± 0.7 |
Definition of abbreviations: NK, natural killer cells; NKT, natural killer T cells; OVA, ovalbumin.
P < 0.05, statistically significant compared with vehicle treatment. Each value represents the mean ± SEM (n = 8).
Figure 4.
IFN-γ+ cells in lungs from sensitized and challenged C57BL/6 mice treated with IL-2 and IL-18. Sensitized mice received intraperitoneal injections of IL-2 and/or IL-18 or vehicle from 1 d before the first OVA challenge to the day of the last OVA challenge. After PI stimulation, the number of intracytoplasmic IFN-γ+ CD4+, CD8+, NKT (NK1.1+, CD3+), and NK (NK1.1+, CD3−) cells per lung was calculated after flow cytometry analysis. Results represent mean ± SEM from eight mice. **P < 0.01 comparing vehicle-treated to combination of IL-2– and IL-18–treated mice.
Effect of Adoptive Transfer of NK Cell from Lungs of Mice Treated with IL-2 and IL-18 In Vivo
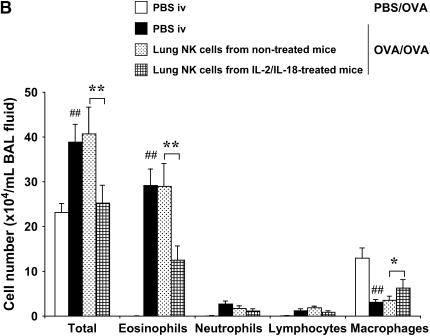
To directly demonstrate the role of IFN-γ+ lung NK cells in mediating the effects of IL-2/IL-18, we isolated and adoptively transferred lung NK cells into sensitized recipients before challenge. Adoptive transfer of lung NK cells (2.0 × 106) from sensitized and challenged mice treated with IL-2 and IL-18 significantly suppressed AHR and the increase of eosinophils in BAL fluid of recipient mice (Figure 5A); transfer of 0.2 × 106 NK cells was without effect (data not shown). In contrast, adoptive transfer of lung NK cells from mice not treated with the combination did not show any inhibitory effects on AHR or airway eosinophilia (Figure 5B). These data establish the role of NK cells in mediating the suppressive effects of the combination of IL-2 and IL-18.
Figure 5.
Effect of transfer of lung NK cells from IL-2– and IL-18–treated mice on allergic AHR (A) and airway inflammation (B). Sensitized donor mice received the combination of IL-2 and IL-18 or vehicle (0.1% BSA containing PBS) from 1 d before the first OVA challenge to the day of the last OVA challenge. Forty-eight hours after the last OVA challenge, lung mononuclear cells were isolated, and NK cells purified as described in Materials and Methods. 2 × 106 cells/mouse were transferred intravenously to sensitized C57BL/6 mice just before the first OVA challenge. Results of airway function are expressed as the percentage change in RL after MCh inhalation. Results represent mean ± SEM from two separate experiments (n = 8). PBS/OVA: nonsensitized and challenged recipients; OVA/OVA: sensitized and challenged recipients. ##P < 0.01 comparing OVA/OVA to PBS/OVA. *P < 0.05, **P < 0.01 comparing PBS-injected to NK cell-recipient mice.
DISCUSSION
In the pathogenesis of bronchial asthma, a number of factors are believed to play a role in the development of this complex allergic airway disease. T lymphocytes and the Th2 type cytokines they produce, IL-4, IL-5, and IL-13, play important roles (27, 28). Previous studies described that the combination of IL-2 and IL-18 supported a Th2 microenvironment in vivo and in vitro (9, 10, 14–16). Somewhat to the contrary, this combination has also been reported to increase release of IFN-γ from mononuclear cells (4). IL-2 is essential to the proliferation and activation of T cells and is involved in the pathogenesis of asthma (29). IL-2 has been shown to induce cytotoxic activity in T cells and NK cells (30, 31), whereas IL-18 has a variety of actions, including induction of IFN-γ.
