INTRODUCTION
The control and treatment of infectious diseases is increasingly complicated by the development of antibiotic-resistant organisms, the emergence and rapid dissemination of new pathogens, and the growing incidence of opportunistic infections. Identification and commercialization of new anti-microbial agents has always been a time-consuming and expensive process, and development of new strategies to control disease are therefore clearly warranted.
The immune system has evolved to help combat various types of infections. As most pathogens enter through mucosal surfaces, the local immune defence may be of particular importance and evolutionary pressure has led to the development of a specialized immunoglobulin class in secretions—IgA. Patients with primary immunodeficiencies involving IgA are known to be highly susceptible to respiratory and gastrointestinal (GI) tract infections (for review see [1]), supporting the notion of a crucial role for antibodies in the mucosal defence.
Infection-prone IgA-deficient patients are currently treated with human IgG given systemically [2]. However, a proportion of these patients still suffer from mucosal infections which necessitates development of additional forms of therapy, and human IgG in high doses has previously in fact been given orally, without adverse effects, to healthy volunteers [3] and to patients suffering from a variety of GI infections (for references see [4]). However, from a theoretical point of view human IgG is expected to be less effective than human IgA due to its comparatively low proteolytic stability, and it may also, due to its interaction with complement and phagocytosing cells, provoke an inflammatory reaction.
Human breast milk, which contains secretory IgA, is known to protect against diarrhoea in infants. Oral administration of purified human immunoglobulin has previously also been suggested to exhibit a prophylactic effect against development of necrotizing enterocolitis in prematurely born children [5] and a therapeutic effect in Campylobacter jejuni [6] and Clostridium difficile [7] induced diarrhoea in immunodeficient patients as well as chronic diarrhoea of unknown aetiology in normal infants [8]. However, the titre against most human pathogens is rather low in the IgA-containing gammaglobulin preparations, and vaccination of volunteers for preparation of hyperimmune products is not feasible. Furthermore, there is always a risk of transmission of infectious agents when using human plasma as a starting material and since the cost for these preparations is high, alternative sources of antibodies must be sought.
The common dairy cow may be one such alternative, as bovine antibodies are actively transported from plasma to milk in cows and are present in high concentrations in colostrum. The antibodies protect the calves from GI infections during the neonatal period and are of major importance for their survival. Bovine immunoglobulin preparations against rotavirus and Escherichia coli have previously been shown to be effective as prophylaxis against infections and have been commercially available for use in farm animals for decades. During the past few years, bovine immunoglobulin preparations have also been shown to protect against natural infection or challenge with a variety of different microorganisms in man. The effect of therapeutically administered bovine antibodies has not been as well documented, but successful intervention has been demonstrated in Helicobacter pylori, rotavirus and cryptosporidial infections (for references see [4,9]). Interaction between bovine antibodies and the human immune system is limited [10] and the preparation can be given without risk of serious adverse events.
The titre of antibodies against many human pathogens is low in colostrum from non-immunized animals [4], and although a therapeutic effect has been suggested by oral administration of high doses of non-specific bovine immunoglobulins to patients with chronic diarrhoea [11–14], it is likely that a future product will have to be derived from immunized animals [10]. In this review, we will summarize published data on the use of bovine immunoglobulin-containing preparations for prophylaxis and therapy against selected mucosal infections in man and suggest directions for its future use.
ADMINISTRATION OF ANTIBODIES AGAINST ORAL PATHOGENS
During the past century, a markedly raised incidence of dental caries has been noted. This has been brought about by the increased standard of living, which in turn has resulted in a diet rich in sucrose. The development of caries is thought to result from an interplay between the host, the diet and the endogenous microflora. The main bacteriological aetiological agent in dental caries is Streptococcus mutans, and several lines of evidence show that the presence of this organism is highly correlated to the incidence of caries.
Although the incidence has been reduced due to prophylactic measures during the past decades, at least in the developed countries, there exist groups of patients where caries remains a major clinical problem. Thus, individuals with xerostomia due to side-effects of certain pharmaceutical products, systemic diseases with engagement of the salivary glands (such as Sjögren's syndrome), the elderly population and patients who have received heavy local irradiation due to tumours in the head and neck region, may all suffer from severe caries as a consequence of the reduction of salivary flow.
Several stages of S. mutans infections are amenable to immune intervention and both active and passive immunization have been used with varying degrees of success during the past two decades. The initial attachment of the bacteria occurs via surface adhesins, and the accumulation of bacteria in the dental plaque is then enhanced by the synthesis of extracellular glucans from sucrose. Salivary antibodies may interrupt this process by aggregation of the bacteria prior to colonization, blocking of surface receptors necessary for colonization, or by inactivation of enzymes necessary for metabolically important functions.
Experimental vaccination against S. mutans has been shown to result in a high level of specific salivary antibodies, which also correlated with prophylaxis against caries development. Numerous attempts have been made to improve the immune response against the bacteria using different adjuvants and various routes of vaccination. However, progress has been hampered by the appearance of antibodies which are cross-reactive with heart tissue in vaccinated animals [15]. Thus, although still possible using selected purified antigens, attempts in this direction have not been vigorously pursued.
Recently, bovine antibodies against S. mutans were shown to inhibit incorporation of glucose by the bacteria in vitro already at low concentrations, and higher concentrations diminished the activity of the glucosyltransferase (GTF) [16]. Passive in vivo immunization against S. mutans has also been tried using oral administration of antibodies from animals vaccinated with whole bacteria or purified components such as the GTF, streptococcal antigen I/II (SA I/II) and serotype carbohydrate. Thus, bovine antibodies against whole bacteria, derived from colostrum of immunized cows, have been used successfully as prophylaxis against dental caries both in an animal experimental model [17] and in healthy volunteers [18]. In the study of Michalek et al. [17], the effect of a multivalent vaccine was examined in a gnotobiotic rat model with rats monoinfected with S. mutans. The group of rats receiving the immune product had reduced plaque scores and less caries than similarly infected animals given a similar product from non-immunized cows. In the study on human subjects, volunteers using a mouth rinse containing milk-derived antibodies against S. mutans resulted in an initial reduction in the amount of recoverable bacteria, and they formed smaller colonies than plaque samples from the control group.
During the last decades the character of caries disease has changed and today it is not a major problem in the general population. However, there are still groups of individuals who suffer from severe dental caries and our future efforts are directed towards optimizing the antibody source and developing a simple form of administration for the preparation, be it a tablet, chewing gum, tooth paste, gel, varnish or mouth rinse. Further, in those areas of the world where fluoridated water and/or routine dental care are not readily available, prophylactically administered bovine antibodies could be used to reduce the normal transmission of cariogenic bacteria from mother to child.
TREATMENT OF ORAL CANDIDIASIS
During the last decade, the incidence of deep fungal infections has risen sharply. Candida constitute the majority of isolates found and Candida albicans alone accounts for approx. 75% of these cases. Candida infections are usually of endogenous origin and are most often derived from colonization of the GI tract. The organism is able to penetrate intact mucosal epithelial cells and the resulting deep infections are most likely due to inability to eliminate the invading fungal cells. This is most often a consequence of prolonged neutropenia in immunocompromised patients, and GI overgrowth of Candida is likely to contribute to the high incidence of invasive infections in this patient category.
Organ and bone-marrow transplanted patients are frequently colonized with Candida and thus highly susceptible to invasive fungal infections. Mortality is quite high in this patient category and up to 70% of bone marrow transplanted patients with candidaemia succumb. In patients with a disseminated disease, the mortality rate may be even higher. Prophylaxis with orally administered polyene antibiotics is routinely given even though this form of treatment has not been documented to be of any beneficial value in preventing invasive fungal infections. Fluconazole, a novel triazole anti-fungal agent, is however highly active against a variety of fungal pathogens and is effective in treating both superficial and systemic candidiasis. Previous studies have shown that prophylactic treatment with fluconazole prevents all types of Candida infections except those caused by Candida krusei. The latter are therefore becoming more frequent among transplanted patients on fluconazole therapy. This form of prophylaxis has also, as expected, been associated with an increasing incidence of infections with drug-resistant Candida species such as Torulopsis glabrata both in bone marrow transplanted and AIDS patients (for review see [19]).
Successful therapeutic intervention against oral candidiasis using bovine antibodies has also previously been suggested in man (referred to in [20]). We therefore initiated a phase I clinical trial in bone marrow transplanted patients using antibodies from cows immunized with whole Candida organisms and purified mannan [21].
Altogether, 59 patients were enrolled in the study, out of which 54% were colonized with Candida. The aim of the study was to evaluate the influence of oral administration of 3.3 g of powder (with an estimated IgG purity of approx. 50%), given three times daily for various time periods before and after transplantation, on the number of yeast in saliva and stool samples. There was a time correlation between the antibodies given and the number of Candida organisms, and in the subgroup of patients with an initial high load of fungi the treatment resulted in low or undetectable levels of the organism in saliva. No adverse events were noted in the trial and we are currently planning an extension of these studies in liver transplanted patients.
Adherence has been shown to play an important role in the pathogenesis of various infections and also plays an important role in colonization with Candida. Multiple interactions have been described, broadly classified into three groups involving protein–protein (such as the integrin analogues) and protein–carbohydrate (such as the fucose binding protein) structures. The product used in our trial apparently contains some antibodies against these adhesins, as blocking of binding to buccal cells could be readily demonstrated (Fig. 1) in a dose-dependent manner (Table 1). The future identification of the structures which confer binding to human cells would facilitate the making of a second generation product of bovine anti-Candida antibodies, where the animals would be immunized with purified adhesins which could greatly enhance the titre of potentially biologically relevant antibodies and thus provide a more cost-effective form of therapy.
Fig. 1.
