Abstract
An investigation of gene expression in Staphylococcus aureus after a switch from aerobic to anaerobic growth was initiated by using the proteomic and transcriptomic approaches. In the absence of external electron acceptors like oxygen or nitrate, an induction of glycolytic enzymes was observed. At the same time the amount of tricarboxylic acid cycle enzymes was very low. NAD is regenerated by mixed acid and butanediol fermentation, as indicated by an elevated synthesis level of fermentation enzymes like lactate dehydrogenases (Ldh1 and Ldh2), alcohol dehydrogenases (AdhE and Adh), α-acetolactate decarboxylase (BudA1), acetolactate synthase (BudB), and acetoin reductase (SACOL0111) as well as an accumulation of fermentation products as lactate and acetate. Moreover, the transcription of genes possibly involved in secretion of lactate (SACOL2363) and formate (SACOL0301) was found to be induced. The formation of acetyl-coenzyme A or acetyl-phosphate might be catalyzed by pyruvate formate lyase, whose synthesis was found to be strongly induced as well. Although nitrate was not present, the expression of genes related to nitrate respiration (NarH, NarI, and NarJ) and nitrate reduction (NirD) was found to be upregulated. Of particular interest, oxygen concentration might affect the virulence properties of S. aureus by regulating the expression of some virulence-associated genes such as pls, hly, splC and splD, epiG, and isaB. To date, the mechanism of anaerobic gene expression in S. aureus has not been fully characterized. In addition to srrA the mRNA levels of several other regulatory genes with yet unknown functions (e.g., SACOL0201, SACOL2360, and SACOL2658) were found to be upregulated during anaerobic growth, indicating a role in the regulation of anaerobic gene expression.
Staphylococcus aureus is one of the leading causative agents of nosocomial infections. Due to the widespread emergence of antibiotic resistance, S. aureus in one of the most feared pathogens in the hospital and, in particular, in intensive care units. For this reason, S. aureus has been a subject of intensive research for many years. As a result, there is a large amount of data available concerning the regulation, function, and structure of various virulence factors and of proteins involved in antibiotic resistance. In addition, epidemiological studies have been of great importance. In contrast, only a few studies have focused on the basic cellular physiology of this pathogen. In particular, in recent years it has become increasingly accepted that this basic cell physiology determines not only growth and survival but pathogenicity as well. Hence, much more knowledge about cell physiology and molecular processes involved in infection is urgently needed to understand staphylococcal pathogenicity, which is a central point for the development of novel tools for diagnosis and successful treatment of S. aureus infections in the future. Functional genomics opens up the opportunity for a new and comprehensive understanding of the cell physiology and infection biology of this bacterium.
Since S. aureus has the ability to invade different tissues, oxygen could be one of the most crucial growth-limiting stimuli (11). It was shown previously that oxygen tension varies between different sites in the host. In particular, in abscesses completely anaerobic conditions have often been found (54). S. aureus can grow under low-oxygen conditions by fermentation or nitrate respiration (6, 19, 68). Previous studies indicate that oxygen plays an important role in virulence gene regulation and in the bacteria's ability to persist and grow in ecological niches similar to those of the host environment (9, 10, 37, 51, 56, 60, 75). In particular, anaerobic conditions and low carbon dioxide levels repress production of tst1 in S. aureus clinical isolates (60, 74, 75). Furthermore, it has been shown that the expression of ica genes involved in cell-to-cell adhesion and biofilm formation is stimulated under anaerobic conditions in S. aureus and Staphylococcus epidermidis (13, 14).
