Abstract
Glucocorticoids (GC) play an important role in the treatment of inflammatory diseases like asthma. However, in selected patients a relative resistance to GC has been reported. Recently, it has been suggested that GC sensitivity of peripheral blood leucocytes may be regulated in a dynamic fashion during exercise, in association with activation of the hypothalamic–pituitary–adrenal (HPA) axis. The aim of the present study was to explore changes in the GC sensitivity of cytokine production by leucocytes following strenuous exercise by well trained oarsmen. These changes were studied using lipopolysaccharide (LPS)-induced and anti-CD2/anti-CD28 MoAb-stimulated cytokine release in whole blood and its modulation by dexamethasone. Following exercise, significant decreases in LPS-induced release of IL-6, tumour necrosis factor-alpha (TNF-α) and IL-10 and anti-CD2/anti-CD28 MoAb-stimulated secretion of interferon-gamma (IFN-γ) were observed. In addition, the inhibitory effect of dexamethasone on both IL-6 and TNF-α secretion was significantly reduced following exercise, whereas that on IL-10 and IFN-γ release was not affected. These exercise-induced changes were accompanied by activation of the HPA axis, as indicated by an increase in circulating adrenocorticotropic hormone (ACTH) levels immediately following exercise. The results from the present study suggest that GC sensitivity of whole blood cytokine release can be regulated in a dynamic fashion and that this can be assessed using an ex vivo stimulation assay. Moreover, since dexamethasone responsiveness of anti-CD2/anti-CD28 MoAb-induced IFN-γ secretion in whole blood is not affected by exercise, it may suggest that exercise differentially affects monocytes and lymphocytes. The dynamic regulation of steroid responsiveness of leucocytes, as observed in the present study, could have important consequences for the effectiveness of GC treatment in inflammatory diseases.
Keywords: cytokines, whole blood, exercise, dexamethasone, glucocorticoids
INTRODUCTION
Glucocorticoid hormones display potent immunosuppressive and anti-inflammatory activity [1], and therefore synthetic glucocorticoids (GC) have been found to be particularly useful in the treatment of various inflammatory diseases, such as asthma, rheumatoid arthritis or systemic lupus erythematosus [1]. The anti-inflammatory actions of GC affect many cell types through a receptor-mediated process, and include profound effects on leucocyte distribution that are mediated in part by inhibition of production and release of many proinflammatory mediators, including cytokines [2]. In addition to these anti-inflammatory effects, GC may also stimulate endogenous anti-inflammatory mechanisms by, for example, stimulating the production of proteinase inhibitors [3] and anti-inflammatory cytokines [4].
However, GC are not always successful in anti-inflammatory therapy. In a minority of patients with chronic inflammatory or immune diseases, an acquired form of GC resistance is reported [5, 6]. In addition to therapeutic unresponsiveness, in vitro effects at the cellular level have been demonstrated. Peripheral blood mononuclear cells (PBMC) of GC-resistant asthmatic patients [7, 8] or patients with Alzheimer's disease [9] are less responsive to dexamethasone inhibition of phytohaemagglutinin (PHA)-induced proliferation. Moreover, the binding affinity of the GC receptor on PBMC in steroid-resistant asthma patients was reported to be markedly reduced [10].
In addition to interindividual differences in GC responsiveness, also intra-individual differences have been reported, suggesting a dynamic regulation of GC sensitivity. Recent in vitro [11] and in vivo [12] data show that GC may down-regulate the expression of their own receptor. In addition, it has been demonstrated that strenuous exercise [13] or academical stress [14] induce a transient decrease in GC sensitivity of leucocytes ex vivo. In both situations, activation of the hypothalamic–pituitary–adrenal (HPA) axis, with elevated levels of cortisol, has been demonstrated. These findings suggest an association between stimulation of the HPA axis and temporal decreases of leucocyte GC sensitivity. One could postulate that other conditions that are known to stimulate HPA activity, such as mycobacterial [15] or viral infections [16], may also affect GC sensitivity. Such decreased responsiveness could have therapeutic consequences when occurring in patients with inflammatory diseases.
It has recently been shown that the production of various cytokines is differentially affected by GC, both in vivo and in vitro [17, 18]. Therefore, the aim of the present study was to investigate the effect of strenuous exercise on the regulation of cytokine production in whole blood ex vivo by dexamethasone. To that end we analysed the production of various lipopolysaccharide (LPS)-induced and anti-CD2/anti-CD28 MoAb-induced pro- and anti-inflammatory cytokines and their modulation by dexamethasone before and after a strenuous exercise challenge in competitive oarsmen.
