Abstract
Cytoadherence of Plasmodium-falciparum-parasitized red blood cells (PRBCs) to host receptors is the key phenomenon in the pathological process of the malaria disease. Some of these interactions can originate poor outcomes responsible for 1 to 3 million annual deaths mostly occurring among children in sub-Saharan Africa. Pregnancy-associated malaria (PAM) represents an important exception of the disease occurring at adulthood in malaria endemic settings. Consequences of this are shared between the mother (maternal anemia) and the baby (low birth weight and infant mortality). Demonstrating that parasites causing PAM express specific variant surface antigens (VSAPAM), including the P. falciparum erythrocyte membrane protein 1 (P f EMP1) variant VAR2CSA, that are targets for protective immunity has strengthened the possibility for the development of PAM-specific vaccine. In this paper, we review the molecular basis of malaria pathogenesis attributable to the erythrocyte stages of the parasites, and findings supporting potential anti-PAM vaccine components evidenced in PAM.
1. THE IMPORTANCE OF CYTOADHERENCE IN THE PATHOPHYSIOLOGY OF PLASMODIUM FALCIPARUM MALARIA
P. falciparum infection encompasses a full range of clinical presentations, from asymptomatic infection to severe disease, including cerebral malaria, severe anemia, acute respiratory failure, hypoglycemia, renal failure, and pulmonary edema. Severe malaria, in particular cerebral malaria and severe anemia, constitutes one of the main causes of hospitalization in nonimmune individual from malaria endemic areas [1]. Patients with cerebral malaria present with a loss of consciousness and a coma, related to vascular obstruction by aggregated parasitized red blood cells (PRBCs), rosettes, and other fibrillous components. Parasite factors, such as GPI anchors elements (glycosylphosphatidylinositol), induce TNF-α and INF-γ, production that in turn will induce overexpression and relocalization of endothelial receptors, such as ICAM-1 and PECAM-1. In severe anemia, human and parasite factors play major roles. Anemia for instance is the consequence of PRBCs destruction, insufficient erythrocyte production, and increased clearance of both infected and noninfected RBCs by the spleen and the macrophages [2–5].
To survive, most microorganisms proceed to evolutionary adjustments in their virulence factors. In Plasmodium spp, these changes allow the parasite to sustain a chronicity inside its host by means of constant antigenic variations, allowing its transmission to the mosquito. Two virulence factors have been described in P. falciparum. Firstly, the growing rate, as parasites isolated from patients presenting with severe malaria express an in vitro multiplication rate higher than that of parasites isolated from nonsevere malaria patients [2].
This suggests that parasites causing severe disease multiply in their host faster than parasites associated with nonsevere disease. Factors controlling parasite multiplication rate still are not identified. The other P. falciparum virulence factor is the cytoadherence phenomenon (for review, see [6–8]). The nature of the P f EMP1 protein expressed on the surface of PRBCs plays a key role in this process. Parasites unable to adhere to vascular endothelium are eliminated from the blood stream by the spleen filter. Indeed, erythrocytes do lose their deformability when parasitized, facilitating their clearance by the spleen. (for review, see [9]). RBCs surface expression of variant antigens constitutes an evasion strategy from the immune system, used by all Plasmodium species studied, and this may probably represent a common feature within the Plasmodium genus. The expression of antigens unknown from the MHC would represent an excellent way to escape the immune system, but would also constitute a threat for species survival. The alternate expression of RBCs surface antigens thus is one of the intrahost mechanisms used by parasites for controlling their own population, while avoiding their host's death related to an excessive parasite multiplication [10, 11].
In P. falciparum, at least two variable surface antigens (VSAs), P f EMP1 and RIFINs, are expressed on the surface of PRBCs [12]. Although members of the STEVORs family have been identified in Maurer's dots, a network of parasite microtubules inside the cytoplasm of PRBCs, these proteins may not be surface-exposed [13, 14]. All these three proteins are encoded by multigene families, and most of the genes composing each family are in a sub-telomeric location, an area subjected to a high level of recombination. Variations affecting VSAs suggest that they are necessary for the parasite survival. Despite the changes needed for immune evasion, the limited number of host receptors imposes the parasite to maintain a minimum stability between structure and function of its surface proteins by maintaining selected amino acids residues.
2. PARASITIZED RED BLOOD CELLS ADHERENCE
Adherence of PRBCs to endothelial receptors is a characteristic of P. falciparum infections [15]. While PRBCs containing young stages (ring) of the parasite do circulate in the blood flow without concern, those RBCs infected by mature stages (trophozoites and schizonts) of the parasite are sequestered in the microvasculature of deep organs [16], thus avoiding passage through the spleen. Parasite-encoded adhesines involved in the RBCs cytoadherence have been associated to protrusions (knobs) at the surface of erythrocytes (Figure 1). Even though adherence under physiological conditions may require knobs, it is now admitted that these knobs are not essential, as knobless parasite lines have been observed in vitro to bind endothelial cells [17, 18]. However Knobs are the location where most parasite ligands are expressed [19]. Following the demonstration of the major role the P f EMP1 protein plays in the mechanisms of PBRCs binding to endothelial cells in 1984 [20], distinct adhesive properties of these parasite proteins to various receptors were also reported. The variations in the P f EMP1 binding properties originate different types of interactions, such as deep organs tropism of PRBCs, agglutination with uninfected RBCs (rosetting) [21] or with other PRBCs (auto-agglutination) [22]. These various facets of PRBCs cytoadherence are in close relation with malaria pathophysiology. The withdrawal of mature forms from the blood flow, and their accumulation in deep organ microvessels may represent a pathologic event more or less well tolerated, according to the target organ and the level of PRBCs accumulation. Sequestration may be the key factor involved in vital organs failure, in particular during cerebral malaria.
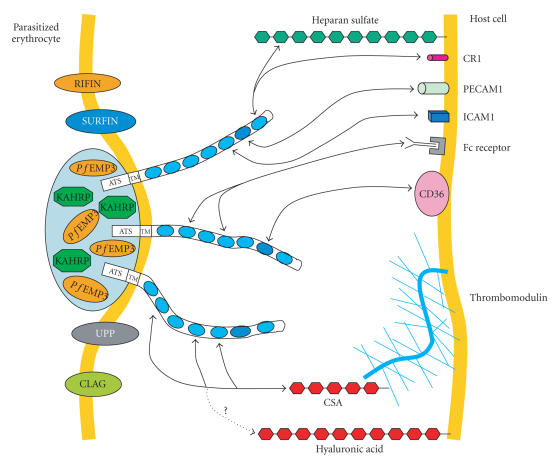
Figure 1.
Schematic diagram of knobs showing potential intermolecular interactions between parasites proteins exported on the surface of PRBC and receptors on the host cell surface. P f EMP, Plasmodium falciparum erythrocyte membrane protein; KHARP, knob-associated histidine-rich protein; RIFIN, repetitive-interspersed family proteins; CLAG, cytoadherence-linked asexual protein; CR1, complement receptor 1; ICAM1, intercellular adhesion molecule 1; PECAM1, platelet endothelial cell adhesion molecule 1; CSA, chondroitin sulphate A; UPP, uncharacterized parasite proteins. The question mark “?” means that the binding to hyaluronic acid is controversial.
3. PREGNANCY-ASSOCIATED MALARIA
In areas endemic for malaria, the pregnant woman is at high risk for malaria. Every year, twenty-five millions of pregnant women are exposed to malaria in sub-Saharan Africa, and pregnancy-associated malaria (PAM) is of serious public health concern [23]. In areas where malaria transmission is intense, its main consequences are a low birth weight (LBW) for the baby and a severe anemia for the mother. During pregnancy, massive sequestration of P. falciparum parasites in the placenta is likely to reduce maternofetal exchanges, explaining the frequency of LBW babies born from infected mothers. However, two recent studies have shown that pregnant women infected with Plasmodium vivax were also likely to give birth to LBW babies, suggesting that local or systemic production of selected inflammatory cytokines may also play a role in the pathological process [24, 25].
Recent data show that P. falciparum parasites infecting pregnant women express an antigenic profile different from that of parasites involved in cerebral malaria, and more generally, from parasites encountered in nonpregnant hosts [26]. This characteristic of PAM parasites is related to placenta-expressed receptors that participate in the selection of parasite phenotypes with a given specificity for these receptors. Chondroitin-sulfate A (CSA) is the major receptor for placenta sequestration [27, 28], and the number of parasite ligands involved in placenta sequestration is consequently highly restricted as compared to those implicated in cerebral malaria where several endothelial receptors may be involved. Although PAM parasites do preferentially bind to CSA, variable abilities were described among different placental isolates [29, 30]. Distinct subpopulations composed of strong and weak binders have been observed in FCR3CSA (a sub-line of FCR3 selected for its adhesion to CSA) using a model of adhesion under flow conditions [31]. Demonstration of different binding abilities among placental isolates showed particular interest as high binders were associated with high risk of LBW [30], and transcribed higher level of var2csa compared to low binders [32], emphasizing the role of var2csa in PAM.