The related gram-positive bacterium Bacillus subtilis performs a mixed-acid fermentation with lactate, acetate, and acetoin under anaerobic conditions (15, 46). In the presence of nitrate, B. subtilis is able to use respiratory processes to reduce nitrate and nitrite (25, 30, 31, 46). The regulatory network that enables the bacteria to adapt gene expression to anaerobic conditions has been intensively studied in recent years. The two-component system ResDE represents one major regulatory system involved in anaerobic gene regulation (30, 46, 47, 49, 69). The mechanism of signal perception by ResDE is still unknown. The N-terminal domain of the histidine kinase ResE contains two membrane subdomains and a large extracytoplasmic loop. Additionally, a cytoplasmic PAS domain was localized between the HAMP linker and the C-terminal kinase domain. A deletion of the PAS domain prevents gene activation under anaerobic conditions (1). ResDE positively regulates genes known to be induced under anaerobic conditions such as fnr, which encodes another main regulator important under anaerobic conditions, the nitrate reductase genes nasDEF, and the flavohemoglobin gene hmp (46, 48, 49). However, most of the effects of ResDE on anaerobic gene regulation in B. subtilis might be mediated through Fnr. A mutant in fnr is not able to use nitrate as a terminal electron acceptor (49). Very recently, the Fnr regulon was defined by using DNA arrays. In total, the expression of 37 genes was found to be changed by a mutation in fnr under anaerobic conditions (57).
A two-component system, SrrAB (for staphylococcal respiratory response AB; synonym, SrhSR), which has a high similarity to the ResDE two-component system of B. subtilis, seems to be one global regulator of the aerobic-anaerobic shift of metabolism in S. aureus (56, 71, 74). Throup et al. (71) provided the first proteomic data indicating that SrrAB controls the upregulation of fermentation enzymes (lactate dehydrogenase and alcohol dehydrogenase) as well as the downregulation of aerobic tricarboxylic acid (TCA) cycle enzymes (succinyl-coenzyme A [succinyl-CoA] synthetase, aconitase, and fumarase) under anaerobic conditions. A mutant in srrAB is characterized by a severe growth defect under these conditions. However, no data are available concerning the mechanism by which SrrAB regulates the transcription of the respective genes. An Fnr protein described in other bacteria is not encoded in the S. aureus genome. Very recently it was shown that the nreABC operon encodes a two-component regulatory system which is involved in the regulation of nitrate/nitrite reduction in S. aureus in response to oxygen (35). The sensor protein NreB is a cytoplasmic protein containing a cysteine cluster which is required for sensing oxygen. The NreB kinase activity depends on iron-sulfur reconstitution and oxygen availability. In the absence of oxygen the autokinase of NreB is activated, and the phosphoryl group is transferred to the response regulator NreC (35).
The proteome map of S. aureus COL already established by Kohler et al. (39) provides an essential tool for a better understanding of cell physiology of S. aureus under defined and nondefined conditions. Most of the metabolic enzymes were visualized in the main proteomic window of pI 4 to 7. In the present study we used this map to identify all those proteins whose synthesis was affected by oxygen concentration. Additionally, the effect of nitrate on protein synthesis was studied. This proteomic approach was complemented by a transcriptomic approach analyzing overall gene expression in the absence of oxygen and nitrate.
MATERIALS AND METHODS
Bacterial strains and growth conditions.
For anaerobic experiments, the S. aureus COL strain (63) was first grown under aerobic conditions in 100 ml of synthetic medium (23) in 500-ml Erlenmeyer flasks under vigorous agitation at 37°C to an optical density at 500 nm (OD500) of 0.5. Cells were then shifted to anaerobic growth by transferring the culture to screw-top ultracentrifugation tubes (10 ml; VWR, Darmstadt, Germany) or Falcon tubes (50 ml) which were completely filled with medium and incubated under vigorous agitation at 37°C.
Anaerobic conditions were verified by using 0.001% resazurin as a redox indicator (42, 65). Microaerophilic and anaerobic conditions were observed at 8 and 23 min after the shift, respectively.
Preparation of pulse-labeled protein extracts.
For the analysis of protein synthesis under aerobic and anaerobic growth conditions, the proteins were pulse-labeled with l-[35S]methionine. Briefly, at time point zero (aerobic growth at an OD500 of 0.5) and at 10, 20, 30, and 60 min after the shift to anaerobic conditions, 500 μCi of l-[35S]methionine was added to the cell culture, which was then cultivated for an additional 5 min to allow the uptake and incorporation of l-[35S]methionine in newly synthesized proteins. To ensure that the pattern of protein synthesis remained unchanged during the course of the experiment in an aerobically grown culture, we also pulse-labeled cells of nontreated culture 60 min after time point zero.