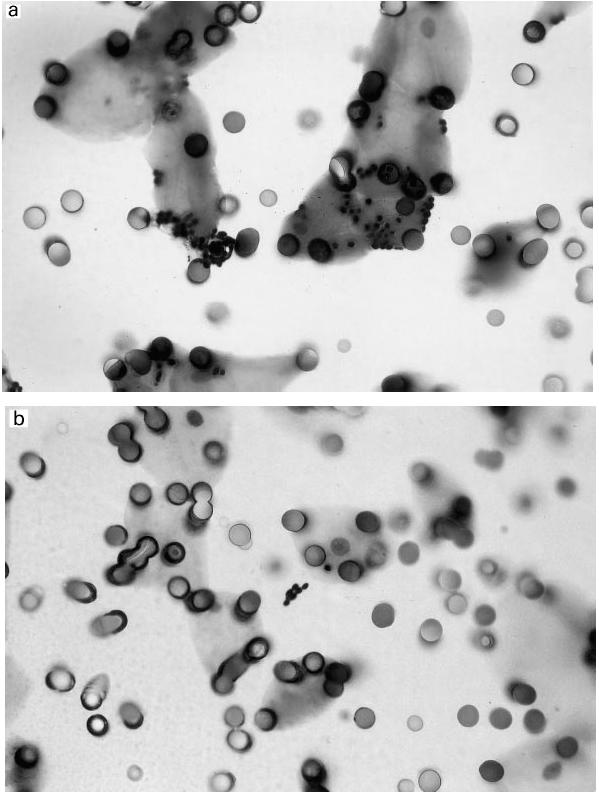
Inhibition of binding of yeast cells to buccal epithelial cells in vitro by addition of bovine immunoglobulins against Candida albicans. Candida organisms were transferred from an agar slant into BHI broth and grown overnight at 23°C. The yeast cells were subsequently washed and resuspended to a concentration of 107/ml and thereafter incubated with different concentrations of the bovine antibody preparations for 1 h at 37°C in a shaking water bath. Epithelial cells were obtained by scraping the mucosal surface of both cheeks with sterile cotton swabs, after which the cells were washed repeatedly and added to the mixture of antibodies and Candida cells for another hour of incubation. The content of the tube was then transferred to a 12-μm pore-sized polycarbonate filter and vacuum was applied. After additional washing, the filter was dried, fixed with methanol and stained. (a) A control slide with an average of 7.3 Candida organisms per buccal epithelial cell. (b) A slide where bovine immunoglobulin has been added, showing an average of 0.3 Candida organisms per buccal cell.
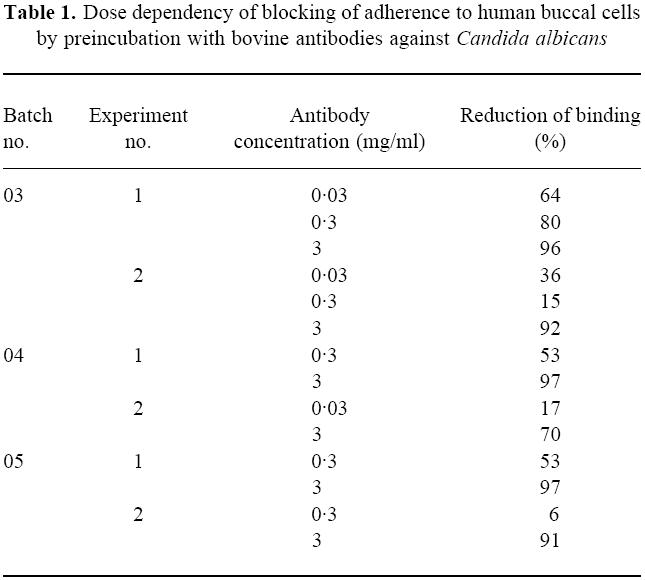
Table 1.
Dose dependency of blocking of adherence to human buccal cells by preincubation with bovine antibodies against Candida albicans
INTERVENTION AGAINST HELICOBACTER INFECTION
Helicobacter pylori, a human-specific gastric pathogen, first isolated in 1982, has emerged as the leading causative agent in chronic active gastritis and peptic ulcer disease. In developing countries, the majority of the population is infected already in early childhood, whereas in the industrialized world < 20% of young people are infected, increasing to about half of the population by 50 years of age. However, infection prevalence is still very significant in view of the medical impact of H. pylori-associated disease. Once established in the host, the bacteria can persist for the lifetime of the individual. Interestingly, most infected subjects show no clinical symptoms, implying the influence of additional factors in the pathogenesis of the disease such as genetic predisposition and the genotypes of the infecting strain. The chronic infection has been correlated to the development of gastric cancer, one of the most common forms of cancer in humans, and H. pylori was recently defined a class 1 carcinogen by the World Health Organization. At present, H. pylori infection can be treated using a combination of bismuth and antibiotics with eradication rates of 90%. Alternatively, omeprazole together with antibiotics such as amoxicillin or clarithromycin can be used. Such therapy appears to be better tolerated and results in eradication rates of 60–80%. The development of antibiotic resistance and the common occurrence of drug intolerance, however, point to a need for therapeutic alternatives.
Helicobacter pylori colonizes the human gastric mucosa by adherence to the mucous epithelial cells and the mucus layer lining the gastric epithelium [22]. These adherence properties protect the bacteria from the extreme acidity of the gastric lumen and displacement from the stomach by forces such as peristalsis and gastric emptying. Central to current thinking in pathogenesis is the importance of adherence for colonization or disease. Bacteria express adhesion molecules that recognize specific carbohydrates on the epithelial cell surface and mucin molecules in the mucus lining. Several putative receptor structures have been described for H. pylori, such as sialylated glycoconjugates, sulphatides and, very recently, the BabA adhesin [23] which binds to the Lewis b blood group antigen.
Previous studies have shown that breast feeding protects from early acquisition of H. pylori in infants [24], suggesting that IgA antibodies in the milk may affect colonization and oral administration of antibodies against Helicobacter may eradicate [25] or reduce [26] the number of bacteria in the gut of experimental animals. In vitro, antibodies from immunized cows are strongly bactericidal against Helicobacter provided that complement is present [27], suggesting that humoral immunity may be of major importance for clearance of the infection. However, the infection is known to be lifelong despite a vigorous immune response including systemically derived, specific IgA in the gastric mucosa, and our own results in patients with IgA deficiency showing that lack of secretory IgA does not seem to have any major effect on the establishment of the infection, argues against a pivotal role for IgA in the defiance against H. pylori [28].
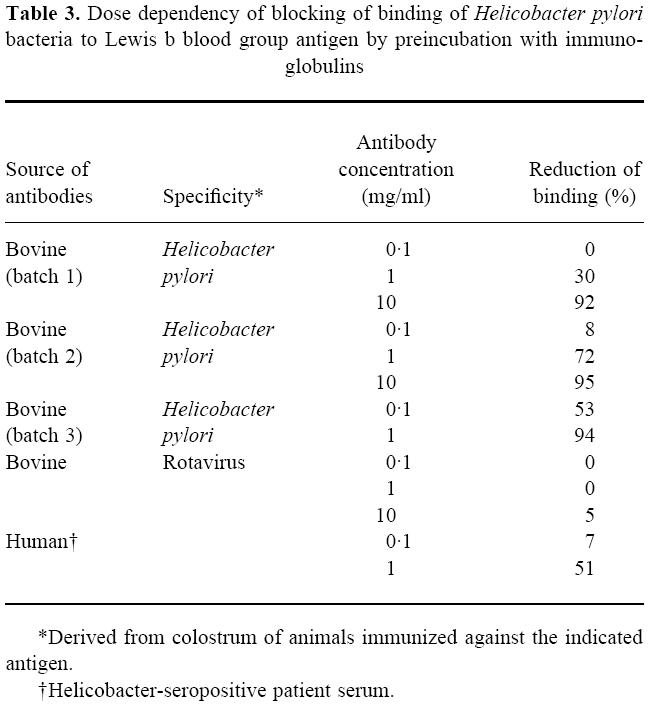
As a major problem in the gut may be related to the low levels of antibodies in the mucus layer (since there is no specific transport mechanism), oral administration would supply sufficient amounts of antibodies for therapeutic efficacy. Topical treatment has also been tried in patients who are chronic carriers of Helicobacter, and although the initial results using bovine antibodies were surprisingly positive [29], subsequent studies [30–33] (summarized in Table 2) have shown modest, albeit encouraging, results. In the first study performed in our own hospital [31], where treatment was given with 8 g daily for 4 weeks using a preparation derived from hyperimmunized cows, 25% of the patients cleared the infection. The preparation contained antibodies against a variety of antigens as detected in Western blot (Fig. 2), including the urease and possibly the CagA and VacA antigens. As these preparations also effectively prevented attachment of bacteria to gut tissue [31] and inhibited binding of a Lewis b conjugate to the bacteria (Table 3), we would anticipate that they may also contain antibodies against adhesin molecules [23].
Table 2.
Passive immunity against Helicobacter pylori infection
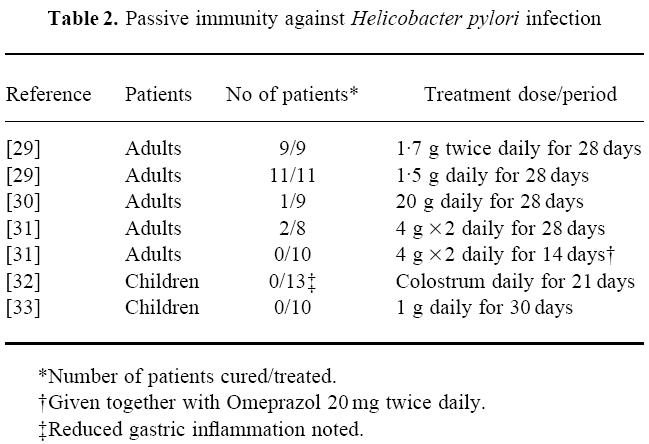
Fig. 2.
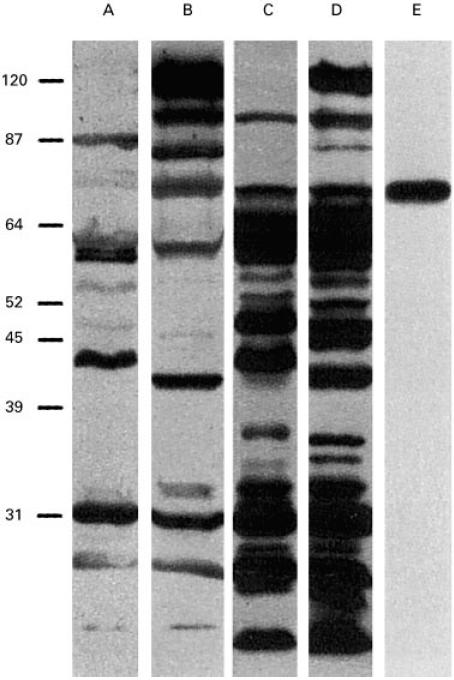
Lanes A, C and B, D, E. SDS–PAGE gel-separated Helicobacter pylori proteins from strains MO19 (Type II) and CCUG 17875 (Type I), respectively. Immunodominant proteins were detected with human sera from an H. pylori-infected individual (lanes A, B), and with bovine colostrum/milk IgG antibodies from cows immunized with strains CCUG17875 and 17874 (lanes C, D) (successfully used in a clinical trial [31]), and with rabbit antisera from immunization experiments with a 20 amino acid synthetic peptide, based on the published 78-kD BabA adhesin sequence (lane E). The top band in lanes B and D corresponds to the 125-kD CagA protein, a member of the cag-PAI (Pathogenicity Island) associated with the more virulent Type I H. pylori strains, and absent from the Type II strains, i.e. strains less associated with acid peptic disease (lanes A, C).
