Abstract
Placental hypoxia is causally implicated in fetal growth restriction and preeclampsia, with both occurring more frequently at high altitude (>2700 m; HA). The nuclear transcription factor hypoxia-inducible factor (HIF) may facilitate placental oxygen transport at HA by increasing erythropoiesis and placental angiogenesis. We therefore investigated HIF expression and its regulatory mechanisms in placentas from normal pregnancies at high (3100 m), moderate (1600 m), and sea level (75 m) altitudes. Moderate-altitude and sea level placentas did not differ, but HIF-1α and the von Hippel-Lindau tumor suppressor protein were overexpressed in HA placentas. The ability of von Hippel-Lindau tumor suppressor protein to form the E3 ubiquitin protein ligase complex, required for HIF-1α degradation, was unaltered in HA placentas. mRNA for factor-inhibiting HIF, a negative modulator of HIF-1α transactivation, was increased, but protein levels were diminished. Elevated HIF-1α likely contributed to the significant increase we report in HIF-1α downstream target proteins, transforming growth factor β3 in the placenta, and vascular endothelial growth factor and erythropoietin in the maternal circulation. These circulating markers and lowered birth to placental weight ratios correlated with increased HIF-1α, thereby linking molecular and systemic physiological data. The HA response to chronic hypoxia resembles preeclampsia in several aspects, illustrating the utility of the HA model in understanding placental pathologies.
The influence of oxygen on fetoplacental development and the role of hypoxia in causing or exacerbating pathological conditions of pregnancy is a major focus of perinatal research. Despite the utility of animal and cell culture models using hypoxic stimuli, there is no in vivo human model with which to compare the results of in vitro and animal experimentation and no means to provide the crucial link to healthy human pregnancy. To date, we do not know whether artificial hypoxic stimuli mimic the pathological conditions attributed to hypoxic stress in vivo, nor can we distinguish the effects of hypoxia from under- or overlying pathologies.
Residence at high altitude (>2700 m) is associated with reduced birth weight and an increased incidence of pregnancy complications, in particular preeclampsia.1 The birth weight effect is independent of socioeconomic status and other risk factors.2,3 High-altitude fetuses do not attain their full genetic growth potential4 and thus represent a natural model of fetal growth restriction. Placental hypoxia is often invoked as an underlying cause or contributor to the development of preeclampsia and/or fetal growth restriction.5,6 Thus the natural experiment of high-altitude residence is a unique opportunity to examine the pathways by which hypoxia, in the absence of pathology, influences placental development and function.
There is increased placental angiogenesis, a reduced distance from the intervillous space to the fetal capillaries,7 and increased maternal and fetal erythropoiesis at high altitude that facilitate oxygen transport. Erythropoiesis and angiogenesis are regulated via the highly conserved hypoxia-inducible factor (HIF) family of transcription factors, implicating HIF as an element of critical importance in placental adaptation to reduced oxygen tension. The HIF family consists of heterodimers composed of one of the three α-subunits (HIF-1α, HIF-2α, or HIF-3α) and a β-subunit known as the aryl hydrocarbon nuclear translocator (ARNT). To become functional, the α-subunit must bind the β-subunit, permitting the dimer to bind to hypoxia response elements in the promoter regions of hypoxia-sensitive genes. HIF regulation is complex, involving a host of factors that can stabilize or destabilize the α-subunit and factors that can modify transcriptional activity. Degradation of HIF-1α protein is inhibited in hypoxia, and it accumulates in the nucleus where, on binding to ARNT, it activates genes with hypoxia response elements. Under normoxic conditions the α-subunit is rapidly ubiquitinated and degraded via the proteosomal pathway,8 a process that requires interaction with the von Hippel-Lindau tumor suppressor protein (VHL).9 VHL in turn functions as the substrate recognition component of an E3 ubiquitin protein ligase complex comprising elongin B, elongin C, and Cullin-2 (the VHLcbc complex).10 Neural precursor cell-expressed developmentally down-regulated 8 (NEDD8) is a ubiquitin-like molecule that binds to Cullin-2 (CUL2) in the VHLcbc complex, and this association is required for VHL-mediated HIF-1α degradation.11,12 Although there is evidence for hypoxic induction of HIF-1α mRNA levels,13 the predominant pathways for HIF-1α regulation occur posttranslationally.14 During normoxia, factor-inhibiting HIF (FIH) promotes hydroxylation of an asparagine residue in the C-terminal transactivation domain of HIF-1α, which in turn inhibits the recruitment of transcriptional coactivator proteins such as CREB binding protein/p300,15,16 thereby inhibiting HIF-1α transactivation activity. Thus, under normoxic conditions HIF-1α is rapidly degraded and/or inactivated.
A number of in vitro and in vivo studies have highlighted the importance of HIF and its activity and degradation in placental development and function.17,18,19,20 However, these studies have focused on more acute low-oxygen insults, largely in vitro, using assumptions about oxygen exposures during established developmental windows or in pathological pregnancy. We have shown that both high altitude and preeclampsia are associated with placental hypoxia and that HIF-regulated proteins are dysregulated in preeclampsia.6,21 Two modes of HIF regulation are independently controlled by oxygen availability, HIF-1α stability, and coactivator recruitment.14,16,22 We therefore focused our studies on HIF-1α expression, the VHLcbc complex, and FIH participation in posttranslational regulation. We further tested the links between placental expression of HIF-1α and clinical parameters such as circulating markers of hypoxia and pregnancy outcome, relationships that have been tested in specific cancers23,24 but not in normal human pregnancy.
Materials and Methods
Participants
Informed consent was obtained in accordance with the Institutional Review Boards/Ethics Committees of the participating institutions. Participants included 11 women at sea level (SL; Toronto, ON, Canada), 13 at 1600 m [moderate altitude (MA); Denver, CO], and 15 at 3100 m [high altitude (HA); Leadville, CO]. At MA and HA, we prospectively evaluated circulating growth factors in women recruited between weeks 9 and 15 of pregnancy. The SL group comprised volunteers who served as normal healthy controls in a prior study of preeclampsia in Toronto, ON, Canada.25 Women were excluded from study if they had any chronic health conditions predisposing to preeclampsia or if they used tobacco, alcohol, or prescription or nonprescription (illicit) drugs other than prenatal vitamins. Women were excluded if they had an abnormal oral glucose tolerance test or developed pregnancy complications. Participants residing at 1600 and 3100 m were included only if they had conceived, gestated, and planned on remaining in the community until after delivery. Women residing at 3100 m were excluded if their work or other obligations required them to travel routinely to lower altitudes.
All participants were enrolled in routine prenatal care and had healthy, normal pregnancies resulting in singleton infants whose birth weights were appropriate for gestational age. Participants did not differ in clinical attributes (Table 1). Near-term arterial blood pressure was normal in all women and did not differ between the three sites. Gestational age was similar in all groups, but the infants born at HA were of lower birth weight than infants born at sea level or moderate altitude (Table 1).