In the present study, the administration of both IL-2 and IL-18, but not either alone, at the time of challenge of sensitized mice effectively prevented the development of AHR, airway eosinophilia, and goblet cell metaplasia. At the same time, this combination significantly altered the cytokine microenvironment, reducing levels of the Th2 cytokines IL-5 and IL-13 and increasing levels of IL-12 and IFN-γ, a major shift from a Th2 to a Th1 predominance.
Several studies have demonstrated the essential role of IL-5 and IL-13 in the development of AHR and airway eosinophilia (32–34). In addition, IL-13 is the major inducer of goblet cell metaplasia and mucus hyperproduction, triggering differentiation of mature goblet cells through induction of the MUC5AC gene (35). In contrast, Th1 cytokines such as IL-12 and IFN-γ have been shown to down-regulate allergic responses in vivo. OVA-sensitized mice, treated with systemic or local administration of IL-12 (36, 37), IFN-γ (38, 39) or IL-12, or IFN-γ gene transfer (40–42), exhibited significant suppression of allergic responses, reducing levels of Th2 cytokines, lung eosinophilia, and AHR. The combined administration of IL-12 and IL-18 was reported to inhibit the development of AHR and airway eosinophilia (19), while co-administration of IL-12 and IL-18 activated NK cells to produce IFN-γ (43, 44). The combination of IL-2 and IL-18 was shown to trigger production of IL-12 from mononuclear cells and dendritic cells (45).
In the present study, IL-12 levels in BAL fluids were increased in sensitized mice treated with IL-2 and IL-18 after challenge. This suggested that the up-regulation of IFN-γ after treatment with IL-2 and IL-18 may be induced not only via a direct stimulation but also indirectly, through the up-regulation of IL-12. IL-12 signaling through specific surface receptors is closely tied to IFN-γ production (46, 47). IL-12 ligation of IL-12 receptors triggers STAT4 phosphorylation and activation. Activated/phosphorylated STAT4 translocates to the nucleus of cells resulting in the increased transcription of IFN-γ and IFN-γ protein production. To determine the role of IFN-γ in the outcomes mediated by combination treatment, OVA-sensitized IFN-γ−/− mice were treated with IL-2 and IL-18 followed by OVA challenge. In the deficient mice, the combination failed to alter the development of AHR and airway eosinophilia, implying that IFN-γ was essential to the down-regulation of these responses triggered by IL-2/IL-18. To further define the pathways involved, we investigated whether the combination of IL-2 and IL-18 was dependent on STAT4 signaling to produce IFN-γ. STAT4−/− mice were sensitized and challenged, treated with IL-2 and IL-18, and AHR and airway inflammation was assessed. In the STAT4−/− mice, similar to the IFN-γ−/− mice, the combination did not alter development of allergen-induced AHR, airway inflammation, or goblet cell metaplasia. In addition, changes in cytokine levels after sensitization and challenges were not influenced by combination treatment; in particular, there was no increase in IFN-γ. These data strongly suggest that IL-12 signaling and STAT4-dependent IFN-γ production are critical to the activities of IL-2 and IL-18 in inhibiting AHR and airway inflammation as well as the skewing of the cytokine response from a Th2 to a Th1 predominance.
Accompanying the changes in allergen-driven responses, the combination of IL-2 and IL-18 elicited increases in numbers of IFN-γ+ NK cells in the lung. Adoptive transfer of these NK cells into sensitized mice before challenge, but not NK cells obtained from non–IL-2/IL-18–treated mice, prevented the development of allergic AHR and airway inflammation. These findings implicate directly NK cells as playing a critical role in the IL-2/IL-18–mediated suppression of allergic airway inflammation and appeared linked to their secretion of IFN-γ. To date, the role of NK cells in the regulation of airway inflammation has been somewhat controversial. Korsgren and coworkers showed that depletion of NK cells abrogated development of allergic airway inflammation (48). They described that NK cells, but not NKT cells, play a critical role in determining whether allergic eosinophilic airway disease develops, and increased NK cell activity exaggerated T cell responses to inhaled antigens. This report appears to be in conflict with our findings, but may suggest that under different conditions NK cells exhibit diverse activities on AHR and airway inflammation. After induction/activation of NK cells by the combination of IL-2 and IL-18, they down-regulated these responses in an IFN-γ–dependent manner.
