Abstract
CD27 molecules are shown to be essential in the regulation of the death, activation and differentiation of T and B cells. However, the influence of CD27 on cytotoxic T-cell function remains obscure. Autologous EBV transformed B-cell lines (LCL), which highly express CD27 ligand CD70, here stimulated T cells and induced the cytotoxic T-lymphocyte (CTL) activity via T-cell antigen receptors (TCR). The cytotoxicity against LCL was diminished when anti-CD70 blocking MoAb was added initially in the culture. Resting T cells killed more CD70-transfected P815 cells than wild type P815 cells in the presence of anti-CD3 MoAb as measured by a 4-h 51Cr release assay, and the cytotoxicity of both of the cell populations completely disappeared in the presence of concanamycin A (CMA). The expression of the perforin by the LCL-induced CTL in the presence of anti-CD70 blocking MoAb was diminished as compared with that without the blockage of CD27/CD70 interactions. The CTL induced by LCL did not kill Fas-transfected WR cells. CD27 signalling in the T cells did not affect Fas ligand (FasL) mRNA expression, LAK activity and IFN-γ synthesis in humans. Our data demonstrate that CD27/CD70 interactions enhance the cytotoxicity of CTL in the induction phase through enhancement of killing activity induced via the perforin-dependent mechanism, but not via the Fas/FasL system.
Keywords: cytotoxic T-lymphocyte, CD27, CD70, perforin
Introduction
Cytotoxic T-lymphocytes (CTL) are deeply involved not only in the elimination of pathogens, such as viruses, but also in the onset of autoimmune diseases or malignant tumours [1]. CTL recognize antigens by T-cell receptors (TCR) and destroy target cells. In recent years, it has been demonstrated that two pathways, perforin/granzyme B and Fas/Fas ligand (FasL), are important in T-cell cytotoxicity [2–5]. Perforin is a 70-kD protein and is found in granules of T and NK cells. Perforin serves to facilitate the entry of apoptosis-inducing granzymes, also stored in the perforin granules, into the target cells [6–8]. In Fas/FasL-mediated apoptosis, FasL expressed on killer T cells stimulates the Fas molecules of the target cells and induces DNA fragmentation, thus leading to their apoptosis [9–11]. The induction and activation of CTL start when TCR recognize a specific antigen presented by antigen-presenting cells. However, sufficient signals are created when various supplemental receptors are stimulated at the same time. CD28; CTLA-4/CD80; CD86 interactions play an important role as a type of costimulatory factor. Azuma et al. [12] reported that the stimulation of CD28 molecule on resting T cells under CD3 stimulation could induce the marked cytotoxic activity.
Recent researches on the TNF/TNF receptor family have demonstrated that TNFs are closely involved in immune responses as costimulatory factors. It is documented that CD27/CD70 interactions are important for T-cell activation in addition to such B-cell functions as plasma cell differentiation, immunoglobulin production and immunological memory [13,14]. It is reported that CD27 signalling enhances NK cell activity [15] and cytotoxicity of T cells in the presence of PHA [16]. In in vivo study, mice expressing constitutive CD70 on B cells had hygher peripheral T cell numbers that showed increased differentiation toward effector-type T cells [17]. A strong inverse correlation of killing activity between perforin expression and CD27 expression has been reported [18–20]. Brown et al. [21] suggest that Ag-specific murine CTL generation is enhanced by recombinant human CD70 expressed on SF9 cell membranes.
The present study utilized an autologous antigen-specific CTL induction system employing EBV transformed B-cell lines (LCL), which strongly express CD70, and analysed the effect of CD27/CD70 interactions on the induction of the MHC-restricted cytotoxicity in humans.
Materials and methods
Abs and reagents
Anti-CD70 blocking MoAb (HNE51; IgG1) and anti-CD3 MoAb (UCHT-1; IgG1) were purchased from Immunotech (Marseille, France). Anti-Fas MoAb (7C11; IgM), which induces apoptosis of Fas-expressing cells, was provided by Dr J. Ritz (Dana-Farber Cancer Institute, Boston, MA, USA). Anti-CD27 MoAb (8H5; IgG1), which does not block the ligation of CD27/CD70 was provided by Dr C. Morimoto (Dana-Farber Cancer Institute, Boston, MA, USA). FITC- and PE-conjugated anti-CD4 MoAb (Leu3a; IgG1), PE-conjugated anti-CD23 MoAb (MHM6; IgG1), and FITC-conjugated anti-CD8 MoAb (Leu2a; IgG1) were purchased from DAKO Japan (Tokyo, Japan). PE-conjugated antiperforin MoAb was purchased from Pharmingen (San Diego, CA, USA). FITC-conjugated anti-Fas MoAb and FITC-conjugated anti-IgG1 MoAb were purchased from MBL (Nagoya, Japan). PerCP-conjugated anti-CD3 MoAb was purchased from Becton Dickinson (San Jose, CA, USA). Conjugation of biotin to anti-CD70 MoAb (HNE51) and anti-CD27 MoAb (8H5) were performed by the standard technique using N-hydroxysuccinimido-biotin (Sigma, St. Louis, MO, USA). G418 was purchased from Life Technologies (Grand Island, NY, USA), human IL-2 from Takeda Seiyaku Co. (Osaka, Japan), and concanamycin A (CMA) from Wako Pure Chemical Industries (Osaka, Japan).
Cell preparation
Human peripheral blood mononuclear cells (PBMNCs) were isolated from healthy donors by Ficoll-Hypaque (Pharmacia, Piscataway, NJ, USA) density gradient centrifugation. They were then separated into sheep erythrocyte rosette-positive (E+) and negative populations [22]. The purity of T cells included in E+ cells was more than 95%.