Table 1.
Maternal and Neonatal Characteristics
| Participant data | 75 m | 1600 m | 3100 m |
|---|---|---|---|
| n | 11 | 13 | 15 |
| Maternal age (years) | 30.2 ± 1.2 | 28.5 ± 0.9 | 27.2 ± 1.6 |
| Primiparous (n) | 6/11 | 13/13 | 15/15 |
| Near-term systolic blood pressure (mm Hg) | 112 ± 7 | 113 ± 8 | 112 ± 12 |
| Diastolic blood pressure (mm Hg) | 68 ± 6 | 73 ± 8 | 74 ± 8 |
| Cesarean deliveries (n) | 4/11 | 2/13 | 3/15 |
| Gestational age (weeks) | 39.1 ± 1.1 | 39.2 ± 0.3 | 39.8 ± 1.7 |
| Birth weight (g) | 3395 ± 300 | 3332 ± 346 | 3066 ± 260*† |
| Infant sex (male/female) | 6/5 | 11/4 | 9/6 |
P < 0.05 SL versus high altitude.
P < 0.05 moderate versus high altitude.
Blood and Tissue Collection
Blood was drawn from the mothers’ antecubital vein at 2- to 4-week intervals throughout pregnancy at MA and HA; serum was separated by centrifugation and stored at −70°C for later analysis. Gestational age at the time the blood sample was obtained was back-calculated using the clinically assessed gestational age of the neonate. Birth weight, gestational age, and laboratory values or clinical observations relevant to the health of the mother were abstracted from their clinical records.
Placentas were collected immediately after delivery and placed in ice-cold phosphate-buffered saline. Villous tissue from five separate regions of the villous core was randomly excised and snap-frozen in liquid nitrogen. A minimum of five random blocks of tissue were taken through the entire thickness of the placenta for formalin fixation and paraffin embedding.
Circulating Markers of Hypoxia
Serial measurements of total serum vascular endothelial growth factor (VEGF) were obtained by competitive radioimmunoassay as previously described.26 Inter- and intra-assay coefficient of variation was <10%. Near-term maternal circulating erythropoietin (EPO) was measured by enzyme-linked immunosorbent assay (R&D Systems, Minneapolis, MN) following the manufacturer’s recommendations. Inter- and intra-assay coefficient of variation was <3%.
RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction
Total RNA extracted from placental tissues was treated with DNase to remove genomic DNA contamination. One microgram of total RNA was reverse transcribed in a total volume of 50 μl using random hexamers (Applied Biosystems, Foster City, CA). The resulting templates (50 ng of cDNA for our target genes and 5 ng for 18S) were quantified by real-time polymerase chain reaction (PCR; ABI Prism 7700). TaqMan probes and primers for human HIF-1α, ARNT, HIF-2α, VHL, FIH, transforming growth factor (TGF)-β3, and ribosomal 18S were purchased from Assays-on-Demand for human genes (Applied Biosystems). For each probe, a dilution series determined the efficiency of amplification of each primer/probe set, and the relative quantification method was used.27 For the relative quantitation, PCR signals were compared between groups after normalization using 18S as an internal reference. Briefly, relative expression was calculated as 2−(Ct gene of interest − Ct 18S). Fold change was calculated according to Livak et al.27
Western Blot Analysis
Western blot analyses were performed as previously described.28 Primary antibodies were mouse monoclonal anti-human HIF-1α (mgc3, 1:250; ABR-Affinity BioReagents Incorporation, Golden, CO), mouse monoclonal anti-human VHL (clone Ig33, 1:250; Oncogene, Cambridge, MA), rabbit polyclonal anti-human CUL2 (1:250; Neomarkers, Fremont, CA), rabbit polyclonal ARNT (1:2000; Novus Biologicals, Littleton, CO), rabbit polyclonal HIF-2α (1:1000; Novus Biologicals), rabbit polyclonal FIH (1:250; Abcam, Cambridge, MA), and goat polyclonal anti-human TGFβ3 (1:500; R&D Systems Inc.). Horseradish peroxidase-conjugated secondary antibodies (1:10,000) were rabbit anti-mouse for HIF-1α and VHL, donkey anti-rabbit for CUL2 and ARNT, and donkey anti-goat for TGFβ3 (Santa Cruz Biotechnology, Santa Cruz, CA).
Immunoprecipitation
Immunoprecipitations were performed as previously described.28 Five hundred micrograms of total protein lysates was pre-cleared, antisera (1 μg) were added, and samples were incubated overnight. Immunoprecipitates were then collected, subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis, and analyzed by immunoblotting using mouse monoclonal anti-human VHL (1:250), rabbit polyclonal antibody NEDD8 (1:500; Alexis Biochemicals, San Diego, CA), or mouse monoclonal anti-human ubiquitin (1:500; Covance Research Products, Berkeley, CA). A 1:10,000 dilution of anti-mouse-Ig-horseradish peroxidase was used as secondary antibody.
Immunohistochemistry
Immunohistochemical analyses were performed as previously described.17 Placental tissues were fixed in buffered formalin and embedded in paraffin. Before immunohistochemical analysis, every 10th section was stained with hematoxylin and eosin to assess the quality of the tissue and select the most representative sections. Mouse monoclonal antibodies against HIF1α and VHL and rabbit polyclonal antibodies against CUL2 were all used at 1:50 dilution. Secondary antibodies (1:300) were either biotinylated goat anti-mouse or goat anti-rabbit IgG. Control experiments were performed by replacing the primary antibody with normal goat serum.
Statistical Analysis
Data are reported as mean ± SEM. The maternal and infant characteristics in Table 1 were compared using χ2 or analysis of variance, as appropriate. For the time course of changes in circulating VEGF, concentrations were compared between low- and high-altitude women and across time during pregnancy using a linear mixed-effects modeling approach as previously described.29 Relationships between variables were assessed using linear regression. For comparison of data between multiple groups, we used Kruskal-Wallis one-way analysis of variance with post hoc Dunn test. Statistical tests were performed using Prism (GraphPad Software, San Diego, CA) and StatView statistical software (StatView Software, Cary, NC). Data are presented as mean ± SEM and reported as statistically significant when P < 0.05.