MATERIALS AND METHODS
Exercise challenge
Nine healthy, well trained competitive oarsmen (six males and three females, 18–25 years old), free of asthmatic and endocrine disorders, participated in the study that was approved by the Medical Ethical Board of Leiden University Medical Centre. Competitive oarsmen obeyed strict rules of their rowing club: no alcohol, no drugs, 8 h of sleep every night. During their visit to our laboratory the oarsmen performed a routine rowing ergometer test (Concept II indoor rower; Morrisville, VT), which is a monthly recurring part of their training programme. During the exercise challenge, on-line measurements of exhaled O2 and CO2 were made, in order to calculate O2 uptake (VO2) and CO2 production (VCO2) (Oxycon Record; Jaeger, Würzburg, Germany). Moreover, heart rate and cardiac function were monitored by electrocardiography. The test consisted of 3-min exercise periods at constant power output, alternated with 0.5-min rest periods, until exhaustion occurred. After every 0.5-min rest period, the power output was increased stepwise by 40 W (males) or 30 W (females) from baseline (males 240 W; females 150 W). The total exercise challenge lasted between 15 and 20 min, depending on personal fitness. The VO2,max was expressed as percentage of predicted to assess the extent of strenuous exercise per individual. An i.v. catheter was placed in one forearm vein 40 min before the start of the exercise challenge. Immediately, blood samples were drawn for the whole blood stimulation assay, the assessment of basal adrenocorticotropic hormone (ACTH) and cortisol plasma levels and total leucocyte count and differentiation. Blood was also collected immediately after cessation of the exercise challenge for determination of ACTH and cortisol plasma levels. Finally, blood was collected 20 min after cessation of the exercise challenge, for a second whole blood stimulation assay and total leucocyte count and differentiation.
Hormone assays
Plasma cortisol and ACTH levels were measured in blood that was collected in iced ethylenediamine tetraacetate (EDTA) tubes (Becton Dickinson, Rutherford, NJ) using a fluorometric assay (cortisol; Abbott, TDx, Abbott Park, IL) and a radioimmunoassay (ACTH; Nichols Diagnostics, San Diego, CA).
Leucocyte differentiation
Venous blood collected in EDTA tubes (Becton Dickinson) was used to assess leucocyte counts and differential cell counts by automated blood count analysis (Technicon H1; Technicon, Tarrytown, NY).
Whole blood stimulation assay and inhibition by dexamethasone
Venous blood was collected in heparinized tubes. Aliquots of 0.5 ml of dexamethasone-21-phosphate (Sigma Chemical Co., St Louis, MO; final concentration 1, 10, 100 nm in pyrogen-free saline) or 0.5 ml of pyrogen-free saline were added to 4.5 ml whole blood, and incubated for 10 min at room temperature. Blood was stimulated with LPS (final concentration 3 ng/ml; LPS from Salmonella typhosa, Sigma) or a mixture of MoAbs to CD2 and CD28 (anti-CD2 MoAbs CLB-T11.1/1 and CLB-T11.1/2, final dilution 1:1000 of murine ascites; anti-CD28 MoAb CLB-CD28/1, 1 μg/ml; CLB, Amsterdam, The Netherlands) dissolved in sterile pyrogen-free saline. Twenty-five microlitres of LPS, anti-CD2/anti-CD28 MoAbs or saline were added to 225 μl whole blood containing dexamethasone in 96-well flat-bottomed plates (Greiner, Alphen a/d Rijn, The Netherlands). After a 24-h incubation period in a humidified atmosphere at 37°C in 5% CO2, the plates were centrifuged for 10 min at 750 g. Plasma samples were collected and stored at −80°C until further analysis.
Cytokine assays
Levels of IL-6, tumour necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ), IL-4 and IL-10 in plasma, harvested from the whole blood assays, were measured by ELISA (CLB) according to the manufacturer's instructions. The lower limits of detection were: 10 pg/ml (IL-6), 10 pg/ml (TNF-α), 20 pg/ml (IFN-γ), 1.5 pg/ml (IL-4) and 5 pg/ml (IL-10). Briefly, 96-well plates were coated with a murine MoAb to IL-6, TNF-α, IFN-γ, IL-4 or IL-10 and subsequently incubated with the sample. Next, the plates were incubated with a biotinylated MoAb to these cytokines. Finally, peroxidase-conjugated streptavidin was added, and 3,3′,5,5′-tetramethylbenzidine (Sigma) was used as a substrate.
Statistic analysis
Cytokine production was expressed as the cytokine level per ml plasma or as the amount of cytokine per monocyte or lymphocyte. Dexamethasone sensitivity was expressed as percentage inhibition of stimulated cytokine production. Differences in the time course and in dexamethasone dose–response curves of LPS-induced or anti-CD2/anti-CD28 MoAb-stimulated whole blood were determined by multiple variate analysis of variance (manova). Significant manova effects were explored using paired Student's t-tests. Likewise, changes in cell counts or plasma ACTH and cortisol concentrations were calculated by Student's paired t-tests. Non-parametric testing (Wilcoxon, matched pairs) was used for data that were not normally distributed. Data are presented as mean and s.e.m. P < 0.05 was considered to be statistically significant.