Table 3.
Dose dependency of blocking of binding of Helicobacter pylori bacteria to Lewis b blood group antigen by preincubation with immunoglobulins
In order to facilitate compliance, we subsequently shortened the treatment period and supplemented the therapy with a proton pump inhibitor. However, this schedule proved ineffective, indicating a need for immunoglobulin preparations with an increased efficacy. As a consequence of the high receptor specificity in the adherence process of Helicobacter and the requirement of biologically conserved receptor binding adhesive motifs, the microbes might have been forced to accept the potential Achilles heel of adhesin proteins as vaccine candidates. Thus, a product enriched for particular antibody specificities, such as those against the BabA adhesin, may be clinically superior, and this possibility needs to be explored in the future.
CLINICAL EFFICACY OF BOVINE ANTIBODIES IN ROTAVIRUS INFECTION IN CHILDREN
Rotavirus is a leading major childhood viral enteropathogen in all areas of the world, causing close to a million deaths annually. In the USA alone, rotavirus is associated with 3% of all hospitalizations in children < 5 years old. Although the nature of protection against infection has not been fully elucidated, it is likely that a major part of the effect is due to neutralization of the virus by specific antibodies. Considerable attention has therefore been directed towards the development of a safe and effective vaccine but unfortunately, to date, such efforts have failed to produce a vaccine which consistently protects children from rotavirus disease. Furthermore, even if a vaccine became available, financial constraints in developing countries may prevent its use and it is likely to be less effective in children with malnutrition and concomitant immunodeficiency.
Marked progress has been achieved during the past decade in reducing the mortality from dehydration in acute diarrhoea with the use of modern oral rehydration solutions. However, these preparations have little or no effect on the nutritional morbidity resulting from the 5–7 days of diarrhoea commonly associated with rotavirus infection.
Breast milk, which contains secretory IgA, has previously been shown to protect against diarrhoea in infants. IgA antibodies directed against rotavirus have also been demonstrated in human milk, the main source of immunoglobulins in the intestine of newborns, although their role in preventing or mitigating rotavirus infection in children has been questioned.
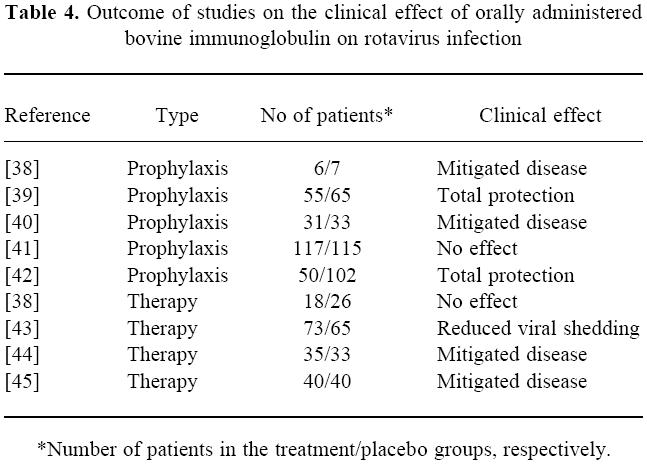
Human IgG has also been given both as effective prophylaxis [34] and therapy [35–37] against rotavirus infection in children, strongly arguing in favour of the possibility of using passive immunity against GI infections, and prophylactic use of immunoglobulins from immunized cows has in fact shown an effect against rotavirus infection in man [38–42](Table 4).
Table 4.
Outcome of studies on the clinical effect of orally administered bovine immunoglobulin on rotavirus infection
The effect of therapeutically administered bovine antibodies has shown variable results, possibly depending on the dose given (Table 5). However, a reduction in the period of shedding of rotavirus after administration of bovine immunoglobulins was published as soon as 1987 [43] (Table 4), followed by a report on successful clinical intervention in rotavirus diarrhoea [44]. In our own recent study [45], performed in Bangladesh, 80 infants with rotavirus diarrhoea were enrolled in a placebo-controlled trial and treated with hyperimmune bovine colostrum-derived antibodies. The results show a substantial reduction in the duration of diarrhoea, stool output and viral shedding in the group of children receiving the ‘active’ preparation.
Table 5.
Dose of bovine immunoglobulins employed in clinical trials in rotavirus-infected children

Recently, a non-structural glycoprotein, NSP4, has been recognized as a rotaviral enterotoxin which causes diarrhoea in an experimental animal model and the symptoms could be eliminated by oral administration of a rabbit antiserum against the NSP4114–135 peptide [46]. Thus, the antitoxin antibodies in the bovine preparations may be crucial for the therapeutic effect of clinically applied bovine immunoglobulins. In fact, these antibodies could be demonstrated in preparations used in previous clinical trials [47] (Fig. 3), although they constituted only a minor proportion of the total reactivity against rotavirus antigens. Thus, a preparation enriched for antitoxin antibodies may be more efficacious, but these products are as yet not available.
Fig. 3.
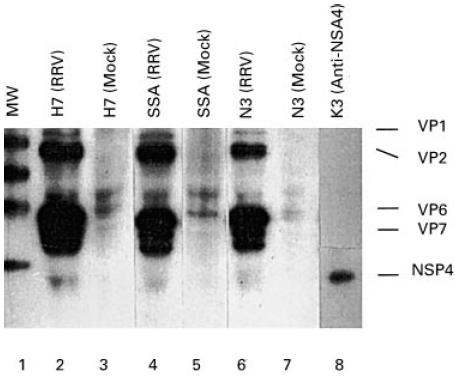
Radioimmunoprecipitation showing the binding of rotavirus antigens by bovine antibodies from various preparations which have been used in clinical trials. Briefly, 5 μl of the bovine immunoglobulin sample were mixed with 10 μl of radiolabelled rotavirus-infected cell lysate or mock-infected cell lysate in a total volume of 600 μl of RIPA buffer. After overnight incubation, protein G-coupled Sepharose beads were added and allowed to incubate for 1 h. After repeated washes and boiling, the samples were analysed by SDS–PAGE. Lane 1 represents molecular size markers and lanes 2–7 represent three immunoglobulin preparations (H7, SSA and N3) which have been incubated with rotavirus antigens or a mock control. Lane 8 represents the result using a MoAb against the NSP4 protein. The relative amounts of anti-NSP4 antibodies were calculated on the intensities of the corresponding band (3%, 1.2% and 2.5%, respectively).
In the developed world, a strategy of treating children with severe rotavirus infection early in the course of their disease could be quite cost-effective given the high cost of hospitalization. In developing countries, the availability of a cheap source of antibodies both for prophylaxis and therapy could possibly help reduce the mortality of the disease.
ROLE OF ANTIBODIES IN EXPERIMENTAL CAMPYLOBACTER JEJUNI INFECTION
Campylobacter jejuni is a Gram-negative rod which is associated with acute GI infection in humans. It is one of the most common causes of enterocolitis world-wide. Several species of animals have been shown to transmit the infection to man. Although the disease is usually self-limiting, severe sequels are occasionally seen and immunodeficient patients may become chronic carriers in spite of aggressive antibiotic therapy. Breast milk-derived IgA antibodies protect against Campylobacter-induced diarrhea [48] and we previously treated a hypogammaglobulinaemic patient with orally administered human IgA with clinical success [7].
Oral administration of antibodies against different GI pathogens has also been shown to be effective both prophylactically and therapeutically in a variety of animal species (for review see [4]) and recently, oral administration of polyclonal chicken antibodies against Campylobacter jejuni was shown to inhibit bacterial colonization [49]. However, as these antibodies were derived from the bile of immunized birds, the availability of material is severely limited.
We recently described our own work on application of bovine immunoglobulins prepared from milk collected day 8–40 post-parturition [50] from cows immunized with a mixture of reference strains and 13 clinical Campylobacter jejuni and Campylobacter coli isolates, against infection in chickens.
Prophylactic administration of bovine antibodies resulted in a very marked reduction in the mean number of bacteria (> 99%) throughout an experiment where bacteria were administered orally to 22-day-old chickens with or without addition of the bovine immunoglobulin preparation (mixed with the bacterial suspension 1 h prior to administration to the chickens). No additional antibodies were given to the animals and the result was followed by daily collection of faecal samples for 5 days At the end of the experiment, three of the five treated animals still remained culture-negative, whereas all of the 10 control animals were heavily infected.
When bovine anti-Campylobacter antibodies were given to infected animals, a 50–80% reduction in the number of bacteria in the faecal samples was observed during the treatment phase compared with control animals. In this experiment, 30-day-old chickens (8 days after the initial infection) were treated daily with an oral administration of bovine antibodies. However, 3 days after discontinuation of the antibody administration, the mean numbers of bacteria were again similar in both groups.
In the final therapeutic experiment, stable infected 34-day-old chickens (6 days after initial infection), were given two oral doses of bovine antibodies or left untreated. Four hours after the final treatment, the chickens were killed and quantitative faecal bacterial cultures performed. There was a marked (62%) and statistically significant reduction in the mean number of Campylobacter after administration of antibodies and a > 90% reduction in the number of bacteria in a majority (six out of 10) of the treated animals. In the control group, the mean number of bacteria increased during the 1-day course of the experiment, resulting in a 82% overall reduction in the number of bacteria in the treatment group at slaughter.
The prophylactic potential of orally administered antibodies against Campylobacter jejuni has hitherto not been explored in man. One possible indication would be as short-term prophylaxis against traveller's diarrhoea, where recent estimates suggest that up to 50% of all cases may be associated with Campylobacter infection [51]. Furthermore, workers in the poultry industry might benefit from prophylactic treatment in order to prevent or mitigate disease during the initial phases of occupational exposure. It may also be applied in immunodeficient patients who are especially prone to infection with Campylobacter jejuni.
ANTIBODY THERAPY IN CLOSTRIDIUM DIFFICILE INFECTION
Clostridium difficile, the causative agent of antibiotic-associated diarrhoea and pseudo-membranous colitis, is a microaerophilic, Gram-positive, spore-forming, rod-shaped organism that is a common cause of nosocomial infection [52]. Pathogenic strains produce two large protein exotoxins. Toxin A, first described in 1980 [53], is responsible for intestinal inflammation, mucosal damage and fluid secretion, whereas toxin B is cytotoxic. Symptoms may range from asymptomatic carriage to potentially fatal pseudomembranous colitis, and there is a substantial incidence of relapsing disease following initial successful therapy [54].