Results
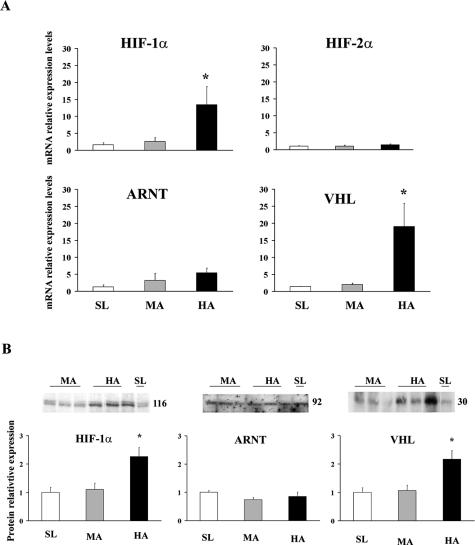
HIF-1α and VHL mRNA expression levels were significantly increased in HA placentas relative to MA and SL placentas (Figure 1A; HIF-1α, 13.4- ± 5.3-fold HA versus MA and SL, P < 0.05; VHL, 16.1- ± 6.0-fold HA versus MA and SL, P < 0.05), whereas mRNA expression for HIF-2α and ARNT were similar across all three altitudes (Figure 1A). Western blots for HIF-1α and VHL were consistent with the findings from quantitative real-time PCR (Figure 1B, representative blots). Densitometric analysis showed that the increased message was associated with increased protein because both HIF-1α (2.26- ± 0.32-fold HA versus SL and MA, P < 0.005) and VHL (2.17- ± 0.30-fold HA versus SL and MA, P < 0.05) were elevated relative to SL and MA (Figure 1B). Consistent with no change in message, ARNT protein levels were similar across all altitudes (Figure 1B). In line with the mRNA findings, HIF-2α protein expression was not significantly altered between altitudes (data not shown) and, therefore, was not further investigated.
Figure 1.
mRNA and protein expression of HIF-related molecules in placentas from different altitudes. A: Real-time PCR analysis for HIF-1α, HIF-2 α, ARNT, and VHL. B: Densitometric analysis of immunoblots for HIF-1α, ARNT, and VHL. Representative Western blots are inserted. *P < 0.05.
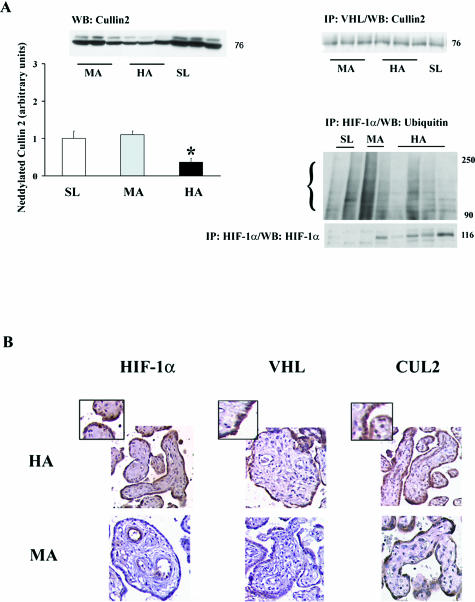
We next examined components of the E3 ligase complex and their interaction. CUL2 protein levels were similar at all three altitudes (Figure 2A, top left blot), whereas neddylated CUL2, identified as an 84-kd band, was reduced in HA placentas relative to SL and MA placentas (0.35- ± 0.1-fold HA versus SL and MA, P < 0.005; Figure 2A, top left blot and panel). The reduction in neddylated CUL2 did not seem to translate into decreased binding of the E3 ligase complex with VHL, because immunoprecipitation of VHL followed by Western blot for CUL2 showed no differences between the three altitudes (Figure 2A, top right blot).
Figure 2.
VHLCBC complex interactions and immunolocalization of HIF-related molecules in placenta at different altitudes. A: Left panel, top, Western blot for Cullin-2 protein, detected at 76 kd, and neddylated Cullin-2 protein. Bottom: Densitometric analysis of neddylated Cullin-2, identified at 84 kd in Western blot (top). Right panel, top: Interaction between VHL and Cullin2 identified by immunoprecipitation with VHL followed by immunoblotting with Cullin-2. Bottom: Immunoprecipitation of HIF-1α followed by Western blotting with ubiquitin or HIF-1α. B: Immunostaining for HIF-1α, VHL, and Cullin-2. Brown color depicts positive immunoreactivity. The insets show greater magnification of the areas of interest. *P < 0.05.
To investigate whether HIF-1α is ubiquitinated to the same degree between altitudes, HIF-1α was precipitated from placental lysate with anti-HIF1α antibody, and immunoprecipitates were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis followed by immunoblotting with an anti-ubiquitin antibody. This experiment was repeated multiple times using all of the samples available, and although variable, the results showed that ubiquitination was either decreased or unchanged at high altitude. Support for a decrease in ubiquitination is provided by Figure 2A, right bottom blot: Immunoprecipitated HIF-1α, probed by Western blot for HIF-1α was markedly increased at high altitude, which is consistent with both decreased ubiquitination and the protein results for HIF-1α.
We also examined the spatial localization of HIF-1α, VHL, and CUL2 (Figure 2B). In HA placentas, HIF-1α immunostaining localized to the syncytiotrophoblast, to the vascular endothelium, and to a lesser extent to the mesenchymal tissue. Staining was virtually absent from syncytium in MA placentas, although low positive immunoreactivity for HIF-1α was present in vascular endothelium. VHL immunostaining localized to the syncytiotrophoblast of HA tissue, whereas no immunoreactivity was present in the MA tissue. Strong positive immunoreactivity for CUL2 also localized to the syncytiotrophoblast but did not differ between MA and HA.
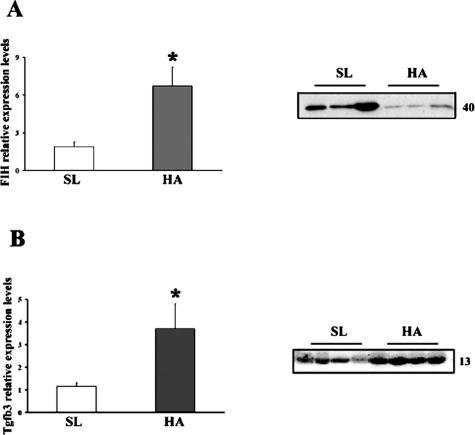
Having established that HIF-1α and VHL are elevated in HA placentas, that the E3 ligase complex binds VHL in a manner similar to controls, and that ubiquitination was decreased or unaltered, we continued the investigation examining a mechanism that contributes to posttranslational modification of HIF-1α activity. FIH is an asparaginyl hydroxylase enzyme known to impede HIF-1α gene transcription via inhibition of recruitment of cofactors for transcription. FIH mRNA was increased in HA versus SL placentas (Figure 3A, left panel), but protein was markedly decreased (Figure 3A, right panel). MA placentas were similar to SL placentas (data not shown). Increased HIF-1α protein production in conjunction with decreased FIH would be expected to lead to greater levels of HIF-1α-dependent gene transactivation relative to sea level. We therefore tested placental expression of TGFβ3, which is regulated via HIF-1α and plays a key role in early placental development.17,30 To extend these placental findings to maternal physiology, we also examined two other well-known HIF-1α-mediated gene products, EPO and VEGF, in the maternal circulation. Placental mRNA and protein for TGFβ3 (Figure 3B, left and right panel, respectively) more than doubled at high altitude relative to sea level, whereas moderate altitude did not differ from sea level (data not shown). EPO (26.0 ± 1.2 mIU at HA, 21.8 ± 1.0 mIU at MA, P < 0.05) and total VEGF (8.6 ± 3.1 at HA versus 5.6 ± 3.1 ng/ml at MA, P < 0.01) concentrations were significantly increased at high altitude in the last serum sample obtained before delivery.