Immunofluoresence
Two- or three-colour flow cytometric analysis was performed by a FACScan cytofluorometer (Becton Dickinson, Mountain View, CA, USA). LCL were stained with anti-CD70-biotin plus streptavidin-PE, anti-CD23-PE, or anti-CD27-PE. Wild/P815 and CD70/P815 were stained with anti-CD70-biotin plus streptavidin-PE. E+ cells were stained with anti-CD8-FITC and anti-CD4-PE, before and after coculturing with LCL in the presence or absence of anti-CD70 blocking MoAb (5 µg/ml). The TCR-Vβ region of the CTL was also studied using the TCR-Vβ repertoire kit (Immunotech). The isotype-matched direct-labelled controls were included in all experiments, always at levels less than 8% positive in cells.
As for the investigation of perforin expression, E+ cells and cultured T cells were stained with anti-CD3-PerCP, or anti-CD4-FITC or anti-CD8-FITC and anti-CD27-biotin plus streptavidin-PerCP before permeabilization. Then, perforin expression was measured by the staining with antiperforin-PE after permeabilization. They were then washed once in PBS, and fixed in 1 ml of PBS containing 4% paraformaldehyde for 15 min at room temperature. After washing twice with 1% FCS in PBS, the cells were incubated with 1 ml of PBS containing 0·1% Triton-X-100 (Sigma Chemical Co., St Louis, MO, USA) and 0·1% BSA for 6 min at room temperature. After washing with 1% FCS in PBS, the cells were stained with PE-conjugated antiperforin MoAb or isotype-matched mouse IgG for 20 min at 4°C. Samples were analysed by flow cytometry.
Preparation of LCL
PBMNCs were separated from whole blood by Ficoll-Hypaque density gradient. PBMNCs (5 × 106) were incubated with supernatant obtained from culture medium of EBV-producing B95·8 marmoset cells and 10 ng/ml of cyclosporin A (Sandoz Pharmaceuticals, Surrey, UK) in RPMI1640 (Life Technologies, Paisley, UK) containing 10% FCS (Hyclone, Logan, UT, USA) and 2 mm l-glutamine at 37°C. After 2 or 3 weeks of culture, large clumps appeared; subsequent subculturing and harvesting of the cells were carried out as needed.
Preparation of CD70-transfectants
Preparation of cDNA of CD70 was described previously [23,24]. Briefly, total RNA was purified by the single-step method from the CD70+ human B cell line (Daudi cells). After amplification by RT-PCR, the cDNA was digested and ligated with the mammalian expression vector BCMGSneo (a gift from Drs H. Karasuyama, A. Kudo and F. Melchers) [25]. The constructed DNA was transfected into the mouse mastocytoma P815 and murine pre-B cell 300–19 by electroporation using a Gene Pulser (Bio-Rad, Hercules, CA, USA). The transfectants were selected by RPMI1640 medium supplemented with 10% FCS in the presence of G418 (1 mg/ml).
Cytotoxic assay
Cytotoxicity against wild/P815 or CD70/P815 cells was tested in a 4-h 51Cr-release assay. Briefly, E+ cells were mixed with 51Cr-labelled wild/P815 or CD70/P815 cells in a total volume of 200 µl in 96-well U-bottom microtitre plates at various effector: target (E: T) ratios ranging from 40 : 1 to 10 : 1 in the presence of anti-CD3 MoAb (0·2 µg/ml). To evaluate the role of perforin, the effector cells were pretreated with 1000 nm CMA for 30 min. After 4 h of incubation at 37°C in a 5% CO2 atmosphere, 100 µl of supernatant was harvested and counted in a gamma counter (Beckman, Palo Alto, CA, USA).
E+ cells were incubated at 2 × 106/well with 8 × 105/well LCL that had been fixed with 1% formaldehyde, in the presence or absence of anti-CD70 blocking MoAb (5 µg/ml) for 12 days. CTL activity was measured against LCL in a 4-h 51Cr-release assay, and against murine T cell lymphoma cell line (wild/WR) and Fas-transfectants (Fas/WR) in a 12-h 51Cr-release assay. Briefly, the effector cells were mixed with 51Cr-labelled LCL or Fas/WR in a total volume of 200 µl in 96-well U-bottom microtitre plates (Nunc, Roskilde, Denmark) at various effector : target (E : T) ratios ranging from 40 : 1 to 20 : 1. To evaluate the role of perforin, the effector cells were pretreated with 1000 nm CMA for 30 min. CMA is an inhibitor of vacuolar type H+-ATPase to perturb the intracellular transport of glycoproteins and inhibit the function of the lytic granules containing perforin and granzymes [26,27]. After 4 h or 12 h of incubation at 37°C in a 5% CO2 atmosphere, 100 µl of supernatant was harvested and counted in a gamma counter.
Cytotoxicity of activated T cells, expressed as percentage cytotoxicity, was calculated by the following formula:
Lynphokine-activated killer (LAK) activity against Fas/WR
E+ cells were incubated at 2 × 106/well with 8 × 105/well mock-transfectants (mock/300–19) or CD70-transfectants (CD70/300–19) fixed with 1% formaldehyde in the presence of IL-2 (25 ng/ml) for 5 days with or without anti-CD70 blocking MoAb (5 µg/ml). These activated T cells were mixed with 51Cr-labelled Fas/WR in a total volume of 200 µl in 96-well U-bottom microtitre plates at effector : target (E : T) ratios ranging from 40 : 1 to 20 : 1. After 12 h of incubation at 37°C in a 5% CO2 atmosphere, 100 µl of supernatant was harvested and counted in a gamma counter.