RESULTS
Exercise challenge
The exercise challenge in females had a duration of 15.5 ± 0.0 min, all reaching the maximal power output of 270 W. Then VO2,max was 138.4 ± 12.3% predicted. The exercise challenge in males had a duration of 16.0 ± 0.92 min and the maximal power output was between 360 W and 440 W, with VO2,max of 135.8 ± 6.6% predicted.
Exercise-induced changes in endogenous hormone levels
Immediately after cessation of the exercise challenge, circulating ACTH levels were significantly increased compared with pre-exercise levels, but cortisol levels were not affected at this time (Fig. 1).
Fig. 1.
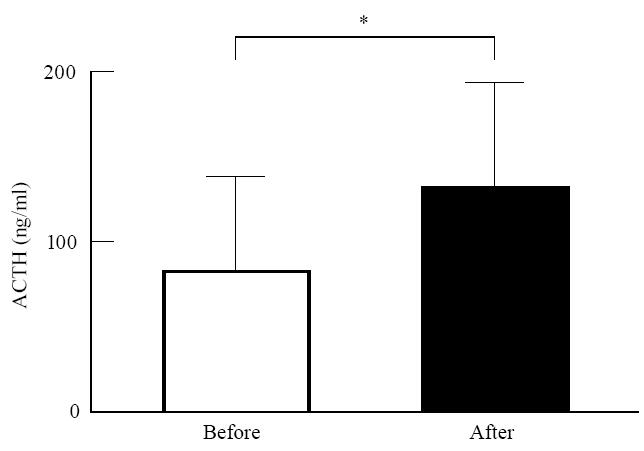
Effect of exercise on circulating adrenocorticotropic hormone (ACTH) levels. Plasma ACTH levels were determined in blood samples obtained 40 min before and immediately after exercise. *P < 0.01versus before exercise.
Changes in leucocyte numbers after exercise
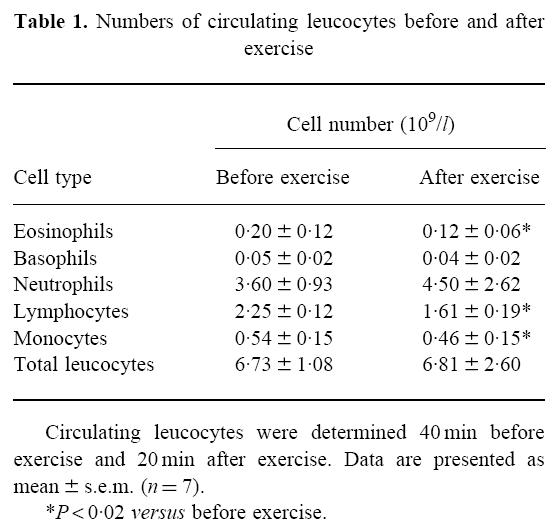
Twenty minutes following cessation of the exercise challenge, there was no change in the total number of circulating leucocytes, neutrophils or basophils, whereas the number of circulating monocytes, lymphocytes and eosinophils was significantly decreased (Table 1).
Table 1.
Numbers of circulating leucocytes before and after exercise
Effect of exercise on LPS-induced cytokine production in whole blood
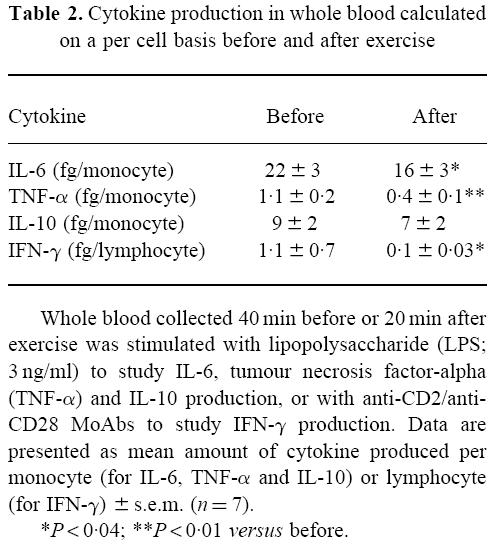
In pilot experiments, we selected a concentration of 3 ng/ml LPS to study cytokine release in whole blood because at this concentration a submaximal effect was observed. Following exercise, a significant decrease in LPS-induced IL-6, TNF-α and IL-10 release was observed (Fig. 2a–c). The main source of IL-6, TNF-α and IL-10 in LPS-stimulated whole blood is the monocyte. Since the number of monocytes was decreased following exercise, we calculated the cytokine production per monocyte. This demonstrated that, also when expressed per monocyte, the decrease in IL-6 and TNF-α release was still significant (Table 2). In contrast, the amount of IL-10 released per monocyte was not decreased following exercise.
