Abstract
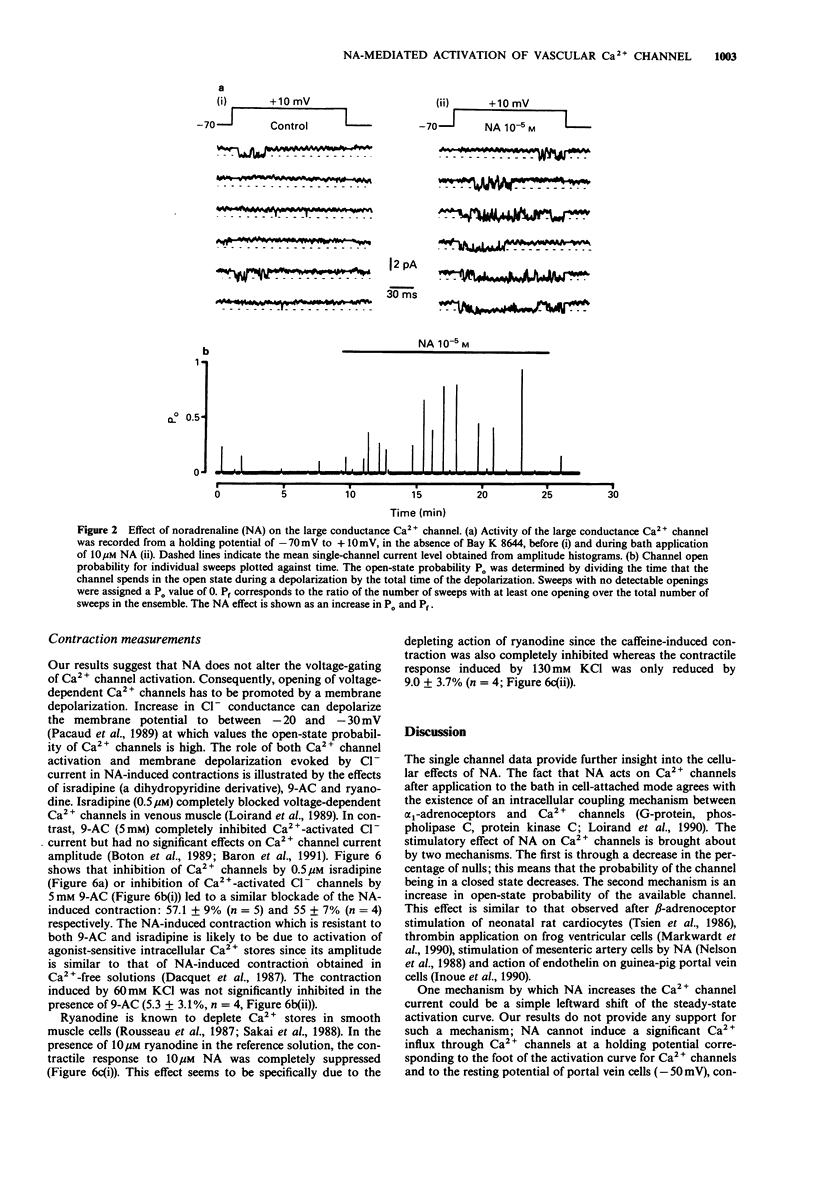
1. The effects of noradrenaline (NA) were studied on vascular smooth muscle cells isolated from rat portal vein. 2. Two types of single-Ca2+ channel currents with conductances of 17 pS and 8 pS were obtained in cell-attached configuration. Bath application of NA increased the open probability of both channels during depolarizing pulses without a change of background membrane conductance. However, NA did not open Ca2+ channels when the membrane patch potential was held at -50 mV, which is about the resting potential in physiological conditions. 3. In the whole-cell configuration, studies of voltage-dependent Ca2+ channel currents showed that the peak conductance curve was not shifted to more negative potentials by NA. 4. Measurements of internal Ca(2+)-concentration ([Ca2+]i) with Indo-1 indicated that NA increased [Ca2+]i at a holding potential of -50 mV and evoked a Ca(2+)-activated Cl- current. These effects were blocked when heparin was included in the pipette solution. 5. A Cl- channel blocker without effect on Ca2+ channels (anthracene-9-carboxylic acid) inhibited the contractions of portal vein strips induced by NA in a manner similar to that produced by a Ca2+ channel inhibitor (isradipine). The NA-induced contraction was completely suppressed in the presence of ryanodine which depletes intracellular Ca2+ stores. 6. The present study suggests that activation of Cl- channels by Ca2+ release produces a membrane depolarization which is a prerequisite for enhanced opening of voltage-dependent Ca2+ channels in response to NA in venous smooth muscle.