4. CHONDROITIN-4-SULFATE (CSA) AND PLACENTAL RECEPTORS FOR SEQUESTRATION
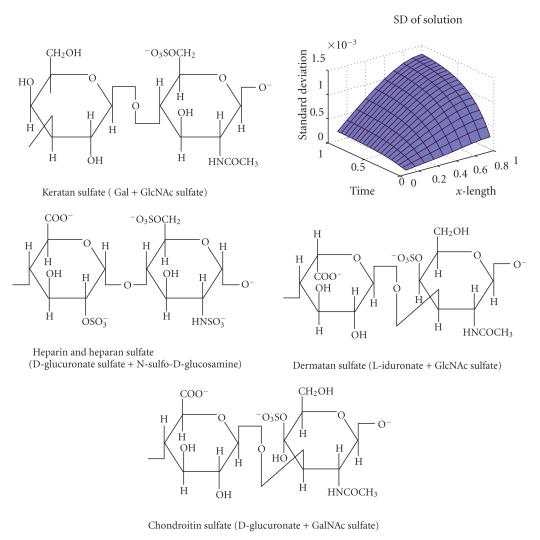
Although glycosaminoglycans (GAG) have previously been shown to be involved in sporozoite adhesion to hepatocytes by binding to heparin-like motifs of the heparan sulfate (HS) [33], CSA is the first such receptor involved in RBCs sequestration. GAGs polysaccharides chains are usually composed of repeats of disaccharides units formed by one hexuronic acid and one hexosamine. At least one of the disaccharide elements has a carboxyl or a sulfate negatively charged. Among major GAGs are hyaluronic acid (HA), chondroitin sulfate, keratin sulfate, heparan sulfate, and heparin. Heparin is mostly a component of intracellular granules of mast cells lining the arteries of the lungs, liver, and skin while heparan sulfate is a component of the cell surface found in the basement membrane. HS contains heparin-like motifs that are enriched with N-sulfated glucosamine and 2-sulfated acids [34]. Classical structure of CSA is tandem repeats of glucuronic acid [1–3] and N-acetylgalactosamine-4-sulfate [1–4]. Figure 2 illustrates structures of the different kinds of disaccharides composing GAGs of physiological significance. It is more and more obvious that GAGs structure is much more heterogeneous than previously thought [36, 37].
Figure 2.
Structure of different kinds of GAG disaccharides of physiological significance.
Chondroitin sulfates (CS) are mosaics formed by C4S (CSA) or C6S (CSC) types of disaccharides. The belonging to a CS type depends on the most abundant disaccharide. In chondroitin sulfate B (CSB), glucuronic acid is changed to iduronic acid, and in CSC, the N-galactosamine sulfate group is in position 6, while CSD and CSE are usually hyaluronate mix. CSB and CSC are not implicated in PBRC adherence [38].
Other GAGs of physiological significance include Type III TGF-β receptor, also called betaglycan, that contains both heparan and chondroitin sulphate chains [39]. CD44 family is composed of molecules that can exist in the proteoglycan and nonproteoglycan forms. CD44 is a cell surface receptor for hyaluronan [40] and is synthesized by lymphocytes, epithelial cells, fibroblasts, glial cells, Kupfer cells, and mesangial cells of the kidney. Like syndecans it has a short intracellular C-terminal and highly-conserved domain and a large extracellular domain [41]. Extracellular domain contains three disulfide-bonded loops, and it has a high homology with the hyaluronan binding region of aggrecan, link protein, neurocan, and versican [42].
The nervous tissue well-characterized proteoglycans include phosphacan, NG2 proteoglycan, agrin, receptor-type protein tyrosine phosphatase, and the aggregating proteoglycans neurocan and brevican. NG2 proteoglycan is a cell membrane-associated chondroitin sulphate proteoglycan present in nervous tissue cells that have not yet specialized into oligodendrocytes [43], but it has been found also in developing mesenchyme and human melanoma cells. The primary structure of NG2 proteoglycan consists of 2325 amino acids that code a 252 kd core protein [43].
In the vascular bed, thrombomodulin (TM) is a transmembrane glycoprotein containing high CSA levels [44]. CSA is involved in TM function, mainly by linking and inactivating the circulating form of thrombin, a coagulation factor [45]. TM is highly present in vascular endothelia (∼100.000 molecules per cell), but also at the syncytiotrophoblast surface [46, 47], to which PRBCs bind in placenta [26]. The specificity of CSA binding of PRBCs is demonstrated by its full inhibition by either a minimum motif of 7 disaccharide units of the 4S type [48] or by chondroitinase ABC treatment. CSA is also present in pulmonary and cerebral vascular endothelium, suggesting a possible role in other severe forms of malaria [49].
Optimal binding of PRBCs in the placenta is observed in the presence of ∼30% 4S disaccharides and ∼70% nonsulfated disaccharides with a minimal motif of 6 disaccharide units [50]. This has been confirmed with a C4S/C6S bovine copolymer, although the minimal motif included 4 C4S units instead of 2 in the case of that from human origin [51]. Chondroitin sulfate proteoglycans (CSPG) isolated from human placenta are low sulfated, with around 8% of chains with a sulfate in position 4 (C4S) and the most part being nonsulfated [37]. This apparent discrepancy is explained by the fact that, in the CSPG structure, sulfated groups are concentrated in domains formed with 6 to 14 disaccharides [52]. These sulfate-rich domains include 20 to 28% of C4S, as opposed to the other regions.
Other studies demonstrate that HA is also involved in placenta sequestration of PRBCs, as most of P. falciparum placental isolates exhibit affinity for this GAG [53, 54]. However, not all agree with this finding [55]. With a similar structure as CSA, N-acetylgalactosamin in HA is not sulfated. Because HA is also present at the surface of syncytiotrophoblasts [56] and endothelial cells from microvessels [57], further studies are required to determine if HA represents a receptor in itself.
Despite the commonly admitted role of CSA in placental sequestration of P. falciparum infected RBCs, the overall process might be more complex involving multiple receptors such as (IgG, IgM, HA, CSA) rather than exclusive interaction with CSA [58]. A P. falciparum line selected according to the IgG-binding phenotype, was also shown to bind strongly to placental syncytiotrophoblasts, with a similar profile as wild isolates [59]. This binding is not inhibited by glycosaminoglycans or by chondroitinase ABC and hyaluronidase treatment, but is inhibited by IgG-binding proteins, suggesting that P f EMP1-containing domains that are able to bind CSA may also harbor IgG and IgM binding sites, offering another linking possibility between the PRBCs surface and Fc receptors expressed in the placenta.
5. VAR GENES FAMILY AND ANTIGENIC VARIATION
In P. falciparum, variant antigens (P f EMP1) expressed at PRBCs surface are encoded by a family of genes called var composed of around 60 copies per haploid genome [60]. Members of this family are distributed among all chromosomes, most being localized in the subtelomeric regions and few in the central region of the chromosomes. Subtelomeric var genes are more vulnerable to the recombination phenomenon that affects their structure. Gene duplication phenomena are also frequent. These phenomena, known since the '30s [61], allow biological evolution and diversity. Selected genes are mutated after duplication or recombined with other members of the family, while transcription of the others is repressed following mutation. In the genome of the 3D7 P. falciparum strain, the high number of truncated var genes (pseudogenes) indicates the high frequency of gene deletion events occurring in the genome.
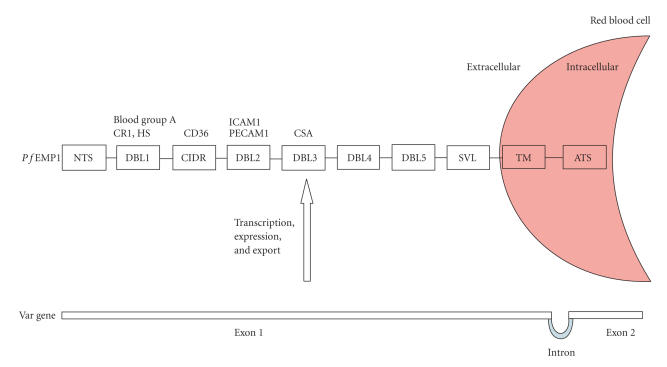
As for primary sequences, the number of domains may vary between var genes, as their size (from 3.9 to 13 kb). P f EMP1 proteins encoded by these genes show differences that originate major antigenic variations at the erythrocyte surface. Each P f EMP1 is constituted by an arrangement of distinct domains. The extracellular part is encoded by var gene exon 1, and possesses a variable N-terminal segment (NTS), several “Duffy-binding like” (DBL) domains (named following the Duffy-binding protein, the first such domain described, that allows P. vivax adherence to the Duffy antigen), and cysteine interdomain rich regions (CIDR) [62]. Each DBL domain is approximately 300 Aa long. Depending on their Aa sequence, DBLs as well as CIDRs have been classified into 5 types (α to ɛ). In selected P f EMP1s, there is a small fragment after DBLβ originating a DBLβC2 structure. At the end of exon 1, there is a sequence of variable length (SVL) and a hydrophobic region with the characteristics of a transmembrane domain (TM) [63]. The entire P f EMP1 molecule is anchored to the erythrocyte membrane by this TM domain, followed by an acidic C-terminal intracellular conserved segment (ATS), encoded by exon 2 (Figure 3).
Figure 3.
Schematic diagram illustrating var gene and P f EMP1 organization. Domains with known binding properties are specified. NTS, N-terminal sequence; DBL, Duffy-binding like; CIDR, cystein-rich interdomain; SVL, sequence of variable length; TM, transmembrane; ATs, acidic terminal sequence.