Fig. 2.
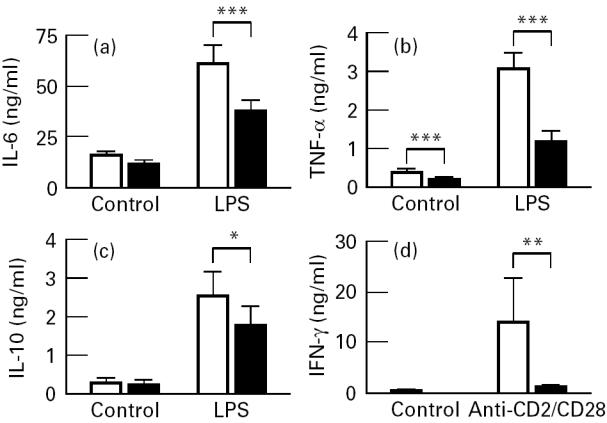
Effect of exercise on lipopolysaccharide (LPS)-induced IL-6 (a), tumour necrosis factor-alpha (TNF-α) (b) and IL-10 synthesis (c); and on anti-CD2/anti-CD28-stimulated IFN-γ release (d). Blood was obtained 40 min before (□) and 20 min after the ending of the exercise (▪) and stimulated with 3 ng/ml LPS or MoAbs against CD2 and CD28 or saline during 24 h. Since the IFN-γ levels were not normally distributed, non-parametric testing (Wilcoxon, matched pairs) was used to analyse statistical significance. *P < 0.05; **P < 0.04; ***P < 0.01 versus before exercise.
Table 2.
Cytokine production in whole blood calculated on a per cell basis before and after exercise
Effect of exercise on anti-CD2/anti-CD28 MoAb-induced cytokine production in whole blood
In addition to LPS, we also employed a lymphocyte-specific stimulus using MoAbs against CD2 and CD28 to study cytokine production in whole blood. In pilot experiments we observed that, compared with LPS, anti-CD2/anti-CD28 stimulation resulted in low levels of IL-6 and TNF-α release, but higher levels of IFN-γ. IL-4 release was detected in only one subject, and therefore not further analysed. Anti-CD2/anti-CD28 stimulation did not increase IL-10 release above the levels observed in unstimulated blood following exercise (Fig. 2c). A significant decrease in IFN-γ production in stimulated blood was observed after exercise (Fig. 2d). When analysing IFN-γ production on a per cell basis, a significant decrease per lymphocyte was found following exercise (Table 2).
Effect of exercise on the modulation of LPS-induced cytokine production in whole blood by dexamethasone
LPS-induced IL-6, TNF-α and IL-10 production was inhibited by dexamethasone in a dose-dependent manner. Following exercise, the effect of dexamethasone on LPS-induced IL-6 and TNF-α release was significantly decreased (manova, P < 0.04 and P < 0.01 versus before exercise; Fig. 3a,b). No changes were detected in the effect of dexamethasone on IL-10 synthesis after exercise (manova, P > 0.28 versus before exercise; Fig. 3c). At 1 nm of dexamethasone, a significant decrease in inhibition of IL-6 production was observed (Fig. 3a). At higher concentrations of dexamethasone, there was no difference in the effect of dexamethasone on IL-6 release before and after exercise. In contrast, the post-exercise dexamethasone sensitivity of LPS-induced TNF-α production was significantly reduced at all three concentrations of dexamethasone tested (Fig. 3b).
Fig. 3.
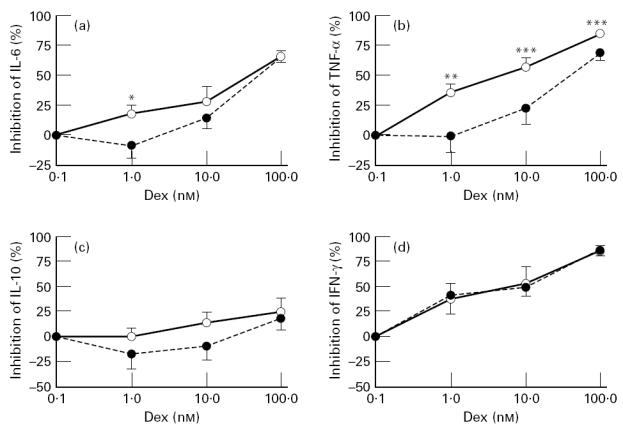
Effect of exercise on dexamethasone sensitivity of lipopolysaccharide (LPS)-induced IL-6 (a), tumour necrosis factor-alpha (TNF-α) (b) and IL-10 synthesis (c) and of anti-CD2/anti-CD28-stimulated IFN-γ release (d). Blood was drawn 40 min before (○) and 20 min after the ending of the exercise (•) and incubated for 24 h with 3 ng/ml LPS or MoAbs against CD2 and CD28 in the presence or absence of various concentrations of dexamethasone (1, 10 or 100 nm). *P < 0.05; **P < 0.04; ***P < 0.02; ****P < 0.01 versus before exercise.