Figure 3.
mRNA and protein expression of FIH (A) and TGFβ3 (B) in placentas from different altitudes. Expression levels were determined by real-time PCR (left panels) and Western blot (right panels) analysis. *P < 0.05.
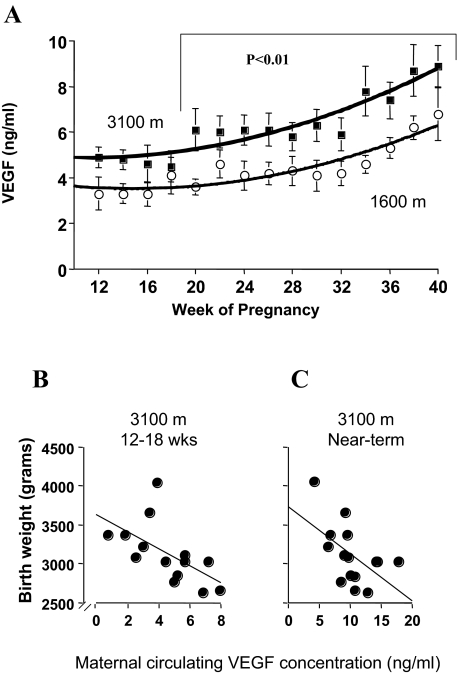
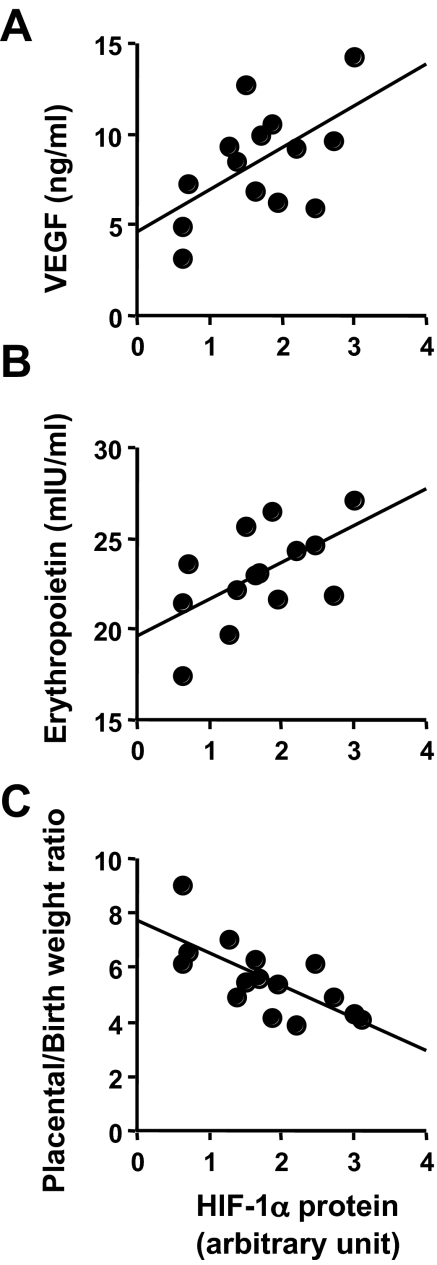
We have previously shown increased mRNA for total VEGF in these same placental samples.21 However, because the placenta at term is a snapshot in time and because human placental samples cannot be taken before delivery, we measured maternal circulating total VEGF throughout pregnancy to see at what time point chronic placental hypoxia might translate into increased serum concentrations of total VEGF in HA mothers. Total VEGF was significantly increased at high altitude from week 20 of gestation onward (Figure 4A). That the placenta was the source of this increased VEGF is supported by samples collected within 24 hours of delivery; these values (1.4 ± 0.5 ng/ml at MA, 1.3 ± 0.6 ng/ml at HA, P = NS) did not differ from those obtained more than 3 months postpartum in the same women (1.0 ± 0.4 ng/ml at MA, 1.0 ± 0.3 ng/ml at HA, P = NS). There was thus no effect of altitude on circulating VEGF in the nonpregnant condition. We also found that circulating VEGF was negatively associated with birth weight both at the time of initial blood sampling in the late first/early second trimester and in the last value obtained before delivery at high altitude (Figure 4, B and C). There was no relationship between VEGF and birth weight at moderate altitude (data not shown). Placental HIF-1α protein, across both altitudes, was positively associated with maternal near-term circulating VEGF and erythropoietin concentrations (Figure 5, A and B) and negatively associated with the birth-to-placental weight ratio (Figure 5C). This last correlation indicates that infants whose body size is small relative to their placental size have greater placental HIF-1α expression and are perhaps exposed to greater hypoxia or are more responsive to a given oxygen tension.
Figure 4.
Circulating VEGF levels and correlations with birth weights at different altitudes. A: Total VEGF concentrations during pregnancy at different altitudes. MA (open circles, hatched line) and HA (closed circles, solid line). The symbols are the mean and SD of the VEGF concentration measured at a particular time point, whereas the lines represent the best-fit regression equations of the time course of change in VEGF (MA, y = 0.004x2 − 0.130x + 4.493, r2 = 0.83; HA, y = 0.005x2 − 0.097x + 5.369, r2 = 0.86). VEGF concentrations were significantly elevated from week 20 onward at high versus moderate altitude. B: Maternal blood VEGF concentrations measured at 14.7 ± 1.3 weeks (range, 12 to 18 weeks) were inversely correlated with birth weight at high altitude (y = 3629.62 − 109.41x, r2 = 0.35, P < 0.05). C: VEGF concentration in the last maternal blood sample obtained before delivery (range, 34 to 39 weeks; mean, 35.7 ± 1.2) was inversely correlated with birth weight at high altitude (y = 3673.28 − 65.15x, r2 = 0.28, P < 0.05).
Figure 5.
Correlations of placental HIF-1α protein with circulating hypoxia markers and birth/placental weights. A and B: Placental HIF-1α protein in HA placenta correlates with both circulating markers of hypoxia, VEGF (A, y = 2.297x + 4.627, r2 = 0.33, P < 0.05) and erythropoietin (B, y = 2.01x + 19.66, r2 = 0.33, P < 0.05) across high and moderate altitudes. C: HIF-1α protein correlated with the birth to placental weight ratio across both altitudes (y = 7.72 − 1.20x, r2 = −0.45, P < 0.05).