Several studies have associated P f EMP1s family to malaria pathogenesis, and the study of the function of the various P f EMP1 molecules represents a research topic of high interest for the development of prevention strategies. The understanding of the mechanisms controlling var gene expression is of utmost importance for the control of their biological role. Var gene expression involves a set of regulation mechanisms implicating activation, switching, and silencing of localization sites. Studies of pre-erythrocytic maturation stages showed that var gene expression operates in a mutually exclusive fashion. Although several transcripts are detectable in a given parasite, a single one is massively transcribed as a full-length (untruncated) mRNA and expressed at the PRBCs surface while the others are kept inactivated (silencing) or give rise to truncated mRNA [64, 65]. Each var gene is a single transcriptional unit that can be activated in situ. The expression of the various members varies according to the development stages [66], but only the expression during the erythrocytic stages seems to play a major role in the parasite development in relation to the immune system escape. Although the expression profile may change in vitro without immune pressure, the expression of the same var gene during long periods of time has often been observed. The phenomenon appears to be highly different in vivo. Recent studies show a total change in the expression profile following passage of the parasite in the mosquito, suggesting a much higher in vivo switching rate [67, 68]. Selected genes with a physical colocalization show a tendency to be activated and expressed during the same development stage [66, 69].
Considering differences in gene structure, chromosomal organization, and sequences of untranslated regions, subgroupings of the var gene family have been proposed [60, 70, 71]. The analysis of gene upstream sequences allowed to define 3 major types of sequences (promoter-like): upsA, upsB, and upsC [60]. Two sequences belonging to the var1 and var2 subfamilies formed independent groups corresponding to upsD and upsE, respectively [71]. These sequences are associated to the localization and the orientation of each var gene. Subtelomeric genes orientated towards the telomere express promoter sequences of the upsA type, those orientated around the centromere express promoter sequences of the upsB type, and those located in the central part of the chromosome, upsC-type sequences. Differential transcription of var genes from different localizations inside chromosomes is likely to be a consequence of the differential expression of promoter-repressing elements. A silencing mechanism associated to the intron has also been suggested [72, 73]. Small size mRNA from some var gene introns, cooperating with 5′-UTR sequences, is able to inhibit expression of these genes. The expression of a conserved var gene lacking intron (varCOMMON) in 3D7 and 60 to 70% of wild isolates strengthens this hypothesis [74]. The high level of similarity in var genes intronic promoter motifs suggests this phenomenon is able to regulate var genes expression. Numerous queries are still remaining unanswered regarding the overall process regulating var genes expression and associated mechanisms. Changes in the chromatin structure have been associated with the switching phenomenon [75, 76]. A recent report by Chookajorn et al. [77] shows that an epigenetic memory that includes histone modifications reminiscent of those associated with gene transcription memory found in the homeotic genes of Drosophila melanogaster is involved in the control of var gene transcription. Specific epigenetic mark consisting in methylation of histone H3 and lysine K9 on chromatin seems to play a major role in transcriptional memory that can provide advantages to the parasites in pathogenesis and immune evasion.
In in vitro cultured P. falciparum strains, switch rate is higher in some lines (as ITG) than in others (as FCR3) [22]. In in vivo conditions, several factors may play a role in a given var gene type selection and expression. In children and malaria naive individuals, parasites tend to express selected VSA (mainly P f EMP1) types at the surface of PRBCs (for review see [78]). In the pregnant woman, the placenta allows to select for parasite subpopulations expressing one (or several) P f EMP1s able to bind receptors that are present on the syncytiotrophoblast surface. Mechanisms for selecting the var gene specifically expressed by parasites binding the placenta is currently unknown. Motifs of nuclear receptor sequences from hormones have been observed in the promoter regions of some var genes, but their putative function remains unknown [79], and the mechanism underlying var genes selection is unclear. Given its importance, there is an obvious need of investigation.
6. PARASITE LIGANDS INVOLVED IN PLACENTAL SEQUESTRATION
Studies of in vitro selected parasite lines evidenced three P f EMP1 molecules that could be involved in placenta sequestration, through interaction with C4S receptors or with nonimmune Ig [59, 80, 81]. Although the CSA binding site has been localized inside the DBLγ domain of P f EMP1 molecules encoded by the FCR3varCSA [81] and varCS2 [80] genes, the role of these DBLγ domains in placenta binding is now questioned. Conversely to CS2var genes, FCR3varCSA genes are much conserved in various isolates and were named var1csa or varCOMMON. Transcription of these genes is not restricted to placental isolates [74, 82, 83], and analyses by Northern blot and real-time quantitative RT-PCR failed to demonstrate any overexpression of the var1csa or varCS2 transcripts in parasite lines selected for CSA adhesion [84–86]. More recent works on laboratory-adapted parasite lines [85, 86] suggested that another var gene is involved in CSA adherence and placenta sequestration. This gene, termed var2csa, is localized on chromosome 12. Another gene with similar, but truncated, sequence is located on chromosome 13. The P f EMP1 protein encoded by var2csa is constituted of 6 DBL domains, among which 3 remain unclassified. The sequence differs phylogenetically from that of other members of the family, and the DBLα and DBLγ are lacking [60]. Var2csa is structurally conserved between isolates and its overexpression by placental isolates is now confirmed [32]. However, the inability to find VAR2CSA P f EMP1 in PRBCs membranes by proteomic approach was unexpected [87]. Moreover, a recent study based on a strategy of cross-linking PRBCs with a radio-iodinated photoactivable C4S dodecasaccharide (representing the minimum requirement for efficient PRBCs binding) rather identified an ∼ 22 kd protein but no protein has been identified within the P f EMP1 molecular weight range as a ligand for C4S [88]. This observation suggests that a low molecular weight PRBCs surface protein is involved in C4S binding. Even though these surprising findings do not exclude the role of VAR2CSA in the binding of PRBCs to C4S as possible technical insufficiencies in the experimental procedures would explain the inability to detect VAR2CSA, it likely appears that parasite binding to C4S not only might involve multiple binding sites within the VAR2CSA [89] but also might necessitate a multiprotein complex possibly comprising VAR2CSA P f EMP1 and other proteins for which identification remains an important goal to achieve.
7. IMMUNITY TO PAM
Initially thought that PAM was due to pregnancy-related immunomodulation and humoral alteration, studies have now established that PAM is caused by P. falciparum which express unique variant surface antigens (VSAPAM) that allow the parasite sequestration in the placenta [90] by binding to CSPG receptors on syncytiotrophoblast [30, 91]. A number of studies have indicated that parasite-encoded VSA in the surface of PRBCs are important targets for acquired protective immunity that develops following exposure to P. falciparum parasites [92–95]. In the case of pregnancy malaria, women who have suffered from PAM develop VSAPAM-specific anti-CSA adhesive antibodies which are associated with protection from malaria in subsequent pregnancies [96, 97]. The difference in susceptibility to PAM between primigravid women and multigravid women is attributed to the lack by primigravidae of antibodies against this particular VSAPAM. These protective antibodies are thought to recognize reasonably conserved parasite antigens, because sera and parasites from pregnant women from different malaria areas cross-react [98]. This has raised hope for development of a vaccine to prevent PAM that should incorporate the PRBCs surface proteins expressed by placental parasites. Recent studies have shown that PAM parasites specifically transcribe high level of var2csa, one of the most conserved subfamily of var genes encoding a member of the P f EMP1 family [32, 99]. Antibodies to this particular VSAPAM was recently shown to specifically label the surface of in vitro adapted CSA-selected parasite [100]. Naturally acquired human monoclonal IgG1 antibodies were recently shown to react exclusively with intact CSA-adhering PRBCs expressing VSAPAM [101]. Plasma samples from individuals from malaria endemic areas recognize VAR2CSA recombinant proteins in a sex- and parity-dependent manner [100, 102] and a kinetic study demonstrated that VAR2CSA-specific antibodies were acquired during pregnancy as an antiparasite response [102]. High plasma levels of anti-VAR2CSA antibodies early in pregnancy are associated with lower risk of LBW [100] and long lasting placental infections [102]. More recently it was shown that mouse antibodies raised against VAR2CSA DBL domains can inhibit adhesion of placental isolates to CSA as up to 60% [103]. These observations demonstrate that the antibody-mediated mechanism of protection against PAM can involve both adhesion-blocking antibodies as well as cytophilic process such as phagocytosis and complement activation. This is consistent with the finding by Megnekou et al. [104] that PAM IgG in Cameroonian women is predominantly composed of IgG1 and IgG3 subclasses.
It was previously suggested that var gene expression is hierarchically structured in field isolates, as the expression of certain var genes was found to be associated with severe malaria in young children [105]. An explanation would be that the progeny of parasites expressing var gene products that mediate the most effective sequestration outgrows the progeny of parasites expressing a molecule mediating less effective binding [105–107]. A similar process was observed in the expression of VAR2CSA molecules as some sequence motifs on DBL3X were more likely to occur distinctly in parasites isolated from primi- and multigravidae [108]. This sequence variation may have great consequence on the development of protective antibodies as PAM severe consequences are observed more among primigravidae.