Effect of exercise on the modulation of anti-CD2/anti-CD28-induced cytokine production in whole blood by dexamethasone
IFN-γ release in anti-CD2/anti-CD28 MoAb-stimulated blood was inhibited by dexamethasone in a dose-dependent fashion. However, the inhibitory effect of dexamethasone on IFN-γ release in whole blood was not changed following exercise (manova, P > 0.96 versus before exercise; Fig. 3d). In contrast to pre-exercise anti-CD2/anti-CD28 MoAb-stimulated IL-10 levels, post-exercise stimulated IL-10 levels were not significantly increased from baseline. Therefore changes in IL-10 dexamethasone sensitivity were not analysed.
DISCUSSION
The results from the present study show that strenuous exercise in trained oarsmen results in a decreased release of IL-6, TNF-α, IL-10 and IFN-γ in whole blood stimulated ex vivo. These exercise-induced changes were accompanied by significant decreases in the number of circulating monocytes, lymphocytes and eosinophils. However, this decrease in cell numbers appeared not to account fully for the changes in IL-6, TNF-α or IFN-γ production, but might explain the decreased IL-10 production. Second, a post-exercise decrease in the effect of dexamethasone on LPS-induced IL-6 and TNF-α production was observed. In contrast, the sensitivity of LPS-induced IL-10 and anti-CD2/anti-CD28 MoAb-induced IFN-γ production to inhibition by dexamethasone was not affected following exercise. These exercise-induced changes were accompanied by activation of the HPA axis, as indicated by the increase in circulating ACTH levels immediately following exercise. Previously, it has been shown that an increase in ACTH levels immediately following exercise is associated with a maximal increase in cortisol 20 min after exercise [13]. Taken together, these findings support the existence of a transient inducible form of GC resistance.
In the present study we observed reduced cytokine production in whole blood stimulated either with LPS or with MoAbs against CD2 and CD28 following exercise, accompanied by activation of the HPA axis. A similar association between activation of the HPA axis and decreased PHA-induced proliferation of PBMC or reduced LPS- or PHA-stimulated cytokine release in whole blood ex vivo has been reported during morning peaks of cortisol circadian rhythm [19], academical stress [14], sepsis [20, 21] and small cell lung cancer [22].
We considered several mechanisms that may explain decreased cytokine release as observed in the present study. First, a decrease in cytokine production might be the direct result of increased levels of endogenous cortisol. Second, changes in specific leucocyte subsets following exercise may explain our observations, since in the present study we observed a decrease in the number of circulating monocytes and lymphocytes post-exercise. However, after correction for the number of monocytes (for IL-6, TNF-α and IL-10 release [23, 24]) or lymphocytes (for IFN-γ release [25]), the release of IL-6, TNF-α and IFN-γ was still significantly decreased following exercise, whereas IL-10 release was not. Third, in septic patients decreased TNF-α and IL-6 production was accompanied by decreased stability of mRNA encoding TNF-α and IL-6 resulting in lower mRNA levels [20]. It is possible that exercise also results in decreased stability of mRNA encoding selected cytokines in circulating leucocytes, and subsequent lower production of these cytokines. Another possibility is that exercise increases the production of compounds such as IL-4, IL-10 or prostaglandin E2 that may suppress cytokine production [26], but this possibility is not supported by our own or other studies. Finally, decreased TNF-α secretion following exercise might be explained by exercise-induced inhibition of processing of membrane-bound precursor TNF-α to the secreted mature protein [27, 28].