Although a majority of adults in the USA are seropositive, the protective role of circulating antitoxin antibodies has not been established. Defective humoral immunity in children has been associated with susceptibility to relapsing Clostridium difficile colitis, and use of gammaglobulin therapy reportedly results in cessation of symptoms [55]. Colonic aspirates containing secretory IgA antitoxin may inhibit binding of toxin A to its intestinal receptor and thereby prevent disease [56]. In animal models, the efficacy of both passive and active immunization has been demonstrated.
Currently, Clostridium difficile ranks as one of the most common causes of bacterial enteritis, and colonizes the intestines of approx. 20% of adult hospital in-patients [54] where chronic asymptomatic carriage is common [57]. Seven percent of patients were culture-positive at admission and 21% of patients acquired Clostridium difficile after admission through oral ingestion of spores from the immediate environment or by contact with shared surfaces contaminated by an infected roommate [54]. Of these, most remained asymptomatic, but approximately one third developed diarrhoea.
Clostridium difficile demonstrates in vitro sensitivity to a variety of anti-microbial agents, but many of the agents which have such activity are also implicated as permitting Clostridium difficile overgrowth. Currently, the most widely accepted agents for the treatment of Clostridium difficile colitis are vancomycin and metronidazole. Although these two drugs demonstrate remarkable efficacy in the treatment of antibiotic-associated diarrhoea, vancomycin in particular has two major drawbacks: cost, and substantial incidence of disease relapse.
The Golden Syrian hamster provides the best characterized animal model of Clostridium difficile colitis [58]. Animals are typically predisposed to disease by the administration of clindamycin. Following oral challenge with viable Clostridium difficile, the hamsters develop inflammation, ulceration and haemorrhage of the intestinal mucosa, lethargy and severe, often fatal, diarrhoea. Treatment with bovine immunoglobulins against Clostridium difficile for 3 days prior to and 10 days post single oral challenge with live bacteria protected six out of nine hamsters, whereas three treated and all non-treated animals died [59].
Purified Clostridium difficile Toxin A induces marked fluid secretion and intestinal inflammation when injected into intestinal loops [53]. However, bovine immunoglobulins against Clostridium difficile substantially inhibited toxin-induced enterotoxicity in the rat ileal loop [60]. Furthermore, histological examination of ileal loop tissues showed significant damage, as reflected by increases in histology score, in tissues exposed to toxin. Enteritis was, however, significantly decreased in animals receiving bovine antibodies against Clostridium difficile, whereas non-immune control immunoglobulins showed no effect [60].
No clinical studies in Clostridium difficile-infected patients have as yet been reported using bovine immunoglobulins. A preparation similar to that described above was, however, given to normal volunteers to evaluate IgG recovery. Data suggested that a proportion of the antibodies (1.6–3.8%) survived the passage through the entire GI tract and this fraction may be increased by simultaneous administration of omeprazol or by encapsulation [61]. An additional observation obtained during these studies was the direct correlation between the total GI transit time and overall bovine IgG recovery, suggesting that colonic degradation also limits the survival of the IgG.
A follow-up study in volunteers with an ileostomy but otherwise apparently normal GI function [62] evaluated the amount of orally administered bovine IgG surviving passage to the small intestine. Almost 50% of the oral dose of bovine IgG was recoverable, and this IgG retained its specific Clostridium difficile antigen-binding activity and Toxin A neutralization capability. Thus, it is not surprising that small bowel pathogens are amenable to bovine antibody therapy, and substantial delivery of intact IgG to the caecum also supports potential efficacy against causes of infectious colonic diarrhoea such as caused by Clostridium difficile.
BOVINE ANTIBODY TREATMENT OF CRYPTOSPORIDIUM PARVUM-INDUCED DIARRHOEA
The obligate parasite Cryptosporidium parvum (C. parvum) is a protozoan that infects the GI tract and, rarely, the biliary tract of humans. Infection with this organism can result in a debilitating diarrhoea with associated dehydration and malnutrition. Typically the organism is found in the small bowel, primarily the jejunum; however, cryptosporidia have been found in the pharynx, bronchi, oesophagus, stomach, duodenum, gallbladder, ileum, appendix, colon, and rectum of immunocompromised hosts. The organism appears to infect most vertebrate groups and can be spread by both zoonotic and person-to-person (primarily faecal–oral) mechanisms. It is also likely that indirect transmission via water, food, and fomites is common [63].
Cryptosporidiosis is a common infection in humans, with high rates of recovery of the Cryptosporidium organism from patients with diarrhoea noted worldwide, as well as evidence of seroprevalence rates of 25–35% in the USA and 64% in South America [64]. In western countries, 0.6–4.3% of small children presenting to medical centres with gastroenteritis have harboured Cryptosporidium oocysts, and in third world countries the rates have ranged from 3% to 30% [63]. In the USA, chronic diarrhoea due to C. parvum infection primarily affects immunocompromised patients, with T lymphocyte dysfunction resulting from infection with HIV being the single most important predisposing factor [65,66]. Other immunocompromised states in which cryptosporidiosis is found include congenital hypogammaglobulinaemia, measles, and patients undergoing cancer chemotherapy or immunosuppressive therapy. In third world countries, marasmus and other forms of childhood malnutrition are the most common predisposing factors [63].
In the immunologically competent patient, cryptosporidiosis may exacerbate and remit, but is generally a self-limiting disease. The incubation period is 4–14 days, and symptoms of profuse watery diarrhoea and abdominal cramps typically persist for 5–11 days or, rarely, as long as 4 weeks [67,68]. In the immunodeficient host, however, the diarrhoea can become irreversible and life-threatening due to profound fluid loss and malabsorption [69,70]. These patients usually present with a prolonged, life-threatening cholera-like illness. Vomiting, weakness, malaise, and low grade fever are often noted; fluid loss in excess of 3–6 l/day is common, and up to 17 l/day of watery faeces has been reported [71].
At present, no effective treatment for cryptosporidiosis exists. Although more than 90 chemotherapeutic, biologic, and anti-diarrhoeal agents have been tested clinically against this disorder, none has proved efficacious. Current therapy is primarily supportive, involving a regimen of anti-diarrhoeal agents, fluid replacement, nutritional supplements, and discontinuation of immunosuppressive drugs in specific patient populations [69,70,72]. Recent studies have shown that bovine immunoglobulins are potentially effective for management of chronic diarrhoea when cryptosporidiosis is identified as the causative organism [11].
The potential efficacy of immune colostrum in treating and/or preventing cryptosporidial infection has been demonstrated in several animal models. Fayer and coworkers [73] demonstrated that treatment with colostral whey obtained from an immunized cow significantly reduced the number of C. parvum organisms found in BALB/c mice after oral inoculation with live oocysts. Further, it appeared that protection was mediated by specific anti-sporozoite activity, since significantly fewer sporozoites were found in mice challenged by rectal administration of isolated sporozoites incubated with immune colostrum than in animals receiving sporozoites incubated in control colostrum or Hanks' balanced salt solution.
Perryman and coworkers [74] reported that MoAbs to C. parvum raised in ascites, anti-C. parvum immune bovine serum, and anti-C. parvum immune bovine colostral whey all neutralized the infectivity of C. parvum sporozoites for mice in a time-dependent manner. Neutralization of sporozoites with MoAbs and bovine serum was significant after 20 min, but greatest following 30 min of incubation. Neutralization of sporozoites with bovine colostral whey, however, was virtually complete after 10 min of incubation. The ability of colostral whey to quickly neutralize sporozoites within the gut lumen may be important in reducing infection following ingestion and in diminishing autoinfection resulting from sporozoite release in the host. In a second experiment, anti-C. parvum immune colostral whey also successfully reduced infectivity scores in mice orally challenged with 20 000 oocysts; neither immune bovine serum nor MoAbs against sporozoite-specific antigens provided significant protection compared with their respective controls.
When sections of mouse intestine were examined histologically at 72 h post-infection, the reduction in parasite numbers in animals treated with purified IgG2 or IgM was approximately half that seen with colostral whey, IgG1, or IgA [75]. All reductions were highly significant (P < 0.0001) for all treatment groups versus control animals. This also confirmed that purified immunoglobulin fractions from immune colostral whey possess distinct anti-parasitic activity, whether or not other active factors are present in bovine whey.
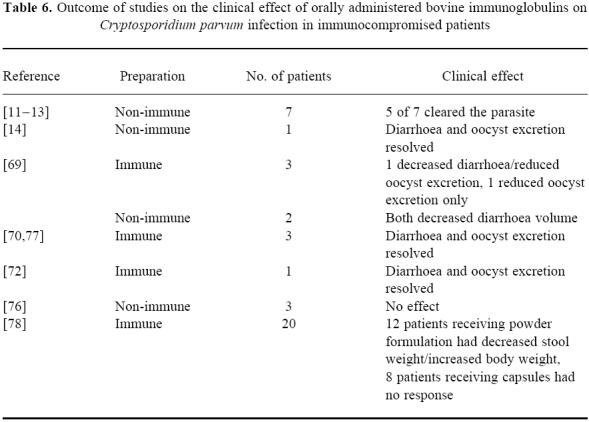
In previous literature reports, a total of 20 C. parvum-infected immunocompromised patients, at least three of whom were paediatric patients, were treated with either non-specific or C. parvum-specific bovine immunoglobulin [11–13,69–71,76,77]. In all of these cases, immunoglobulin was administered by an oral, nasogastric or nasoduodenal route, and the duration of treatment was from 1 to 21 days. In the three studies using specific anti-C. parvum bovine colostrum and in one study using non-specific bovine immunoglobulin concentrate, most patients experienced successful reduction or elimination of clinical symptoms (primarily diarrhoea). In another study in which a non-specific colostrum product was administered to three patients, no clinical improvement was noted (Table 6).
Table 6.
Outcome of studies on the clinical effect of orally administered bovine immunoglobulins on Cryptosporidium parvum infection in immunocompromised patients
In a recently reported study [78] of a bovine immunoglobulin concentrate preparation specific for the treatment of C. parvum diarrhoea associated with AIDS in adult patients, there was a significant decrease in stool weight, and a decrease in stool frequency in response to antibody therapy. The study enrolled 24 total patients: 16 patients with C. parvum infection only, four patients with C. parvum plus other non-C. parvum infection, three patients with non-specific diarrhoea with no identifiable pathogen, and one with an untreatable opportunistic infection. The 16 patients in the C. parvum infection only group were randomized to receive the product in either a powdered formulation or an encapsulated formulation; all other patients received the powdered formulation. Mean body weight also increased by 1.6 kg compared with baseline in patients with C. parvum receiving the powdered bovine antibody concentrate.