The contribution of cell-mediated immunity in protection against PAM remains unclear. The maternofetal interface is a complex network where numerous cytokines are secreted. Immunomodulation during pregnancy was first considered to result from a Th1/Th2 bias to facilitate the fetal allograft development, and resulting in a decrease of Th1-type cytokines (TFN-α and IFN-γ) [109] and an increase of Th2-type cytokines (IL-4, IL-10, TGF-β) [110, 111]. Later, it was thought to result from monocyte activation and relative lymphocyte inhibition [112]. Placental cytokines modulate the antigen-presenting cell function by inhibiting or increasing the expression of various molecules on the monocyte surface. The development of P. falciparum in the placenta causes an immune imbalance with an increase of inflammatory cytokines, IFN-γ and TNF-α [113–115], explaining that immunomodulation is more important in the placental blood than in the peripheral blood [116]. This inflammatory response is responsible for functional damages in placental villi, and disturbances of the fetomaternal exchanges, leading to low birth weight [113, 117]. IFN-γ secretion by mononuclear cells of the intervillous blood is associated with protection against PAM [118], demonstrating the implication of the cell response. High level of anti-inflammatory cytokines is observed in multigravidae compared to primigravidae, suggesting that involvement of cell-mediated immunity in the mechanism of protection would necessitate a fine balance in timing and production of pro- and anti-inflammatory cytokines [119, 120].
8. TREATMENT AND PREVENTION: PERSPECTIVES FOR HUMAN APPLICATION
Initially the World Health Organization (WHO) recommended that pregnant women living in malaria-endemic areas receive chemoprophylaxis with a safe and effective antimalarial drug as part of routine antenatal care. Although this policy was widely adopted across sub-Saharan Africa, program implementation was often poor or nonexistent, especially in East Africa. Due to a number of difficulties encountered including the difficult deliverability of this strategy (poor adherence with weekly drug dosing) and rising rates of resistance to most chemoprophylaxis regimens, including chloroquine [121], WHO had to change its recommendations. In 2002, after studies conducted in Malawi and Kenya demonstrated that two treatment doses of sulfadoxine-pyrimethamine (SP) administered as intermittent preventive treatment (IPTp) during routine antenatal care decreased maternal anemia and diminished the frequency of low birthweight [122–124], WHO developed a strategic framework for the control of malaria during pregnancy in Africa [23]. The document recommends that pregnant women receive at least two doses of IPTp during the second and third trimesters at routine antenatal care visits. The prevention strategies of malaria during pregnancy include IPTp, insecticide-treated nets, and effective case management of clinical malaria. The new policy leading to the adoption of IPTp is unique to pregnancy and is still under evaluation. In contrast, insecticide-treated nets and case management are strategies that are in use for all age and gender strata.
A recent study using depolymerized heparin demonstrated that these modified glycosaminoglycans (dGAGs) are able to disrupt binding properties of P. falciparum that form rosettes and employ heparan sulfate as a host receptor. Intravenous injection of these dGAGs could block up to 80% of PRBCs from binding in the microvasculature and release already sequestered parasites into the circulation in an in vivo model of severe malaria [125].
GAGs are structures contributing to host cell recognition and invasion by various infectious agents, including viruses (herpes simplex, viral hepatitis, HIV) and parasites (Babesia, Leishmania, Plasmodium) (for review see [126]). Three types of GAGs interact with P. falciparum endo-erythrocytic cycle: heparane sulfates [127] involved in rosette formation, CSA in PRBCs adherence, and heparine in RBCs invasion inhibition. As regards PRBCs adherence, it was shown that soluble CSA is involved both in vivo and in vitro. Intravenous injections of soluble CSA to monkeys infected with a CSA-binding Plasmodium strain are followed by the release of mature stages of the parasite in the peripheral blood [128]. Moreover, PBRCs in vitro binding is inhibited by almost 90% by purified CSA. These observations originated works related to the inhibitory capacity of CS. Hitherto, various polysaccharides have been tested for their ability to inhibit human erythrocyte invasion by P. falciparum merozoites, and PBRCs binding to various receptors [129–132]. Numerous sulfated polysaccharides, such as heparines, sulfate dextrans, fucoidans, and hyaluronates all exhibit inhibitory properties, but at different levels. Two carraghenate derivatives and cellulose sulfate (CS10) inhibit PRBCs binding to CSA [133]. Chondroitin-4-sulfate, a molecule already marketed (Chondrosulf, Structum), was unable to inhibit PRBCs binding to Saimiri endothelium [128], but this was probably related to the oral administration of the drug. Intravenous administration of Structum was effective in Saimiri, but the drug of bovine origin is not anymore available. Industrial synthesis of glycosaminoglycans cannot be currently achieved. It is necessary to conduct additional structural and toxicologic studies of CS, for this to be considered as a potential candidate for treating PAM.
ACKNOWLEDGMENTS
This work was supported by grants from the French Agence Nationale de la Recherche (MIME 2006), and the Institut de Médecine et d'Epidémiologie Appliquée. N. T. Ndam was the recipient of a postdoctoral fellowship from the Fondation pour la Recherche Médicale.
References
- 1.Marsh K, Snow RW. Malaria transmission and morbidity. Parassitologia. 1999;41(1–3):241–246. [PubMed] [Google Scholar]
- 2.Chotivanich K, Udomsangpetch R, Simpson JA, et al. Parasite multiplication potential and the severity of falciparum malaria. The Journal of Infectious Diseases. 2000;181(3):1206–1209. doi: 10.1086/315353. [DOI] [PubMed] [Google Scholar]
- 3.Wickramasinghe SN, Abdalla SH. Blood and bone marrow changes in malaria. Bailliere's Best Practice and Research in Clinical Haematology. 2000;13(2):277–299. doi: 10.1053/beha.1999.0072. [DOI] [PubMed] [Google Scholar]
- 4.Chotivanich K, Udomsangpetch R, Dondorp A, et al. The mechanisms of parasite clearance after antimalarial treatment of Plasmodium falciparum malaria. The Journal of Infectious Diseases. 2000;182(2):629–633. doi: 10.1086/315718. [DOI] [PubMed] [Google Scholar]
- 5.Ekvall H. Malaria and anemia. Current Opinion in Hematology. 2003;10(2):108–114. doi: 10.1097/00062752-200303000-00002. [DOI] [PubMed] [Google Scholar]
- 6.Miller LH, Good MF, Milon G. Malaria pathogenesis. Science. 1994;264(5167):1878–1883. doi: 10.1126/science.8009217. [DOI] [PubMed] [Google Scholar]
- 7.Chen Q, Schlichtherle M, Wahlgren M. Molecular aspects of severe malaria. Clinical Microbiology Reviews. 2000;13(3):439–450. doi: 10.1128/cmr.13.3.439-450.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kyes S, Horrocks P, Newbold C. Antigenic variation at the infected red cell surface in malaria. Annual Review of Microbiology. 2001;55:673–707. doi: 10.1146/annurev.micro.55.1.673. [DOI] [PubMed] [Google Scholar]
- 9.Engwerda CR, Beattie L, Amante FH. The importance of the spleen in malaria. Trends in Parasitology. 2005;21(2):75–80. doi: 10.1016/j.pt.2004.11.008. [DOI] [PubMed] [Google Scholar]
- 10.Saul A. The role of variant surface antigens on malaria-infected red blood cells. Parasitology Today. 1999;15(11):455–457. doi: 10.1016/s0169-4758(99)01534-3. [DOI] [PubMed] [Google Scholar]
- 11.Paget-McNicol S, Gatton M, Hastings I, Saul A. The Plasmodium falciparum var gene switching rate, switching mechanism and patterns of parasite recrudescence described by mathematical modelling. Parasitology. 2002;124(3):225–235. doi: 10.1017/s0031182001001160. [DOI] [PubMed] [Google Scholar]
- 12.Craig A, Scherf A. Molecules on the surface of the Plasmodium falciparum infected erythrocyte and their role in malaria pathogenesis and immune evasion. Molecular and Biochemical Parasitology. 2001;115(2):129–143. doi: 10.1016/s0166-6851(01)00275-4. [DOI] [PubMed] [Google Scholar]
