Abstract
Neutralizing antibody responses against heterologous isolates in human immunodeficiency virus type 1 (HIV-1) and HIV-2 infections were compared, and their relationships with established clinical markers of progression were examined. Neutralizing responses against 7 heterologous primary isolates and 1 laboratory strain were compared between 32 untreated HIV-1-infected subjects and 35 untreated HIV-2-infected subjects using a pseudotyped reporter virus assay. The breadth of the neutralizing response, defined as the proportion of panel viruses positively neutralized by patient plasma, was significantly greater among HIV-2-infected subjects than among HIV-1-infected subjects. Notably, for fully one-third of HIV-2 subjects, all viruses were effectively neutralized in our panel. Magnitudes of responses, defined as reciprocal 50% inhibitory concentration (IC50) titers for positive reactions, were significantly greater among HIV-1-infected subjects than among HIV-2-infected subjects. When plasma samples from HIV-1 patients were tested for cross-neutralization of HIV-2 and vice versa, we found that these intertype responses are very rare and their prevalences comparable in both HIV-1 and HIV-2 infection. The significantly higher magnitude of heterologous responses for HIV-1 compared to HIV-2 prompted us to examine associations with viremia, which is known to be significantly higher in HIV-1 infection. Importantly, there was a significant positive correlation between the IC50 titer and viral load within both the HIV-1 and HIV-2 groups, suggesting heterologous antibodies may be driven by viral replication. We conclude that HIV-2 infection is characterized by a broad, low-magnitude intratype neutralization response, while HIV-1 is characterized by a narrower but higher-magnitude intratype response and that a significant positive association between the IC50 titer and viremia is common to both HIV-1 and HIV-2 infections.
Human immunodeficiency virus type 2 (HIV-2), like HIV-1, causes AIDS in humans. Although they are highly related lentiviruses, HIV-1 and HIV-2 maintain some distinct epidemiological and biological characteristics. For example, HIV-2 is largely confined to West Africa, while HIV-1 infection is prevalent worldwide. Importantly, disease progression occurs much more slowly with HIV-2 (29, 30, 53), with more than 95% of infected individuals followed for at least 8 years fitting a clinical definition of long-term nonprogression (29). This difference makes HIV-2 a potentially important model of attenuated HIV infection that might provide unique insight into the pathogenesis of HIV-1. To date, the precise mechanisms responsible for this attenuated phenotype of HIV-2 remain unclear. Previous studies have shown that proviral loads are comparable between HIV-1- and HIV-2-infected individuals (8, 36, 39). Plasma viral loads, by contrast, are significantly higher in people infected with HIV-1 (2, 4, 39, 40, 47). This suggests that a key difference between the two HIV types may lie in the degree of viral replication (39), and it is presumed that host immunity largely contributes to the more successful control of HIV-2 infection.
Neutralizing antibody responses are critical mediators of host defense against many human pathogens, although their role in HIV infection remains unclear. One of the difficulties in assessing the importance of neutralizing antibody responses in HIV infection has been the lack of standardized neutralization assays. This problem has been addressed by the increasing use of HIV reporter viruses bearing primary HIV env clones, initially applied to neutralizing antibody studies by Richman et al. (42) and Wei et al. (51). These reporter virus-based neutralization assays are based on single-round infections by envelope pseudotyped viruses, and the readout is more directly related to the number of infection events than is the case with traditional p24 antigen-based assays, which are sensitive to viral and target cell factors that are influenced by virus replication and dependent on multiple rounds of virus infection.
Most of what is known about the role of neutralizing antibody responses in HIV infection is the result of studies of HIV-1-infected populations or studies using nonhuman primate models. Passive-transfer experiments with simian-human immunodeficiency virus-infected macaques demonstrated that preexisting neutralizing antibodies can prevent AIDS virus infection (22). Additionally, some studies suggest that potent heterologous neutralizing antibody responses contribute to the control of HIV-1 in patients classified as long-term nonprogressors (12, 13, 33, 38), although other studies have failed to replicate these findings (7, 10, 21, 26). Most individuals recently infected with HIV-1 mount a vigorous neutralizing antibody response directed against autologous virus; however, antibody escape often emerges during early infection (3, 5, 18, 34, 42, 51). By contrast, neutralizing antibody responses to heterologous primary isolates or to laboratory strains have been shown to be negligible or nonexistent during the first year or two of HIV-1 infection (1, 32, 34, 42). The breadth and magnitude of these heterologous responses may be largely driven by HIV diversification (20, 43) and viral replication (6, 16). Given the purported differences in in vivo viral replication between HIV-1 and HIV-2 (39), it is tempting to speculate that qualitative differences in heterologous neutralizing antibody responses might also exist. This is of interest, since a major focus of the HIV vaccine effort is the development of broadly reactive neutralizing antibodies able to retain potent and broad activities against heterologous primary isolates (19).
To date, there have been few comprehensive studies of the neutralizing antibody response in natural HIV-2 infection. Some early studies suggested that autologous neutralizing antibody responses to contemporaneous isolates may be more common in people with HIV-2 infection than in those infected with HIV-1 (9, 17, 19, 48). Few studies have comprehensively examined the neutralizing antibody response against heterologous primary isolates in HIV-2-infected individuals. One recent study demonstrated potent neutralization against heterologous primary HIV-2 isolates among a small number of HIV-2-infected subjects (46); however, this study was limited to patients who were antiretroviral therapy-experienced and/or had clinically progressed and utilized a nonstandard neutralization assay.
Potent heterologous neutralizing antibody responses in HIV-2 infection might suggest that antibodies are more easily able to neutralize HIV-2 or that people infected with HIV-2 possess a uniquely flexible host antibody repertoire. Either interpretation would support the hypothesis that neutralizing antibody responses are especially robust in HIV-2 infection, correlating with its suppressed replication and pathogenesis. In order to directly compare plasma neutralizing activity against heterologous primary isolates between HIV-1- and HIV-2-infected subjects, plasma samples from 32 antiretroviral therapy-naive HIV-1-infected subjects were compared to those from 35 antiretroviral therapy-naive HIV-2-infected subjects using a pseudotyped reporter virus assay.
MATERIALS AND METHODS
Study population.
This study was conducted with women enrolled in an ongoing prospective clinical cohort of registered female sex workers in Dakar, Senegal, which has been followed since 1985. Various epidemiological and clinical aspects of this cohort have been reported previously (25). All subjects signed informed consent and participated in protocols approved by the Counseil National de Lutte Contre le Sida Comite Ethique et Juridique and the Harvard School of Public Health Human Subjects Committee, in accordance with the Federal Office of Human Research Protections regulations. HIV serostatus and CD4+ T-cell counts were determined as described previously (25). All subjects were free of dual HIV-1/HIV-2 infection. Plasma samples for viral RNA were collected beginning in 1996; blood was collected in EDTA-containing tubes and the plasma stored at −70°C within 6 h of collection, following separation by use of Ficoll-Hypaque (Organon Teknika Cappel; Durham, NC). Viral loads were determined as described below. All subjects had been infected with HIV for >6 years. The time of infection for seroincident women was estimated to be the midpoint between the dates of their last seronegative and first seropositive bleeds. All subjects enrolled in this study were antiretroviral therapy naive and had CD4+ T-cell counts of >200/μl at the time of sample acquisition.