Exactly 5 min after the addition of radioactively labeled methionine, protein synthesis was blocked by the addition of 1 ml of stop solution (90 mg/ml chloramphenicol and 136 mg/ml unlabeled methionine). The cells were separated from the medium by centrifugation (9,000 × g at 4°C for 5 min), washed twice with TE buffer (10 mM Tris, 1 mM EDTA, pH 7.5), and stored at −20°C. Protein extracts from these cells were prepared by resuspension of the cell pellet in 400 μl of TE buffer containing 0.01 mg of lysostaphin, followed by incubation on ice for 10 min and disruption by sonication. The cell lysate was cleared by a first centrifugation step (9,000 × g for 10 min at 4°C) in order to remove cell debris, followed by a second centrifugation step (21,000 × g for 30 min at 4°C) to remove insoluble and aggregated proteins which could interfere with isoelectric focusing. The radioactive labels of proteins were determined by standard procedures (2). The protein concentration was determined using Roti Nanoquant reagent (Roth, Germany), and the protein solution was stored at −20°C.
Preparation of cytoplasmic proteins for preparative two-dimensional (2D) gel electrophoresis.
Cells of 50-ml cultures were harvested on ice and centrifuged for 10 min at 7,000 × g and 4°C. Cells were washed twice with ice-cold TE buffer and resuspended in 2 ml of lysis buffer (10 mM Tris, 1 mM EDTA, 1 mM phenylmethylsulfonyl fluoride, pH 7.5) containing 50 μg of lysostaphin. For cell lysis, the cell suspension was incubated on ice for 10 min, and afterwards the cells were disrupted using a French press (SLM Instruments). The lysate was centrifuged for 10 min at 9,000 × g (4°C). In order to remove membrane fragments and insoluble proteins, the lysate was centrifuged for 30 min at 21,000 × g (4°C). The protein concentration was determined using Roti Nanoquant (Roth, Germany), and the protein solution was stored at −20°C.
Analytical and preparative 2D-PAGE.
2D polyacrylamide gel electrophoresis (2D-PAGE) was performed using the immobilized pH gradient (IPG) technique described previously (7). In the first dimension, the protein samples were separated on IPG strips (GE-Healthcare, Little Chalfont, United Kingdom) in the pH range of 4 to 7. For analytical 2D-PAGE 80 μg of radioactively labeled protein extracts was loaded onto the IPG strips. The resulting 2D gels were fixed with 40% (vol/vol) ethanol and 10% (vol/vol) acetic acid for 1 to 2 h and subsequently stained with silver nitrate (3). The stained gels were scanned. Afterwards the gels were dried in a vacuum dryer and fixed onto Whatman paper. The dried gels were exposed to storage phosphor screens (Molecular Dynamics, Krefeld, Germany) and scanned with a Storm 840 PhosphorImager (Molecular Dynamics, Krefeld, Germany). The exposure time of each gel depended on the signal intensity.
For identification of proteins by mass spectrometry (MS), preparative 2D-PAGE was performed. A total of 350 μg of protein extract was loaded onto the IPG strips (GE-Healthcare, Little Chalfont, United Kingdom) in a pH range of 4 to 7. The resulting 2D gels were fixed as described above and stained with colloidal Coomassie Brillant Blue (8).
Protein identification by MS.
For identification of proteins by matrix-assisted laser desorption ionization (MALDI)-time of flight (TOF) MS, Coomassie-stained protein spots were cut from gels using a spot cutter (Proteome Work) with a picker head of 2 mm and transferred into 96-well microtiter plates. Digestion with trypsin and subsequent spotting of peptide solutions onto the MALDI targets were performed automatically in an Ettan Spot Handling Workstation (GE-Healthcare, Little Chalfont, United Kingdom) using a modified standard protocol (20). MALDI-TOF MS analyses of spotted peptide solutions were carried out on a Proteome-Analyzer 4700 (Applied Biosystems, Foster City, CA). The spectra were recorded in a reflector mode in a mass range from 900 to 3,700 Da. Automatic or manual calibration was performed as described previously (20). After calibration the peak lists were created using the “peak to mascot” script of the 4700 Explorer software. The resulting peak lists were analyzed by using the mascot search engine (Matrix Science, London, United Kingdom), GPMAW 4.1 (Lighthouse Data). The annotation of S. aureus COL was used for protein identification and denotation. Peptide mixtures that at least twice yielded a Mowes score of at least 50 and a sequence coverage of at least 30% were regarded as positive identifications. Proteins that failed to exceed the 30% sequence coverage cutoff were subjected to MALDI tandem MS (20). Database searches were performed using the Mascot search engine with the protein databases of the S. aureus COL strain.