Full text
PDF

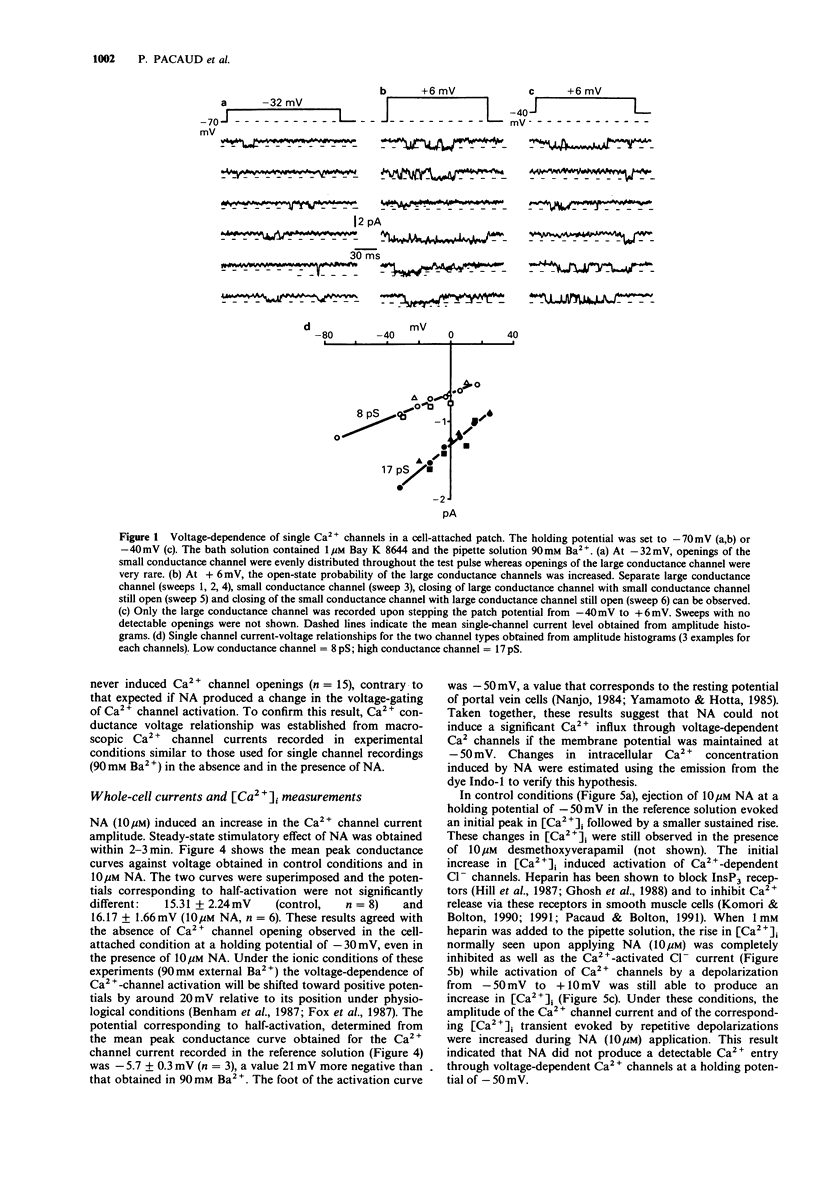
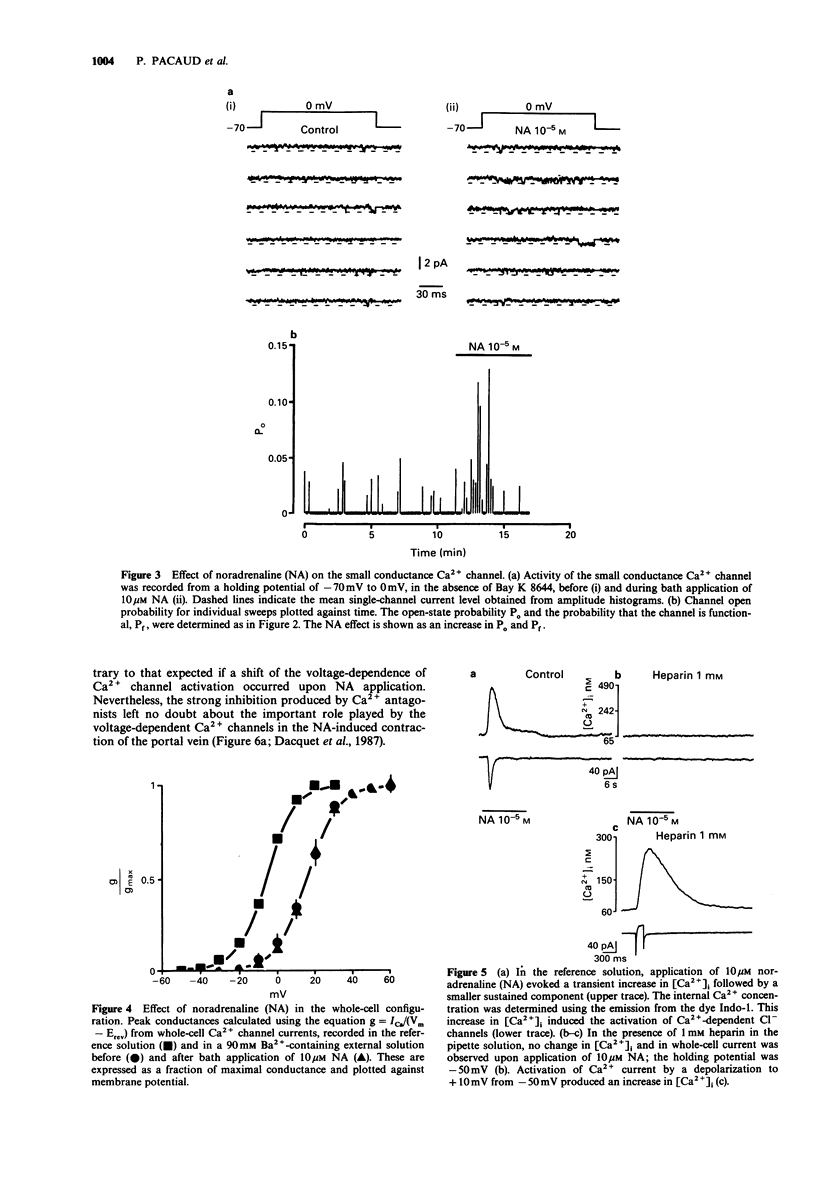


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Almers W., Neher E. The Ca signal from fura-2 loaded mast cells depends strongly on the method of dye-loading. FEBS Lett. 1985 Nov 11;192(1):13–18. doi: 10.1016/0014-5793(85)80033-8. [DOI] [PubMed] [Google Scholar]
- Benham C. D., Tsien R. W. Noradrenaline modulation of calcium channels in single smooth muscle cells from rabbit ear artery. J Physiol. 1988 Oct;404:767–784. doi: 10.1113/jphysiol.1988.sp017318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benham C. D. Voltage-gated and agonist-mediated rises in intracellular Ca2+ in rat clonal pituitary cells (GH3) held under voltage clamp. J Physiol. 1989 Aug;415:143–158. doi: 10.1113/jphysiol.1989.sp017716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolton T. B. Mechanisms of action of transmitters and other substances on smooth muscle. Physiol Rev. 1979 Jul;59(3):606–718. doi: 10.1152/physrev.1979.59.3.606. [DOI] [PubMed] [Google Scholar]
- Boton R., Dascal N., Gillo B., Lass Y. Two calcium-activated chloride conductances in Xenopus laevis oocytes permeabilized with the ionophore A23187. J Physiol. 1989 Jan;408:511–534. doi: 10.1113/jphysiol.1989.sp017473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byrne N. G., Large W. A. Mechanism of action of alpha-adrenoceptor activation in single cells freshly dissociated from the rabbit portal vein. Br J Pharmacol. 1988 Jun;94(2):475–482. doi: 10.1111/j.1476-5381.1988.tb11550.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Byrne N. G., Large W. A. Membrane ionic mechanisms activated by noradrenaline in cells isolated from the rabbit portal vein. J Physiol. 1988 Oct;404:557–573. doi: 10.1113/jphysiol.1988.sp017306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bülbring E., Tomita T. Catecholamine action on smooth muscle. Pharmacol Rev. 1987 Mar;39(1):49–96. [PubMed] [Google Scholar]