Quantification of plasma viral loads.
HIV-1 plasma viral loads were determined using a commercially available assay (Amplicor HIV-1 monitor test, version 1.5; Roche, Basel, Switzerland). To determine HIV-2 viral loads, an in-house real-time PCR viral load assay was developed. Full-length HIV-2 genomic RNA from a previously described in vitro infection (28) was extracted (QIAamp viral RNA mini-kit; QIAGEN, Valencia, CA), treated with DNase (New England Biolabs; Ipswich, MA), and repurified (MEGAclear; Ambion, Foster City, CA). Quantification of this genomic HIV-2 RNA was determined using in vitro-transcribed HIV-2 RNA generated as follows. Briefly, to construct a quantitative standard, a fragment of the HIV-2 long terminal repeat-gag gene was amplified (from a previously cloned HIV-2 DNA sample) using primers HIV2gagF (CCA ACC ACG ACG GAG TGC TC) and AM2gag1r (CAA TTC ATT CGC TGC CCA CAC) and cloned into an in vitro transcription construct using commercially available reagents (BLOCK-iT RNAi TOPO transcription kit; Invitrogen, Carlsbad, CA). RNA was generated by in vitro transcription (MEGAshortscript; Ambion, Foster City, CA), purified (MEGAclear; Ambion, Foster City, CA), treated with DNase (New England Biolabs, Ipswich, MA), and repurified. The concentration of in vitro-transcribed RNA was determined from an optical density reading at 260 nm and used to generate a quantitative standard of full-length HIV-2 genomic RNA in a two-step real-time reverse transcription-PCR (RT-PCR) assay (Taqman reverse transcription reagents and Taqman universal PCR master mix; Applied Biosystems, Foster City, CA) using primers HIV2gagF and HIV2gagR (CTC TCA AGA CGG AGT TTC TCG C), and RNA was detected using the probe HIV2gagP (AGG CCT CCG GGT GAA GGT AAG), which contained a 5′ 6-carboxyfluorescein fluorescent reporter and a 3′ MGB nonfluorescent quencher. For quantification of plasma viral loads, RNA standard curves were generated from the full-length HIV-2 genomic RNA. This standard-curve RNA was added to noninfected lysis buffer and extracted in parallel with HIV-2 plasma samples (QIAamp viral RNA mini-kit; QIAGEN, Valencia, CA). Viral RNA was quantified relative to the HIV-2 genomic RNA standard curve in the two-step real-time RT-PCR assay described above.
env amplification.
HIV genomic RNA or DNA was isolated from either plasma or peripheral blood mononuclear cells (QIAamp viral RNA mini-kit and blood and cell culture DNA mini-kit; QIAGEN, Valencia, CA). env was amplified in a nested RT-PCR from plasma RNA using a one-step RT-PCR assay kit (SuperScript One-Step RT-PCR kit; Invitrogen, Carlsbad, CA) according to the manufacturer's instructions and using first-round primers H1R1F (TAG GCA TCT CCT ATG GCA GGA AGA A) and H1R1R (TAA ATC TTG AGA TRC TGC TCC TAC TC) for HIV-1 and H2R1F (CTY ATT TTC CAG GTS TGG TST GGC ARA GGT) and H2R1R (TCT CCA TGG GGT GTT TTC AT) for HIV-2. Second-round PCRs were performed using primers H1R2F (CAC CGA ATA AGA GAA AGA GCA GAA GAY A) and H1R2R (CTT TTT GAC CAC TTG CCM CCC AT) for HIV-1 and H2R2F (CTA GGT ACC GTA TGA TGT GTG GTA AGA GT) and H2R2R (TGC GGA TCC TCA CAG GAG GGC GAT TTC TGC) for HIV-2. Where proviral DNA was used as a template, the same primer pairs described above were used in a nested PCR. Blunt-ended PCR products, representing env pools, were cloned into the pcDNA 3.1 directional cloning vector according to the manufacturer's instructions (Invitrogen, Carlsbad, CA). env expression vectors were expanded in 300-ml cultures, purified (QIAGEN MaxiPrep kit; QIAGEN, Valencia, CA), and quantified and purity assessed by using a spectrophotometer.
Neutralizing antibody assay.
Reporter virus particles containing subject Env proteins were produced by cotransfecting 293T cells with env expression vectors (10 μg) plus an env-deficient HIV-1 genomic vector, pNL4-3.Luc.R-E- (20 μg), that contains a firefly luciferase indicator gene (obtained through the NIH AIDS Research and Reference Reagent Program).
A panel of eight HIV-1 reporter viruses was used. Seven of these carried primary patient-derived Env proteins from epidemiologically unlinked HIV-1 CRF02_AG-infected individuals (CRF02_AG is the most prevalent HIV-1 strain in our cohort (31, 44), and one carried the Env protein from the neutralization-sensitive laboratory strain NL4-3. A panel of eight HIV-2 reporter viruses was used. Seven of these carried primary patient-derived Env proteins from epidemiologically unlinked HIV-2-infected individuals, and one carried the Env protein of laboratory-strain CBL23 (initially described in reference 45).
Recombinant viruses pseudotyped with patient-derived virus Env proteins were harvested 48 h posttransfection, centrifuged, and quantified for p24 content using a commercial enzyme-linked immunosorbent assay (Alliance HIV-1 p24 Antigen ELISA kit; Perkin-Elmer, Wellesley, MA). All viruses were screened to ensure they were functional for infection and yielded high luciferase readings in lysates of target cells prior to use in neutralization assays. The neutralizing antibody assay used was based on the pseudotyped reporter virus system as described by Richman et al. (42). Briefly, 750 pg of HIV pseudoviruses (based on p24 content) were incubated for 1 h at 37°C with fivefold dilutions of heat-inactivated patient plasma samples. Repeated controls using a range of virus input values were run to verify that luciferase expression correlated directly with the amount of virus inoculum used but that the neutralization titer was largely independent of the virus inoculum. All dilutions were normalized to contain 10% total human plasma using normal donor plasma. U87 cells expressing CD4 and the CCR5 and CXCR4 coreceptors were then inoculated with virus-plasma dilutions in the absence of added cations. Virus infectivity was determined 72 h postinoculation by measuring the amount of luciferase activity expressed in infected cells. Background luminescence was measured using control wells that contained only target cells and medium. Luciferase activity was quantified using a commercial luciferase assay kit (Steady-Glo luciferase assay system; Promega), and plates were read on a microplate luminometer.