Protein quantitation approaches.
The 2D gel image analysis was performed with the software DELTA 2D (Decodon GmbH, Greifswald, Germany). Three different data sets were analyzed in order to screen for differences in the synthesis of cytoplasmic proteins identified on 2D gels under anaerobic growth conditions.
Detection of glucose, lactate, and acetate.
The concentrations of glucose, lactate, and acetate were determined in the supernatant of aerobically and anaerobically grown cells. After sampling, the cells were separated from the supernatant by centrifugation, and the amounts of the respective metabolites were measured by using the test combinations d-glucose, dl-lactate, and acetate (Boehringer Mannheim) according to the manufacturer's instructions. The test combinations are offered for measuring the respective metabolites in complex mixtures such as food. The determinations are based on enzymatic reactions resulting in the formation of NADH and NADPH. The amount of NADH or NADPH formed in these reactions can be measured at 340 nm and is stoichiometric to the amount of glucose and lactate, respectively. For acetate, the amount of the formed NADH was not directly proportional to the acetate concentration because of the equilibrium of a preceding indicator reaction. The calculation was done as recommended by the manufacturer. Immediately after inoculation the amount of glucose added to the medium could be confirmed, and no acetate and lactate was measured.
RNA preparation.
Total RNA from S. aureus was isolated using the acid-phenol method (23, 43) with some modifications. Samples (20 ml) from exponentially growing cultures (OD500 of 0.5) and from anaerobically growing cultures at different times after the shift were treated with 10 ml of ice-cold killing buffer (20 mM Tris-HCl [pH 7.5], 5 mM MgCl2, 20 mM NaN3). The cells were immediately separated from the supernatant by centrifugation (for 5 min at 7,155 × g at 4°C), washed with ice-cold killing buffer, and resuspended in lysis buffer (3 mM EDTA, 200 mM NaCl). For mechanical disruption, the cell suspension was transferred into screw-top tubes containing 500 μl of glass beads (diameter, 0.1 to 0.11 mm; Sartorius, Goettingen, Germany) and 500 μl of water-saturated phenol-chloroform-isoamyl alcohol (25:24:1, vol/vol/vol); the cells were then disrupted by homogenization using a Ribolyser (Thermo Electron Corporation) for 30 s at 6.5 m/s. Afterwards, the resulting RNA solution was extracted once with water-saturated phenol-chloroform-isoamyl alcohol (25:24:1, vol/vol/vol), twice with chloroform-isoamyl alcohol (24:1, vol/vol), and once with water-saturated ether. RNA was precipitated by using 70% ethanol and resuspended in deionized water. The quality of RNA was ensured by gel electrophoresis and by analysis with a Bioanalyzer (Agilent Technologies, Palo Alto, CA).
Northern blot analyses.
Digoxigenin-labeled RNA probes were prepared by in vitro transcription with T7 RNA polymerase by using PCR fragments as templates (23, 33). The PCR fragments were generated by using chromosomal DNA of S. aureus COL isolated with a chromosomal DNA isolation kit (Promega, Madison, WI), according to the manufacturer's recommendations, and the respective oligonucleotides (Table 1). Northern blot analyses were carried out as previously described (73). The digoxigenin-labeled RNA marker I (Roche, Indianapolis, IN) was used to calculate the sizes of the transcripts. The hybridization signals were detected using a Lumi-Imager (Roche Diagnostics, Mannheim, Germany) and analyzed using the software package Lumi-Analyst (Roche Diagnostics, Mannheim, Germany).
TABLE 1.