In addition to effects on stimulated cytokine production in whole blood, we also observed marked effects of exercise on the sensitivity of cytokine production to dexamethasone. Similar results regarding steroid responsiveness of LPS-induced IL-6 production in whole blood have been reported by DeRijk et al. using treadmill exercise in moderately trained subjects [13]. In addition, Sauer et al. reported a decreased cortisol sensitivity in vitro of PHA-induced PBMC proliferation in subjects undergoing academical stress [14]. We extended these observations by demonstrating not only a decreased sensitivity for dexamethasone of LPS-induced IL-6 secretion, but also of TNF-α secretion, whereas the sensitivity of LPS-induced IL-10 secretion was not affected following strenuous exercise in well trained oarsmen. In addition, we also used the monocyte-independent T cell-specific stimulus anti-CD2/anti-CD28. Using this stimulus, IFN-γ secretion was markedly reduced following exercise, whereas its sensitivity to dexamethasone was not affected. This suggests that exercise differentially affects monocytes and T cells. Alternatively, the CD2/CD28 stimulatory route of IFN-γ production itself may be resistant to exercise-induced changes in dexamethasone sensitivity [29]. Since we have not used alternative routes for T cell stimulation, we can not discriminate between these alternative explanations.
Several mechanisms may explain transient changes in GC sensitivity as observed in the present study. First, GC may influence the expression of the glucocorticoid receptor (GR) as demonstrated both in vitro [11] and in vivo [12]. Endogenous GC may thus contribute to GC sensitivity, since low circulating cortisol levels in neonates are associated with increased dexamethasone sensitivity of PBMC proliferation ex vivo [30]. Conversely, increased levels of endogenous GC may have contributed to the decreased GC sensitivity following exercise, as observed in our study. A second mechanism that may be involved in regulation of GC sensitivity is the modulatory activity of cytokines such as IL-2 and IL-4, that in vitro have been shown to reduce GR-binding activity and steroid responsiveness of PBMC [31]. Finally, recent studies suggest that changes in GC sensitivity may be the direct result of differential expression of α- and β-isoforms of the GR. Since the β-isoform is able to inhibit hormone-induced, GRα-mediated stimulation of gene expression, it may function as a dominant negative inhibitor of GRα activity [32, 33]. Whether such mechanisms underlie the changes in GC sensitivity following exercise remains to be determined.
We observed very rapid changes in GR-sensitivity of monocytes that are probably not explained by altered GR expression, since this would require de novo mRNA and protein synthesis. Rapid changes in GC sensitivity may be the result of an interaction of the activated GR with transcription factors such as AP-1 and NF-κB [17, 33]. An alternative explanation for the rapid changes is that exercise results in differential migration of subsets of monocytes from the circulating pool, resulting in depletion of the subset most sensitive to GC from the circulation. To our knowledge, subsets of monocytes that differ in their sensitivity to GC have not been described.
Our results may have clinical implications, since a post-exercise decrease in the number of circulating monocytes and lymphocytes, together with a decreased cytokine release, are indicative of the existence of temporal immunosuppression. This is in accordance with observations on decreased immune responses in overtrained athletes accompanied by an increased susceptibility to infections [34, 35]. Our findings indicate that there are no clear changes in monocyte-derived IL-10 production and GC sensitivity of IL-10 release. In contrast, the sensitivity to GC of the proinflammatory cytokines IL-6 and TNF-α is decreased following exercise. Whether this implies that GC treatment in vivo differentially affects the balance between pro- and anti-inflammatory cytokines before and after exercise (or other conditions that result in HPA axis activation) remains to be determined. In view of the important role of GC in the treatment of inflammatory disorders, mechanisms leading to a change in GC sensitivity as described in the present study may contribute to effectiveness of GC treatment.
Acknowledgments
The authors thank J. J. Brahim for assistance during the exercise challenges. This study was supported in part by the Netherlands Asthma Foundation (grant 93.17).