Neutralizing activity was displayed as the percent inhibition of viral infection (luciferase activity) at each antibody dilution compared to an antibody-negative control: percent inhibition = [1 − (luciferase with antibody/luciferase without antibody)] × 100. Fifty percent inhibitory concentration (IC50) titers were calculated using the BioDataFit HTP 1.2 program as the reciprocal of the plasma dilution conferring 50% inhibition relative to negative plasma controls. To monitor the amount of neutralization activity that is not HIV Env protein specific, each plasma sample was also tested against a pseudovirus carrying the vesicular stomatitis virus (VSV) envelope protein (obtained through the NIH AIDS Research and Reference Reagent Program). These VSV envelope proteins are able to mediate virus entry into the target cells used but are not inhibited by anti-HIV Env antibodies. Sera from 10 HIV-negative individuals from the same cohort were also used as controls against all pseudoviruses tested. The reproducibility of the assay was examined by repeatedly testing the laboratory strain viruses HIV-1 NL4.3 and HIV-2 CBL23 using the same HIV-1 (for NL4.3) and HIV-2 (for CBL23) pooled plasma samples composed of samples from five randomly selected subjects. Using this control, the reproducibility of the assay within and between runs was found to be within 2.8-fold.
Statistical analysis.
Statistical analysis was performed using the STATA statistical software package (STATA, version 9.0; StataCorp, College Station, TX). IC50 titers were calculated using the BioDataFit HTP 1.2 program (Chang Bioscience, Castro Valley, CA). Unadjusted comparisons in breadth and magnitude of neutralizing antibody responses between groups were performed using the Mann-Whitney test. To adjust for CD4+ T-cell counts and age when comparing HIV-1-infected subjects to HIV-2-infected subjects, multivariate regression analyses were performed. All correlations were examined using either the Pearson's or the Spearman's correlation coefficient.
RESULTS
Subject characteristics.
We examined heterologous neutralizing antibody responses for 32 HIV-1-infected individuals and 35 HIV-2-infected individuals (Table 1). The HIV-1-infected subjects (median age [interquartile range {IQR}] = 36 [31 to 42]) were younger than the HIV-2-infected subjects (median age [IQR] = 40 [36 to 42.5]; P = 0.005). Although all subjects were in the asymptomatic phase of HIV infection, CD4+ T-cell counts were lower for HIV-1-infected subjects (median cells/μl [IQR] = 575 [448 to 805]) than for HIV-2-infected subjects (median cells/μl [IQR] = 760 [531 to 969]; P = 0.0315). Consistent with previous reports (2, 4, 39, 40, 47), plasma viral loads were significantly higher for HIV-1-infected subjects (median log10 copies/ml [IQR] = 4.01 [3.62 to 4.57]) than for HIV-2-infected subjects (median log10 copies/ml [IQR] = 2.6 [2.6 to 3.1]; P = 0.009).
TABLE 1.
Study population characteristics by groupa
| Patient cohort (nb) | Age (yr) | CD4+ T-cell count (cells/mm3) | Plasma HIV RNA level (log10 copies/ml) | Duration of HIV infection (yr) |
|---|---|---|---|---|
| HIV-1 infected (32) | 36 (31-42) | 575 (448-805) | 4.01 (3.62-4.57) | 8.6 (7.8-10.5) |
| HIV-2 infected (35) | 40 (36-42.5) | 760 (531-969) | 2.6 (2.6-3.1) | 9.1 (6-10.5) |
Values are medians, with IQRs given in parentheses.
n, no. of patients.
Pseudotyped-virus characteristics.
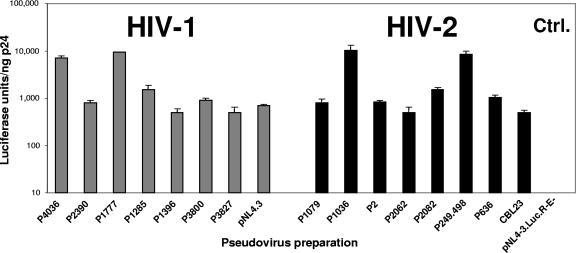
Patient-derived HIV-1 and HIV-2 env genes were used to produce reporter-pseudotyped virus with an env-deficient HIV-1 genomic vector (pNL4-3.Luc.R-E-) engineered to express a luciferase reporter gene (14). The use of the pNL4.3.Luc-R-E backbone with HIV-2 Env proteins to produce infection-competent pseudoviruses has been reported previously (23, 24). To ensure that all pseudotyped viruses were functional and capable of mediating infection into target cells, purified virus preparations were analyzed in a single-round infectivity assay (37, 42). Figure 1 demonstrates that all HIV-1- and HIV-2-pseudotyped viruses used in the study showed infectivity for U87/CD4/CCR5/CXCR4 cells, albeit to various levels. Overall, HIV-1 and HIV-2 pseudoviruses demonstrated comparable infectivity efficiencies in our assay system.
FIG. 1.
Infectivities of pseudoviruses carrying HIV-1 or HIV-2 Env proteins. Virus stocks were generated by cotransfecting 293T cells with vectors carrying HIV-1 and HIV-2 env genes with pNL4.3.Luc-R-E. Infectivity was determined by infection of U87/CD4/CCR5/CXCR4-expressing target cells and measurement of luciferase light signals in target cell lysates per nanogram of p24. Error bars represent standard errors from three independent experiments. Infectivity values for pNL4.3- and CBL23-bearing cells as well as the backbone pNL4.3.Luc-R-E are shown for controls.
Although there was variability within subjects (to different pseudoviruses) and between subjects, the neutralization assay consistently generated neutralization curves similar in shape and slope for the HIV-1 and HIV-2 groups (data not shown). IC50s are reported as they are taken at the inflection of a sigmoid curve and can therefore be estimated with greater confidence than 90% inhibitory concentrations. Importantly, comparable IC50s resulted in comparable nonneutralized fractions between the HIV-1 and HIV-2 groups (i.e., inhibition curves typically plateaued at comparable percent neutralization).
Breadth of heterologous neutralizing antibody responses.
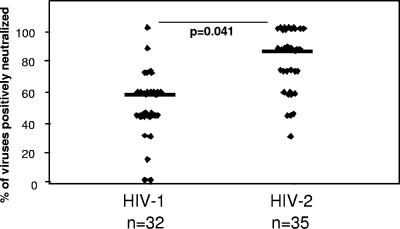
We first examined the breadth of neutralizing antibody responses against our panel of heterologous reporter viruses. A response was considered positive for neutralization if the IC50 titer against the HIV reporter virus was >3-fold higher than that against the VSV control.