In summary, administration of the combination of IL-2 and IL-18 suppressed allergic AHR and airway inflammation in vivo. The mechanism underlying these effects appears to be through an up-regulation of IL-12 production and IL-12/STAT4–dependent cell signaling, resulting in increased IFN-γ production in NK cells. Manipulation of this inducible and effective regulatory pathway may be therapeutically beneficial in the treatment of allergic airway disease.
Acknowledgments
The authors thank L. N. Cunningham and D. Nabighian (National Jewish Medical and Research Center, Denver, CO) for their assistance.
This work was supported by NIH grants HL-36577, HL-42246, and HL-61005, and by EPA grant R825702.
Originally Published in Press as DOI: 10.1165/rcmb.2006-0231OC on October 12, 2006
Conflict of Interest Statement: None of the authors has a financial relationship with a commercial entity that has an interest in the subject of this manuscript.
References
- 1.Morgan DA, Ruscetti FW, Gallo R. Selective in vitro growth of T lymphocytes from normal human bone marrows. Science 1976;193:1007–1008. [DOI] [PubMed] [Google Scholar]
- 2.Nelson BH. IL-2, regulatory T cells, and tolerance. J Immunol 2004;172:3983–3988. [DOI] [PubMed] [Google Scholar]
- 3.Gaffen SL, Liu KD. Overview of interleukin-2 function, production and clinical applications. Cytokine 2004;28:109–123. [DOI] [PubMed] [Google Scholar]
- 4.Son YI, Dallal RM, Mailliard RB, Egawa S, Jonak ZL, Lotze MT. Interleukin-18 (IL-18) synergizes with IL-2 to enhance cytotoxicity, interferon-gamma production, and expansion of natural killer cells. Cancer Res 2001;61:884–888. [PubMed] [Google Scholar]
- 5.Lotze MT, Chang AE, Seipp CA, Simpson C, Vetto JT, Rosenberg SA. High-dose recombinant interleukin 2 in the treatment of patients with disseminated cancer. Responses, treatment-related morbidity, and histologic findings. JAMA 1986;256:3117–3124. [PubMed] [Google Scholar]
- 6.Lindsey KR, Rosenberg SA, Sherry RM. Impact of the number of treatment courses on the clinical response of patients who receive high-dose bolus interleukin-2. J Clin Oncol 2000;18:1954–1959. [DOI] [PubMed] [Google Scholar]
- 7.Dinarello CA, Novick D, Puren AJ, Fantuzzi G, Shapiro L, Muhl H, Yoon DY, Reznikov LL, Kim SH, Rubinstein M. Overview of interleukin-18: more than an interferon-gamma inducing factor. J Leukoc Biol 1998;63:658–664. [PubMed] [Google Scholar]
- 8.Dinarello CA. Interleukin-18. Methods 1999;19:121–132. [DOI] [PubMed] [Google Scholar]
- 9.Nakanishi K. Innate and acquired activation pathways in T cells. Nat Immunol 2001;2:140–142. [DOI] [PubMed] [Google Scholar]
- 10.Nakanishi K, Yoshimoto T, Tsutsui H, Okamura H. Interleukin-18 regulates both Th1 and Th2 responses. Annu Rev Immunol 2001;19:423–474. [DOI] [PubMed] [Google Scholar]
- 11.Wigginton JM, Lee JK, Wiltrout TA, Alvord WG, Hixon JA, Subleski J, Back TC, Wiltrout RH. Synergistic engagement of an ineffective endogenous anti-tumor immune response and induction of IFN-gamma and Fas-ligand-dependent tumor eradication by combined administration of IL-18 and IL-2. J Immunol 2002;169:4467–4474. [DOI] [PubMed] [Google Scholar]