Oligonucleotides used in this study
| Gene identifiera | Gene namea | Primer
|
|
|---|---|---|---|
| Direction | Sequence (5′ to 3′)b | ||
| SACOL0222 | ldh1 | Forward | CATGCCACACCATATTCTCC |
| Reverse | CTAATACGACTCACTATAGGGAGATCTGCTTCAGCCATAATATC | ||
| SACOL0301 | Forward | GGACGACTGGGTAAATAACG | |
| Reverse | CTAATACGACTCACTATAGGGAGAGGCGAATATGGTAACACCGT | ||
| SACOL0838 | gapA1 | Forward | GTTTCACAGGTGAAGTAGAGG |
| Reverse | CTAATACGACTCACTATAGGGAGAACACGAGTTTGTGTAGCGTC | ||
| SACOL1746 | pfkA | Forward | ACTAGTGGTGGAGATTCACC |
| Reverse | CTAATACGACTCACTATAGGGAGAACCAACTGATAATCCAGCCC | ||
| SACOL2363 | Forward | GTATCGCTTCTGGTGCAGTA | |
| Reverse | CTAATACGACTCACTATAGGGAGACTGCTAATTCAGGTCCACTG | ||
| SACOL2399 | nirR | Forward | TTGTTGCACATGGCATGAGG |
| Reverse | CTAATACGACTCACTATAGGGAGATTCTCGCATATGTCGGCTTG | ||
Based on TIGR annotation (http://www.tigr.org).
The underlined sequences at the 5′ end represent the recognition sequence for the T7 RNA polymerase (33).
Microarray analyses.
The DNA microarrays used in this study (sciTRACER S. aureus N315) were purchased from Scienion (Berlin, Germany) and contain 2,338 PCR products of 200 to 500 bp in length covering about 90% of the S. aureus N315 genes (NCBI NC_002745). PCR products were designed by the manufacturer (Scienion AG, Berlin, Germany) and are optimized for similar thermodynamic hybridization parameters and display no significant cross-hybridization with other open reading frames of the N315 genome. Each PCR product was spotted in duplicate on glass slides.
For DNA microarray analysis, RNA of two independent experiments was prepared. Samples were taken immediately before the shift as well as at 10, 20, 30, and 60 min after the shift to anaerobiosis. The control pool was generated by mixing the same RNA amounts of each sample within one biological replicate. In one experiment the control pool was labeled Cy5, whereas the cDNA synthesized from RNA of shifted cells was labeled Cy3. In the second experiment the Cye dye labeling of the control pool and the samples was switched.
For cDNA synthesis 10 μg of total RNA was mixed with 0.5 μg of random hexamers (GE Healthcare, Little Chalfont, United Kingdom) in a total volume of 15 μl and denatured at 70°C for 2 min. The denatured RNA was then added to the labeling mixture (8 μl of 5× first-strand buffer [Invitrogen, Karlsruhe, Germany], 4 μl of Cy3-dUTP or Cy5-dUTP [GE Healthcare, Little Chalfont, United Kingdom], 4 μl of 0.1 M dithiothreitol [Invitrogen, Karlsruhe, Germany], 1 μl of RNasin [40 U/μl; Promega, Madison, WI] and 4 μl of deoxynucleoside triphosphate mix (dATP, dCTP, and dGTP at a 5 mM concentration and dTTP at 2 mM; GE Healthcare, Little Chalfont, United Kingdom]) and incubated for 25 min at 42°C after the addition of 2 μl of Superscript II (200 U/μl; Invitrogen, Karlsruhe, Germany). This first incubation was followed by a second incubation at 42°C for 35 min in the presence of 2 μl of freshly added Superscript II. The reaction was stopped by the addition of 5 μl of EDTA (0.5 M), and the RNA was hydrolyzed by the addition of 10 μl of NaOH (1 M) and incubation at 65°C for 15 min. The solution was neutralized with 25 μl of 1 M Tris-HCl (pH 7.5). Purification of labeled cDNA was done with a CyScribe Purification Kit (GE Healthcare, Little Chalfont, United Kingdom) according to the manufacturer's instruction. The purified cDNA was then concentrated in a SpeedVac, and the concentrate (ca. 2 μl) was dissolved in 40 μl of hybridization buffer provided by Scienion (Berlin, Germany). Prior to the hybridization, the labeled cDNA hybridization buffer mixture was incubated for 2 min at 65°C. The slides were hybridized overnight (16 h) in a water bath at 42°C. After hybridization and three washing steps (0.03% sodium dodecyl sulfate and 2× SSC [1× SSC is 0.15 M NaCl plus 0.015 M sodium citrate] for 5 min at room temperature; 0.2× SSC for 5 min 42°C; and 0.06× SSC for 1 min at room temperature), the slides were dried by centrifugation. Slides were scanned using a ScanArray scanner (PE Biosystems, Weiterstadt, Germany).