The breadths of the responses were generally lower for HIV-1-infected subjects than for HIV-2-infected subjects (P value = 0.041) after adjusting for age and CD4+ T-cell counts (Fig. 2). Plasma from HIV-1-infected subjects neutralized a median of 57% (IQR, 43% to 57%) of viruses in the panel, while plasma from HIV-2-infected subjects neutralized a median of 86% (IQR, 57% to 100%) of the viruses in their panel. Among our HIV-2-infected subjects, all 35 subjects (100%) neutralized at least 1 virus in the panel and, surprisingly, almost one-third (31%) neutralized all 7 primary heterologous viruses in the panel.
FIG. 2.
Breadth of neutralizing antibody responses against a panel of heterologous HIV pseudoviruses among patients with asymptomatic chronic-stage HIV-1 (n = 32) or HIV-2 (n = 35) infection. Breadth was defined as the percentage of viruses in the panel neutralized by subject plasma, where a positive response is defined as an IC50 titer against the HIV pseudovirus that is >3-fold the response against a VSV pseudovirus. Plasma from HIV-2-infected subjects neutralized a significantly greater proportion of heterologous viruses than did plasma from HIV-1-infected subjects (P = 0.041).
There was no significant association between breadth of response (in terms of proportion of viruses neutralized) and CD4+ T-cell count for either the HIV-1-infected (r = 0.11; P = 0.532) or HIV-2-infected (r = 0.06; P = 0.845) infected group. However, within the HIV-1-infected group, there was a positive association between breadth of response and viremia (r = 0.41; P = 0.039).
Together these results suggest that compared to the case with HIV-1 infection, the heterologous neutralizing antibody response in asymptomatic HIV-2 infection is broad, with most serum samples able to neutralize a majority of the viruses in the panel.
Magnitude of heterologous neutralizing antibody responses.
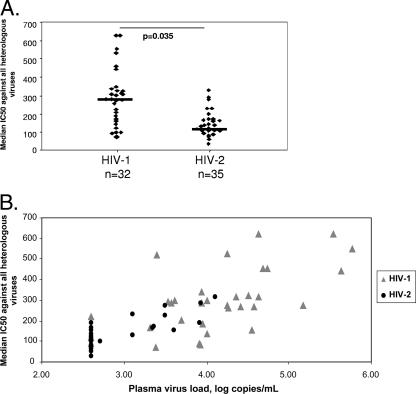
The magnitude of the responses was defined based on the median IC50 titer against all primary heterologous viruses. Although responses varied widely between subjects, the magnitudes of the responses were significantly greater (P = 0.035) among HIV-1-infected subjects (median IC50 titer [IQR] = 285 [186 to 340]) than among HIV-2-infected subjects (median IC50 titer [IQR] = 117 [100 to 167]) after adjusting for age and CD4+ T-cell counts (Fig. 3A). Within the HIV-1 group, IC50 titers against the laboratory strain NL4.3 were significantly higher (p = 0.008) than responses against the reporter viruses carrying patient-derived Env proteins. Within the HIV-2 group, IC50 titers against the CBL23 isolate were only slightly higher (P = 0.082) than responses against the patient-derived reporter viruses.
FIG. 3.
Magnitude of neutralizing antibody responses against a panel of heterologous HIV pseudoviruses among patients with asymptomatic HIV-1 (n = 32) or HIV-2 (n = 35) infection. (A) Comparison of the magnitude of neutralizing antibody responses against HIV reporter viruses. The magnitude of the responses was defined based on the median IC50 titer against all primary heterologous viruses, where a response was positive for neutralization. For HIV-1-infected subjects, the magnitude of neutralizing response against heterologous viruses was significantly higher than that for HIV-2-infected subjects (P = 0.035). (B) Relationship between magnitude of neutralizing antibody response and viral load for HIV-1-infected (triangles) or HIV-2-infected (circles) subjects. There was a significant positive correlation between viremia and the magnitude of the neutralizing response for HIV-1-infected (r = 0.63; P = 0.011) and HIV-2-infected (r = 0.59; P = 0.017) groups.
As shown in Fig. 3B, there was a significant positive association between the magnitude of responses (in terms of average IC50 titer against all viruses) and plasma viral load both within the HIV-1-infected group (r = 0.63; P = 0.011) and within the HIV-2-infected group (r = 0.59; P = 0.017). No association was found between the magnitude of the neutralizing responses and CD4+ T-cell counts in either the HIV-1-infected (r = 0.38; P = 0.182) or HIV-2-infected (r = 0.22; P = 0.374) population.
These results suggest that although HIV-2-infected individuals have broad heterologous neutralizing antibody responses, these responses are generally of low titer compared to those in HIV-1 infection. Further, the significant positive associations we found between the magnitude of neutralizing responses and viremia in both HIV-1- and HIV-2-infected groups suggest that heterologous neutralizing antibody responses may be driven by viral replication in HIV infection.
Cross-neutralization responses.
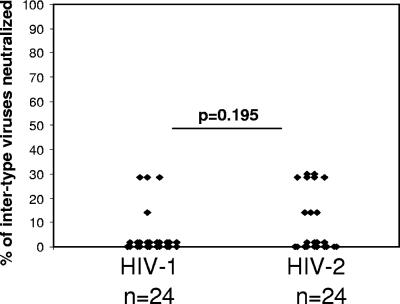
The significant breadth of the heterologous neutralizing antibody response we observed in our HIV-2-infected subjects prompted us to investigate whether plasmas from these individuals are able to neutralize primary HIV-1 reporter viruses and vice versa. Cross-neutralizing antibodies were assessed in a subgroup of 24 HIV-1-infected subjects and 24 HIV-2-positive subjects, randomly selected from the larger study groups in the cross-sectional analysis, using the same panel of HIV-1 and HIV-2 reporter viruses described above.
As shown in Fig. 4, among the 24 samples from HIV-1-infected subjects, only 4 (25%) scored positive for neutralization against any of the HIV-2 viruses in the panel. In total, of the 168 HIV-1 plasma/HIV-2 virus combinations, only 7 (4%) were positive for cross-neutralization. Among the 24 samples from HIV-2-infected subjects, only 9 (38%) were positive for neutralization against any of the HIV-1 viruses in the panel. In total, of the 168 HIV-2 plasma/HIV-1 virus combinations, only 15 (9%) were positive for neutralization. There was not a significant difference in the prevalence of intertype cross-neutralizing responses between HIV-1- and HIV-2-infected subjects (P = 0.195). Notably, the HIV-1 laboratory strain NL4-3 was significantly more sensitive to cross-neutralization than were the patient-derived viruses (P = 0.008). Sixty-five percent of the HIV-2 subject samples tested were positive for neutralization against this virus, with an average IC50 titer of 325. This result is consistent with the high sensitivity of this virus to neutralization and suggests that HIV-1 and HIV-2 share common neutralization epitopes that may be uniquely exposed on the NL4.3 envelope.