- 13.Kaviratne M, Khan SM, Jarra W, Preiser PR. Small variant STEVOR antigen is uniquely located within Maurer's clefts in Plasmodium falciparum-infected red blood cells. Eukaryotic Cell. 2002;1(6):926–935. doi: 10.1128/EC.1.6.926-935.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hui GSN, Siddiqui WA. Characterization of a Plasmodium falciparum polypeptide associated with membrane vesicles in the infected erythrocytes. Molecular and Biochemical Parasitology. 1988;29(2-3):283–293. doi: 10.1016/0166-6851(88)90083-7. [DOI] [PubMed] [Google Scholar]
- 15.MacPherson GG, Warrell MJ, White NJ, Looareesuwan S, Warrell DA. Human cerebral malaria. A quantitative ultrastructural analysis of parasitized erythrocyte sequestration. American Journal of Pathology. 1985;119(3):385–401. [PMC free article] [PubMed] [Google Scholar]
- 16.Luse SA, Miller LH. Plasmodium falciparum malaria. Ultrastructure of parasitized erythrocytes in cardiac vessels. American Journal of Tropical Medicine and Hygiene. 1971;20(5):655–660. [PubMed] [Google Scholar]
- 17.Biggs BA, Culvenor JG, Ng JS, Kemp DJ, Brown GV. Plasmodium falciparum: cytoadherence of a knobless clone. Experimental Parasitology. 1989;69(2):189–197. doi: 10.1016/0014-4894(89)90187-2. [DOI] [PubMed] [Google Scholar]
- 18.Udomsangpetch R, Aikawa M, Berzins K, Wahlgren M, Perlmann P. Cytoadherence of knobless Plasmodium falciparum-infected erythrocytes and its inhibition by a human monoclonal antibody. Nature. 1989;338(6218):763–765. doi: 10.1038/338763a0. [DOI] [PubMed] [Google Scholar]
- 19.Nakamura K-I, Hasler T, Morehead K, Howard RJ, Aikawa M. Plasmodium falciparum-infected erythrocyte receptor(s) for CD36 and thrombospondin are restricted to knobs on the erythrocyte surface. Journal of Histochemistry and Cytochemistry. 1992;40(9):1419–1422. doi: 10.1177/40.9.1380530. [DOI] [PubMed] [Google Scholar]
- 20.Leech JH, Barnwell JW, Miller LH, Howard RJ. Identification of a strain-specific malarial antigen exposed on the surface of Plasmodium falciparum-infected erythrocytes. Journal of Experimental Medicine. 1984;159(6):1567–1575. doi: 10.1084/jem.159.6.1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.David PH, Handunnetti SM, Leech JH, Gamage P, Mendis KN. Rosetting: a new cytoadherence property of malaria-infected erythrocytes. American Journal of Tropical Medicine and Hygiene. 1988;38(2):289–297. doi: 10.4269/ajtmh.1988.38.289. [DOI] [PubMed] [Google Scholar]
- 22.Roberts DJ, Craig AG, Berendt AR, et al. Rapid switching to multiple antigenic and adhesive phenotypes in malaria. Nature. 1992;357(6380):689–692. doi: 10.1038/357689a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.WHO. A strategic framework for malaria prevention and control during pregnancy in the African region. Brazzaville, Congo: World Health Organization; Report ARF/MAL/04/01. 2004
- 24.Nosten F, McGready R, Simpson JA, et al. Effects of Plasmodium vivax malaria in pregnancy. The Lancet. 1999;354(9178):546–549. doi: 10.1016/s0140-6736(98)09247-2. [DOI] [PubMed] [Google Scholar]
- 25.Singh N, Shukla MM, Sharma VP. Epidemiology of malaria in pregnancy in central India. Bulletin of the World Health Organization. 1999;77(7):567–572. [PMC free article] [PubMed] [Google Scholar]
- 26.Beeson JG, Brown GV, Molyneux ME, Mhango C, Dzinjalamala F, Rogerson SJ. Plasmodium falciparum isolates from infected pregnant women and children are associated with distinct adhesive and antigenic properties. The Journal of Infectious Diseases. 1999;180(2):464–472. doi: 10.1086/314899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fried M, Duffy PE. Adherence of Plasmodium falciparum to chondroitin sulfate A in the human placenta. Science. 1996;272(5267):1502–1504. doi: 10.1126/science.272.5267.1502. [DOI] [PubMed] [Google Scholar]
- 28.Fried M, Fried M, Domingo GJ, Mutabingwa TK, Duffy PE. Plasmodium falciparum: chondroitin sulfate A is the major receptor for adhesion of parasitized erythrocytes in the placenta. Experimental Parasitology. 2006;113(1):36–42. doi: 10.1016/j.exppara.2005.12.003. [DOI] [PubMed] [Google Scholar]
- 29.Beeson JG, Mann EJ, Elliott SR, et al. Antibodies to variant surface antigens of Plasmodium falciparum-infected erythrocytes and adhesion inhibitory antibodies are associated with placental malaria and have overlapping and distinct targets. The Journal of Infectious Diseases. 2004;189(3):540–551. doi: 10.1086/381186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tuikue Ndam N, Fievet N, Bertin G, Cottrell G, Gaye A, Deloron P. Variable adhesion abilities and overlapping antigenic properties in placental Plasmodium falciparum isolates. The Journal of Infectious Diseases. 2004;190(11):2001–2009. doi: 10.1086/425521. [DOI] [PubMed] [Google Scholar]
- 31.Pouvelle B, Traoré B, Nogueira PA, Pradines B, LéPolard C, Gysin J. Modeling of Plasmodium falciparum-infected erythrocyte cytoadhesion in microvascular conditions: chondroitin-4-sulfate binding, a competitive phenotype. The Journal of Infectious Diseases. 2003;187(2):292–302. doi: 10.1086/346050. [DOI] [PubMed] [Google Scholar]
- 32.Tuikue Ndam N, Salanti A, Bertin G, et al. High level of var2csa transcription by Plasmodium falciparum isolated from the placenta. The Journal of Infectious Diseases. 2005;192(2):331–335. doi: 10.1086/430933. [DOI] [PubMed] [Google Scholar]
- 33.Pancake SJ, Holt GD, Mellouk S, Hoffman SL. Malaria sporozoites and circumsporozoite proteins bind specifically to sulfated glycoconjugates. The Journal of Cell Biology. 1992;117(6):1351–1357. doi: 10.1083/jcb.117.6.1351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pinzon-Ortiz C, Friedman J, Esko J, Sinnis P. The binding of the circumsporozoite protein to cell surface heparan sulfate proteoglycans is required for plasmodium sporozoite attachment to target cells. The Journal of Biological Chemistry. 2001;276(29):26784–26791. doi: 10.1074/jbc.M104038200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pradel G, Garapaty S, Frevert U. Proteoglycans mediate malaria sporozoite targeting to the liver. Molecular Microbiology. 2002;45(3):637–651. doi: 10.1046/j.1365-2958.2002.03057.x. [DOI] [PubMed] [Google Scholar]
- 36.Karamanos NK, Syrokou A, Vanky P, Nurminen M, Hjerpe A. Determination of 24 variously sulfated galactosaminoglycan- and hyaluronan-derived disaccharides by high-performance liquid chromatography. Analytical Biochemistry. 1994;221(1):189–199. doi: 10.1006/abio.1994.1396. [DOI] [PubMed] [Google Scholar]
- 37.Achur RN, Valiyaveettil M, Alkhalil A, Ockenhouse CF, Gowda DC. Characterization of proteoglycans of human placenta and identification of unique chondroitin sulfate proteoglycans of the intervillous spaces that mediate the adherence of Plasmodium falciparum-infected erythrocytes to the placenta. The Journal of Biological Chemistry. 2000;275(51):40344–40356. doi: 10.1074/jbc.M006398200. [DOI] [PubMed] [Google Scholar]
- 38.Gysin J, Pouvelle B, Le Tonqueze M, Edelman L, Boffa M-C. Chondroitin sulfate of thrombomodulin is an adhesion receptor for Plasmodium falciparum-infected erythrocytes. Molecular and Biochemical Parasitology. 1997;88(1-2):267–271. doi: 10.1016/s0166-6851(97)00082-0. [DOI] [PubMed] [Google Scholar]
- 39.Cheifetz S, Massague J. Transforming growth factor-β (TGF-β) receptor proteoglycan. Cell surface expression and ligand binding in the absence of glycosaminoglycan chains. The Journal of Biological Chemistry. 1989;264(20):12025–12028. [PubMed] [Google Scholar]
- 40.Miyake K, Underhill CB, Lesley J, Kincade PW. Hyaluronate can function as a cell adhesion molecule and CD44 participates in hyaluronate recognition. Journal of Experimental Medicine. 1990;172(1):69–75. doi: 10.1084/jem.172.1.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jalkanen S, Bargatze RF, de los Toyos J, Butcher EC. Lymphocyte recognition of high endothelium: antibodies to distinct epitopes of an 85-95-kD glycoprotein antigen differentially inhibit lymphocyte binding to lymph node, mucosal, or synovial endothelial cells. The Journal of Cell Biology. 1987;105(2):983–990. doi: 10.1083/jcb.105.2.983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Goldstein LA, Zhou DFH, Picker LJ, et al. A human lymphocyte homing receptor, the hermes antigen, is related to cartilage proteoglycan core and link proteins. Cell. 1989;56(6):1063–1072. doi: 10.1016/0092-8674(89)90639-9. [DOI] [PubMed] [Google Scholar]