The obtained images were quantified with the ScanArray Express software, version 3.0, (PE Biosystems, Weiterstadt, Germany) using the adaptive threshold as the quantitation method. The measured fluorescence signals were subjected to local background subtraction followed by LOWESS subgrid normalization (ArrayInformatics software; Perkin Elmer). Negative spot intensities were replaced by 1.00E-10.
To analyze the degree of variation in signal intensity within each spot pair, we applied the following criteria. Ideally, the fluorescence values for two spots within a spot pair should be equal. In such a case, the log2 value of the ratios of the two spots (1 and 2, identified by the prefix R or G for red or green channel) of a spot pair should be zero for the red channel [log2 (R1/R2)], the green channel [log2 (G1/G2)], and the composite image (log2 [(R1/G1)/(R2/G2)]). By applying these criteria to each spot pair, a normal curve of distribution was found (see Fig. S1 in the supplemental material). Five percent of the spot pairs had a log2 ratio ≥0.7 or ≤−0.7 and were defined as outliers. The respective spot pairs were excluded from further analyses.
To test whether genes were differentially expressed during anaerobiosis, we used an analysis of variance test (level of significance α of <0.05 and a false discovery rate of 0.1). Only genes for which the main effect of time was significant in both biological replicates were considered differentially expressed.
RESULTS
The influence of oxygen availability on global gene transcription in S. aureus.
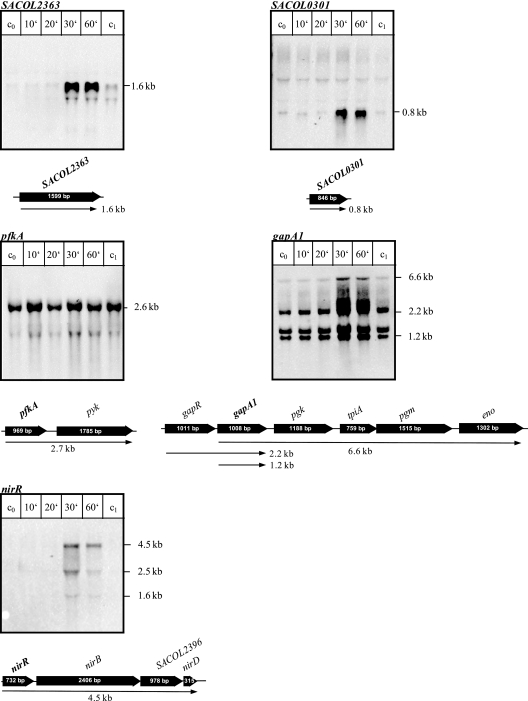
In order to get a global view on the influence of oxygen on gene regulation, transcriptomic studies were carried out by using full-genome DNA microarrays. Although S. aureus is a facultative anaerobic bacterium, the growth rate was drastically reduced after the shift from aerobic to anaerobic growth conditions. To establish appropriate time points for DNA microarray experiments under anaerobic conditions, we monitored the expression of ldh1, known to be induced under anaerobic conditions (22), by Northern blot experiments. Using an ldh1-specific probe, a transcript of 0.95 kb was detected about 20 min after cells were shifted to anaerobic conditions. The transcript was not present in aerobically growing cells (data not shown).
For DNA microarray experiments, total RNA was isolated from aerobically grown cells and from cells 10, 20, 30, and 60 min after the shift to anaerobic conditions. By comparing gene transcription under aerobic and anaerobic conditions, we were able to identify differently expressed genes (see Tables S1 and S2 in the supplemental material). In all experiments, a gene was considered to be regulated by the availability of oxygen if its transcription was induced or repressed at least twofold at one time point. Accordingly, the mRNA levels of 207 genes significantly changed under anaerobic conditions, including 130 genes with an increased transcription rate (see Table S1 in the supplemental material) and 77 genes with a decreased transcription rate (see Table S2 in the supplemental material).