Discussion
An intricate balance between HIF-1α and its regulatory molecules was revealed within the high-altitude model. The 10- and 20-fold increases in HIF-1α and VHL mRNA, respectively, were associated with a doubling of protein levels. FIH showed a fivefold increase in mRNA but reduced protein levels. VHL binding to CUL2 was unchanged, and HIF-1α ubiquitination was similar or lower at high altitude. These alterations in HIF-1α and its regulatory machinery translated into significant increases in HIF-1α downstream targets (TGFβ3, EPO, and VEGF) in the placenta and maternal circulation. This may be due to greater placental HIF-1α expression and decreased inhibition of HIF-1α transactivation. To our knowledge, these data are the first to directly correlate placental molecular findings with circulating growth factors during pregnancy and with pregnancy outcome.
Apart from one report, all of the high-altitude data point to excess HIF-1α expression and activity in the high-altitude placenta. The other report measured HIF-1 activity using an enzyme-linked immunosorbent assay, which showed no differences, and concluded that the placentas were well adapted.31 However, global patterns of gene expression are similar in high-altitude versus in vitro low-oxygen treatment.21 A number of HIF-regulated genes clustered with greater expression levels in both hypoxic conditions relative to control.21 Increased capillary density and enhanced ballooning of the capillaries within the tertiary villi are characteristics of placental adaptation at high altitude.32,33 Placental erythropoietin receptor is increased, and at least four gene products known to be HIF-regulated and produced by the placenta (VEGF, EPO, sFLT-1, and endothelin) are elevated in the placenta and/or maternal circulation at high altitude.25,34,35
The β-subunit of HIF-1 is constitutively produced, and our data, like that from most other cell and tissue systems, show no change in ARNT mRNA or protein with hypoxia.14,36 The decrement in oxygen tension in the blood entering the intervillous space at an altitude of 3100 m is ∼20%.7 Because intervillous oxygen tension is already quite low near term (40 mm Hg or approximately 6%),37 the altitude-associated decrement to approximately 4 to 5% is close to what is used in cell and organ culture to mimic placental “hypoxia.” That such subtle differences can translate into differences in expression of HIF-1α and its regulatory molecules is supported by prior work from our laboratories showing that HIF-regulatory proteins such as prolyl hydroxylases respond to changes in oxygen encompassing 2 to 8%.20 This same subtlety is now well documented in human placental development: HIF-1α mRNA and protein expression are elevated early in gestation when oxygen tension is low (2 to 3%) and decline with the onset of higher oxygen tension (>8%) in the late first trimester.38 Because FIH is regulated by oxygen independent of HIF1α stability,16 elevated transcripts suggest either that that this enzyme is capable of sensing the lowered oxygen present in the high-altitude placenta or that factors as yet unidentified inhibit translation under conditions of hypoxia. Of particular importance is that the decreased FIH protein levels likely contributed to the increased downstream placental gene products we examined, including TGFβ3, which is known to be directly regulated by HIF-1α.20,30
We recognize that in many models, hypoxia does not increase HIF-1α mRNA but operates at the level of protein stabilization. Conversely, in some models, sustained increase in HIF-1α mRNA is related to mRNA stabilization.36 Our data on these term samples are consistent with those obtained in organ culture from first trimester placentas.17 This suggests that in vivo, the trophoblast can maintain higher levels of HIF-1α mRNA expression, just as is observed in some cancer cells.39,40 However, we cannot exclude that other mechanisms unrelated to hypoxia such as prostanoids,41 norepinephrine,42 and cytokines43 may contribute to the higher levels of HIF-1α reported here.
The increase in VHL at HA begs the question of why HIF-1α protein remained elevated. An increase in VHL protein does not reflect the amount of VHL that is functionally capable of binding HIF-1α and targeting it for degradation. Leaving aside potential modification of the binding dynamics between the VHL complex and HIF-1α, our data suggest that similar amounts of HIF-1α would be bound and targeted for degradation. Thus an increase in HIF-1α protein, uncompensated by an equivalent increase in the functional VHLcbc complex would lead to an increase in HIF1α protein and gene activation, which is what we observed. With respect to the question of VHL complex and HIF-1α binding dynamics, there is strong evidence to support that the action of prolyl hydroxylases, required for VHLcbc recognition of the HIF-1α-subunit, is reduced under conditions of hypoxia.44,45,46 Future studies are warranted to examine the expression of these enzymes in placentas from high-altitude pregnancies. The results on HIF-1α ubiquitination showed that it was decreased or unchanged at high altitude. However, the literature and the balance of the other data acquired, especially the reduction in neddylated CUL2, which is required in vitro for full ubiquitin ligase activity of the VHLCBC complex,12 would support that decreased ubiquitination was present at high altitude.
The observation that VHL expression is elevated in conditions of chronic hypoxia is in agreement with our recent observation showing increased VHL in early gestation20 and with other in vitro studies showing oxygen-dependent changes in VHL expression both in cell lines and villous explants.47,48 In addition, the elevated level of VHL may contribute to other features noted in the high-altitude placenta, eg, cytotrophoblast proliferation,49 because VHL plays a role in regulating a variety of cellular events including cell proliferation and extracellular matrix assembly.50,51 The finding of less neddylated CUL2 in HA placentas is also consistent with the prior literature. As noted above, NEDD8 conjugation to CUL2 is required for full ubiquitin ligase activity of the VHLCBC complex12 but is not essential for VHLCBC complex formation.52 Neddylated CUL2 first appears in human placentas during the developmental window in which intervillous oxygen tension rises.20 Its reduction in the present report is consistent with lowered oxygen tension.
We examined circulating total VEGF as a serial marker of placental hypoxia during pregnancy. Free VEGF is virtually absent in human pregnancy, and hence the total (bound) VEGF that we measured in the circulation is unlikely to have significant biological function. It may exert paracrine effects within the placenta before binding with its soluble receptor sFlt-1, including vasodilation.53,54 Our measures immediately postpartum and 3 months after delivery indicate that the elevated total VEGF in human pregnancy is of placental origin. Placental HIF protein was positively correlated with near-term maternal circulating VEGF and erythropoietin, supporting the idea that placental HIF activation can measurably impact on maternal physiology. In turn, excess VEGF in the maternal circulation was associated with lower birth weight. The opposite is found at mid-gestation at sea level: greater total VEGF was correlated with greater birth weight.55 Wheeler et al55 speculated that the correlations earlier in pregnancy at sea level may reflect maternal cardiovascular adaptation to pregnancy. If this were the case, it would not necessarily be surprising to find a negative relationship at high altitude. Maternal physiological changes at high altitude are intermediate between normal and preeclamptic pregnancy at sea level.56
Finally, our findings may provide the opportunity to refine our understanding of pregnancy pathologies. Dysregulation of the HIF system is implicated in preeclamptic pregnancies.6,17,57,58 The increase in HIF-1α reported here is similar to or even greater than the rise reported in preeclampsia, but it differs in that we see no increase in HIF-2α.59 HIF-2α is implicated in oxidative stress,60 which is increased in preeclamptic placentas61 but decreased in the high-altitude model.62 We have shown that there are similar patterns of gene expression in high-altitude and preeclamptic placentas. However, the preeclamptic placentas had greater increases in the HIF-regulated genes VEGF, TGFβ3, and ceruloplasmin than those from high altitude.21 These data suggest that for a given level of HIF-1α, transactivation may be increased in preeclampsia. Hence, posttranscriptional mechanisms would bear closer inspection in the pathology of preeclamptic pregnancies.