- 43.Nishiyama A, Dahlin KJ, Prince JT, Johnstone SR, Stallcup WB. The primary structure of NG2, a novel membrane-spanning proteoglycan. The Journal of Cell Biology. 1991;114(2):359–371. doi: 10.1083/jcb.114.2.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gerlitz B, Hassell T, Vlahos CJ, Parkinson JF, Bang NU, Grinnell BW. Identification of the predominant glycosaminoglycan-attachment site in soluble recombinant human thrombomodulin: potential regulation of functionality by glycosyltransferase competition for serine474. Biochemical Journal. 1993;295(1):131–140. doi: 10.1042/bj2950131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bourin MC, Boffa MC, Björk I, Lindahl U. Functional domains of rabbit thrombomodulin. Proceedings of the National Academy of Sciences of the United States of America. 1986;83(16):5924–5928. doi: 10.1073/pnas.83.16.5924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Maruyama I, Bell CE, Majerus PW. Thrombomodulin is found on endothelium of arteries, veins, capillaries, and lymphatics, and on syncytiotrophoblast of human placenta. The Journal of Cell Biology. 1985;101(2):363–371. doi: 10.1083/jcb.101.2.363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Maubert B, Guilbert LJ, Deloron P. Cytoadherence of Plasmodium falciparum to intercellular adhesion molecule 1 and chondroitin-4-sulfate expressed by the syncytiotrophoblast in the human placenta. Infection and Immunity. 1997;65(4):1251–1257. doi: 10.1128/iai.65.4.1251-1257.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Beeson JG, Chai W, Rogerson SJ, Lawson AM, Brown GV. Inhibition of binding of malaria-infected erythrocytes by a tetradecasaccharide fraction from chondroitin sulfate A. Infection and Immunity. 1998;66(7):3397–3402. doi: 10.1128/iai.66.7.3397-3402.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Prudhomme JG, Sherman IW, Land KM, Moses AV, Stenglein S, Nelson JA. Studies of plasmodium falciparum cytoadherence using immortalized human brain capillary endothelial cells. International Journal for Parasitology. 1996;26(6):647–655. doi: 10.1016/0020-7519(96)00027-6. [DOI] [PubMed] [Google Scholar]
- 50.Alkhalil A, Achur RN, Valiyaveettil M, Ockenhouse CF, Gowda DC. Structural requirements for the adherence of Plasmodium falciparum-infected erythrocytes to chondroitin sulfate proteoglycans of human placenta. The Journal of Biological Chemistry. 2000;275(51):40357–40364. doi: 10.1074/jbc.M006399200. [DOI] [PubMed] [Google Scholar]
- 51.Chai W, Beeson JG, Lawson AM. The structural motif in chondroitin sulfate for adhesion of Plasmodium falciparum-infected erythrocytes comprises disaccharide units of 4-O-sulfated and non-sulfated N-acetylgalactosamine linked to glucuronic acid. The Journal of Biological Chemistry. 2002;277(25):22438–22446. doi: 10.1074/jbc.M111401200. [DOI] [PubMed] [Google Scholar]
- 52.Achur RN, Valiyaveettil M, Gowda DC. The low sulfated chondroitin sulfate proteoglycans of human placenta have sulfate group-clustered domains that can efficiently bind Plasmodium falciparum-infected erythrocytes. The Journal of Biological Chemistry. 2003;278(13):11705–11713. doi: 10.1074/jbc.M211015200. [DOI] [PubMed] [Google Scholar]
- 53.Beeson JG, Rogerson SJ, Cooke BM, et al. Adhesion of Plasmodium falciparum-infected erythrocytes to hyaluronic acid in placental malaria. Nature Medicine. 2000;6(1):86–90. doi: 10.1038/71582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chai W, Beeson JG, Kogelberg H, Brown GV, Lawson AM. Inhibition of adhesion of Plasmodium falciparum-infected erythrocytes by structurally defined hyaluronic acid dodecasaccharides. Infection and Immunity. 2001;69(1):420–425. doi: 10.1128/IAI.69.1.420-425.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Valiyaveettil M, Achur RN, Alkhalil A, Ockenhouse CF, Gowda DC. Plasmodium falciparum cytoadherence to human placenta: evaluation of hyaluronic acid and chondroitin 4-sulfate for binding of infected erythrocytes. Experimental Parasitology. 2001;99(2):57–65. doi: 10.1006/expr.2001.4642. [DOI] [PubMed] [Google Scholar]
- 56.Sunderland CA, Bulmer JN, Luscombe M, Redman CWG, Stirrat GM. Immunohistological and biochemical evidence for a role for hyaluronic acid in growth and development of the placenta. Journal of Reproductive Immunology. 1985;8(2-3):197–212. doi: 10.1016/0165-0378(85)90041-5. [DOI] [PubMed] [Google Scholar]
- 57.Mohamadzadeh M, DeGrendele H, Arizpe H, Estess P, Siegelman M. Proinflammatory stimuli regulate endothelial hyaluronan expression and CD44/HA-dependent primary adhesion. Journal of Clinical Investigation. 1998;101(1):97–108. doi: 10.1172/JCI1604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rasti N, Namusoke F, Chêne A, et al. Nonimmune immunoglobulin binding and multiple adhesion characterize Plasmodium falciparum-infected erythrocytes of placental origin. Proceedings of the National Academy of Sciences of the United States of America. 2006;103(37):13795–13800. doi: 10.1073/pnas.0601519103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Flick K, Scholander C, Chen Q, et al. Role of nonimmune IgG bound to PfEMP1 in placental malaria. Science. 2001;293(5537):2098–2100. doi: 10.1126/science.1062891. [DOI] [PubMed] [Google Scholar]
- 60.Gardner MJ, Hall N, Fung E, et al. Genome sequence of the human malaria parasite Plasmodium falciparum. Nature. 2002;419(6906):498–511. doi: 10.1038/nature01097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhang J. Evolution by gene duplication: an update. Trends in Ecology and Evolution. 2003;18(6):292–298. [Google Scholar]
- 62.Smith JD, Kyes S, Craig AG, et al. Analysis of adhesive domains from the A4VAR Plasmodium falciparum erythrocyte membrane protein-1 identifies a CD36 binding domain. Molecular and Biochemical Parasitology. 1998;97(1-2):133–148. doi: 10.1016/s0166-6851(98)00145-5. [DOI] [PubMed] [Google Scholar]
- 63.Engelman DM, Steitz TA, Goldman A. Identifying nonpolar transbilayer helices in amino acid sequences of membrane proteins. Annual Review of Biophysics and Biophysical Chemistry. 1986;15:321–353. doi: 10.1146/annurev.bb.15.060186.001541. [DOI] [PubMed] [Google Scholar]
- 64.Chen Q, Fernandez V, Sundström A, et al. Developmental selection of var gene expression in Plasmodium falciparum. Nature. 1998;394(6691):392–395. doi: 10.1038/28660. [DOI] [PubMed] [Google Scholar]
- 65.Scherf A, Hernandez-Rivas R, Buffet P, et al. Antigenic variation in malaria: in situ switching, relaxed and mutually exclusive transcription of var genes during intra-erythrocytic development in Plasmodium falciparum. The EMBO Journal. 1998;17(18):5418–5426. doi: 10.1093/emboj/17.18.5418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Florens L, Washburn MP, Raine JD, et al. A proteomic view of the Plasmodium falciparum life cycle. Nature. 2002;419(6906):520–526. doi: 10.1038/nature01107. [DOI] [PubMed] [Google Scholar]
- 67.Peters J, Fowler E, Gatton M, Chen N, Saul A, Cheng Q. High diversity and rapid changeover of expressed var genes during the acute phase of Plasmodium falciparum infections in human volunteers. Proceedings of the National Academy of Sciences of the United States of America. 2002;99(16):10689–10694. doi: 10.1073/pnas.162349899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gatton ML, Peters JM, Fowler EV, Cheng Q. Switching rates of Plasmodium falciparum var genes: faster than we thought? Trends in Parasitology. 2003;19(5):202–208. doi: 10.1016/s1471-4922(03)00067-9. [DOI] [PubMed] [Google Scholar]
- 69.Deitsch KW, del Pinal A, Wellems TE. Intra-cluster recombination and var transcription switches in the antigenic variation of Plasmodium falciparum. Molecular and Biochemical Parasitology. 1999;101(1-2):107–116. doi: 10.1016/s0166-6851(99)00062-6. [DOI] [PubMed] [Google Scholar]
- 70.Kraemer SM, Smith JD. Evidence for the importance of genetic structuring to the structural and functional specialization of the Plasmodium falciparum var gene family. Molecular Microbiology. 2003;50(5):1527–1538. doi: 10.1046/j.1365-2958.2003.03814.x. [DOI] [PubMed] [Google Scholar]
- 71.Lavstsen T, Salanti A, Jensen AT, Arnot DE, Theander TG. Sub-grouping of Plasmodium falciparum 3D7 var genes based on sequence analysis of coding and non-coding regions. Malaria Journal. 2003;2(1):27. doi: 10.1186/1475-2875-2-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Calderwood MS, Gannoun-Zaki L, Wellems TE, Deitsch KW. Plasmodium falciparum var genes are regulated by two regions with separate promoters, one upstream of the coding region and a second within the intron. The Journal of Biological Chemistry. 2003;278(36):34125–34132. doi: 10.1074/jbc.M213065200. [DOI] [PubMed] [Google Scholar]