As expected, several genes that are expressed at higher levels under oxygen depletion conditions belong to the functional categories of glycolysis, fermentation, and anaerobic respiration (see Table S1 in the supplemental material). Therefore, the transcription of six genes taking part in glycolysis was upregulated—gapA1, eno, pfkA, fbaA, pgm, and fdaB—indicating enhanced glycolytic activity under anaerobic conditions (Fig. 1A; see Table S1 in the supplemental material). Similarly, eight genes coding for proteins involved in fermentation processes are highly transcribed. As expected, among these genes are ldh1 and ldh2 encoding lactate dehydrogenases. Moreover, the transcription of genes that might be involved in butanediol synthesis was increased under fermentative conditions. These are budB and the SACOL0111 gene. Other genes taking part in acetate and ethanol formation such as pflB and pflA and the SACOL0135 and SACOL0660 genes had high levels of transcription as well (Fig. 1C; see Table S1 in the supplemental material). Similar results were obtained for genes of the arc operon, arcA and arcB2, which are probably involved in arginine catabolism under anaerobic conditions (see Table S1 in the supplemental material).
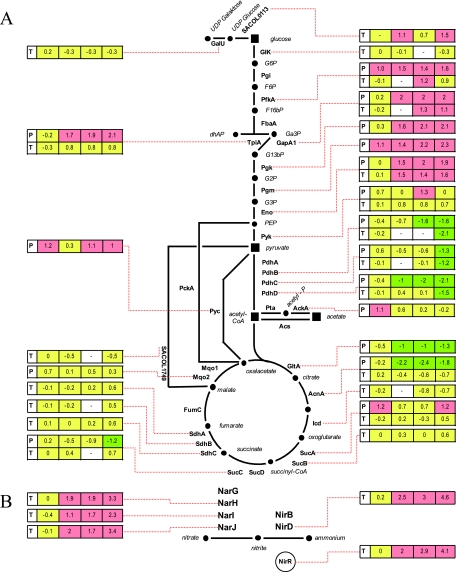
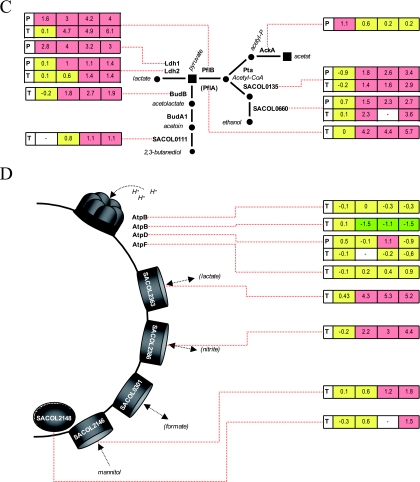
FIG. 1.
Assignment of genes and proteins in S. aureus COL to biochemical pathways and transport processes. (A) Glycolysis and citric acid cycle. (B) Nitrate respiration. (C) Fermentation. (D) Transport processes. Transcriptomic (T) and proteomic (P) data for the respective enzymes are presented (induction ratios as logarithm to the base 2 are shown). Green boxes denote time points when the respective transcript or protein was significantly upregulated, red boxes denote time points when the respective transcript or protein was significantly downregulated under anaerobic conditions, and yellow boxes indicate that there were no significant changes. For some enzymes of the pathways, no results could be shown since either the proteins could not be identified on the gels or no microarray results were available. AckA, acetate kinase; AcnA, aconitate hydratase; Acs, acetyl-CoA synthetase; AtpB, ATP synthase F0, subunit A; AtpD, ATP synthase F0, subunit D; AtpF, ATP synthase F0, subunit F; BudA1, α-acetolactate decarboxylase; BudB, acetolactate synthase; Eno, enolase; Fba, fructose-bisphosphate aldolase; FumC, fumarate hydratase; GalU, UTP-glucose-1-phosphate uridylyltransferase; GapA1, glyceraldehyde-3-phosphate dehydrogenase; GlcK, glucokinase; GltA, citrate synthase; Icd, isocitrate dehydrogenase; Ldh1, lactate dehydrogenase; Ldh2, lactate dehydrogenase; Mqo1, malate:quinone oxidoreductase; Mqo2, malate:quinine oxidoreductase; NarG, respiratory nitrate reductase, alpha subunit; NarH, respiratory nitrate reductase, beta subunit; NarI, respiratory nitrate reductase, gamma subunit; NarJ, respiratory nitrate reductase, delta subunit; NirB, nitrite reductase, subunit; NirD, nitrite reductase, small subunit; NirR, transcriptional regulator; PckA, phosphoenolpyruvate carboxykinase; PdhA, pyruvate dehydrogenase complex E1 component, alpha subunit; PdhB, pyruvate dehydrogenase component E1; PdhC, pyruvate dehydrogenase component E2; PdhD, pyruvate dehydrogenase component E3; PfkA, phosphofructokinase; PflA, pyuvate formate lyase-activating enzyme; PflB, pyruvate formate lyase; Pgi, glucose-6-phosphate isomerase; Pgk, phosphoglycerate kinase; Pgm, phosphoglycerate mutase; Pta, phosphate acetyltransferase; Pyc, pyruvate carboxylase; Pyk, pyruvate