- Dacquet C., Mironneau C., Mironneau J. Effects of calcium entry blockers on calcium-dependent contractions of rat portal vein. Br J Pharmacol. 1987 Sep;92(1):203–211. doi: 10.1111/j.1476-5381.1987.tb11313.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox A. P., Nowycky M. C., Tsien R. W. Kinetic and pharmacological properties distinguishing three types of calcium currents in chick sensory neurones. J Physiol. 1987 Dec;394:149–172. doi: 10.1113/jphysiol.1987.sp016864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh T. K., Eis P. S., Mullaney J. M., Ebert C. L., Gill D. L. Competitive, reversible, and potent antagonism of inositol 1,4,5-trisphosphate-activated calcium release by heparin. J Biol Chem. 1988 Aug 15;263(23):11075–11079. [PubMed] [Google Scholar]
- Grynkiewicz G., Poenie M., Tsien R. Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem. 1985 Mar 25;260(6):3440–3450. [PubMed] [Google Scholar]
- Hamill O. P., Marty A., Neher E., Sakmann B., Sigworth F. J. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981 Aug;391(2):85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Inoue R., Isenberg G. Effect of membrane potential on acetylcholine-induced inward current in guinea-pig ileum. J Physiol. 1990 May;424:57–71. doi: 10.1113/jphysiol.1990.sp018055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue R., Isenberg G. Intracellular calcium ions modulate acetylcholine-induced inward current in guinea-pig ileum. J Physiol. 1990 May;424:73–92. doi: 10.1113/jphysiol.1990.sp018056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inoue Y., Oike M., Nakao K., Kitamura K., Kuriyama H. Endothelin augments unitary calcium channel currents on the smooth muscle cell membrane of guinea-pig portal vein. J Physiol. 1990 Apr;423:171–191. doi: 10.1113/jphysiol.1990.sp018017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klöckner U., Isenberg G. Endothelin depolarizes myocytes from porcine coronary and human mesenteric arteries through a Ca-activated chloride current. Pflugers Arch. 1991 Mar;418(1-2):168–175. doi: 10.1007/BF00370467. [DOI] [PubMed] [Google Scholar]