- 73.Deitsch KW, Calderwood MS, Wellems TE. Malaria: cooperative silencing elements in var genes. Nature. 2001;412(6850):875–876. doi: 10.1038/35091146. [DOI] [PubMed] [Google Scholar]
- 74.Winter G, Chen Q, Flick K, Kremsner P, Fernandez V, Wahlgren M. The 3D7var5.2 (varCOMMON) type var gene family is commonly expressed in non-placental Plasmodium falciparum malaria. Molecular and Biochemical Parasitology. 2003;127(2):179–191. doi: 10.1016/s0166-6851(03)00004-5. [DOI] [PubMed] [Google Scholar]
- 75.Freitas LH, Jr, Hernandez-Rivas R, Ralph SA, et al. Telomeric heterochromatin propagation and histone acetylation control mutually exclusive expression of antigenic variation genes in malaria parasites. Cell. 2005;121(1):25–36. doi: 10.1016/j.cell.2005.01.037. [DOI] [PubMed] [Google Scholar]
- 76.Duraisingh MT, Voss TS, Marty AJ, et al. Heterochromatin silencing and locus repositioning linked to regulation of virulence genes in Plasmodium falciparum. Cell. 2005;121(1):13–24. doi: 10.1016/j.cell.2005.01.036. [DOI] [PubMed] [Google Scholar]
- 77.Chookajorn T, Dzikowski R, Frank M, et al. Epigenetic memory at malaria virulence genes. Proceedings of the National Academy of Sciences of the United States of America. 2007;104(3):899–902. doi: 10.1073/pnas.0609084103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hviid L, Staalsoe T. Malaria immunity in infants: a special case of a general phenomenon? Trends in Parasitology. 2004;20(2):66–72. doi: 10.1016/j.pt.2003.11.009. [DOI] [PubMed] [Google Scholar]
- 79.Voss TS, Kaestli M, Vogel D, Bopp S, Beck H-P. Identification of nuclear proteins that interact differentially with Plasmodium falciparum var gene promoters. Molecular Microbiology. 2003;48(6):1593–1607. doi: 10.1046/j.1365-2958.2003.03528.x. [DOI] [PubMed] [Google Scholar]
- 80.Reeder JC, Cowman AF, Davern KM, et al. The adhesion of Plasmodium falciparum-infected erythrocytes to chondroitin sulfate A is mediated by P. falciparum erythrocyte membrane protein 1. Proceedings of the National Academy of Sciences of the United States of America. 1999;96(9):5198–5202. doi: 10.1073/pnas.96.9.5198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Buffet PA, Gamain B, Scheidig C, et al. Plasmodium falciparum domain mediating adhesion to chondroitin sulfate A: a receptor for human placental infection. Proceedings of the National Academy of Sciences of the United States of America. 1999;96(22):12743–12748. doi: 10.1073/pnas.96.22.12743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Rowe JA, Kyes SA, Rogerson SJ, Babiker HA, Raza A. Identification of a conserved Plasmodium falciparum var gene implicated in malaria in pregnancy. The Journal of Infectious Diseases. 2002;185(8):1207–1211. doi: 10.1086/339684. [DOI] [PubMed] [Google Scholar]
- 83.Fried M, Duffy PE. Two DBLγ subtypes are commonly expressed by placental isolates of Plasmodium falciparum. Molecular and Biochemical Parasitology. 2002;122(2):201–210. doi: 10.1016/s0166-6851(02)00103-2. [DOI] [PubMed] [Google Scholar]
- 84.Kyes SA, Christodoulou Z, Raza A, et al. A well-conserved Plasmodium falciparum var gene shows an unusual stage-specific transcript pattern. Molecular Microbiology. 2003;48(5):1339–1348. doi: 10.1046/j.1365-2958.2003.03505.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Salanti A, Staalsoe T, Lavstsen T, et al. Selective upregulation of a single distinctly structured var gene in chondroitin sulphate A-adhering Plasmodium falciparum involved in pregnancy-associated malaria. Molecular Microbiology. 2003;49(1):179–191. doi: 10.1046/j.1365-2958.2003.03570.x. [DOI] [PubMed] [Google Scholar]
- 86.Duffy MF, Byrne TJ, Elliott SR, et al. Broad analysis reveals a consistent pattern of var gene transcription in Plasmodium falciparum repeatedly selected for a defined adhesion phenotype. Molecular Microbiology. 2005;56(3):774–788. doi: 10.1111/j.1365-2958.2005.04577.x. [DOI] [PubMed] [Google Scholar]
- 87.Fried M, Wendler JP, Mutabingwa TK, Duffy PE. Mass spectrometric analysis of Plasmodium falciparum erythrocyte membrane protein-1 variants expressed by placental malaria parasites. Proteomics. 2004;4(4):1086–1093. doi: 10.1002/pmic.200300666. [DOI] [PubMed] [Google Scholar]
- 88.Gowda AS, Madhunapantula SV, Achur RN, Valiyaveettil M, Bhavanandan VP, Gowda DC. Structural basis for the adherence of Plasmodium falciparum-infected erythrocytes to chondroitin 4-sulfate and design of novel photoactivable reagents for the identification of parasite adhesive proteins. The Journal of Biological Chemistry. 2007;282(2):916–928. doi: 10.1074/jbc.M604741200. [DOI] [PubMed] [Google Scholar]
- 89.Gamain B, Trimnell AR, Scheidig C, Schert A, Miller LH, Smith JD. Identification of multiple chondroitin sulfate A (CSA)-binding domains in the var2CSA gene transcribed in CSA-binding parasites. The Journal of Infectious Diseases. 2005;191(6):1010–1013. doi: 10.1086/428137. [DOI] [PubMed] [Google Scholar]
- 90.Fried M, Duffy PE. Adherence of Plasmodium falciparum to chondroitin sulfate A in the human placenta. Science. 1996;272(5267):1502–1504. doi: 10.1126/science.272.5267.1502. [DOI] [PubMed] [Google Scholar]
- 91.Achur RN, Valiyaveettil M, Alkhalil A, Ockenhouse CF, Gowda DC. Characterization of proteoglycans of human placenta and identification of unique chondroitin sulfate proteoglycans of the intervillous spaces that mediate the adherence of Plasmodium falciparum-infected erythrocytes to the placenta. The Journal of Biological Chemistry. 2000;275(51):40344–40356. doi: 10.1074/jbc.M006398200. [DOI] [PubMed] [Google Scholar]
- 92.Bull PC, Lowe BS, Kortok M, Marsh K. Antibody recognition of Plasmodium falciparum erythrocyte surface antigens in Kenya: evidence for rare and prevalent variants. Infection and Immunity. 1999;67(2):733–739. doi: 10.1128/iai.67.2.733-739.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Fried M, Nosten F, Brockman A, Brabin BJ, Duffy PE. Maternal antibodies block malaria. Nature. 1998;395(6705):851–852. doi: 10.1038/27570. [DOI] [PubMed] [Google Scholar]
- 94.Marsh K, Otoo L, Hayes RJ, Carson DC, Greenwood BM. Antibodies to blood stage antigens of Plasmodium falciparum in rural Gambians and their relation to protection against infection. Transactions of the Royal Society of Tropical Medicine and Hygiene. 1989;83(3):293–303. doi: 10.1016/0035-9203(89)90478-1. [DOI] [PubMed] [Google Scholar]
- 95.Nielsen MA, Vestergaard LS, Lusingu J, et al. Geographical and temporal conservation of antibody recognition of Plasmodium falciparum variant surface antigens. Infection and Immunity. 2004;72(6):3531–3535. doi: 10.1128/IAI.72.6.3531-3535.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Staalsoe T, Shulman CE, Bulmer JN, Kawuondo K, Marsh K, Hviid L. Variant surface antigen-specific IgG and protection against clinical consequences of pregnancy-associated Plasmodium falciparum malaria. The Lancet. 2004;363(9405):283–289. doi: 10.1016/S0140-6736(03)15386-X. [DOI] [PubMed] [Google Scholar]
- 97.Duffy PE, Fried M. Antibodies that inhibit Plasmodium falciparum adhesion to chondroitin sulfate A are associated with increased birth weight and the gestational age of newborns. Infection and Immunity. 2003;71(11):6620–6623. doi: 10.1128/IAI.71.11.6620-6623.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Fried M, Duffy PE. Maternal malaria and parasite adhesion. Journal of Molecular Medicine. 1998;76(3-4):162–171. doi: 10.1007/s001090050205. [DOI] [PubMed] [Google Scholar]
- 99.Duffy MF, Caragounis A, Noviyanti R, et al. Transcribed var genes associated with placental malaria in Malawian women. Infection and Immunity. 2006;74(8):4875–4883. doi: 10.1128/IAI.01978-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Salanti A, Dahlbäck M, Turner L, et al. Evidence for the involvement of VAR2CSA in pregnancy-associated malaria. Journal of Experimental Medicine. 2004;200(9):1197–1203. doi: 10.1084/jem.20041579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Barfod L, Bernasconi NL, Dahlbäck M, et al. Human pregnancy-associated malaria-specific B cells target polymorphic, conformational epitopes in VAR2CSA. Molecular Microbiology. 2007;63(2):335–347. doi: 10.1111/j.1365-2958.2006.05503.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Tuikue Ndam N, Salanti A, Le-Hesran J-Y, et al. Dynamics of anti-VAR2CSA immunoglobulin G response in a cohort of senegalese pregnant women. The Journal of Infectious Diseases. 2006;193(5):713–720. doi: 10.1086/500146. [DOI] [PubMed] [Google Scholar]