References
- 1.Cohen JJ, Scheimer HN, Claman HN, Oronsky AL. San Diego: Academic Press; 1989. Anti-inflammatory steroid action. Basic and clinical aspects; pp. 111–31. [Google Scholar]
- 2.Schwiebert LA, Beck LA, Stellato C, Bickel CA, Bochner BS, Schleimer RP. Glucocorticosteroid inhibition of cytokine production: relevance to antiallergic actions. J Allergy Clin Immunol. 1996;97:143–52. doi: 10.1016/s0091-6749(96)80214-4. [DOI] [PubMed] [Google Scholar]
- 3.Abbinante-Nissen JM, Simpson LG, Leikauf GD. Corticosteroids increase secretory leukocyte protease inhibitor transcript levels in airway epithelial cells. Am J Physiol. 1995;12:L601–L606. doi: 10.1152/ajplung.1995.268.4.L601. [DOI] [PubMed] [Google Scholar]
- 4.van Furth AM, Seijmonsbergen EM, Langermans JA, van der Meide PH, van Furth R. Effect of xanthine derivates and dexamethasone on Streptococcus pneumoniae-stimulated production of tumor-necrosis factor alpha, interleukin-1 beta (IL-1 beta), and IL-10 by human leukocytes. Clin Diagn Lab Immunol. 1995;2:689–92. doi: 10.1128/cdli.2.6.689-692.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cypcar D, Busse WW. Steroid-resistant asthma. J Allergy Clin Immunol. 1993;92:362–72. doi: 10.1016/0091-6749(93)90114-u. [DOI] [PubMed] [Google Scholar]
- 6.Kirkham B, Corkill MM, Davison SC, Panayi GS. Response to glucocorticoid treatment in rheumatoid arthritis: in vitro cell-mediated immune assay predicts in vivo response. J Rheumatol. 1991;18:1130. [PubMed] [Google Scholar]
- 7.Corrigan CJ, Brown PH, Barnes NC, et al. Glucocorticoid resistance in chronic asthma: glucocorticoid pharmacokinetics, glucocorticoid receptor characteristics, and inhibition of peripheral blood T cell proliferation by glucocorticoids in vitro. Am Rev Res Dis. 1991;144:101–25. doi: 10.1164/ajrccm/144.5.1016. [DOI] [PubMed] [Google Scholar]
- 8.Spahn JD, Landwehr LP, Nimmagadda S, Susrs W, Leung DYM, Szefler SJ. Effects of glucocorticoids on lymphocyte activation in patients with steroid-sensitive and steroid-resistant asthma. J Allergy Clin Immunol. 1996;98:1073–9. doi: 10.1016/s0091-6749(96)80194-1. [DOI] [PubMed] [Google Scholar]
- 9.Nijhuis E, Hinloopen B, Van Duijn C, Hofman A, Rozing J, Nagelkerken L. Decreased sensitivity to dexamethasone in lymphocytes from patients with Alzheimer's disease. Clin Immunol Immunopathol. 1994;73:45–52. doi: 10.1006/clin.1994.1168. [DOI] [PubMed] [Google Scholar]
- 10.Adcock IM, Lane SJ, Brown CR, Peters MJ, Lee TH, Barnes PJ. Differences in binding of glucocorticoid receptor to DNA in steroid-resistant asthma. J Immunol. 1995;154:3500–5. [PubMed] [Google Scholar]
- 11.Rosewicz S, McDonald AR, Maddux BA, Goldfine ID, Miesfeld RL, Logsdon CD. Mechanism of glucocorticoid receptor down-regulation by glucocorticoids. J Biol Chem. 1988;263:2581–4. [PubMed] [Google Scholar]
- 12.Korn SH, Wouters EFM, Wesseling G, Arends J, Thunnissen BJM. In vitro and in vivo modulation of α- and β-glucocorticoidreceptor mRNA in human bronchial epithelium. Am J Resp Crit Care Med. 1997;155:1117–22. doi: 10.1164/ajrccm.155.3.9116996. [DOI] [PubMed] [Google Scholar]
- 13.DeRijk RH, Petrides J, Deuster P, Gold PW, Sternberg EM. Changes in corticosteroid sensitivity of peripheral blood lymphocytes after strenuous exercise in humans. J Clin Endo Metabol. 1996;81:228–35. doi: 10.1210/jcem.81.1.8550757. [DOI] [PubMed] [Google Scholar]
- 14.Sauer J, Polack E, Wikinski S, Holsboer F, Stalla GK, Arzt E. The glucocorticoid sensitivity of lymphocytes changes according to the activity of the hypothalamic–pituitary–adrenocortical system. Psychoneuroendocrinol. 1995;20:269–80. doi: 10.1016/0306-4530(94)00058-i. [DOI] [PubMed] [Google Scholar]
- 15.Brown DH, Sheridan JF, Pearl D, Zwillig BS. Regulation of mycobacterial growth by the hypothalamus–pituitary–adrenal axis: differential responses of Mycobacterium bovis BCC-resistant and -susceptible mice. Infect Immun. 1993;61:4793–800. doi: 10.1128/iai.61.11.4793-4800.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dunn AJ, Vickers SL. Neurochemical and neuroendocrine responses to Newcastle disease virus administration in mice. Brain Res. 1994;645:103–12. doi: 10.1016/0006-8993(94)91643-8. [DOI] [PubMed] [Google Scholar]