Footnotes
Address reprint requests to Stacy Zamudio, Ph.D., New Jersey Medical School, Department of Obstetrics, Gynecology and Women’s Health, 185 South Orange Ave., MSB E605, Newark, NJ 07103. E-mail: stacy.zamudio@umdnj.edu.
Supported by the Canadian Institutes of Health Research (grant MT-14096 to I.C.), by the National Science Foundation (grant BCS 0309142 to S.Z.), and the National Institutes of Health (grants HD42737 to S.Z. and HD46982 to N.I.). I.C. is the recipient of a mid-career Canadian Institutes of Health Research award administered through the Ontario Women’s Health Council. M.P. is the holder of a Canadian Research Chair (tier 1) in Respiration.
References
- Zamudio S. High-altitude hypoxia and preeclampsia. Front Biosci. 2007;12:2967–2977. doi: 10.2741/2286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen GM, Moore LG. The effect of high altitude and other risk factors on birthweight: independent or interactive effects? Am J Public Health. 1997;87:1003–1007. doi: 10.2105/ajph.87.6.1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giussani DA, Phillips PS, Anstee S, Barker DJ. Effects of altitude versus economic status on birth weight and body shape at birth. Pediatr Res. 2001;49:490–494. doi: 10.1203/00006450-200104000-00009. [DOI] [PubMed] [Google Scholar]
- Zamudio S, Droma T, Norkyel KY, Acharya G, Zamudio JA, Niermeyer SN, Moore LG. Protection from intrauterine growth retardation in Tibetans at high altitude. Am J Phys Anthropol. 1993;91:215–224. doi: 10.1002/ajpa.1330910207. [DOI] [PubMed] [Google Scholar]
- Genbacev O, Joslin R, Damsky CH, Polliotti BM, Fisher SJ. Hypoxia alters early gestation human cytotrophoblast differentiation/invasion in vitro and models the placental defects that occur in preeclampsia. J Clin Invest. 1996;97:540–550. doi: 10.1172/JCI118447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caniggia I, Grisaru-Gravnosky S, Kuliszewsky M, Post M, Lye SJ. Inhibition of TGF-beta 3 restores the invasive capability of extravillous trophoblasts in preeclamptic pregnancies. J Clin Invest. 1999;103:1641–1650. doi: 10.1172/JCI6380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamudio S. The placenta at high altitude. High Alt Med Biol. 2003;4:171–191. doi: 10.1089/152702903322022785. [DOI] [PubMed] [Google Scholar]
- Cockman ME, Masson N, Mole DR, Jaakkola P, Chang GW, Clifford SC, Maher ER, Pugh CW, Ratcliffe PJ, Maxwell PH. Hypoxia inducible factor-alpha binding and ubiquitylation by the von Hippel-Lindau tumor suppressor protein. J Biol Chem. 2000;275:25733–25741. doi: 10.1074/jbc.M002740200. [DOI] [PubMed] [Google Scholar]
- Kibel A, Iliopoulos O, DeCaprio JA, Kaelin WG., Jr Binding of the von Hippel-Lindau tumor suppressor protein to Elongin B and C. Science. 1995;269:1444–1446. doi: 10.1126/science.7660130. [DOI] [PubMed] [Google Scholar]
- Ohh M, Park CW, Ivan M, Hoffman MA, Kim TY, Huang LE, Pavletich N, Chau V, Kaelin WG. Ubiquitination of hypoxia-inducible factor requires direct binding to the beta-domain of the von Hippel-Lindau protein. Nat Cell Biol. 2000;2:423–427. doi: 10.1038/35017054. [DOI] [PubMed] [Google Scholar]
- Kamitani T, Kito K, Nguyen HP, Yeh ET. Characterization of NEDD8, a developmentally down-regulated ubiquitin-like protein. J Biol Chem. 1997;272:28557–28562. doi: 10.1074/jbc.272.45.28557. [DOI] [PubMed] [Google Scholar]
- Ohh M, Kim WY, Moslehi JJ, Chen Y, Chau V, Read MA, Kaelin WG., Jr An intact NEDD8 pathway is required for Cullin-dependent ubiquitylation in mammalian cells. EMBO Rep. 2002;3:177–182. doi: 10.1093/embo-reports/kvf028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang GL, Jiang BH, Rue EA, Semenza GL. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci USA. 1995;92:5510–5514. doi: 10.1073/pnas.92.12.5510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruick RK. Oxygen sensing in the hypoxic response pathway: regulation of the hypoxia-inducible transcription factor. Genes Dev. 2003;17:2614–2623. doi: 10.1101/gad.1145503. [DOI] [PubMed] [Google Scholar]
- Mahon PC, Hirota K, Semenza GL. FIH-1: a novel protein that interacts with HIF-1alpha and VHL to mediate repression of HIF-1 transcriptional activity. Genes Dev. 2001;15:2675–2686. doi: 10.1101/gad.924501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lando D, Peet DJ, Gorman JJ, Whelan DA, Whitelaw ML, Bruick RK. FIH-1 is an asparaginyl hydroxylase enzyme that regulates the transcriptional activity of hypoxia-inducible factor. Genes Dev. 2002;16:1466–1471. doi: 10.1101/gad.991402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caniggia I, Mostachfi H, Winter J, Gassmann M, Lye SJ, Kuliszewski M, Post M. Hypoxia-inducible factor-1 mediates the biological effects of oxygen on human trophoblast differentiation through TGFbeta(3). J Clin Invest. 2000;105:577–587. doi: 10.1172/JCI8316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajakumar A, Conrad KP. Expression, ontogeny, and regulation of hypoxia-inducible transcription factors in the human placenta. Biol Reprod. 2000;63:559–569. doi: 10.1095/biolreprod63.2.559. [DOI] [PubMed] [Google Scholar]
- Rajakumar A, Doty K, Daftary A, Markovic N, Conrad KP. Expression of von Hippel Lindau (pVHL) protein in placentae from normal pregnant women and women with preeclampsia. Placenta. 2006;27:411–421. doi: 10.1016/j.placenta.2005.04.002. [DOI] [PubMed] [Google Scholar]