Reverse transcription polymerase chain reaction (RT-PCR) of FasL mRNA
Total RNA was extracted from E+ cells (1 × 106 cells) by the acid-guanidium thiocyanate-phenol-chloroform (AGPC) method using a RNAzol rapid RNA purification kit (Biotecx, Houston, TX, USA) as previously described [28]. Briefly, first-stranded cDNA copies were synthesized by using M-MLV reverse transcriptase (Superscript II; GibcoBRL, Gaithersburg, MD, USA) with oligo (dT) (GibcoBRL) as a primer in a total volume of 20 µl, and was used for PCR reaction. Primer sequences were as follows: FasL sense primer 5′-AAGGGTGGCCTTGTGATC-3′ and antisense primer 5′-GACCAGAGAGAGCTCAGA-3′, β2-microglobulin sense primer 5′-GCTATGTGTCTGGGTTTCAT-3′ and antisense primer 5′-CCCACTTAACTATCTTGGGC-3′. 2 µl of cDNA was amplified in PCR under the following conditions using respective primers and 5 U of Taq DNA polymerase (GibcoBRL): 94°C for 1 minute, 55°C for 2 min and 72°C for 3 min; 30 cycles. After amplification, PCR products were separated by electrophoresis on 1·2% agarose gel containing ethidium bromide and visualized by UV light illumination.
Statistical analysis
Statistical analysis of the specific cytotoxicity in each experiment was performed by two-sample t-test. Differences with P < 0·05 were considered significant.
Results
Effects of CD27/CD70 interactions on resting T-cell cytotoxicity
To investigate whether or not CD27/CD70 interactions affect the cytotoxicity of resting T cells, we transfected CD70 gene into P815 cells and prepared CD70-transfectants (CD70/P815). CD70 expression was not seen on wild type P815 cells (wild/P815), but after the transfection of CD70, the expression became marked (data not shown). We measured the ability of resting T cells to kill wild/P815 and CD70/P815 for 4 h in the presence of anti-CD3 MoAb. In the presence of anti-CD3 MoAb, resting T cells killed wild/P815, and the cytotoxicity against CD70/P815 was stronger than that against wild/P815 (Fig. 1a). This enhancement in the cytotoxicity was mostly negated by the addition of CMA (Fig. 1a). These findings confirm that CD27/CD70 interactions enhance the cytotoxicity of resting T cells in the presence of CD3 signalling via perforin-mediated mechanism.
Fig. 1.
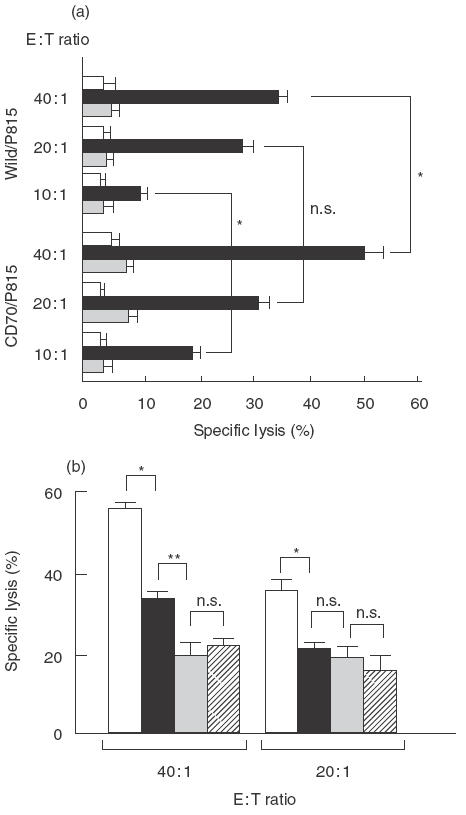
(a)CD27-mediated cytotoxicity by resting T cells. E+ cells were preincubated with or without CMA (1000 nm) for 30 min The pretreatment cells were then tested for the cytotoxicity against wild/P815 (upper) or CD70/P815 (lower) (1 × 104/well) in the presence of 0·2 µg/ml anti-CD3 MoAb at E : T ratios of 40 : 1, 20 : 1, and 10 : 1 in a 4-h 51Cr-release assay. □ Medium, ▪ anti-CD3 MoAb,  anti-CD3MoAb + CMA. (b) Effects of CD27/CD70 interactions on cytotoxic T-lymphocyte activity. E+ cells (2 × 106/well) were cocultured with autologous LCL (8 × 105/well) in the presence (▪,
anti-CD3MoAb + CMA. (b) Effects of CD27/CD70 interactions on cytotoxic T-lymphocyte activity. E+ cells (2 × 106/well) were cocultured with autologous LCL (8 × 105/well) in the presence (▪,  ) of 5 µg/ml anti-CD70 MoAb or in the absence (□,
) of 5 µg/ml anti-CD70 MoAb or in the absence (□,  ) of anti-CD70 MoAb for 12 days. The pretreated cells were tested for the cytotoxicity against autologous LCL (1 × 104/well) at E : T ratios of 40 : 1 and 20 : 1 with (
) of anti-CD70 MoAb for 12 days. The pretreated cells were tested for the cytotoxicity against autologous LCL (1 × 104/well) at E : T ratios of 40 : 1 and 20 : 1 with ( ,
,  ) or without (□, ▪) CMA (1000 nm) in a standard 4-h 51Cr-release assay. Results are expressed as the mean percentage of cytotoxicity ± SD of triplicates. Representative data from three experiments using different donors are shown. Statistical analysis: two-sample t-test; n.s., not significant; *P < 0·01; **P < 0·05.
) or without (□, ▪) CMA (1000 nm) in a standard 4-h 51Cr-release assay. Results are expressed as the mean percentage of cytotoxicity ± SD of triplicates. Representative data from three experiments using different donors are shown. Statistical analysis: two-sample t-test; n.s., not significant; *P < 0·01; **P < 0·05.