- 12.Acres B, Gantzer M, Remy C, Futin N, Accart N, Chaloin O, Hoebeke J, Balloul JM, Paul S. Fusokine interleukin-2/interleukin-18, a novel potent innate and adaptive immune stimulator with decreased toxicity. Cancer Res 2005;65:9536–9546. [DOI] [PubMed] [Google Scholar]
- 13.Redlinger RE Jr, Mailliard RB, Lotze MT, Barksdale EM Jr. Synergistic interleukin-18 and low-dose interleukin-2 promote regression of established murine neuroblastoma in vivo. J Pediatr Surg 2003;38:301–307. (discussion 301–307). [DOI] [PubMed] [Google Scholar]
- 14.El-Mezayen RE, Matsumoto T. In vitro responsiveness to IL-18 in combination with IL-12 or IL-2 by PBMC from patients with bronchial asthma and atopic dermatitis. Clin Immunol 2004;111:61–68. [DOI] [PubMed] [Google Scholar]
- 15.Hoshino T, Yagita H, Ortaldo JR, Wiltrout RH, Young HA. In vivo administration of IL-18 can induce IgE production through Th2 cytokine induction and up-regulation of CD40 ligand (CD154) expression on CD4+ T cells. Eur J Immunol 2000;30:1998–2006. [DOI] [PubMed] [Google Scholar]
- 16.Hoshino T, Wiltrout RH, Young HA. IL-18 is a potent coinducer of IL-13 in NK and T cells: a new potential role for IL-18 in modulating the immune response. J Immunol 1999;162:5070–5077. [PubMed] [Google Scholar]
- 17.Busse WW, Lemanske RF Jr. Asthma. N Engl J Med 2001;344:350–362. [DOI] [PubMed] [Google Scholar]
- 18.Ahn HJ, Maruo S, Tomura M, Mu J, Hamaoka T, Nakanishi K, Clark S, Kurimoto M, Okamura H, Fujiwara H. A mechanism underlying synergy between IL-12 and IFN-gamma-inducing factor in enhanced production of IFN-gamma. J Immunol 1997;159:2125–2131. [PubMed] [Google Scholar]
- 19.Hofstra CL, Van Ark I, Hofman G, Kool M, Nijkamp FP, Van Oosterhout AJ. Prevention of Th2-like cell responses by coadministration of IL-12 and IL-18 is associated with inhibition of antigen-induced airway hyperresponsiveness, eosinophilia, and serum IgE levels. J Immunol 1998;161:5054–5060. [PubMed] [Google Scholar]
- 20.Kumano K, Nakao A, Nakajima H, Hayashi F, Kurimoto M, Okamura H, Saito Y, Iwamoto I. Interleukin-18 enhances antigen-induced eosinophil recruitment into the mouse airways. Am J Respir Crit Care Med 1999;160:873–878. [DOI] [PubMed] [Google Scholar]
- 21.Tsutsui H, Yoshimoto T, Hayashi N, Mizutani H, Nakanishi K. Induction of allergic inflammation by interleukin-18 in experimental animal models. Immunol Rev 2004;202:115–138. [DOI] [PubMed] [Google Scholar]
- 22.Takeda K, Hamelmann E, Joetham A, Shultz LD, Larsen GL, Irvin CG, Gelfand EW. Development of eosinophilic airway inflammation and airway hyperresponsiveness in mast cell-deficient mice. J Exp Med 1997;186:449–454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Matsubara S, Li G, Takeda K, Loader JE, Pine P, Masuda ES, Miyahara N, Miyahara S, Lucas JJ, Dakhama A, et al. Inhibition of spleen tyrosine kinase prevents mast cell activation and airway hyperresponsiveness. Am J Respir Crit Care Med 2006;173:56–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Matsubara S, Koya T, Takeda K, Joetham A, Miyahara N, Pine P, Masuda ES, Swasey CH, Gelfand EW. Syk activation in dendritic cells is