- Komori S., Bolton T. B. Calcium release induced by inositol 1,4,5-trisphosphate in single rabbit intestinal smooth muscle cells. J Physiol. 1991 Feb;433:495–517. doi: 10.1113/jphysiol.1991.sp018440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komori S., Bolton T. B. Role of G-proteins in muscarinic receptor inward and outward currents in rabbit jejunal smooth muscle. J Physiol. 1990 Aug;427:395–419. doi: 10.1113/jphysiol.1990.sp018178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loirand G., Mironneau C., Mironneau J., Pacaud P. Two types of calcium currents in single smooth muscle cells from rat portal vein. J Physiol. 1989 May;412:333–349. doi: 10.1113/jphysiol.1989.sp017619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loirand G., Pacaud P., Mironneau C., Mironneau J. Evidence for two distinct calcium channels in rat vascular smooth muscle cells in short-term primary culture. Pflugers Arch. 1986 Nov;407(5):566–568. doi: 10.1007/BF00657519. [DOI] [PubMed] [Google Scholar]
- Loirand G., Pacaud P., Mironneau C., Mironneau J. GTP-binding proteins mediate noradrenaline effects on calcium and chloride currents in rat portal vein myocytes. J Physiol. 1990 Sep;428:517–529. doi: 10.1113/jphysiol.1990.sp018225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markwardt F., Franke T., Albitz R., Nilius B. Effects of thrombin on single calcium channels in frog ventricular cells. Pflugers Arch. 1990 Feb;415(5):547–553. doi: 10.1007/BF02583505. [DOI] [PubMed] [Google Scholar]
- Mironneau J., Gargouil Y. M. Action of indapamide on excitation-contraction coupling in vascular smooth muscle. Eur J Pharmacol. 1979 Jul 15;57(1):57–67. doi: 10.1016/0014-2999(79)90103-1. [DOI] [PubMed] [Google Scholar]
- Nanjo T. Effects of noradrenaline and acetylcholine on electro-mechanical properties of the guinea-pig portal vein. Br J Pharmacol. 1984 Mar;81(3):427–440. doi: 10.1111/j.1476-5381.1984.tb10095.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson M. T., Patlak J. B., Worley J. F., Standen N. B. Calcium channels, potassium channels, and voltage dependence of arterial smooth muscle tone. Am J Physiol. 1990 Jul;259(1 Pt 1):C3–18. doi: 10.1152/ajpcell.1990.259.1.C3. [DOI] [PubMed] [Google Scholar]
- Nelson M. T., Standen N. B., Brayden J. E., Worley J. F., 3rd Noradrenaline contracts arteries by activating voltage-dependent calcium channels. Nature. 1988 Nov 24;336(6197):382–385. doi: 10.1038/336382a0. [DOI] [PubMed] [Google Scholar]