Effects of CD27/CD70 interactions on the induction of cytotoxic activity
In order to ascertain effects of CD27/CD70 interactions on CTL activity, we assessed CTL activity induced by autologous LCL on which CD70 were strongly expressed (data not shown), with or without anti-CD70 blocking MoAb. At 12 days after the coculturing of T cells and fixed LCL, the ratio of CD4/CD8 populations of activated T cells was similar to that of resting T cells, and there was no change in the CD4/CD8 ratio with or without anti-CD70 blocking MoAb, and CD3−/CD56+ NK cells were <1% (data not shown). CTL activity induced by LCL with the initial addition of anti-CD70 blocking MoAb in the culture was lower than that without the antibodies (Fig. 1b). These findings suggest that CD27/CD70 interactions act during the CTL induction to influence their cytotoxicity. When CMA, an inhibitor of vacuolar type H+-ATPase, was added to the effector cells before the contact with the target cells, their cytotoxicity was greatly suppressed with or without anti-CD70 blocking MoAb (Fig. 1b). Therefore, perforin/granzyme B appears to be closely involved in CD27/CD70-mediated cytotoxicity.
Perforin expression in CTL
We investigated perforin expression in the induced T cells by autologous LCL using flow cytometry with or without anti-CD70 blocking MoAb. Very low levels of perforin expression in resting CD4+ and CD8+ T cells were found (data not shown). 12 days after the stimulation, the levels of the perforin expression with anti-CD70 blocking MoAb were lower than that without anti-CD70 blocking MoAb (Fig. 2). Therefore, CD27/CD70 interactions appear to enhance the cytotoxicity by, at least in some part, facilitating the production of perforin.
Fig. 2.
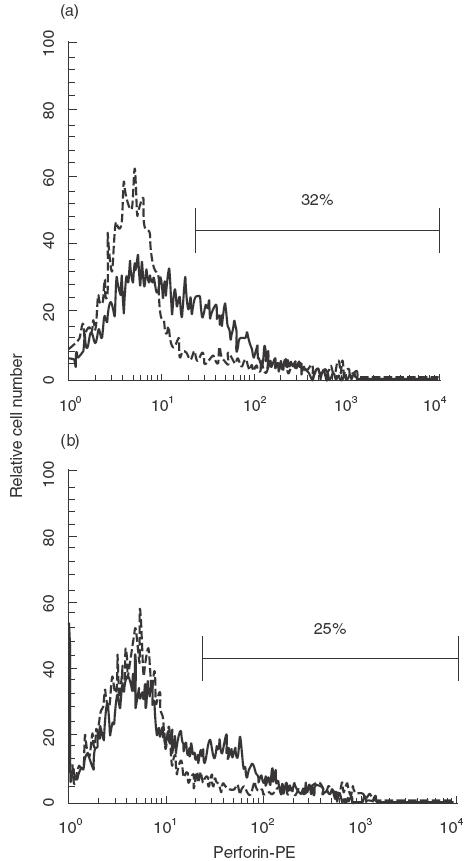
The expression of perforin in cytotoxic T lymphocyte. E+ cells were cocultured with autologous LCL in the absence of or in the presence of (b) 5 µg/ml of anti-CD70 MoAb for 12 days. The pretreated cells were stained with antiperforin-PE (——) after permeabilization. Isotype-matched PE-labelled mouse IgG MoAb was used as the negative control (·········). Representative data from three experiments using different donors are shown.
Inverse correlation of the expression of CD27 and perforin in CTL
We next determined the influence of CD27/CD70 interactions on perforin expression in CTL. Since there was a strong inverse correlation between perforin expression and CD27 expression [18], we analysed the expression of CD27 and perforin in CTL. For this purpose, E+ cells were cocultured with LCL in the presence or absence of anti-CD70 blocking MoAb, and perforin expression was assayed in CD27+ and CD27− cells. At 12 days after the coculture with LCL, perforin expression in the cells without anti-CD70 blocking MoAb was more increased than those in the presence of anti-CD70 blocking MoAb, and perforin expression was especially recognized in CD8+CD27− T cells (Fig. 3).
Fig. 3.
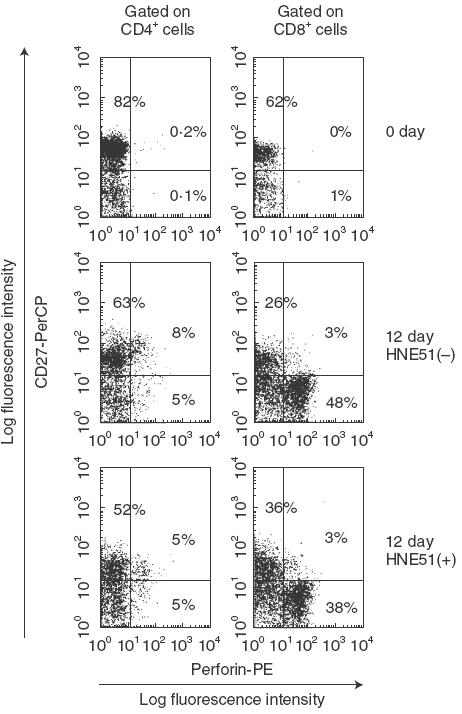
Correlation of the expression of CD27 and perforin on CTL. E+ cells were cocultured with autologous LCL in the absence of or in the presence of 5 µg/ml of anti-CD70 MoAb for 12 days. E+ cells were stained with anti-CD27-biotin plus streptavidin-PerCP and anti-CD4-FITC or anti-CD8-FITC before and after cocultured with LCL. Then, these cells were permeabilized as described in Materials and Methods, and stained with antiperforin-PE. Similar data were obtained in three independent experiments.
These data indicate that CD27 expression is diminished with the increase of CTL activity and the increased expression of perforin is found particulary in CD8+CD27− T cells.