THE ROLE OF BOVINE ANTIBODIES IN INFECTIONS CAUSED BY ESCHERICHIA COLI
Diarrhoea may result from infection with one or more strains of Escherichia coli. The enterotoxigenic E. coli (ETEC) strains are the most common cause of diarrhoea in travellers to the less developed countries [79,80]. Regrettably, large scale use of prophylactic antibiotics by travellers has resulted in the development of multiresistant organisms. Enteropathogenic E. coli (EPEC) are an important cause of acute infectious diarrhoea in young children throughout the world, and a major contributor to infant mortality in developing nations [41]. The enterohaemorrhagic E. coli (EHEC) are recognized as key intestinal pathogens in the development of extra-intestinal sequelae due to their elaboration of Shiga-like toxins [81]. Severe complications such as haemolytic uraemia syndrome due to E. coli O157:H7 have recently been attributed to food-borne contamination in countries around the world.
To cause disease, E. coli must first adhere to colonize the upper small bowel. ETEC strains then elicit diarrhoea by secreting enterotoxins that have a direct fluid-secreting effect on the intestinal mucosa. While antibiotic agents can be successfully used to treat this generally mild to moderate diarrhoea in travellers, fear of promulgating wide-spread anti-microbial resistant strains now limits their prophylactic use. A substitute therapeutic with virtually no side-effects would be a welcome alternative. The effectiveness of bovine antibodies in protecting volunteer subjects against oral pathogen challenge has been demonstrated in two separate, double blind studies. In the first study [81], a multiple-antigen E. coli-specific milk immunoglobulin concentrate was 100% effective in preventing diarrhoea in 10 volunteers orally challenged with one of the ETEC strains used for immunization, whereas nine of 10 controls developed diarrhoea (P < 0.0001). In the second trial [80], bovine antibodies against purified ETEC colonization factor antigens (CFA) were evaluated. The milk-derived antibodies protected 14 of 15 subjects from clinical diarrhoea, whereas seven of 10 volunteers receiving a placebo preparation developed symptoms (P < 0.002).
In newborn infants, the protective effect of breast feeding in limiting E. coli infection is well recognized. Passively transferred factors in human milk include antibodies, lactoferrin and lactoperoxidase [82]. Supplemental bovine milk-derived antibody preparations in infant feeding formulas have been used to treat active EPEC disease in infants in one of the earliest controlled studies using antibodies [83]. Sixty hospitalized infants (10 days to 18 months of age) with active EPEC diarrhoea were treated for 10 days with 1 g/kg body weight/day of a specific milk immunoglobulin concentrate (MIC). Among 51 patients infected with E. coli strains also present in the vaccine which was used to immunize the cows used for their production of the antibody preparation, 84% became stool culture-negative following treatment, whereas only one of nine infants infected with strains not included in the vaccine (controls) became negative (P < 0.01). Faecal recovery of bovine immunoglobulins from infant feeding studies has proved that these antibodies survive GI passage as intact molecules, and active fragments and stool extracts from these infants provided protection in a mouse E. coli challenge model [84].
A prophylactic, double-blinded, placebo-controlled field study in 232 infants receiving an average of 0.12 MIC/kg body weight/day (as a 1% admixture in the formula) was not successful in reducing the incidence of diarrhoea or impacting the isolation of EPEC organisms from stool in symptomatic subjects [41]. The isolation rate of EPEC bacteria was significantly lower in asymptomatic than symptomatic subjects. Inadequate intake of a sufficient amount of specific anti-E. coli antibodies, due to the dilution with a similar product derived from non-immunized cows, may have accounted for the negative results in this particular study.
Recent outbreaks of E. coli O157:H7 in the USA and Japan have resulted in the deaths of a number of children due to a haemolytic uraemic syndrome. Antibiotic therapy is not recommended early in the infectious process, as disruption of the bacteria in the gut may cause release of Shiga-like toxins (SLT) and/or exacerbate toxin absorption. As the prevalence of EHEC in cattle is high, colostral antibodies are thought to contribute to the protection of newborn calves against these organisms. Lissner and coworkers [81] have demonstrated an in vitro efficacy of specific bovine immunoglobulins from non-immunized cows in neutralizing the cytotoxic activity of SLT-1 and SLT-II EHEC toxins on Vero cells and inhibition of the cytolytic activity on human erythrocytes in vitro. Therefore, antibody neutralization of EHEC toxins may be protective if administered sufficiently early in the course of the disease.
DISCUSSION
Bovine colostrum has been used therapeutically in man for decades. However, as the active component resides in the IgG fraction, it is being replaced by purified immunoglobulin preparations. This also leads to a transition from using a poorly defined biological product to applying a defined pharmaceutical drug. Still, there is a need for standardization if it is to be recognized by the regulatory agencies, and as only the genes encoding the heavy chain constant region genes of bovine IgG1 and IgG2 were previously known, we recently cloned the bovine μ [85], γ3 [86], α [87] and ε [88] genes. The sequences will allow us to make peptides against suitable epitopes and thus to generate antisera which recognize the different isotypes. This work is performed as a part of an ongoing IUIS effort to characterize immunoglobulins in different animal species and to supply monoclonal bovine antibodies of different classes and subclasses as ‘golden’ standards.
The differing results which have been obtained in the clinical studies published to date may cast some doubt on the concept of using bovine immunoglobulins for human therapy. This is likely to be due to differences in study design or in the quality of the preparation itself where the specificity, titre and subclass composition of the antibodies may differ.
The class and subclass composition of a given product may, at least theoretically, be of importance for the clinical outcome. In colostrum, IgG1 antibodies constitute the vast majority of all antibodies (due to a selective transport mechanism at the time of parturition), whereas in milk-derived immunoglobulin products, the proportion of IgG1 is reduced. Antibodies against an antigen which induces mainly or exclusively IgG2 or IgG3 would then be present in minute amounts in colostrum or a colostrum-derived product. Thus, as bovine antibodies against rotavirus are largely restricted to IgG1 [89,90], a reduced proportion of IgG1 in milk-derived products may actually result in a product which is inferior from a therapeutic point of view. The poor response in the therapeutic trial against rotavirus using milk-derived antibodies [34] may thus be a result of both the low amount of antibodies used and the proportionally reduced content of IgG1 antibodies.
Specific antibodies against a variety of pathogens are present in non-immunized cows, but the amount of antibodies is clearly not sufficient for a therapeutic effect in man. For example, while non-immunized cows possess moderate levels of antibodies against rotavirus, acquired through natural exposure, these antibodies are directed against bovine serotypes (mainly serotypes 6 and 10) and can therefore not effectively neutralize the most common serotypes [1–4] in man. Through immunization, a 100-fold increase in the amount of relevant anti-rotavirus antibodies can be achieved [10], but when these antibodies are diluted with immunoglobulins from non-immunized animals (1% admixture of the specific immunoglobulin preparations), no prophylactic effect is either to be expected or actually observed in clinical trials [32].
The titre of antibodies against different pathogens is usually measured by ELISA, but unfortunately, neutralizing titres for the preparations used for therapy are not always determined. Furthermore, these titres are usually not adjusted for the immunoglobulin content of the products and comparison between different clinical trials is therefore difficult. Although there exists a relationship between the ELISA titre and the neutralizing titre, the correlation is far from perfect and conclusions on the potential efficacy of the product cannot be drawn from ELISA titres alone. It should also be noted that differences in the neutralizing antibody titre against the various serotypes may affect the outcome of a clinical trial depending on the circulating serotype at the time of the study. This is also relevant for the vaccination strategy employed, where one would have to try to reach a high titre against all serotypes and a high amount and affinity of the antibodies may thus be crucial for success.
The concentration of immunoglobulins in the intestine of normal healthy adults is in the order of 10–100 mg/l. The proportion of specific antibodies would be expected to constitute a very small fraction thereof (< 1/1000) even in individuals who are immune to the pathogen in question. In the normal human adult, approx. 5 g of IgA are produced and secreted at mucosal surfaces each day. The corresponding level in an infant would thus be expected to be in the order of 0.5–1 g/day which, provided that the child has previously been vaccinated or has suffered from infection, is sufficient to render him or her immune against subsequent attacks by the same pathogen. Indeed, the daily oral administration of 600 mg of human immunoglobulin from normal donors (containing approx. 360 mg of IgA), presumably immune against a variety of pathogens, has been reported to be sufficient as prophylaxis against necrotizing enterocolitis in premature infants [5].
The dose of immunoglobulin needed for oral prophylaxis or therapy has not been clearly defined as yet and may be dependent on the survival of the antibodies in the GI tract. When using human IgG, approx. 10–25% of orally administered human IgG survives the passage through the GI tract in children or adults with diarrhoea [91,92], and IgA is also detectable in the stool of premature children treated prophylactically with orally administered human IgA [5]. A number of clinical studies have also shown detectable amounts of bovine IgG in faeces after oral administration [34,61,86,93–99], where a GI survival of 10–20% of bovine IgG in a macromolecular form has been shown in adult patients with diarrhoea and in new-born infants. If the survival could be increased, by protecting the proteins from the acidic content of the stomach and/or proteolytic enzymes, the immunoglobulin dose required could be lowered and thereby also the cost.
Digestion of proteins begins with the hydrochloric acid in the stomach. The low pH denatures the protein which makes peptide bonds inside the protein accessible for the endopeptidases, the pepsins, secreted in the stomach. These are only active below pH 3.0 and 15% of the peptide bonds are hydrolysed in the stomach. In the duodenum endopeptidases (trypsin and chymotrypsin), and exopeptidases from the pancreas, hydrolyse 50% of the peptide bonds. The secretion of these enzymes seems to be regulated by a feedback mechanism, i.e. excess of enzymes (all proteins digested) turns off the secretion. Free amino acids and very short peptides are then absorbed through the epithelial wall in the small intestine, where the remaining 35% of the peptide bonds are hydrolysed by epithelial brush-border endo- and exopeptidases.
When bovine antibodies are used for prophylaxis, similar or lower doses compared with human antibodies would be expected to be effective provided that the cows are properly immunized. In challenge studies performed by Tacket and coworkers [81], complete protection against E. coli challenge in adult volunteers was seen using a daily oral administration of 5 g of bovine immunoglobulin from hyperimmunized animals. In a piglet model of human rotavirus infection, 0.6–1 g/day also sufficed to afford protection [100], suggesting that for a child, a daily dose of 500 mg of immunoglobulins from an immunized cow might be adequate for prophylaxis. This is clearly within the range given in the hitherto published trials, but as the quality of the specific antibodies within the preparation will also be of marked importance for the clinical outcome, the results obtained have not always been positive.