- Pacaud P., Loirand G., Mironneau C., Mironneau J. Noradrenaline activates a calcium-activated chloride conductance and increases the voltage-dependent calcium current in cultured single cells of rat portal vein. Br J Pharmacol. 1989 May;97(1):139–146. doi: 10.1111/j.1476-5381.1989.tb11934.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rousseau E., Smith J. S., Meissner G. Ryanodine modifies conductance and gating behavior of single Ca2+ release channel. Am J Physiol. 1987 Sep;253(3 Pt 1):C364–C368. doi: 10.1152/ajpcell.1987.253.3.C364. [DOI] [PubMed] [Google Scholar]
- Sakai T., Terada K., Kitamura K., Kuriyama H. Ryanodine inhibits the Ca-dependent K current after depletion of Ca stored in smooth muscle cells of the rabbit ileal longitudinal muscle. Br J Pharmacol. 1988 Dec;95(4):1089–1100. doi: 10.1111/j.1476-5381.1988.tb11743.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki H. Effects of endogenous and exogenous noradrenaline on the smooth muscle of guinea-pig mesenteric vein. J Physiol. 1981 Dec;321:495–512. doi: 10.1113/jphysiol.1981.sp013999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takata Y. Regional differences in electrical and mechanical properties of guinea-pig mesenteric vessels. Jpn J Physiol. 1980;30(5):709–728. doi: 10.2170/jjphysiol.30.709. [DOI] [PubMed] [Google Scholar]
- Tsien R. W., Bean B. P., Hess P., Lansman J. B., Nilius B., Nowycky M. C. Mechanisms of calcium channel modulation by beta-adrenergic agents and dihydropyridine calcium agonists. J Mol Cell Cardiol. 1986 Jul;18(7):691–710. doi: 10.1016/s0022-2828(86)80941-5. [DOI] [PubMed] [Google Scholar]
- Van Helden D. F. An alpha-adrenoceptor-mediated chloride conductance in mesenteric veins of the guinea-pig. J Physiol. 1988 Jul;401:489–501. doi: 10.1113/jphysiol.1988.sp017174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto Y., Hotta K. Mechanisms involved in contraction of smooth muscles of the rat portal vein as induced by sodium depletion. Jpn J Physiol. 1985;35(5):717–727. doi: 10.2170/jjphysiol.35.717. [DOI] [PubMed] [Google Scholar]