Fas/FasL-mediated cytotoxicity
We investigated whether or not Fas/FasL interactions are involved in LCL-induced T-cell cytotoxicity using Fas-transfectants as target cells. Fas/WR strongly expressed Fas antigen, whereas Fas expression was not seen on wild/WR (Fig. 4a). When Fas molecules on LCL or wild/WR were stimulated with anti-Fas antibodies, the killing of LCL or wild/WR (data not shown) was mild, whereas Fas/WR killing was marked (about 90%) (Fig. 4b). Although LCL-induced CTL were cytotoxic against LCL, they showed no cytotoxicity against Fas/WR (Fig. 4b) or wild/WR (data not shown). These findings clarify that the CD27/CD70-mediated cytotoxicity induced by LCL is not mediated via the Fas/FasL pathway.
Fig. 4.
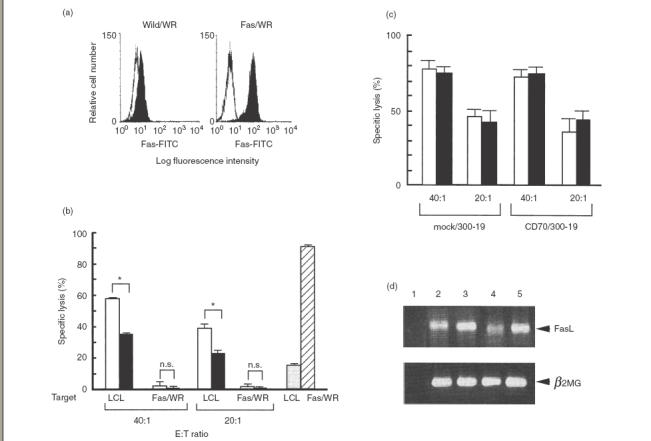
FasL-mediated cytotoxicity of CTL and effects of CD27/CD70 interaction on LAK activity. (a) The expression of Fas antigen on Fas/WR. Wild/WR and Fas/WR were stained with anti-Fas-FITC (——) and analysed by a FACScan. Isotype-matched FITC-labelled mouse IgG MoAb was used as the negative control (··········). (b) FasL-mediated cytotoxicity of CTL. E+ cells (2 × 106/well) were cocultured with autologous LCL (8 × 105/well) in the presence (▪) of or in the absence (□) of 5 µg/ml anti-CD70 MoAb for 12 days. The pretreated cells were tested for the cytotoxicity against autologous LCL in a 4-h 51Cr-release assay or against Fas/WR in a 12-h 51Cr-release assay at E : T ratios of 40 : 1 and 20 : 1. Fas/WR ( ) and LCL (
) and LCL ( ) were mixed with anti-Fas MoAb (7C11) which induced apotosis of the Fas-expressing cells in a 12- h 51Cr-release assay for positive controls. Results are expressed as the mean percentage of cytotoxicity ± SD of triplicates. Similar results were obtained in three independent experiments. (c) IL-2 (25 ng/ml) activated E+ cells (2 × 106/well) were cocultured with mock/300–19 or CD70/300–19 (8 × 105/well) in the absence (□) of anti-CD70 MoAb or in the presence (▪) for 5 days. The pretreated cells were tested for the cytotoxicity against Fas/WR at E : T ratios of 40 : 1 and 20 : 1 in a 12-h 51Cr-release assay. Results are expressed as the mean percentage of cytotoxicity ± SD of triplicates. Similar results were obtained in three independent experiments. (d) The expression of FasL and β2-microglobulin were analysed by RT-PCR as described in the Materials and Methods. Lane 1: no DNA, lane 2 : 24 h IL-2 activated E+ cells with mock/300–19, lane 3 : 48 h IL-2 activated E+ cells with mock/300–19, lane 4 : 24 h IL-2 activated E+ cells with CD70/300–19, and lane 5 : 48 h IL-2 activated E+ cells with CD70/300–19. Representative data from three independent experiments are shown. Statistical analysis: two-sample t-test; n.s., not significant; *P < 0·01.
) were mixed with anti-Fas MoAb (7C11) which induced apotosis of the Fas-expressing cells in a 12- h 51Cr-release assay for positive controls. Results are expressed as the mean percentage of cytotoxicity ± SD of triplicates. Similar results were obtained in three independent experiments. (c) IL-2 (25 ng/ml) activated E+ cells (2 × 106/well) were cocultured with mock/300–19 or CD70/300–19 (8 × 105/well) in the absence (□) of anti-CD70 MoAb or in the presence (▪) for 5 days. The pretreated cells were tested for the cytotoxicity against Fas/WR at E : T ratios of 40 : 1 and 20 : 1 in a 12-h 51Cr-release assay. Results are expressed as the mean percentage of cytotoxicity ± SD of triplicates. Similar results were obtained in three independent experiments. (d) The expression of FasL and β2-microglobulin were analysed by RT-PCR as described in the Materials and Methods. Lane 1: no DNA, lane 2 : 24 h IL-2 activated E+ cells with mock/300–19, lane 3 : 48 h IL-2 activated E+ cells with mock/300–19, lane 4 : 24 h IL-2 activated E+ cells with CD70/300–19, and lane 5 : 48 h IL-2 activated E+ cells with CD70/300–19. Representative data from three independent experiments are shown. Statistical analysis: two-sample t-test; n.s., not significant; *P < 0·01.