FIG. 4.
Intertype cross-neutralization of HIV-2 pseudoviruses by plasma from HIV-1-infected subjects (n = 24) and neutralization of HIV-1 pseudoviruses by plasma from HIV-2-infected subjects (n = 24). There was no significant difference in the proportions of HIV-1- and HIV-2-infected subjects, demonstrating positive neutralization against intertype viruses (P = 0.195).
DISCUSSION
Comparing neutralizing antibody responses between HIV-1- and HIV-2-infected individuals using standardized assays is important to our understanding of the mechanisms responsible for the attenuated pathogenesis of HIV-2 relative to that of HIV-1 and may shed light on the role of these responses in HIV-1 infection. This is the first study, to our knowledge, to apply the HIV reporter virus system to directly compare the heterologous neutralizing antibody responses for HIV-1- and HIV-2-infected individuals. Our findings demonstrate several important characteristics of the heterologous neutralizing antibody response in HIV-1 and HIV-2 infection and highlight some interesting differences between the two groups.
In this study we demonstrate that individuals with chronic HIV-2 infection maintain very broad neutralizing antibodies, with about one-third of the subjects able to neutralize all seven of the primary viruses in the panel. By comparison, HIV-1-infected subjects neutralized a median of three of the viruses in our panel. The ability of an HIV-2-infected subject to neutralize viruses from other individuals suggests that important neutralizing determinants must be shared between viruses isolated from different individuals. Homogeneity at important neutralization domains would, in essence, reduce the distinction between heterologous and autologous viruses and result in extensive cross-reactivity. This interpretation is also supported by our results demonstrating that none of the HIV-2 reporter viruses in our panel, derived from epidemiologically unlinked subjects at different stages of infection, was atypically resistant or sensitive to neutralization. On the other hand, two HIV-1 viruses in our panel seemed to be particularly resistant to neutralization by heterologous plasma. Considering the selective pressure neutralizing antibodies can impose on viral populations, our results suggest that functional constraints may exist to limit evolution of neutralizing domains of the HIV-2 envelope and that this property may be different for the HIV-1 virus. Elucidating this property may provide important information for the design of immunogens able to induce broadly reactive neutralizing antibodies in HIV infection.
Beyond envelope sequence diversity, higher CD4+ T-cell counts (13) and longer durations of HIV infection (34) have also been associated with the breadth of the heterologous neutralizing antibody response. Although our HIV-2-infected individuals did have higher median CD4+ T-cell counts than our HIV-1-infected subjects, we did not find any association between this measure and either the breadth or the magnitude of the neutralizing antibody response. Additionally, all of our comparisons between HIV-1-infected subjects and HIV-2-infected subjects were performed using multivariate regression models in which we adjusted for CD4+ T-cell counts. Further, although our HIV-2-infected subjects were older, there was no significant difference in the duration of HIV infection between our HIV-1-infected subjects and our HIV-2-infected subjects. Therefore, we feel that these issues have not significantly affected our interpretation of the data from this study.
It remains unclear from our results whether these broad responses represent a uniquely diverse antibody repertoire or that the HIV-2 envelope is especially prone to neutralization. It is possible that, similar to what was shown with T-cell-receptor flexibility and broad cytotoxic-T-lymphocyte responses in HIV-2 infection (27), broad antibody responses represent B-cell receptor heterogeneity. Reeves and Doms have hypothesized (41) that HIV-2 may be prone to neutralization due to a more “open” coreceptor binding site conformation or that the functionally important positions (neutralizing domains) in the HIV-2 envelope are more genetically constrained. However, the results from the work of Decker et al. (15) demonstrate that HIV-1 patients maintain robust cross-neutralizing antibodies that are almost exclusively dependent on CD4 binding, suggesting that the HIV-2 coreceptor binding site is not accessible to neutralizing antibodies in its normal conformation.
In 1995, we first reported a retrospective epidemiologic analysis of the Dakar cohort that suggested that HIV-2 infection provided approximately 70% protection from subsequent HIV-1 infection (49, 50). It has been hypothesized that a potent cross-reactive immune response might contribute to this protection (41), with neutralizing antibodies being an obvious prime candidate. Our results in this report, however, suggest that such cross-neutralizing antibodies are extremely rare in HIV-2- or HIV-1-infected individuals. This is contrary to reports from some early studies that plasma from HIV-2-infected individuals was able to neutralize HIV-1 isolates (17, 52). This discrepancy is likely the result of methodological differences, since early studies used laboratory-adapted HIV isolates that are generally uniquely sensitive to plasma neutralization (11, 35) and used replication-competent viruses capable of multiple rounds of replication. It is possible, however, that HIV-2 patients do maintain CD4-induced antibodies able to neutralize HIV-1, since this relationship was shown to exist in the opposite direction by Decker et al. (15).
Although broad, our study demonstrates that heterologous neutralizing antibody responses in HIV-2 infection are generally of very low magnitude compared to those in HIV-1 infection. The low-titer response in HIV-2 infection may likely result from the suppressed viral replication and antigenic stimulation in HIV-2 infection in vivo compared to those of HIV-1 infection (MacNeil et al., submitted for publication). Lending strong support to this interpretation, we demonstrate here a significant positive correlation between IC50 titers and plasma viral load in both the HIV-1 and HIV-2 groups. A similar relationship was recently demonstrated in a study of HIV-1-infected individuals (16), suggesting that the association between viremia and the magnitude of the heterologous neutralizing response is a common characteristic of HIV infection.
It is difficult to interpret the clinical importance of heterologous neutralizing antibody responses to the control of HIV-1 or HIV-2 infection based on our findings. Against the argument that heterologous responses are clinically important in HIV infection, we demonstrate a strong positive association between IC50 titers and viral load levels in both HIV-1- and HIV-2-infected groups. This suggests that the magnitude of the heterologous response is driven by viral replication, which would be expected to inversely correlate with clinical status. However, since all of our subjects fell within the asymptomatic phase of HIV infection, any conclusions extending to symptomatic patients should be made with caution.
Our study has several shortcomings that must be mentioned. First, our study lacks an analysis of neutralizing antibody responses against autologous viruses. This information is critical to understanding the role of neutralizing antibodies and viral evolution in HIV infection and how they may differ between HIV-1 and HIV-2 infection. Such an analysis is technically difficult in HIV-2 due undetectable viral loads in a majority of HIV-2-infected individuals, making the cloning of env sequences from the plasma compartment very difficult for a majority of asymptomatic individuals. Bailey et al. (6) demonstrated that env cloning can be accomplished with patients with undetectable viral loads, but this technique utilized large volumes of patient plasma that are unattainable in our cohort design. Second, we utilized a cross-sectional analysis in this study, which makes it difficult to elucidate cause and effect relationships between neutralization responses and clinical markers. Third, the low numbers of subjects who progress clinically from HIV-2 infection limits our ability to dissect and compare the differences in neutralizing antibody responses between HIV-1- and HIV-2-infected individuals able to control their infection and those who progress to AIDS.