- 17.Barnes P. Mechanisms of action of glucocorticoids in asthma. Am J Resp Crit Care Med. 1996;154:S21–S27. doi: 10.1164/ajrccm/154.2_Pt_2.S21. [DOI] [PubMed] [Google Scholar]
- 18.Brattsand R, Linden M. Cytokine modulation by glucocorticoids: mechanisms and actions in cellular studies. Aliment Pharmacol. 1996;10:81–90. doi: 10.1046/j.1365-2036.1996.22164025.x. [DOI] [PubMed] [Google Scholar]
- 19.DeRijk R, Michelson D, Karp B, et al. Exercise and circadian rhythm-induced variations in plasma cortisol differentially regulate interleukin-1β (IL-1β), IL-6 and tumor necrosis factor-α (TNFα) productions in humans: high sensitivity of TNFα and resistance of IL-6. J Clin Endo Metabol. 1997;82:2182–91. doi: 10.1210/jcem.82.7.4041. [DOI] [PubMed] [Google Scholar]
- 20.Ertel W, Kremer J, Kennedy J, et al. Downregulation of proinflammatory cytokine release in whole blood from septic patients. Blood. 1995;85:1341–7. [PubMed] [Google Scholar]
- 21.Kremer J, Jarrar D, Steckholzer U, Ertel W. Interleukin-1, -6 and tumor necrosis factor-α release is down-regulated in whole blood from septic patients. Acta Haematol. 1996;95:268–73. doi: 10.1159/000203895. [DOI] [PubMed] [Google Scholar]
- 22.Fischer JR, Schindel M, Stein N, et al. Selective suppression of cytokine secretion in patients with small-cell lung cancer. Anal Oncol. 1995;6:921–6. doi: 10.1093/oxfordjournals.annonc.a059360. [DOI] [PubMed] [Google Scholar]
- 23.Heinrich PC, Castell JV, Andus T. Interleukin-6 and the acute phase response. Biochem J. 1990;265:621–36. doi: 10.1042/bj2650621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dinarello CA. Interleukin-1 and interleukin-1 antagonism. Blood. 1991;77:1627–52. [PubMed] [Google Scholar]
- 25.Van der Pouw-Kraan T, Van Kooten C, Rensink I, Aarden L. Interleukin (IL)-4 production by human T cells: differential regulation of IL-4 vs. IL-2 production. Eur J Immunol. 1992;22:1237–41. doi: 10.1002/eji.1830220519. [DOI] [PubMed] [Google Scholar]
- 26.Grbic JT, Mannick JA, Gough DB, Rodrick ML. The role of prostaglandin E2 in immune suppression following injury. Ann Surg. 1991;214:253–63. doi: 10.1097/00000658-199109000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gearing AJH, Beckett P, Christodoulou M, et al. Processing of tumour necrosis factor-α precursor by metalloproteinases. Nature. 1994;370:555–7. doi: 10.1038/370555a0. [DOI] [PubMed] [Google Scholar]
- 28.McGeehan GM, Becherer JD, Bast RC, Jr, et al. Regulation of tumour necrosis factor-α processing by metalloproteinase inhibitor. Nature. 1994;370:558–61. doi: 10.1038/370558a0. [DOI] [PubMed] [Google Scholar]
- 29.Nijhuis EWP, Hinloopen B, Odding J, Nagelkerken L. Abrogation of the suppressive effects of dexamethasone by PKC activation or CD28 triggering. Cell Immunol. 1994;156:438–47. doi: 10.1006/cimm.1994.1188. [DOI] [PubMed] [Google Scholar]
- 30.Kavelaars A, Zijlstra J, Bakker JM, et al. Increased dexamethasone sensitivity of neonatal leukocytes: different mechanisms of glucocorticoid inhibition of T cell proliferation in adult and neonatal cells. Eur J Immunol. 1995;25:1346–51. doi: 10.1002/eji.1830250532. [DOI] [PubMed] [Google Scholar]
- 31.Kam JC, Szefler SJ, Surs W, Sher ER, Leung DYM. Combination IL-2 and IL-4 reduces glucocorticoid receptor-binding affinity and T cell response to glucocorticoids. J Immunol. 1993;151:346–6. [PubMed] [Google Scholar]
- 32.Bamberger CM, Bamberger A, de Castro M, Chrousos GP. Glucocorticoid receptor β, a potential endogenous inhibitor of glucocorticoid action in humans. J Clin Invest. 1995;95:2435–41. doi: 10.1172/JCI117943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bamberger CM, Schulte HM, Chrousos GP. Molecular determinants of glucocorticoid receptor function and tissue sensitivity to glucocorticoids. Endoc Rev. 1996;17:245–61. doi: 10.1210/edrv-17-3-245. [DOI] [PubMed] [Google Scholar]
- 34.Espersen GT, Elbaek A, Ernst E, et al. Effect of physical exercise on cytokines and lymphocyte subpopulations in human peripheral blood. APMIS. 1990;98:395–400. doi: 10.1111/j.1699-0463.1990.tb01049.x. [DOI] [PubMed] [Google Scholar]
- 35.Field CJ, Gougeon R, Marliss EB. Circulating mononuclear cell numbers and function during intense exercise and recovery. J Appl Physiol. 1991;71:1089–97. doi: 10.1152/jappl.1991.71.3.1089. [DOI] [PubMed] [Google Scholar]