Effects of CD27/CD70 interactions on lymphokine-activated killer (LAK) activity
LAK, stimulated by IL-2, are known to kill Fas/WR [29]. Therefore, we investigated whether or not CD27/CD70 interactions affect Fas-mediated LAK activity. In other words, we added either mock/300–19 or CD70/300–19 when LAK were induced by IL-2, and then assessed the ability of LAK to kill Fas/WR. There was no difference in the killing activity against Fas/WR between the initial addition of mock/300–19 and CD70/300–19, when LAK were induced in the presence of IL-2 (Fig. 4c). Also, the initial addition of anti-CD70 blocking MoAb did not have any effect on the killing in the presence of mock/300–19 or CD70/300–19 (Fig. 4c). In addition, although IL-2 induces the expression of FasL mRNA by T cells, CD27 signalling did not affect the FasL mRNA expression (Fig. 4d).
These findings indicate that CD27 signalling is not involved in LAK activities via Fas and FasL expression.
Discussion
The results of the present study indicate that CD27/CD70 interactions play an important role as a costimulatory factor in the cytotoxic activity of T cells, and that this T-cell cytotoxicity involves the perforin/granzyme B pathway, but not the Fas/FasL pathway in human.
When resting T cells are stimulated by the P815 cell line having Fc receptors in the presence of anti-CD3 MoAb, P815 cells are killed within a short period of time via the perforin/granzyme B-mediated pathway [12,30–32]. As with CD80-transfected P815 cells described previously [12], the ability of resting T cells to kill P815 cells was enhanced by the contact with CD70-transfected P815 cells in the presence of anti-CD3 MoAb, which was completely negated by CMA. On the basis of the findings, CD27/CD70 interactions appear to promptly enhance the perforin/granzyme B-mediated killing (Fig. 1a).
Since CD80 and CD86 are expressed on LCL [33], it is reasonable to assume that CD80; CD86/CTLA-4; CD28 interactions are also involved in CTL activity [34,35]. EBV-activated B cells, LCL strongly expressed CD70 as well as other activation markers, indicating that EBV infection causes the activation of B cells and induces strong CD70 expression on their surface (data not shown). The MHC-restricted cytotoxicity of CTL induced by LCL was weakened, when CD27/CD70 interactions were inhibited by adding anti-CD70 blocking MoAb in the induction phase (Fig. 1b). CD27/CD70 interactions play a crucial role not only in the immunoglobulin production attributable to T–B interaction [14] and enhancement of NK or T cell activity [15] but also in the augmentation of CTL activity. CTL activity was not completely suppressed by CMA, and the activity with CMA did not decrease further compared to the activity with the presence of anti-CD70 blocking MoAb in the induction phase (Fig. 1b), thus suggesting that the enhancement of CTL activity by CD27/CD70 interactions is largely perforin/granzyme B dependent. In addition, levels of perforin expression by CTL were decreased by the initial addition of anti-CD70 blocking MoAb in the induction phase, which supports the view that perforin production is involved in the enhancement of CTL activity via CD27 signalling (Fig. 2). The relation between perforin and CD27 expression on CTL has been investigated [18–21]. CMV-specific CD8+ T cells, which are both CD27− and CD28−, appear to be fully mature effector cells and express perforin [19]. There is a strong inverse correlation between perforin and CD27 expression, even when comparing HIV-specific and CMV-specific populations from the same donors, implying that perforin-producing cells are CD27−[18]. Our data also suggested that the increase of CTL activity induced by LCL was mainly involved in CD8+CD27− T cells (Fig. 3). The enhancement of the CTL-cytotoxicity may be involved in CD27 down-regulation induced via CD27 signalling by CD70.
As CMA did not completely suppress the cytotoxic activity of induced CTL (Fig. 1b), Fas/FasL and/or TRAIL [36] may be involved. In our experiments, CTL induced by LCL did not kill Fas/WR at all, indicating that the cytotoxic activity of CTL is not mediated by Fas (Fig. 4b). We analysed the FasL expression on CTL by flow cytometry, but failed to detect FasL because it disappeared from the surface due to cleavage ([37] and our unpublished data). Unfortunately, we could not confirm the sufficient FasL surface expression even with the addition of metalloproteinase [38].
The system using the Fas/WR cell line as a target is specific for Fas/FasL-mediated cytotoxicity; FasL-transfectants lyse Fas/WR cells but not wild/WR cells in our experiments (data not shown). LAK cells induced by IL-2 can kill Fas/WR [28]. This observation prompts us to ascertain whether CD27/CD70 interactions affect LAK activity via Fas/FasL-mediated killing. CD27 signalling did not enhance the Fas-mediated cytotoxicity against Fas/WR cells or the FasL mRNA expression in the presence of IL-2, thus clarifying that CD27 signalling is not involved in Fas/FasL-mediated cytotoxicity even in the LAK system (Fig. 4c,d).
It is reported that prestimulation via CD27 enhanced the cytotoxic activity of murine NK cells in an IFN-γ-dependent manner [39]. But in our experiment in humans, IFN-γ was not involved in the CD27-mediated T-cell cytotoxicity (data not shown).
Brown et al. [21] suggest that CD27/CD70 interactions have an effect on CTL activation or differentiation events occurring later in alloantigen-induced responses in mice. In our study, the enhancement of CTL activity induced by CD27/CD70 interactions was analysed in autologous antigen-specific CTL in humans. However it is unknown what is the specific antigen of autologous LCL. Recently, Savoldo et al. [40] analysed TCR-combinatorial diversity of EBV-specific CTL by MoAb recognizing TCR-Vβ specific regions. They reported EBV-specific CTL presented considerable heterogeneity, with no specific over- or under-representation of any Vβ family. Our data also showed heterogeneous repertoire of TCR-Vβ subsets in CTL induced by autologous LCL (data not shown).
In the future, it will be necessary to analyse effects of CD27/CD70 interactions on CTL that have been cultured for a longer time, and to investigate cytotoxic mechanisms other than perforin/granzyme B and Fas/FasL.
Acknowledgments
We thank Drs H. Yagita, K. Okumura and A. Komiyama for their support in this study.