- Ietta F, Wu Y, Winter J, Xu J, Wang J, Post M, Caniggia I. Dynamic HIF1A regulation during human placental development. Biol Reprod. 2006;75:112–121. doi: 10.1095/biolreprod.106.051557. [DOI] [PubMed] [Google Scholar]
- Soleymanlou N, Jurisica I, Nevo O, Ietta F, Zhang X, Zamudio S, Post M, Caniggia I. Molecular evidence of placental hypoxia in preeclampsia. J Clin Endocrinol Metab. 2005;90:4299–4308. doi: 10.1210/jc.2005-0078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semenza GL. HIF-1, O(2), and the 3 PHDs: how animal cells signal hypoxia to the nucleus. Cell. 2001;107:1–3. doi: 10.1016/s0092-8674(01)00518-9. [DOI] [PubMed] [Google Scholar]
- Axelson H, Fredlund E, Ovenberger M, Landberg G, Pahlman S. Hypoxia-induced dedifferentiation of tumor cells: a mechanism behind heterogeneity and aggressiveness of solid tumors. Semin Cell Dev Biol. 2005;16:554–563. doi: 10.1016/j.semcdb.2005.03.007. [DOI] [PubMed] [Google Scholar]
- Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3:721–732. doi: 10.1038/nrc1187. [DOI] [PubMed] [Google Scholar]
- Nevo O, Soleymanlou N, Wu Y, Xu J, Kingdom J, Many A, Zamudio S, Caniggia I. Increased expression of sFlt-1 in in vivo and in vitro models of human placental hypoxia is mediated by HIF-1. Am J Physiol. 2006;291:R1085–R1093. doi: 10.1152/ajpregu.00794.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anthony FW, Evans PW, Wheeler T, Wood PJ. Variation in detection of VEGF in maternal serum by immunoassay and the possible influence of binding proteins. Ann Clin Biochem. 1997;34:276–280. doi: 10.1177/000456329703400309. [DOI] [PubMed] [Google Scholar]
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- MacPhee DJ, Mostachfi H, Han R, Lye SJ, Post M, Caniggia I. Focal adhesion kinase is a key mediator of human trophoblast development. Lab Invest. 2001;81:1469–1483. doi: 10.1038/labinvest.3780362. [DOI] [PubMed] [Google Scholar]
- Young DA, Zerbe GO., Jr HWW: Fieller’s theorem, Scheffe’s simultaneous confidence intervals, and ratios of the parameters of linear and nonlinear mixed-effects models. Biometrics. 1997;53:114–123. [PubMed] [Google Scholar]
- Nishi H, Nakada T, Hokamura M, Osakabe Y, Itokazu O, Huang LE, Isaka K. Hypoxia-inducible factor-1 transactivates transforming growth factor-beta3 in trophoblast. Endocrinology. 2004;145:4113–4118. doi: 10.1210/en.2003-1639. [DOI] [PubMed] [Google Scholar]
- Tissot van Patot MC, Bendrick-Peart J, Beckey VE, Serkova N, Zwerdlinger L. Greater vascularity, lowered HIF-1/DNA binding, and elevated GSH as markers of adaptation to in vivo chronic hypoxia. Am J Physiol. 2004;287:L525–L532. doi: 10.1152/ajplung.00203.2003. [DOI] [PubMed] [Google Scholar]
- Starzyk KA, Salafia CM, Pezzullo JC, Lage JM, Parkash V, Vercruysse L, Hanssens M, Pijnenborg R. Quantitative differences in arterial morphometry define the placental bed in preeclampsia. Hum Pathol. 1997;28:353–358. doi: 10.1016/s0046-8177(97)90135-0. [DOI] [PubMed] [Google Scholar]
- Ogino S, Redline RW. Villous capillary lesions of the placenta: distinctions between chorangioma, chorangiomatosis, and chorangiosis. Hum Pathol. 2000;31:945–954. doi: 10.1053/hupa.2000.9036. [DOI] [PubMed] [Google Scholar]
- Zamudio S, Baumann MU, Illsley NP. Effects of chronic hypoxia in vivo on the expression of human placental glucose transporters. Placenta. 2006;27:49–55. doi: 10.1016/j.placenta.2004.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore LG, Shriver M, Bemis L, Hickler B, Wilson M, Brutsaert T, Parra E, Vargas E. Maternal adaptation to high-altitude pregnancy: an experiment of nature. A review. Placenta. 2004;25(Suppl A):S60–S71. doi: 10.1016/j.placenta.2004.01.008. [DOI] [PubMed] [Google Scholar]
- Semenza GL. Regulation of mammalian O2 homeostasis by hypoxia-inducible factor 1. Annu Rev Cell Dev Biol. 1999;15:551–578. doi: 10.1146/annurev.cellbio.15.1.551. [DOI] [PubMed] [Google Scholar]
- Soothill PW, Nicolaides KH, Rodeck CH, Campbell S. Effect of gestational age on fetal and intervillous blood gas and acid-base values in human pregnancy. Fetal Ther. 1986;1:168–175. doi: 10.1159/000262264. [DOI] [PubMed] [Google Scholar]
- Jauniaux E, Watson AL, Burton G. Evaluation of respiratory gases and acid-base gradients in fetal tissue and uteroplacental tissue between 7–16 weeks. Am J Obstet Gynecol. 2001;184:998–1003. doi: 10.1067/mob.2001.111935. [DOI] [PubMed] [Google Scholar]
- Søndergaard KL, Hilton DA, Penney M, Ollerenshaw M, Demaine AG. Expression of hypoxia-inducible factor 1alpha in tumours of patients with glioblastoma. Neuropathol Appl Neurobiol. 2002;28:210–217. doi: 10.1046/j.1365-2990.2002.00391.x. [DOI] [PubMed] [Google Scholar]
- Koshikawa N, Iyozumi A, Gassmann M, Takenaga K. Constitutive upregulation of hypoxia-inducible factor-1alpha mRNA occurring in highly metastatic lung carcinoma cells leads to vascular endothelial growth factor overexpression upon hypoxic exposure. Oncogene. 2003;22:6717–6724. doi: 10.1038/sj.onc.1206765. [DOI] [PubMed] [Google Scholar]
- Critchley HO, Osei J, Henderson TA, Boswell L, Sales KJ, Jabbour HN, Hirani N. Hypoxia-inducible factor-1alpha expression in human endometrium and its regulation by prostaglandin E-series prostanoid receptor 2 (EP2). Endocrinology. 2006;147:744–753. doi: 10.1210/en.2005-1153. [DOI] [PubMed] [Google Scholar]
- Nikami H, Nedergaard J, Fredriksson JM. Norepinephrine but not hypoxia stimulates HIF-1alpha gene expression in brown adipocytes. Biochem Biophys Res Commun. 2005;337:121–126. doi: 10.1016/j.bbrc.2005.09.011. [DOI] [PubMed] [Google Scholar]