essential for airway hyperresponsiveness and inflammation. Am J Respir Cell Mol Biol 2006;34:426–433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Oshiba A, Hamelmann E, Takeda K, Bradley KL, Loader JE, Larsen GL, Gelfand EW. Passive transfer of immediate hypersensitivity and airway hyperresponsiveness by allergen-specific immunoglobulin (Ig) E and IgG1 in mice. J Clin Invest 1996;97:1398–1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Miyahara N, Takeda K, Kodama T, Joetham A, Taube C, Park JW, Miyahara S, Balhorn A, Dakhama A, Gelfand EW. Contribution of antigen-primed CD8+ T cells to the development of airway hyperresponsiveness and inflammation is associated with IL-13. J Immunol 2004;172:2549–2558. [DOI] [PubMed] [Google Scholar]
- 27.Wills-Karp M. Immunologic basis of antigen-induced airway hyperresponsiveness. Annu Rev Immunol 1999;17:255–281. [DOI] [PubMed] [Google Scholar]
- 28.Zhang DH, Yang L, Cohn L, Parkyn L, Homer R, Ray P, Ray A. Inhibition of allergic inflammation in a murine model of asthma by expression of a dominant-negative mutant of GATA-3. Immunity 1999;11:473–482. [DOI] [PubMed] [Google Scholar]
- 29.Virchow JC Jr, Kroegel C, Walker C, Matthys H. Inflammatory determinants of asthma severity: mediator and cellular changes in bronchoalveolar lavage fluid of patients with severe asthma. J Allergy Clin Immunol 1996;98:S27–S33. (discussion S33–40). [PubMed] [Google Scholar]
- 30.Cortes JE, Kantarjian HM, O'Brien S, Giles F, Keating MJ, Freireich EJ, Estey EH. A pilot study of interleukin-2 for adult patients with acute myelogenous leukemia in first complete remission. Cancer 1999;85:1506–1513. [PubMed] [Google Scholar]
- 31.Farace F, Angevin E, Dietrich PY, Leboullaire C, Vanderplancke J, Escudier B, Triebel F. Low-dose IL-2 treatment: activation of discrete T- and NK-cell sub-populations in vivo. Int J Cancer 1995;62:523–528. [DOI] [PubMed] [Google Scholar]
- 32.Wills-Karp M, Luyimbazi J, Xu X, Schofield B, Neben TY, Karp CL, Donaldson DD. Interleukin-13: central mediator of allergic asthma. Science 1998;282:2258–2261. [DOI] [PubMed] [Google Scholar]
- 33.Webb DC, Mahalingam S, Cai Y, Matthaei KI, Donaldson DD, Foster PS. Antigen-specific production of interleukin (IL)-13 and IL-5 cooperate to mediate IL-4Ralpha-independent airway hyperreactivity. Eur J Immunol 2003;33:3377–3385. [DOI] [PubMed] [Google Scholar]
- 34.Kuperman DA, Huang X, Koth LL, Chang GH, Dolganov GM, Zhu Z, Elias JA, Sheppard D, Erle DJ. Direct effects of interleukin-13 on epithelial cells cause airway hyperreactivity and mucus overproduction in asthma. Nat Med 2002;8:885–889. [DOI] [PubMed] [Google Scholar]
- 35.Zuhdi Alimam M, Piazza FM, Selby DM, Letwin N, Huang L, Rose MC. Muc-5/5ac mucin messenger RNA and protein expression is a marker of goblet cell metaplasia in murine airways. Am J Respir Cell Mol Biol 2000;22:253–260. [DOI] [PubMed] [Google Scholar]