REFERENCES
- 1.Gustafsson A, Levitsky V, Zou JZ, et al. Epstein-Barr virus (EBV) load in bone marrow transplant recipients at risk to develop posttransplant lymphoproliferative disease. prophylactic infusion of EBV-specific cytotoxic T cells. Blood. 2000;95:807–14. [PubMed] [Google Scholar]
- 2.Lowin B, Hahne M, Mattmann C, Tschopp J. Cytolytic T-cell cytotoxicity is mediated through perforin and Fas lytic pathways. Nature. 1994;370:650–2. doi: 10.1038/370650a0. [DOI] [PubMed] [Google Scholar]
- 3.Kagi D, Vignaux F, Ledermann B, Burki K, Depraetere V, Nagata S, Hengartner H, Golstein P. Fas and perforin pathways as major mechanisms of T cell-mediated cytotoxicity. Science. 1994;265:528–30. doi: 10.1126/science.7518614. [DOI] [PubMed] [Google Scholar]
- 4.Berke G. The CTL's kiss of death. Cell. 1995;81:9–12. doi: 10.1016/0092-8674(95)90365-8. [DOI] [PubMed] [Google Scholar]
- 5.Schroter M, Lowin B, Borner C, Tschopp J. Regulation of Fas (Apo-1/CD95)- and perforin-mediated lytic pathways of primary cytotoxic T lymphocytes by the protooncogene bcl-2. Eur J Immunol. 1995;25:3509–13. doi: 10.1002/eji.1830251245. [DOI] [PubMed] [Google Scholar]
- 6.Motyka B, Korbutt G, Pinkoski MJ, et al. Mannose 6-phosphate/insulin-like growth factor II receptor is a death receptor for granzyme B during cytotoxic T cell-induced apoptosis. Cell. 2000;103:491–500. doi: 10.1016/s0092-8674(00)00140-9. [DOI] [PubMed] [Google Scholar]
- 7.Froelich CJ, Orth K, Turbov J, et al. New paradigm for lymphocyte granule-mediated cytotoxicity. J Biol Chem. 1996;271:29073–9. doi: 10.1074/jbc.271.46.29073. [DOI] [PubMed] [Google Scholar]
- 8.Shi L, Mai S, Israels S, Browne K, Trapani JA, Greenberg AH. Granzyme B (GraB) autonomously crosses the cell membrane and perforin initiates apoptosis and GraB nuclear localization. J Exp Med. 1997;185:855–66. doi: 10.1084/jem.185.5.855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Suda T, Takahashi T, Golstein P, Nagata S. Molecular cloning and expression of the Fas ligand, a novel member of the tumor necrosis factor family. Cell. 1993;75:1169–78. doi: 10.1016/0092-8674(93)90326-l. [DOI] [PubMed] [Google Scholar]
- 10.Suda T, Nagata S. Purification and characterization of the Fas-ligand that induces apoptosis. J Exp Med. 1994;179:873–9. doi: 10.1084/jem.179.3.873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Itoh N, Yonehara S, Ishii A, et al. The polypeptide encoded by the cDNA for human cell surface antigen Fas can mediate apoptosis. Cell. 1991;66:233–43. doi: 10.1016/0092-8674(91)90614-5. [DOI] [PubMed] [Google Scholar]
- 12.Azuma M, Cayabyab M, Buck D, Phillips JH, Lanier LL. CD28 interaction with B7 costimulates primary allogeneic proliferative responses and cytotoxicity mediated by small, resting T lymphocytes. J Exp Med. 1992;175:353–60. doi: 10.1084/jem.175.2.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Agematsu K, Nagumo H, Yang FC, et al. B cell subpopulations separated by CD27 and crucial collaboration of CD27+ B cells and helper T cells in immunoglobulin production. Eur J Immunol. 1997;27:2073–9. doi: 10.1002/eji.1830270835. [DOI] [PubMed] [Google Scholar]
- 14.Agematsu K, Hokibara S, Nagumo H, Komiyama A. CD27: a memory B-cell marker. Immunol Today. 2000;21:204–6. doi: 10.1016/s0167-5699(00)01605-4. [DOI] [PubMed] [Google Scholar]
- 15.Yang FC, Agematsu K, Nakazawa T, et al. CD27/CD70 interaction directly induces natural killer cell killing activity. Immunology. 1996;88:289–93. doi: 10.1111/j.1365-2567.1996.tb00017.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Goodwin RG, Alderson MR, Smith CA, et al. Molecular and biochemical characterization of a ligand for CD27 defines a new family of cytokines with homology to tumor necrosis factor. Cell. 1993;73:447–56. doi: 10.1016/0092-8674(93)90133-b. [DOI] [PubMed] [Google Scholar]
- 17.Arens R, Tesselaar K, Baars PA, et al. Constitutive CD27/CD70 interaction induces expansion of effector-type T cells and results in IFNgamma-mediated B cell depletion. Immunity. 2001;15:801–12. doi: 10.1016/s1074-7613(01)00236-9. [DOI] [PubMed] [Google Scholar]
- 18.Appay V, Nixon DF, Donahoe SM, et al. HIV-specific CD8 (+) T cells produce antiviral cytokines but are impaired in cytolytic function. J Exp Med. 2000;192:63–75. doi: 10.1084/jem.192.1.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hamann D, Baars PA, Rep MH, Hooibrink B, Kerkhof-Garde SR, Klein MR, Van Lier RA. Phenotypic and functional separation of memory and effector human CD8+ T cells. J Exp Med. 1997;186:1407–18. doi: 10.1084/jem.186.9.1407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Baars PA, Ribeiro Do Couto LM, Leusen JH, Hooibrink B, Kuijpers TW, Lens SM, Van Lier RA. Cytolytic mechanisms and expression of activation-regulating receptors on effector-type CD8+CD45RA+CD27− human T cells. J Immunol. 2000;165:1910–7. doi: 10.4049/jimmunol.165.4.1910. [DOI] [PubMed] [Google Scholar]