- Scharte M, Han X, Bertges DJ, Fink MP, Delude RL. Cytokines induce HIF-1 DNA binding and the expression of HIF-1-dependent genes in cultured rat enterocytes. Am J Physiol. 2003;284:G373–G384. doi: 10.1152/ajpgi.00076.2002. [DOI] [PubMed] [Google Scholar]
- Bruick RK, McKnight SL. A conserved family of prolyl-4-hydroxylases that modify HIF. Science. 2001;294:1337–1340. doi: 10.1126/science.1066373. [DOI] [PubMed] [Google Scholar]
- Berra E, Benizri E, Ginouves A, Volmat V, Roux D, Pouyssegur J. HIF prolyl-hydroxylase 2 is the key oxygen sensor setting low steady-state levels of HIF-1alpha in normoxia. EMBO J. 2003;22:4082–4090. doi: 10.1093/emboj/cdg392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D’Angelo G, Duplan E, Boyer N, Vigne P, Frelin C. Hypoxia up-regulates prolyl hydroxylase activity: a feedback mechanism that limits HIF-1 responses during reoxygenation. J Biol Chem. 2003;278:38183–38187. doi: 10.1074/jbc.M302244200. [DOI] [PubMed] [Google Scholar]
- Turcotte S, Desrosiers RR, Beliveau R. Hypoxia upregulates von Hippel-Lindau tumor-suppressor protein through RhoA-dependent activity in renal cell carcinoma. Am J Physiol. 2004;286:F338–F348. doi: 10.1152/ajprenal.00254.2003. [DOI] [PubMed] [Google Scholar]
- Genbacev O, Krtolica A, Kaelin W, Fisher SJ. Human cytotrophoblast expression of the von Hippel-Lindau protein is downregulated during uterine invasion in situ and upregulated by hypoxia in vitro. Dev Biol. 2001;233:526–536. doi: 10.1006/dbio.2001.0231. [DOI] [PubMed] [Google Scholar]
- Ali KZ. Stereological study of the effect of altitude on the trophoblast cell populations of human term placental villi. Placenta. 1997;18:447–450. doi: 10.1016/s0143-4004(97)80046-x. [DOI] [PubMed] [Google Scholar]
- Pause A, Lee S, Lonergan KM, Klausner RD. The von Hippel-Lindau tumor suppressor gene is required for cell cycle exit upon serum withdrawal. Proc Natl Acad Sci USA. 1998;95:993–998. doi: 10.1073/pnas.95.3.993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohh M, Yauch RL, Lonergan KM, Whaley JM, Stemmer-Rachamimov AO, Louis DN, Gavin BJ, Kley N, Kaelin WG, Jr, Iliopoulos O. The von Hippel-Lindau tumor suppressor protein is required for proper assembly of an extracellular fibronectin matrix. Mol Cell. 1998;1:959–968. doi: 10.1016/s1097-2765(00)80096-9. [DOI] [PubMed] [Google Scholar]
- Liakopoulos D, Busgen T, Brychzy A, Jentsch S, Pause A. Conjugation of the ubiquitin-like protein NEDD8 to cullin-2 is linked to von Hippel-Lindau tumor suppressor function. Proc Natl Acad Sci USA. 1999;96:5510–5515. doi: 10.1073/pnas.96.10.5510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ni Y, May V, Braas K, Osol G. Pregnancy augments uteroplacental vascular endothelial growth factor gene expression and vasodilatoreffects. Am J Physiol. 1997;273:H938–H944. doi: 10.1152/ajpheart.1997.273.2.H938. [DOI] [PubMed] [Google Scholar]
- He H, Venema VJ, Gu X, Venema RC, Marrero MB, Caldwell RB. Vascular endothelial growth factor signals endothelial cell production of nitric oxide and prostacyclin through flk-1/KDR activation of c-Src. J Biol Chem. 1999;274:25130–25135. doi: 10.1074/jbc.274.35.25130. [DOI] [PubMed] [Google Scholar]
- Wheeler T, Evans PW, Anthony FW, Godfrey KM, Howe DT, Osmond C. Relationship between maternal serum vascular endothelial growth factor concentration in early pregnancy and fetal and placental growth. Hum Reprod. 1999;14:1619–1623. doi: 10.1093/humrep/14.6.1619. [DOI] [PubMed] [Google Scholar]
- Zamudio S. Lyall F, editor. Cambridge, UK: Cambridge University Press,; High Altitude and Hypertension in Pregnancy. 2006 [Google Scholar]
- Rajakumar A, Doty K, Daftary A, Harger G, Conrad KP. Impaired oxygen-dependent reduction of HIF-1alpha and -2alpha proteins in pre-eclamptic placentae. Placenta. 2003;24:199–208. doi: 10.1053/plac.2002.0893. [DOI] [PubMed] [Google Scholar]
- Caniggia I, Winter J, Lye SJ, Post M. Oxygen and placental development during the first trimester: implications for the pathophysiology of pre-eclampsia. Placenta. 2000;21(Suppl A):S25–S30. doi: 10.1053/plac.1999.0522. [DOI] [PubMed] [Google Scholar]
- Rajakumar A, Whitelock KA, Weissfeld LA, Daftary AR, Markovic N, Conrad KP. Selective overexpression of the hypoxia-inducible transcription factor, HIF-2alpha, in placentas from women with preeclampsia. Biol Reprod. 2001;64:499–506. doi: 10.1093/biolreprod/64.2.499. [DOI] [PubMed] [Google Scholar]
- Scortegagna M, Ding K, Oktay Y, Gaur A, Thurmond F, Yan LJ, Marck BT, Matsumoto AM, Shelton JM, Richardson JA, Bennett MJ, Garcia JA. Multiple organ pathology, metabolic abnormalities and impaired homeostasis of reactive oxygen species in Epas1−/− mice. Nat Genet. 2003;35:331–340. doi: 10.1038/ng1266. [DOI] [PubMed] [Google Scholar]
- Vanderlelie J, Venardos K, Clifton VL, Gude NM, Clarke FM, Perkins AV. Increased biological oxidation and reduced anti-oxidant enzyme activity in pre-eclamptic placentae. Placenta. 2005;26:53–58. doi: 10.1016/j.placenta.2004.04.002. [DOI] [PubMed] [Google Scholar]
- Zamudio S, Kovalenko O, Vanderlelie J, Illsley NP, Heller D, Belliappa S, Perkins AV. Chronic hypoxia in vivo reduces placental oxidative stress. Placenta. 2007 doi: 10.1016/j.placenta.2006.11.010. DOI: 10.1016/j.placenta.2006.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]