Therapeutic intervention in patients with an ongoing infection might theoretically require the use of higher amounts of antibodies. In trials on rotavirus infection [35–37], 300 mg/kg body weight per day of human immunoglobulin, i.e. an estimated 1000–4000 mg daily, has been used, and in other infections such as Clostridium difficile [6] and Clostridium jejuni [7], similar doses have been employed. Successful intervention has also been obtained when treating H. pylori [29–33] and cryptosporidial [69,72,76,78] infections with daily administration of 3–15 g of immunoglobulin from immunized cows. In the study by Mitra et al. [44], approx. 9 g of immunoglobulin were given daily to rotavirus-infected children. Although these studies all point to the necessity of using high doses of immunoglobulins when trying to combat an ongoing infection, the amount of antibodies needed still remains to be determined.
Emerging antibiotic resistance constitutes an increasing threat to society which necessitates development of alternative anti-microbial strategies. This is particularly pertinent as large scale application of antibiotics is precluded on grounds of microbial ecology, and a product containing polyclonal bovine antibodies should be acceptable from an environmental point of view. Polyclonal bovine antibodies contain a very small amount of ‘relevant’ antibodies, and if MoAbs against targeted structures were to be applied therapeutically, the daily doses needed would probably be in the μg range. However, in most cases resistance against the effect of MoAbs due to mutations would be expected to emerge within a short period of time and there is thus a need, in the predictable future, for a polyclonal product.
It has taken hundreds of million of years to perfect the antibody system, and passive immunity is a concept which is utilized by most mammals. We feel that the time has come to establish that bovine antibodies also have a role to play in human therapy and what remains to be improved is the quality of the antibodies and the dosage and timing of their administration.
Acknowledgments
This work was supported by the Swedish Medical Research Council, the Sigurd and Elsa Golje Memorial Foundation, the Ruth and Richard Julin Foundation and the Swedish Patent Fund for Reserach in Preventive Dentistry.
REFERENCES
- 1.Rosen F, Wedgwood RJP, Eibl M, et al. Primary immunodeficiency diseases. Report from a WHO Scientific Group Clin Exp Immunol. 1997;109(Suppl. 1):28. [PubMed] [Google Scholar]
- 2.Gustafson R, Gardulf A, Granert C, Hansen S, Hammarström L. New strategy for prophylactic therapy to patients with ‘selective’ IgA deficiency. Lancet. 1997;350:865. doi: 10.1016/S0140-6736(05)62034-X. [DOI] [PubMed] [Google Scholar]
- 3.Janson A, Nord C-E, Hammarström L. Effects of orally administered human immunoglobulin on the gastrointestinal bacterial flora. Eur J Clin Microbiol. 1995;14:61–64. doi: 10.1007/BF02112623. [DOI] [PubMed] [Google Scholar]
- 4.Hammarström L, Gardulf A, Hammarström V, Janson A, Lindberg K, Smith CIE. Systemic and topical immunoglobulin treatment in immunocompromised patients. Immunol Rev. 1994;139:43–70. doi: 10.1111/j.1600-065x.1994.tb00856.x. [DOI] [PubMed] [Google Scholar]
- 5.Eibl MM, Wolf HM, Fürnkranz H, Rosenkranz A. Prevention of necrotizing enterocolitis in low-birth-weight infants by IgA-IgG feeding. N Engl J Med. 1988;319:1–7. doi: 10.1056/NEJM198807073190101. [DOI] [PubMed] [Google Scholar]
- 6.Tjellström B, Stenhammar L, Eriksson S, Magnusson K-E. Oral immunoglobulin A supplement in treatment of Clostridium difficile enteritis. Lancet. 1993;341:701–2. doi: 10.1016/0140-6736(93)90477-x. [DOI] [PubMed] [Google Scholar]
- 7.Hammarström V, Smith CIE, Hammarström L. Oral immunoglobulin treatment in Campylobacter jejuni enteritis. Lancet. 1993;341:1036. [PubMed] [Google Scholar]
- 8.Casswall T, Hammarström L, Veress B, Nord C-E, Bogstedt A, Brockstedt U, Dahlström K-A. Oral IgA treatment of chronic non-specific diarrhoea in infants and children. Acta Paediat. 1996;85:1126–8. doi: 10.1111/j.1651-2227.1996.tb14231.x. [DOI] [PubMed] [Google Scholar]
- 9.Bogstedt AK, Johansen K, Hatta H, Kim M, Casswall T, Svensson L, Hammarström L. Passive immunity against diarrhoea. Acta Paediat. 1996;85:125–8. doi: 10.1111/j.1651-2227.1996.tb13975.x. [DOI] [PubMed] [Google Scholar]
- 10.Janson A, Nava S, Brüssow H, Mahanalabis D, Hammarström L. Indigenous antimicrobial agents of milk–recent developments. International Dairy Federation Press; 1994. Titers of specific antibodies in immunised and non-immunised cow colostrum. Implications for their use in patients with gastro-intestinal infections; pp. 221–8. [Google Scholar]
- 11.Rump JA, Arndt R, Arnold A, et al. Treatment of diarrhoea in human immunodeficiency virus-infected patients with immunoglobulins from bovine colostrum. Clin Invest. 1992;70:588–94. doi: 10.1007/BF00184800. [DOI] [PubMed] [Google Scholar]
- 12.Plettenberg A, Stoehr A, Stellbrink H-J, Albrecht H, Meigel W. A preparation from bovine colostrum in the treatment of HIV-positive patients with chronic diarrhea. Clin Invest. 1993;71:42–45. doi: 10.1007/BF00210962. [DOI] [PubMed] [Google Scholar]
- 13.Stephan W, Dichtelmüller H, Lissner R. Antibodies from colostrum in oral immunotherapy. J Clin Chem Clin Biochem. 1990;28:19–23. [PubMed] [Google Scholar]
- 14.Shield J, Melville C, Novelli V, Anderson G, Scheimberg I, Gibb D, Milla P. Bovine colostrum immunoglobulin concentrate for cryptosporidiosis in AIDS. Arch Dis Child. 1993;69:451–3. doi: 10.1136/adc.69.4.451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hughes M, MacHardy SM, Sheppard AJ, Woods NC. Evidence for an immunological relationship between Streptococcus mutans and human cardiac tissue. Infect Immun. 1980;27:576–88. doi: 10.1128/iai.27.2.576-588.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Loimaranta V, Tenovuo J, Virtanen S, Marnila P, Syväoja E-L, Tupasela T, Korhonen H. Generation of bovine immune colostrum against Streptococcus mutans and Streptococcus sobrinus and its effect on glucose uptake and extracellular polysaccharide formation by mutans streptococci. Vaccine. 1997;15:1261–8. doi: 10.1016/s0264-410x(97)00027-3. [DOI] [PubMed] [Google Scholar]
- 17.Michalek SM, Gregory RL, Harmon CC, Katz J, Richardson GJ, Hilton T, Filler SJ, McGhee JF. Protection of gnotobiotic rats against dental caries by passive immunization with bovine milk antibodies to Streptococcus mutans. Infect Immun. 1987;55:2341–7. doi: 10.1128/iai.55.10.2341-2347.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Filler SJ, Gregory RL, Michalek SM, Katz J, McGhee JR. Effect of immune bovine milk on Streptococcus mutans in human dental placque. Archs Oral Biol. 1991;36:41–47. doi: 10.1016/0003-9969(91)90052-v. [DOI] [PubMed] [Google Scholar]
- 19.Como JA, Dismukes WE. Drug therapy. N Engl J Med. 1994;330:263–72. doi: 10.1056/NEJM199401273300407. [DOI] [PubMed] [Google Scholar]
- 20.Faber V. Cows make antibodies against AIDS infections. New Scientist. 1988:35. [Google Scholar]
- 21.Tollemar J, Gross N, Dolgiras N, Jarstrand C, Ringdén O, Hammarström L. Passive immunity against Candida in bone-marrow transplanted patients: reduction of salivary yeast counts by oral administration of bovine anti-Candida antibodies. Bone Marrow Transplantation. 1999;23:283–90. doi: 10.1038/sj.bmt.1701560. [DOI] [PubMed] [Google Scholar]
- 22.Borén T, Falk P, Roth KA, Larson G, Normark S. Attachment of Helicobacter pylori to gastric epithelium mediated by blood group antigens. Sci. 1993;262:1892–5. doi: 10.1126/science.8018146. [DOI] [PubMed] [Google Scholar]
- 23.Ilver D, Arnqvist A, Ögren J, et al. The Helicobacter pylori Lewis b blood group antigen binding adhesin revealed by re-tagging. Sci. 1998;279:373–7. doi: 10.1126/science.279.5349.373. [DOI] [PubMed] [Google Scholar]
- 24.Thomas JE, Austin S, Dale A, McClean P, Harding M, Coward WA, Weaver LT. Protection by human milk IgA against Helicobacter pylori infection in infancy. Lancet. 1993;342:121. doi: 10.1016/0140-6736(93)91327-i. [DOI] [PubMed] [Google Scholar]
- 25.Czinn SJ, Nedrud JG. Protection of germ-free mice from infection by Helicobacter felis after active oral or passive IgA immunization. Vaccine. 1993;11:637–42. doi: 10.1016/0264-410x(93)90309-l. [DOI] [PubMed] [Google Scholar]
- 26.Cordle CT, Schaller JP, Krakowa S. Indigenous antimicrobial agents of milk–recent developments. International Dairy Federation Press; 1994. Bovine antibodies to Helicobacter pylori as a possible treatment for gastritis and peptic ulcer disease in humans; pp. 131–2. [Google Scholar]
- 27.Korhonen H, Syväoja E-L, Ahola-Luttila H, Sievelä S, Kopola S, Husu J, Kosunen TU. Bactericidal effect of bovine normal and immune serum, colostrum and milk against Helicobacter pylori. J Appl Bacteriol. 1995;78:655–62. doi: 10.1111/j.1365-2672.1995.tb03112.x. [DOI] [PubMed] [Google Scholar]