In conclusion, we demonstrate that heterologous neutralizing antibody responses in HIV-2 infection are generally broad, suggesting that circulating HIV-2 viruses in Senegal share common neutralization domains. We suggest these results may be indicative of constraints on viral evolution that may differ for HIV-1. Additionally, the significantly lower magnitudes of neutralizing responses in HIV-2 infection seem to represent the suppressed in vivo viral replication of this virus compared to the case with HIV-1 infection. Similarly, the positive association between heterologous responses and viral load further suggest these responses are driven by viral replication. Finally, we demonstrate that intertype cross-neutralizing responses are extremely rare in both HIV-1 and HIV-2 infections.
Acknowledgments
We thank Beth Chaplin and Christopher Mullins for technical assistance and all members of the Kanki laboratory for helpful discussions.
This work was supported by National Institute of Allergy and Infectious Diseases grant AI46274-04 from the National Institutes of Health.
Footnotes
Published ahead of print on 14 February 2007.
REFERENCES
- 1.Aasa-Chapman, M. M., A. Hayman, P. Newton, D. Cornforth, I. Williams, P. Borrow, P. Balfe, and A. McKnight. 2004. Development of the antibody response in acute HIV-1 infection. AIDS 18:371-381. [DOI] [PubMed] [Google Scholar]
- 2.Alabi, A. S., S. Jaffar, K. Ariyoshi, T. Blanchard, M. Schim van der Loeff, A. A. Awasana, T. Corrah, S. Sabally, R. Sarge-Njie, F. Cham-Jallow, A. Jaye, N. Berry, and H. Whittle. 2003. Plasma viral load, CD4 cell percentage, HLA and survival of HIV-1, HIV-2, and dually infected Gambian patients. AIDS 17:1513-1520. [DOI] [PubMed] [Google Scholar]
- 3.Albert, J., B. Abrahamsson, K. Nagy, E. Aurelius, H. Gaines, G. Nystrom, and E. M. Fenyo. 1990. Rapid development of isolate-specific neutralizing antibodies after primary HIV-1 infection and consequent emergence of virus variants which resist neutralization by autologous sera. AIDS 4:107-112. [DOI] [PubMed] [Google Scholar]
- 4.Andersson, S., H. Norrgren, Z. da Silva, A. Biague, S. Bamba, S. Kwok, C. Christopherson, G. Biberfeld, and J. Albert. 2000. Plasma viral load in HIV-1 and HIV-2 singly and dually infected individuals in Guinea-Bissau, West Africa: significantly lower plasma virus set point in HIV-2 infection than in HIV-1 infection. Arch. Intern. Med. 160:3286-3293. [DOI] [PubMed] [Google Scholar]
- 5.Arendrup, M., C. Nielsen, J. E. Hansen, C. Pedersen, L. Mathiesen, and J. O. Nielsen. 1992. Autologous HIV-1 neutralizing antibodies: emergence of neutralization-resistant escape virus and subsequent development of escape virus neutralizing antibodies. J. Acquir. Immune. Defic. Syndr. 5:303-307. [PubMed] [Google Scholar]
- 6.Bailey, J. R., K. G. Lassen, H. C. Yang, T. C. Quinn, S. C. Ray, J. N. Blankson, and R. F. Siliciano. 2006. Neutralizing antibodies do not mediate suppression of human immunodeficiency virus type 1 in elite suppressors or selection of plasma virus variants in patients on highly active antiretroviral therapy. J. Virol. 80:4758-4770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barker, E., K. N. Bossart, and J. A. Levy. 1998. Primary CD8+ cells from HIV-infected individuals can suppress productive infection of macrophages independent of beta-chemokines. Proc. Natl. Acad. Sci. USA 95:1725-1729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Berry, N., K. Ariyoshi, O. Jobe, P. T. Ngum, T. Corrah, A. Wilkins, H. Whittle, and R. Tedder. 1994. HIV type 2 proviral load measured by quantitative polymerase chain reaction correlates with CD4+ lymphopenia in HIV type 2-infected individuals. AIDS Res. Hum. Retrovir. 10:1031-1037. [DOI] [PubMed] [Google Scholar]
- 9.Bjorling, E., G. Scarlatti, A. von Gegerfelt, J. Albert, G. Biberfeld, F. Chiodi, E. Norrby, and E. M. Fenyo. 1993. Autologous neutralizing antibodies prevail in HIV-2 but not in HIV-1 infection. Virology 193:528-530. [DOI] [PubMed] [Google Scholar]
- 10.Bradney, A. P., S. Scheer, J. M. Crawford, S. P. Buchbinder, and D. C. Montefiori. 1999. Neutralization escape in human immunodeficiency virus type 1-infected long-term nonprogressors. J. Infect. Dis. 179:1264-1267. [DOI] [PubMed] [Google Scholar]
- 11.Burton, D. R., and D. C. Montefiori. 1997. The antibody response in HIV-1 infection. AIDS 11(Suppl. A):S87-S98. [PubMed] [Google Scholar]
- 12.Cao, Y., L. Qin, L. Zhang, J. Safrit, and D. D. Ho. 1995. Virologic and immunologic characterization of long-term survivors of human immunodeficiency virus type 1 infection. N. Engl. J. Med. 332:201-208. [DOI] [PubMed] [Google Scholar]
- 13.Carotenuto, P., D. Looij, L. Keldermans, F. de Wolf, and J. Goudsmit. 1998. Neutralizing antibodies are positively associated with CD4+ T-cell counts and T-cell function in long-term AIDS-free infection. AIDS 12:1591-1600. [DOI] [PubMed] [Google Scholar]
- 14.Connor, R. I., B. K. Chen, S. Choe, and N. R. Landau. 1995. Vpr is required for efficient replication of human immunodeficiency virus type-1 in mononuclear phagocytes. Virology 206:935-944. [DOI] [PubMed] [Google Scholar]