- 103.Avril M, Gamain B, LéPolard C, Viaud N, Scherf A, Gysin J. Characterization of anti-var2CSA-PfEMP1 cytoadhesion inhibitory mouse monoclonal antibodies. Microbes and Infection. 2006;8(14-15):2863–2871. doi: 10.1016/j.micinf.2006.09.005. [DOI] [PubMed] [Google Scholar]
- 104.Megnekou R, Staalsoe T, Taylor DW, Leke R, Hviid L. Effects of pregnancy and intensity of Plasmodium falciparum transmission on immunoglobulin G subclass responses to variant surface antigens. Infection and Immunity. 2005;73(7):4112–4118. doi: 10.1128/IAI.73.7.4112-4118.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Jensen ATR, Magistrado P, Sharp S, et al. Plasmodium falciparum associated with severe childhood malaria preferentially expresses PfEMP1 encoded by group A var genes. Journal of Experimental Medicine. 2004;199(9):1179–1190. doi: 10.1084/jem.20040274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Hviid L, Staalsoe T. Malaria immunity in infants: a special case of a general phenomenon? Trends in Parasitology. 2004;20(2):66–72. doi: 10.1016/j.pt.2003.11.009. [DOI] [PubMed] [Google Scholar]
- 107.Lavstsen T, Magistrado P, Hermsen CC, et al. Expression of Plasmodium falciparum erythrocyte membrane protein 1 in experimentally infected humans. Malaria Journal. 2005;4(1):21. doi: 10.1186/1475-2875-4-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Dahlbäck M, Rask TS, Andersen PH, et al. Epitope mapping and topographic analysis of VAR2CSA DBL3X involved in P. falciparum placental sequestration. PLoS Pathogens. 2006;2(11):e124. doi: 10.1371/journal.ppat.0020124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Krishnan L, Guilbert LJ, Wegmann TG, Belosevic M, Mosmann TR. T helper 1 response against Leishmania major in pregnant C57BL/6 mice increases implantation failure and fetal resorptions: correlation with increased IFN-γ and TNF and reduced IL-10 production by placental cells. The Journal of Immunology. 1996;156(2):653–662. [PubMed] [Google Scholar]
- 110.Wegmann TG, Lin H, Guilbert L, Mosmann TR. Bidirectional cytokine interactions in the maternal-fetal relationship: is successful pregnancy a TH2 phenomenon? Immunology Today. 1993;14(7):353–356. doi: 10.1016/0167-5699(93)90235-D. [DOI] [PubMed] [Google Scholar]
- 111.Chaouat G, Cayol V, Mairovitz V, Dubanchet S. Localization of the Th2 cytokines IL-3, IL-4, IL-10 at the fetomaternal interface during human and murine pregnancy and lack of requirement for Fas/Fas ligand interaction for a successful allogeneic pregnancy. American Journal of Reproductive Immunology. 1999;42(1):1–13. doi: 10.1111/j.1600-0897.1999.tb00459.x. [DOI] [PubMed] [Google Scholar]
- 112.Sacks GP, Studena K, Sargent IL, Redman CWG. Normal pregnancy and preeclampsia both produce inflammatory changes in peripheral blood leukocytes akin to those of sepsis. American Journal of Obstetrics and Gynecology. 1998;179(1):80–86. doi: 10.1016/s0002-9378(98)70254-6. [DOI] [PubMed] [Google Scholar]
- 113.Fried M, Muga RO, Misore AO, Duffy PE. Malaria elicits type 1 cytokines in the human placenta: IFN-γ and TNF-α associated with pregnancy outcomes. The Journal of Immunology. 1998;160(5):2523–2530. [PubMed] [Google Scholar]
- 114.Moormann AM, Sullivan AD, Rochford RA, et al. Malaria and pregnancy: placental cytokine expression and its relationship to intrauterine growth retardation. The Journal of Infectious Diseases. 1999;180(6):1987–1993. doi: 10.1086/315135. [DOI] [PubMed] [Google Scholar]
- 115.Fievet N, Moussa M, Tami G, et al. Plasmodium falciparum induces a Th1/Th2 disequilibrium, favoring the Th1-type pathway, in the human placenta. The Journal of Infectious Diseases. 2001;183(10):1530–1534. doi: 10.1086/320201. [DOI] [PubMed] [Google Scholar]
- 116.Diouf I, Fievet N, Doucouré S, et al. Monocyte activation and T cell inhibition in Plasmodium falciparum-infected placenta. The Journal of Infectious Diseases. 2004;189(12):2235–2242. doi: 10.1086/420791. [DOI] [PubMed] [Google Scholar]
- 117.Ordi J, Ismail MR, Ventura PJ, et al. Massive chronic intervillositis of the placenta associated with malaria infection. American Journal of Surgical Pathology. 1998;22(8):1006–1011. doi: 10.1097/00000478-199808000-00011. [DOI] [PubMed] [Google Scholar]
- 118.Moore JM, Nahlen BL, Misore A, Lal AA, Udhayakumar V. Immunity to placental malaria. I. Elevated production of interferon-γ by placental blood mononuclear cells is associated with protection in an area with high transmission of malaria. The Journal of Infectious Diseases. 1999;179(5):1218–1225. doi: 10.1086/314737. [DOI] [PubMed] [Google Scholar]
- 119.Beeson JG, Rogerson SJ, Elliott SR, Duffy MF. Targets of protective antibodies to malaria during pregnancy. The Journal of Infectious Diseases. 2005;192(9):1647–1650. doi: 10.1086/496895. [DOI] [PubMed] [Google Scholar]
- 120.Chaisavaneeyakorn S, Lucchi N, Abramowsky C, et al. Immunohistological characterization of macrophage migration inhibitory factor expression in Plasmodium falciparum-infected placentas. Infection and Immunity. 2005;73(6):3287–3293. doi: 10.1128/IAI.73.6.3287-3293.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Bertin G, Tuikue Ndam N, Jafari-Guemouri S, et al. High prevalence of Plasmodium falciparum pfcrt K76T mutation in pregnant women taking chloroquine prophylaxis in Senegal. Journal of Antimicrobial Chemotherapy. 2005;55(5):788–791. doi: 10.1093/jac/dki097. [DOI] [PubMed] [Google Scholar]
- 122.Schultz LJ, Steketee RW, Chitsulo L, Macheso A, Nyasulu Y, Ettling M. Malaria and childbearing women in Malawi: knowledge, attitudes and practices. Tropical Medicine and Parasitology. 1994;45(1):65–69. [PubMed] [Google Scholar]
- 123.Parise ME, Ayisi JG, Nahlen BL, et al. Efficacy of sulfadoxine-pyrimethamine for prevention of placental malaria in an area of Kenya with a high prevalence of malaria and human immunodeficiency virus infection. American Journal of Tropical Medicine and Hygiene. 1998;59(5):813–822. doi: 10.4269/ajtmh.1998.59.813. [DOI] [PubMed] [Google Scholar]
- 124.Shulman CE, Dorman EK, Cutts F, et al. Intermittent sulphadoxine-pyrimethamine to prevent severe anaemia secondary to malaria in pregnancy: a randomised placebo-controlled trial. The Lancet. 1999;353(9153):632–636. doi: 10.1016/s0140-6736(98)07318-8. [DOI] [PubMed] [Google Scholar]
- 125.Vogt AM, Pettersson F, Moll K, et al. Release of sequestered malaria parasites upon injection of a glycosaminoglycan. PLoS Pathogens. 2006;2(9):e100. doi: 10.1371/journal.ppat.0020100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Spillmann D. Carbohydrates in cellular recognition: from leucine-zipper to sugar-zipper? Glycoconjugate Journal. 1994;11(3):169–171. doi: 10.1007/BF00731214. [DOI] [PubMed] [Google Scholar]
- 127.Barragan A, Spillmann D, Carlson J, Wahlgren M. Role of glycans in Plasmodium falciparum infection. Biochemical Society Transactions. 1999;27(4):487–493. doi: 10.1042/bst0270487. [DOI] [PubMed] [Google Scholar]
- 128.Pouvelle B, Meyer P, Robert C, Bardel L, Gysin J. Chondroitin-4-sulfate impairs in vitro and in vivo cytoadherence of Plasmodium falciparum infected erythrocytes. Molecular Medicine. 1997;3(8):508–518. [PMC free article] [PubMed] [Google Scholar]
- 129.Carlson J, Wahlgren M. Plasmodium falciparum erythrocyte rosetting is mediated by promiscuous lectin-like interactions. Journal of Experimental Medicine. 1992;176(5):1311–1317. doi: 10.1084/jem.176.5.1311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Clark DL, Su S, Davidson EA. Saccharide anions as inhibitors of the malaria parasite. Glycoconjugate Journal. 1997;14(4):473–479. doi: 10.1023/a:1018551518610. [DOI] [PubMed] [Google Scholar]
- 131.Rowe A, Berendt AR, Marsh K, Newbold CI. Plasmodium falciparum: a family of sulphated glycoconjugates disrupts erythrocyte rosettes. Experimental Parasitology. 1994;79(4):506–516. doi: 10.1006/expr.1994.1111. [DOI] [PubMed] [Google Scholar]
- 132.Xiao L, Yang C, Patterson PS, Udhayakumar V, Lal AA. Sulfated polyanions inhibit invasion of erythrocytes by plasmodial merozoites and cytoadherence of endothelial cells to parasitized erythrocytes. Infection and Immunity. 1996;64(4):1373–1378. doi: 10.1128/iai.64.4.1373-1378.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Andrews KT, Klatt N, Adams Y, Mischnick P, Schwartz-Albiez R. Inhibition of chondroitin-4-sulfate-specific adhesion of Plasmodium falciparum-infected erythrocytes by sulfated polysaccharides. Infection and Immunity. 2005;73(7):4288–4294. doi: 10.1128/IAI.73.7.4288-4294.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]