- 28.Bogstedt A, Nava S, Wadström T, Hammarström L. Helicobacter pylori infections in IgA deficiency: lack of role for the secretory immune system. Clin Exp Immunol. 1996;105:202–4. doi: 10.1046/j.1365-2249.1996.d01-745.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ando K, Nakamura T. A method for producing a new medicine for both treating and preventing peptic ulcer diseases and gastritis and thus formulated medicines. European Patent Application 91310049.1; 1991 [Google Scholar]
- 30.Tarpila S, Korhonen H, Salminen S. 24th International Dairy Congress. Australia, Melbourne: 1994. Immune colostrum in the treatment of Helicobacter pylori gastritis; p. 293. Abstr. [Google Scholar]
- 31.Bogstedt AK, Casswall T, Björck L, et al. Generation of bovine anti-Helicobacter pylori antibodies for oral therapy. Submitted. doi: 10.1080/003655202762671242. [DOI] [PubMed] [Google Scholar]
- 32.Oona M, Maaroos HI, Rago T, Korhonen H, Salminen S. 9th Medical Symposium of the Yrjö Jahnsson Foundation. Sannäs, Finland: 1994. Use of bovine immune colostrum in the treatment of Helicobacter pylori infection in children. [Google Scholar]
- 33.Casswall TH, Sarker SA, Albert MJ, Bergström M, Björck L, Hammarström L. Treatment of Helicobacter pylori infections in rural Bangladesh with oral immunoglobulins from hyperimmune bovine colostrum: a pilot trial. Aliment Pharmacol Ther. 1998;12:563–6. doi: 10.1046/j.1365-2036.1998.00335.x. [DOI] [PubMed] [Google Scholar]
- 34.Barnes GL, Hewson PH, McLellan JA, Doyle LW, Knoches AML, Kitchen WH, Bishop RF. A randomised trial of oral gammaglobulin in low-birth-weight infants infected with rotavirus. Lancet. 1982;i:1371–3. doi: 10.1016/s0140-6736(82)92496-5. [DOI] [PubMed] [Google Scholar]
- 35.Guarino A, Guandalini S, Albano F, Mascia A, De Ritis G, Rubino A. Enteral immunoglobulins for treatment of protracted rotaviral diarrhea. Pediatr Inf Dis J. 1991;10:612–4. doi: 10.1097/00006454-199108000-00010. [DOI] [PubMed] [Google Scholar]
- 36.Guarino A, Cabani RB, Russo S, Albano F, Canani MB, Ruggeri FM, Donelli G, Rubino A. Oral immunoglobulins for treatment of acute rotaviral gastroenteritis. Pediatr. 1994;93:12–16. [PubMed] [Google Scholar]
- 37.Ventura A, Nassimbeni G, Martelossi S, Bohm P, D'Agaro PL. Experience with gamma globulins per os in the therapy and prevention of infectious diarrhea. Pediatr Med Chir. 1993;15:343–6. [PubMed] [Google Scholar]
- 38.Ebina T, Sato A, Umezu K, et al. Prevention of rotavirus infection by oral administration of cow colostrum containing anti-human rotavirus antibody. Med Microbiol Immunol. 1985;174:177–85. doi: 10.1007/BF02123694. [DOI] [PubMed] [Google Scholar]
- 39.Davidson GP, Daniels E, Nunan H, Moore AG, Whyte PBD, Franklin K, McCloud PI, Moore DJ. Passive immunization of children with bovine colostrum containing antibodies to human rotavirus. Lancet. 1989;2:709–12. doi: 10.1016/s0140-6736(89)90771-x. [DOI] [PubMed] [Google Scholar]
- 40.Turner RB, Kelsey DK. Abstract, 27th joint Conference on cholera and related diarrheal diseases. Virginia: 1991. Passive immunization for prevention of rotavirus infection in young infants. [Google Scholar]
- 41.Brunser O, Espinoza J, Figueroa G, Asaya M, Spencer E, Hilpert H, Link-Amster H, Brüssow H. Field trial of an infant formula containing anti-rotavirus and anti-Escherichia coli milk antibodies from hyperimmunized cows. J Pediatr Gastroenterol Nutr. 1992;15:63–72. doi: 10.1097/00005176-199207000-00010. [DOI] [PubMed] [Google Scholar]
- 42.Davidson G, Tam J, Kiruhakaran C. Passive protection against hospital acquired symptomatic rotavirus gastroenteritis in India and Hong Kong. Abstract 90, NASPGN-ESPGAN Meeting; 1994; Houston. [Google Scholar]
- 43.Hilpert H, Brüssow H, Mietens C, Sidoti J, Lerner L, Werchau H. Use of bovine milk concentrate containing antibody to rotavirus to treat rotavirus gastroenteritis in infants. J Infect Dis. 1987;156:158–66. doi: 10.1093/infdis/156.1.158. [DOI] [PubMed] [Google Scholar]
- 44.Mitra AK, Mahalanabis D, Ashraf H, Unicomb L, Eeckels R, Tzipori S. Hyperimmune cow colostrum reduces diarrhea due to rotavirus: a double-blind, controlled clinical trial. Acta Paediatr. 1995;84:996–1001. doi: 10.1111/j.1651-2227.1995.tb13814.x. [DOI] [PubMed] [Google Scholar]
- 45.Sarker SA, Casswall TH, Alam NH, Albert MJ, Fuchs GJ, Mahalanabis D, Hammarström L. Successful treatment of Rotavirus diarrhoea in children with immunoglobulin from bovine colostrum: a double blind, placebo-controlled clinical trial. Ped Inf Dis J. 1998;17:1149–54. doi: 10.1097/00006454-199812000-00010. [DOI] [PubMed] [Google Scholar]
- 46.Ball J, Tiann P, Zeng Q-Y, Morris A, Estes M. Age-dependent diarrhoea induced by a rotavirus nonstructural glycoprotein. Sci. 1996;276:101–4. doi: 10.1126/science.272.5258.101. [DOI] [PubMed] [Google Scholar]
- 47.Pan Q, Johansen K, Svensson L, Hammarström L. Antibody against rotaviral enterotoxin in bovine antibody preparations used in oral therapy. Immunol Cell Biol. 1997;75(Suppl.):A59. [Google Scholar]
- 48.Torres O, Cruz JR. Protection against Campylobacter diarrhea: role of milk IgA antibodies against bacterial surface antigens. Acta Paediatr. 1993;82:835–8. doi: 10.1111/j.1651-2227.1993.tb17622.x. [DOI] [PubMed] [Google Scholar]
- 49.Stern NJ, Meinersmann RJ, Dickerson HW. Influence of antibody treatment of Campylobacter jejuni on the dose required to colonize chicks. Avian Dis. 1990;34:595–601. [PubMed] [Google Scholar]
- 50.Tsubokura K, Berndtson E, Bogstedt A, Kaijser B, Kim M, Ozeki M, Hammarström L. Oral administration of antibodies as prophylaxis and therapy in Campylobacter jejuni infected chickens. Clin Exp Immunol. 1997;108:451–6. doi: 10.1046/j.1365-2249.1997.3901288.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Petruccelli BP, Murphy GS, Sanchez JL, et al. Treatment of traveler's diarrhea with ciprofloxacin and loperamide. J Inf Dis. 1992;165:557–60. doi: 10.1093/infdis/165.3.557. [DOI] [PubMed] [Google Scholar]
- 52.Kelly CP, Pothoulakis C, LaMont JT. Current concepts: Clostridium difficile colitis. N Engl J Med. 1994;330:257–62. doi: 10.1056/NEJM199401273300406. [DOI] [PubMed] [Google Scholar]
- 53.Lyerly D, Frivan H, Wilkins T. Clostridium difficile: its disease and toxins. Clin Microbiol Rev. 1988;1:1–88. doi: 10.1128/cmr.1.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kelly CP, LaMont JT. Treatment of Clostridium difficile diarrhea and colitis. In: Wolfe WM, LaMont JT, editors. Gastrointestinal pharmacotherapy. Philadelphia: W. B. Saunders Co; 1993. pp. 199–212. [Google Scholar]
- 55.Leung DYM, Kelly CP, Boguniewicz M, Pothoulakis C, LaMont JT, Flores A. Treatment with intravenously administered gamma globulin of chronic relapsing colitis induced by Clostridium difficile toxin. J Pediat. 1991;118:633–7. doi: 10.1016/s0022-3476(05)83393-1. [DOI] [PubMed] [Google Scholar]
- 56.Kelly CP, Pothoulakis C, Orellana J, LaMont JT. Human colonic aspirates containing immunoglobulin A antibody to Clostridium difficile toxin A inhibit toxin A-receptor binding. Gastroenterol. 1992;102:35–40. doi: 10.1016/0016-5085(92)91781-x. [DOI] [PubMed] [Google Scholar]
- 57.McFarland LV, Mulligan ME, Kwok RYY, Stamm WE. Nosocomial acquisition of Clostridium difficile infection. N Engl J Med. 1989;320:204–10. doi: 10.1056/NEJM198901263200402. [DOI] [PubMed] [Google Scholar]
- 58.Fekety R. Zak O, Sande M, editors. Animal models of antibiotic-induced colitis. Experimental models in antimicrobial chemotherapy. 1986;2:61–72. [Google Scholar]
- 59.Lyerly DM, Bostwick EF, Binion SB, Wilkins TD. Passive immunization against disease caused by Clostridium difficile by use of bovine immunoglobulin G concentrate. Infect Immun. 1991;59:2215–8. doi: 10.1128/iai.59.6.2215-2218.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kelly CP, Pothoulakis C, Vavva F, Castagliuolo I, Bostwick EF, O'Keane JC, Keates S, LaMont JT. Anti-Clostridium difficile bovine immunoglobulin concentrate inhibits cytotoxicity and enterotoxicity of C. difficile toxins. Antimicrob Agents Chemother. 1996;40:373–9. doi: 10.1128/aac.40.2.373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kelly CP, Chetham S, Keates S, Bostwick EF, Roush AM, Castagliuolo I, LaMont JT, Pothoulakis C. Survival of anti-Clostridium difficile bovine immunoglobulin concentrate in the human gastrointestinal tract. Antimicrob Agents Chemother. 1997;41:236–41. doi: 10.1128/aac.41.2.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Warny M, Fatimi A, Bostwick EF, Laine DC, LaMont JT, Pothoulakis C, Kelly CP. Bovine immunoglobulin concentrate—C. difficile retains C. difficile toxin neutralizing activity after passage through the human stomach and small intestine. Gut. 1999 doi: 10.1136/gut.44.2.212. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Plorde JJ. Cryptosporidiosis. In: Wilson JD, Braunwald E, Isselbacher KJ, editors. Principles of internal medicine. 12. New York: McGraw-Hill; 1991. pp. 803–5. [Google Scholar]