- 15.Decker, J. M., F. Bibollet-Ruche, X. Wei, S. Wang, D. N. Levy, W. Wang, E. Delaporte, M. Peeters, C. A. Derdeyn, S. Allen, E. Hunter, M. S. Saag, J. A. Hoxie, B. H. Hahn, P. D. Kwong, J. E. Robinson, and G. M. Shaw. 2005. Antigenic conservation and immunogenicity of the HIV coreceptor binding site. J. Exp. Med. 201:1407-1419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Deeks, S. G., B. Schweighardt, T. Wrin, J. Galovich, R. Hoh, E. Sinclair, P. Hunt, J. M. McCune, J. N. Martin, C. J. Petropoulos, and F. M. Hecht. 2006. Neutralizing antibody responses against autologous and heterologous viruses in acute versus chronic human immunodeficiency virus (HIV) infection: evidence for a constraint on the ability of HIV to completely evade neutralizing antibody responses. J. Virol. 80:6155-6164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fenyo, E. M., and P. Putkonen. 1996. Broad cross-neutralizing activity in serum is associated with slow progression and low risk of transmission in primate lentivirus infections. Immunol. Lett. 51:95-99. [DOI] [PubMed] [Google Scholar]
- 18.Geffin, R., C. Hutto, C. Andrew, and G. B. Scott. 2003. A longitudinal assessment of autologous neutralizing antibodies in children perinatally infected with human immunodeficiency virus type 1. Virology 310:207-215. [DOI] [PubMed] [Google Scholar]
- 19.Haigwood, N. L., and L. Stamatatos. 2003. Role of neutralizing antibodies in HIV infection. AIDS 17(Suppl. 4):S67-S71. [DOI] [PubMed] [Google Scholar]
- 20.Halapi, E., T. Leitner, M. Jansson, G. Scarlatti, P. Orlandi, A. Plebani, L. Romiti, J. Albert, H. Wigzell, and P. Rossi. 1997. Correlation between HIV sequence evolution, specific immune response and clinical outcome in vertically infected infants. AIDS 11:1709-1717. [DOI] [PubMed] [Google Scholar]
- 21.Harrer, T., E. Harrer, S. A. Kalams, T. Elbeik, S. I. Staprans, M. B. Feinberg, Y. Cao, D. D. Ho, T. Yilma, A. M. Caliendo, R. P. Johnson, S. P. Buchbinder, and B. D. Walker. 1996. Strong cytotoxic T cell and weak neutralizing antibody responses in a subset of persons with stable nonprogressing HIV type 1 infection. AIDS Res. Hum. Retrovir. 12:585-592. [DOI] [PubMed] [Google Scholar]
- 22.Heeney, J. L., V. J. Teeuwsen, M. van Gils, W. M. Bogers, C. De Giuli Morghen, A. Radaelli, S. Barnett, B. Morein, L. Akerblom, Y. Wang, T. Lehner, and D. Davis. 1998. Beta-chemokines and neutralizing antibody titers correlate with sterilizing immunity generated in HIV-1 vaccinated macaques. Proc. Natl. Acad. Sci. USA 95:10803-10808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hill, C. M., H. Deng, D. Unutmaz, V. N. Kewalramani, L. Bastiani, M. K. Gorny, S. Zolla-Pazner, and D. R. Littman. 1997. Envelope glycoproteins from human immunodeficiency virus types 1 and 2 and simian immunodeficiency virus can use human CCR5 as a coreceptor for viral entry and make direct CD4-dependent interactions with this chemokine receptor. J. Virol. 71:6296-6304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hill, C. M., D. Kwon, M. Jones, C. B. Davis, S. Marmon, B. L. Daugherty, J. A. DeMartino, M. S. Springer, D. Unutmaz, and D. R. Littman. 1998. The amino terminus of human CCR5 is required for its function as a receptor for diverse human and simian immunodeficiency virus envelope glycoproteins. Virology 248:357-371. [DOI] [PubMed] [Google Scholar]
- 25.Kanki, P., S. M'Boup, R. Marlink, K. Travers, C. C. Hsieh, A. Gueye, C. Boye, J. L. Sankale, C. Donnelly, W. Leisenring, et al. 1992. Prevalence and risk determinants of human immunodeficiency virus type 2 (HIV-2) and human immunodeficiency virus type 1 (HIV-1) in west African female prostitutes. Am. J. Epidemiol. 136:895-907. [DOI] [PubMed] [Google Scholar]
- 26.Loomis-Price, L. D., J. H. Cox, J. R. Mascola, T. C. VanCott, N. L. Michael, T. R. Fouts, R. R. Redfield, M. L. Robb, B. Wahren, H. W. Sheppard, and D. L. Birx. 1998. Correlation between humoral responses to human immunodeficiency virus type 1 envelope and disease progression in early-stage infection. J. Infect. Dis. 178:1306-1316. [DOI] [PubMed] [Google Scholar]
- 27.Lopes, A. R., A. Jaye, L. Dorrell, S. Sabally, A. Alabi, N. A. Jones, D. R. Flower, A. De Groot, P. Newton, R. M. Lascar, I. Williams, H. Whittle, A. Bertoletti, P. Borrow, and M. K. Maini. 2003. Greater CD8+ TCR heterogeneity and functional flexibility in HIV-2 compared to HIV-1 infection. J. Immunol. 171:307-316. [DOI] [PubMed] [Google Scholar]
- 28.MacNeil, A., J. L. Sankale, S. T. Meloni, A. D. Sarr, S. Mboup, and P. Kanki. 2006. Genomic sites of human immunodeficiency virus type 2 (HIV-2) integration: similarities to HIV-1 in vitro and possible differences in vivo. J. Virol. 80:7316-7321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Marlink, R., P. Kanki, I. Thior, K. Travers, G. Eisen, T. Siby, I. Traore, C. C. Hsieh, M. C. Dia, E. H. Gueye, et al. 1994. Reduced rate of disease development after HIV-2 infection as compared to HIV-1. Science 265:1587-1590. [DOI] [PubMed] [Google Scholar]
- 30.Marlink, R. G., D. Ricard, S. M'Boup, P. J. Kanki, J. L. Romet-Lemonne, I. N′Doye, K. Diop, M. A. Simpson, F. Greco, M. J. Chou, et al. 1988. Clinical, hematologic, and immunologic cross-sectional evaluation of individuals exposed to human immunodeficiency virus type-2 (HIV-2). AIDS Res. Hum. Retrovir. 4:137-148. [DOI] [PubMed] [Google Scholar]
- 31.Meloni, S. T., J. L. Sankale, D. J. Hamel, G. Eisen, A. Gueye-Ndiaye, S. Mboup, and P. J. Kanki. 2004. Molecular epidemiology of human immunodeficiency virus type 1 sub-subtype A3 in Senegal from 1988 to 2001. J. Virol. 78:12455-12461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Meylan, P. R., R. S. Kornbluth, I. Zbinden, and D. D. Richman. 1994. Influence of host cell type and V3 loop of the surface glycoprotein on susceptibility of human immunodeficiency virus type 1 to polyanion compounds. Antimicrob. Agents Chemother. 38:2910-2916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Montefiori, D. C., G. Pantaleo, L. M. Fink, J. T. Zhou, J. Y. Zhou, M. Bilska, G. D. Miralles, and A. S. Fauci. 1996. Neutralizing and infection-enhancing antibody responses to human immunodeficiency virus type 1 in long-term nonprogressors. J. Infect. Dis. 173:60-67. [DOI] [PubMed] [Google Scholar]