- 21.Brown GR, Meek K, Nishioka Y, Thiele DL. CD27-CD27 ligand/CD70 interactions enhance alloantigen-induced proliferation and cytolytic activity in CD8+ T lymphocytes. J Immunol. 1995;154:3686–95. [PubMed] [Google Scholar]
- 22.Morimoto C, Letvin NL, Distaso JA, Aldrich WR, Schlossman SF. The isolation of characterization of the human suppressor inducer T cell subset. J Immunol. 1985;134:1508–15. [PubMed] [Google Scholar]
- 23.Agematsu K, Kobata T, Sugita K, Hirose T, Schlossmann SF, Morimoto C. Direct cellular communication between CD45RO and CD45RA T cell subset via CD27/CD70. J Immunol. 1995;154:3627–35. [PubMed] [Google Scholar]
- 24.Peltz G, Trounstine M, Moore K. Cloned and expressed human Fc receptor for IgG mediates anti-CD3-dependent lymphoproliferation. J Immunol. 1988;141:1891–6. [PubMed] [Google Scholar]
- 25.Karasuyama H, Kudo A, Melchers F. The protein encoded by the VpreB and λ5 Pre-B cell-specific genes can associated with earch other and with µ heavy chain. J Exp Med. 1990;172:969–72. doi: 10.1084/jem.172.3.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Togashi K, Kataoka T, Nagai K. Characterization of a series of vacuolar type H(+)-ATPase inhibitors on CTL-mediated cytotoxicity. Immunol Lett. 1997;55:139–44. doi: 10.1016/s0165-2478(97)02698-9. [DOI] [PubMed] [Google Scholar]
- 27.Kataoka T, Togashi K, Takayama H, Takaku K, Nagai K. Inactivation and proteolytic degradation of perforin within lytic granules upon neutralization of acidic pH. Immunology. 1997;91:493–500. doi: 10.1046/j.1365-2567.1997.00257.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nakazawa T, Agematsu K, Yabuhara A. Later development of Fas ligand-mediated cytotoxicity as compared with granule-mediated cytotoxicity during the maturation of natural killer cells. Immunology. 1997;92:180–7. doi: 10.1046/j.1365-2567.1997.00343.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nakazawa T, Agematsu K, Yasui K, et al. Cytolytic mechanisms involved in non-MHC-restricted cytotoxicity in Chediak–Higashi syndrome. Clin Exp Immunol. 1999;118:108–14. doi: 10.1046/j.1365-2249.1999.01025.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mentzer SJ, Barbosa JA, Burakoff SJ. T3 monoclonal antibody activation of nonspecific cytolysis: a mechanism of CTL inhibition. J Immunol. 1985;135:34–8. [PubMed] [Google Scholar]
- 31.Spits H, Yssel H, Leeuwenberg J, De Vries JE. Antigen-specific cytotoxic T cell and antigen-specific proliferating T cell clones can be induced to cytolytic activity by monoclonal antibodies against T3. Eur J Immunol. 1985;15:88–91. doi: 10.1002/eji.1830150117. [DOI] [PubMed] [Google Scholar]
- 32.Leeuwenberg JF, Spits H, Tax WJ, Capel PJ. Induction of nonspecific cytotoxicity by monoclonal anti-T3 antibodies. J Immunol. 1985;134:3770–5. [PubMed] [Google Scholar]
- 33.Shinozaki K, Yasui K, Agematsu K. Direct B/B–cell interactions in immunoglobulin synthesis. Clin Exp Immunol. 2001;124:386–91. doi: 10.1046/j.1365-2249.2001.01518.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vandenborre K, Van Gool SW, Kasran A, Ceuppens JL, Boogaerts MA, Vandenberghe P. Interaction of CTLA-4 (CD152) with CD80 or CD86 inhibits human T-cell activation. Immunology. 1999;98:413–21. doi: 10.1046/j.1365-2567.1999.00888.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Aicher A, Hayden-Ledbetter M, Brady WA, et al. Characterization of human inducible costimulator ligand expression and function. J Immunol. 2000;164:4689–96. doi: 10.4049/jimmunol.164.9.4689. [DOI] [PubMed] [Google Scholar]
- 36.Khanolkar A, Yagita H, Cannon MJ. Preferential utilization of the perforin/granzyme pathway for lysis of epstein-barr virus-transformed lymphoblastoid cells by virus-specific CD4 (+) T cells. Virology. 2001;287:79–88. doi: 10.1006/viro.2001.1020. [DOI] [PubMed] [Google Scholar]
- 37.Tanaka M, Suda T, Takahashi T, Nagata S. Expression of the functional soluble form of human fas ligand in activated lymphocytes. EMBO J. 1995;14:1129–35. doi: 10.1002/j.1460-2075.1995.tb07096.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kayagaki N, Kawasaki A, Ebata T, et al. Metalloproteinase-mediated release of human Fas ligand. J Exp Med. 1995;182:1777–83. doi: 10.1084/jem.182.6.1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Takeda K, Yagita H, Okumura K. CD27-mediated activation of murine NK cells. J Immunol. 2000;116:1741–5. doi: 10.4049/jimmunol.164.4.1741. [DOI] [PubMed] [Google Scholar]
- 40.Savoldo B, Cubbage ML, Durett AG, et al. Generation of EBV-specific CD4+ cytotoxic T cells from virus naive individuals. J Immunol. 2002;168:909–18. doi: 10.4049/jimmunol.168.2.909. [DOI] [PubMed] [Google Scholar]