- 64.Petersen C. Cryptosporidiosis in patients infected with the human immuno-deficiency virus. Clin Infect Dis. 1992;15:903–9. doi: 10.1093/clind/15.6.903. [DOI] [PubMed] [Google Scholar]
- 65.Soave R, Weikel C. Cryptosporidium and other protozoa including Isospora, Sarcocystis, Balantidium coli and Blastocystis. In: Mandall GL, Douglas RG, Bennett JE, editors. Principles and practices of infectious disease. 3. New York: Churchill Livingstone Inc; 1990. pp. 2122–30. [Google Scholar]
- 66.Laughon BE, Druckman DA, Vernon A, Quin TC, Polk BF, Modlin JF. Prevalence of enteric pathogens in homosexual men with and without acquired immunodeficiency syndrome. Gastroenterol. 1988;94:984–92. doi: 10.1016/0016-5085(88)90557-4. [DOI] [PubMed] [Google Scholar]
- 67.Crawford FG, Vermund SH. Human cryptosporidiosis. Crit Rev Microbiol. 1988;16:113–59. doi: 10.3109/10408418809104469. [DOI] [PubMed] [Google Scholar]
- 68.Janoff EN, Reller LB. Cryptosporidium species, a protean protozoan. J Clin Microbiol. 1987;25:967–75. doi: 10.1128/jcm.25.6.967-975.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Nord J, Ma P, DiJohn D, Tzipori S, Tacket CO. Treatment with bovine hyperimmune colostrum of cryptosporidial diarrhea in AIDS patients. AIDS. 1990;4:581–4. doi: 10.1097/00002030-199006000-00015. [DOI] [PubMed] [Google Scholar]
- 70.Tzipori S, Roberton D, Cooper DA, White L. Chronic cryptosporidial diarrhea and hyperimmune cow colostrum. Lancet. 1987;2:344–5. doi: 10.1016/s0140-6736(87)90944-5. [DOI] [PubMed] [Google Scholar]
- 71.Angus KW. Cryptosporidiosis and AIDS. Baillers Clin Gastroenterol. 1990;4:425–41. doi: 10.1016/0950-3528(90)90010-e. [DOI] [PubMed] [Google Scholar]
- 72.Ungar BLP, Ward DJ, Fayer R, Quinn CA. Cessation of cryptosporidium-associated diarrhea in an acquired immunodeficiency syndrome patient after treatment with hyperimmune bovine colostrum. Gastroenterol. 1990;98:486–9. doi: 10.1016/0016-5085(90)90842-o. [DOI] [PubMed] [Google Scholar]
- 73.Fayer R, Perryman LE, Riggs MW. Hyperimmune bovine colostrum neutralizes cryptosporidium sporozoites and protects mice against oocyst challenge. J Parasitol. 1989;75:151–3. [PubMed] [Google Scholar]
- 74.Perryman LE, Riggs MW, Mason PH, Fayer R. Kinetics of Cryptosporidium parvum sporozoite neutralization by monoclonal antibodies, immune bovine serum, and immune bovine colostrum. Infect Immun. 1990;58:257–9. doi: 10.1128/iai.58.1.257-259.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Fayer R, Guidry A, Blackburn BL. Immunotherapeutic efficacy of bovine colostral immunoglobulins from a hyperimmunized cow against cryptosporidiosis in neonatal mice. Infect Immun. 1990;58:2962–5. doi: 10.1128/iai.58.9.2962-2965.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Saxon A, Weinstein W. Oral administration of bovine colostrum anti-cryptosporidia antibody fails to alter the course of human cryptosporidiosis. J Parasitol. 1987;73:413–5. [PubMed] [Google Scholar]
- 77.Tzipori S, Roberton D, Chapman C. Remission of diarrhoea due to cryptosporidiosis in an immunodeficient child treated with hyperimmune bovine colostrum. Br Med J. 1986;293:1276–7. doi: 10.1136/bmj.293.6557.1276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Greenberg PD, Cello JP. Treatment of severe diarrhea caused by Cryptosporidium parvum with oral bovine immunoglobulin concentrate in patients with AIDS. J AIDS. 1996;13:348–54. doi: 10.1097/00042560-199612010-00008. [DOI] [PubMed] [Google Scholar]
- 79.Tacket CO, Losonsky G, Link H, Hoang Y, Guesry P, Hilpert H, Levine MM. Protection by milk immunoglobulin concentrate against oral challenge with enterotoxigenic Escherichia coli. N Engl J Med. 1988;318:1240–3. doi: 10.1056/NEJM198805123181904. [DOI] [PubMed] [Google Scholar]
- 80.Freedman DJ, Tacket CO, Delehanty A, Maneval DR, Nataro J, Crabb JH. Milk immunoglobulin with specific activity against purified colonization factor antigens can protect against oral challenge with enterotoxigenic Escherichia coli. J Infect Dis. 1998;177:662–7. doi: 10.1086/514227. [DOI] [PubMed] [Google Scholar]
- 81.Lissner R, Schmidt H, Karch H. A standard immunoglobulin press produced from bovine colostra shows antibody reactivity and neutralization activity against Shiga-like toxins and EHEC-hemolysin of Escherichia coli O157:H7. Infect. 1996;24:378–83. doi: 10.1007/BF01716084. [DOI] [PubMed] [Google Scholar]
- 82.Guerrant RL. Principles and syndromes of enteric infection. In: Mandall GL, Bennett JE, Dolin R, editors. Principles and practices of infectious disease. 4. New York: Churchill Li vingstone Inc; 1995. pp. 945–62. [Google Scholar]
- 83.Mietens C, Keinhorst H, Hilpert H, Gerber H, Amster& H, Pahud JJ. Treatment of infantile E. coli gastroenteritis with specific bovine anti-E. coli milk immunoglobulins. Eur J Pediatr. 1979;132:239–52. doi: 10.1007/BF00496847. [DOI] [PubMed] [Google Scholar]
- 84.Hilpert H, Gerber H, De Peyer E, Nusslé D. Gastrointestinal passage of bovine anti-E. coli milk immunoglobulins (Ig) in infants. Nestle Res News. 1974;75:134–8. [Google Scholar]
- 85.Mousavi M, Rabbani H, Pihlstedt L, Hammarström L. Characterization of the gene for the membrane and secretory form of the IgM heavy chain constant region gene of the cow. Immunol. 1998;93:581–8. doi: 10.1046/j.1365-2567.1998.00444.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rabbani H, Brown WR, Butler JEB, Hammarström L. Genetic polymorphism of bovine IgG3. Immunogenet. 1997;46:326–31. doi: 10.1007/s002510050279. [DOI] [PubMed] [Google Scholar]
- 87.Brown W, Rabbani H, Butler J, Hammarström L. Characterization of the gene for bovine IgA. Immunol. 1997;91:1–6. doi: 10.1046/j.1365-2567.1997.00227.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mousavi M, Rabbani H, Hammarström L. Characterization of the bovine ε gene. Immunol. 1997;92:369–73. doi: 10.1046/j.1365-2567.1997.00352.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Saif LJ, Smith KL, Landmeier BJ, Bohl EH, Theil KW, Todhunter DA. Immune response of pregnant cows to bovine rotavirus immunization. Am J Vet Res. 1984;45:49–58. [PubMed] [Google Scholar]
- 90.Yolken RH, Losonsky GA, Vonderfecht S, Leister F, Wee SB. Antibody to human rotavirus in cow's milk. N Engl J Med. 1985;312:605–10. doi: 10.1056/NEJM198503073121002. [DOI] [PubMed] [Google Scholar]
- 91.Blum PM, Phelps DL, Ank BJ, Krantman HJ, Stiehm ER. Survival of oral human serum globulin in the gastrointestinal tract of low birth weight infants. Pediat Res. 1981;15:1256–60. doi: 10.1203/00006450-198109000-00006. [DOI] [PubMed] [Google Scholar]
- 92.Losonsky GA, Johnson JP, Winkelstein JA, Yolken RH. Oral administration of human serum immunoglobulin in immunodeficient patients with viral gastroenteritis. J Clin Invest. 1985;76:2362–7. doi: 10.1172/JCI112248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Leresche E, Paunier L, Andrejevic G. Stabilité dans le tractus digestif du nourrisson et tolerance clinique dímmunoglobulines bovines a teneur anticorps elevee contre les E. coli pathogenes, administrees par voie orale. Comp Immunol Microbiol Infect Dis. 1979;1:205–20. doi: 10.1016/0147-9571(79)90049-3. [DOI] [PubMed] [Google Scholar]
- 94.Tacket CO, Binion SB, Bostwick E, Losonsky G, Roy MJ, Edelman R. Efficacy of bovine milk immunoglobulin concentrate in preventing illness after Shigella flexneri challenge. Am J Trop Med Hyg. 1992;17:276–83. doi: 10.4269/ajtmh.1992.47.276. [DOI] [PubMed] [Google Scholar]
- 95.Zinkernagel RM, Hilpert H, Gerber H. The digestion of colostral bovine immunoglobulins in infants. Experentia. 1972;28:741. [Google Scholar]
- 96.Lodinova-Zadníkova R, Korych B, Bartakova Z. Treatment of gastrointestinal infections in infants by oral administration of colostral antibodies. Die Nahrung. 1987;31:465–7. doi: 10.1002/food.19870310537. [DOI] [PubMed] [Google Scholar]
- 97.McClead RE, Butler T, Rabbani GH. Orally administered bovine colostral anti-cholera toxin antibodies: results of two clinical trials. Am J Med. 1988;85:811–6. doi: 10.1016/s0002-9343(88)80026-3. [DOI] [PubMed] [Google Scholar]
- 98.Bogstedt AK, Robertson AK, Hammarström L. Survival of immunoglobulins from different species through the gastrointestinal passage of healthy adult volunteers: implications for human therapy. Antimicrob Agents Chemother. 1997;41:2320. doi: 10.1128/aac.41.10.2320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Roos N, Mahe S, Benamouzig R, Sick H, Rautureau J, Tome D. 15N-labeled immunoglobulins from bovine colostrum are partially resistant to digestion in human intestine. J Nutr. 1995;125:1238–44. doi: 10.1093/jn/125.5.1238. [DOI] [PubMed] [Google Scholar]
- 100.Schaller JP, Saif LJ, Cordle CT, Candler EL, Winship TR, Smith KL. Prevention of human rotavirus-induced diarrhea in gnotobiotic piglets using bovine antibody. J Inf Dis. 1992;165:623–30. doi: 10.1093/infdis/165.4.623. [DOI] [PMC free article] [PubMed] [Google Scholar]