- 36.Kodama T, Kuribayashi K, Nakamura H, Fujita M, Fujita T, Takeda K, Dakhama A, Gelfand EW, Matsuyama T, Kitada O. Role of interleukin-12 in the regulation of CD4+ T cell apoptosis in a mouse model of asthma. Clin Exp Immunol 2003;131:199–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schwarze J, Hamelmann E, Cieslewicz G, Tomkinson A, Joetham A, Bradley K, Gelfand EW. Local treatment with IL-12 is an effective inhibitor of airway hyperresponsiveness and lung eosinophilia after airway challenge in sensitized mice. J Allergy Clin Immunol 1998;102:86–93. [DOI] [PubMed] [Google Scholar]
- 38.Lack G, Gelfand EW. The role of nebulized IFN-gamma in the modulation of allergic responses. Adv Exp Med Biol 1996;409:17–23. [DOI] [PubMed] [Google Scholar]
- 39.Lack G, Bradley KL, Hamelmann E, Renz H, Loader J, Leung DY, Larsen G, Gelfand EW. Nebulized IFN-gamma inhibits the development of secondary allergic responses in mice. J Immunol 1996;157:1432–1439. [PubMed] [Google Scholar]
- 40.Dow SW, Schwarze J, Heath TD, Potter TA, Gelfand EW. Systemic and local interferon gamma gene delivery to the lungs for treatment of allergen-induced airway hyperresponsiveness in mice. Hum Gene Ther 1999;10:1905–1914. [DOI] [PubMed] [Google Scholar]
- 41.Behera AK, Kumar M, Lockey RF, Mohapatra SS. Adenovirus-mediated interferon gamma gene therapy for allergic asthma: involvement of interleukin 12 and STAT4 signaling. Hum Gene Ther 2002;13:1697–1709. [DOI] [PubMed] [Google Scholar]
- 42.Hogan SP, Foster PS, Tan X, Ramsay AJ. Mucosal IL-12 gene delivery inhibits allergic airways disease and restores local antiviral immunity. Eur J Immunol 1998;28:413–423. [DOI] [PubMed] [Google Scholar]
- 43.Zhang T, Kawakami K, Qureshi MH, Okamura H, Kurimoto M, Saito A. Interleukin-12 (IL-12) and IL-18 synergistically induce the fungicidal activity of murine peritoneal exudate cells against Cryptococcus neoformans through production of gamma interferon by natural killer cells. Infect Immun 1997;65:3594–3599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lauwerys BR, Renauld JC, Houssiau FA. Synergistic proliferation and activation of natural killer cells by interleukin 12 and interleukin 18. Cytokine 1999;11:822–830. [DOI] [PubMed] [Google Scholar]
- 45.Mailliard RB, Alber SM, Shen H, Watkins SC, Kirkwood JM, Herberman RB, Kalinski P. IL-18-induced CD83+CCR7+ NK helper cells. J Exp Med 2005;202:941–953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Verhagen CE, de Boer T, Smits HH, Verreck FA, Wierenga EA, Kurimoto M, Lammas DA, Kumararatne DS, Sanal O, Kroon FP, et al. Residual type 1 immunity in patients genetically deficient for interleukin 12 receptor beta1 (IL-12Rbeta1): evidence for an IL-12Rbeta1-independent pathway of IL-12 responsiveness in human T cells. J Exp Med 2000;192:517–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lammas DA, Casanova JL, Kumararatne DS. Clinical consequences of defects in the IL-12-dependent interferon-gamma (IFN-gamma) pathway. Clin Exp Immunol 2000;121:417–425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Korsgren M, Persson CG, Sundler F, Bjerke T, Hansson T, Chambers BJ, Hong S, Van Kaer L, Ljunggren HG, Korsgren O. Natural killer cells determine development of allergen-induced eosinophilic airway inflammation in mice. J Exp Med 1999;189:553–562. [DOI] [PMC free article] [PubMed] [Google Scholar]