- 34.Moog, C., H. J. Fleury, I. Pellegrin, A. Kirn, and A. M. Aubertin. 1997. Autologous and heterologous neutralizing antibody responses following initial seroconversion in human immunodeficiency virus type 1-infected individuals. J. Virol. 71:3734-3741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Moore, J. P., and D. D. Ho. 1995. HIV-1 neutralization: the consequences of viral adaptation to growth on transformed T cells. AIDS 9(Suppl. A):S117-S136. [PubMed] [Google Scholar]
- 36.Norrgren, H., A. N. Cardoso, Z. J. da Silva, S. Andersson, F. Dias, G. Biberfeld, and A. Naucler. 1997. Increased prevalence of HIV-2 infection in hospitalized patients with severe bacterial diseases in Guinea-Bissau. Scand. J. Infect. Dis. 29:453-459. [DOI] [PubMed] [Google Scholar]
- 37.Petropoulos, C. J., N. T. Parkin, K. L. Limoli, Y. S. Lie, T. Wrin, W. Huang, H. Tian, D. Smith, G. A. Winslow, D. J. Capon, and J. M. Whitcomb. 2000. A novel phenotypic drug susceptibility assay for human immunodeficiency virus type 1. Antimicrob. Agents Chemother. 44:920-928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pilgrim, A. K., G. Pantaleo, O. J. Cohen, L. M. Fink, J. Y. Zhou, J. T. Zhou, D. P. Bolognesi, A. S. Fauci, and D. C. Montefiori. 1997. Neutralizing antibody responses to human immunodeficiency virus type 1 in primary infection and long-term-nonprogressive infection. J. Infect. Dis. 176:924-932. [DOI] [PubMed] [Google Scholar]
- 39.Popper, S. J., A. D. Sarr, A. Gueye-Ndiaye, S. Mboup, M. E. Essex, and P. J. Kanki. 2000. Low plasma human immunodeficiency virus type 2 viral load is independent of proviral load: low virus production in vivo. J. Virol. 74:1554-1557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Popper, S. J., A. D. Sarr, K. U. Travers, A. Gueye-Ndiaye, S. Mboup, M. E. Essex, and P. J. Kanki. 1999. Lower human immunodeficiency virus (HIV) type 2 viral load reflects the difference in pathogenicity of HIV-1 and HIV-2. J. Infect. Dis. 180:1116-1121. [DOI] [PubMed] [Google Scholar]
- 41.Reeves, J. D., and R. W. Doms. 2002. Human immunodeficiency virus type 2. J. Gen. Virol. 83:1253-1265. [DOI] [PubMed] [Google Scholar]
- 42.Richman, D. D., T. Wrin, S. J. Little, and C. J. Petropoulos. 2003. Rapid evolution of the neutralizing antibody response to HIV type 1 infection. Proc. Natl. Acad. Sci. USA 100:4144-4149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rybarczyk, B. J., D. Montefiori, P. R. Johnson, A. West, R. E. Johnston, and R. Swanstrom. 2004. Correlation between env V1/V2 region diversification and neutralizing antibodies during primary infection by simian immunodeficiency virus sm in rhesus macaques. J. Virol. 78:3561-3571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sankale, J. L., D. Hamel, A. Woolsey, T. Traore, T. C. Dia, A. Gueye-Ndiaye, M. Essex, T. Mboup, and P. Kanki. 2000. Molecular evolution of human immunodeficiency virus type 1 subtype A in Senegal: 1988-1997. J. Hum. Virol. 3:157-164. [PubMed] [Google Scholar]
- 45.Schulz, T. F., D. Whitby, J. G. Hoad, T. Corrah, H. Whittle, and R. A. Weiss. 1990. Biological and molecular variability of human immunodeficiency virus type 2 isolates from The Gambia. J. Virol. 64:5177-5182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Shi, Y., E. Brandin, E. Vincic, M. Jansson, A. Blaxhult, K. Gyllensten, L. Moberg, C. Brostrom, E. M. Fenyo, and J. Albert. 2005. Evolution of human immunodeficiency virus type 2 coreceptor usage, autologous neutralization, envelope sequence and glycosylation. J. Gen. Virol. 86:3385-3396. [DOI] [PubMed] [Google Scholar]
- 47.Simon, F., S. Matheron, C. Tamalet, I. Loussert-Ajaka, S. Bartczak, J. M. Pepin, C. Dhiver, E. Gamba, C. Elbim, J. A. Gastaut, et al. 1993. Cellular and plasma viral load in patients infected with HIV-2. AIDS 7:1411-1417. [DOI] [PubMed] [Google Scholar]
- 48.Tamalet, C., F. Simon, C. Dhiver, S. Matheron, P. de Micco, J. A. Gastao, and F. Brun-Vezinet. 1995. Autologous neutralizing antibodies and viral load in HIV-2-infected individuals. AIDS 9:90-91. [PubMed] [Google Scholar]
- 49.Travers, K., S. Mboup, R. Marlink, A. Gueye-Nidaye, T. Siby, I. Thior, I. Traore, A. Dieng-Sarr, J. L. Sankale, C. Mullins, et al. 1995. Natural protection against HIV-1 infection provided by HIV-2. Science 268:1612-1615. [DOI] [PubMed] [Google Scholar]
- 50.Travers, K. U., G. E. Eisen, R. G. Marlink, M. E. Essex, C. C. Hsieh, S. Mboup, and P. J. Kanki. 1998. Protection from HIV-1 infection by HIV-2. AIDS 12:224-225. [PubMed] [Google Scholar]
- 51.Wei, X., J. M. Decker, S. Wang, H. Hui, J. C. Kappes, X. Wu, J. F. Salazar-Gonzalez, M. G. Salazar, J. M. Kilby, M. S. Saag, N. L. Komarova, M. A. Nowak, B. H. Hahn, P. D. Kwong, and G. M. Shaw. 2003. Antibody neutralization and escape by HIV-1. Nature 422:307-312. [DOI] [PubMed] [Google Scholar]
- 52.Weiss, R. A., P. R. Clapham, J. N. Weber, D. Whitby, R. S. Tedder, T. O'Connor, S. Chamaret, and L. Montagnier. 1988. HIV-2 antisera cross-neutralize HIV-1. AIDS 2:95-100. [DOI] [PubMed] [Google Scholar]
- 53.Whittle, H., J. Morris, J. Todd, T. Corrah, S. Sabally, J. Bangali, P. T. Ngom, M. Rolfe, and A. Wilkins. 1994. HIV-2-infected patients survive longer than HIV-1-infected patients. AIDS 8:1617-1620. [DOI] [PubMed] [Google Scholar]