kinase; SACOL0111, acetoin reductase; SACOL0113, NAD-dependent epimerase/dehydratase family protein; SACOL0135, alcohol dehydrogenase; SACOL0301, formate/nitrite transporter protein; SACOL0660, alcohol dehydrogenase; SACOL1749, malic enzyme, flavoprotein chain; SACOL2146, manitol specific IIBC component; SACOL2148, manitol specific IIA component; SACOL2363, L-lactate permease; SACOL2386, nitrite extrusion protein; SdhA, succinate dehydrogenase; SdhB, succinate dehydrogenase, iron-sulfur protein; SdhC, succinate dehydrogenase, cytochrome b558 subunit; SucA, 2-oxoglutarate dehydrogenase component E1; SucB, 2-oxoglutarate dehydrogenase component E2; SucC, succinyl-CoA synthetase, beta subunit; SucD, succinyl-CoA synthetase, alpha subunit; TpiA, triosephosphate isomerase.
S. aureus is able to use nitrate or nitrite as alternative electron acceptors (6). Accordingly, transcripts of the nar and nir operon were found to be present at elevated levels under anaerobic conditions (Fig. 1B; see Table S1 in the supplemental material). This is of particular interest since nitrate seems to be unnecessary for anaerobic expression of these genes.
Moreover, among the induced genes are also genes encoding several transport proteins (see Table S1 in the supplemental material). In particular, the transcription of genes encoding a putative formate/nitrite transporter family protein (SACOL0301), a putative l-lactate permease (SACOL2363), and a putative nitrite transport protein (SACOL2386), which might be involved in extrusion of either fermentation end products or nitrite produced by anaerobic respiration (Fig. 1D). Interestingly, among the genes with the highest levels of induction were those that might encode components of a mannitol-specific phosphotransferase system and a permease specific for maltose (see Table S1 in the supplemental material).
During anaerobic growth, the mRNA levels of several regulatory genes were found to be upregulated compared to aerobically grown cells, indicating a role in the regulation of anaerobic gene expression (see Table S1 in the supplemental material). One of the genes with the highest level of induction among this functional group codes for a transcriptional regulator of the MerR family (SACOL2388). Also, the two-component system SrrAB already shown to be involved in oxygen regulation in S. aureus (56, 71, 74) was induced under oxygen-limiting conditions. Additionally, genes known to affect virulence gene expression such as rot and sarZ were induced.
Of particular interest, the transcription of several genes coding for virulence factors was found to be upregulated under oxygen depletion conditions as well. These are the genes pls, hly, splD and splC, epiG, and isaB and the SACOL0470, SACOL2004, and SACOL2006 genes (see Table S1 in the supplemental material). Moreover, the transcription of two genes involved in capsular polysaccharide synthesis was also upregulated (see Table S1 in the supplemental material).
About one-third of the genes in the S. aureus genome have not yet been functionally characterized. Interestingly, in our DNA microarray experiments the amount of mRNA of 32 genes belonging to this group was increased (see Table S1 in the supplemental material). This strongly implies a function of the corresponding gene products in the adaptive process to oxygen-limiting conditions. The specific role of these proteins, however, has to be elucidated by further experiments.
As expected, among the downregulated genes are three genes of the pdhABCD operon encoding the pyruvate dehydrogenase (PDH) complex that converts pyruvate to acetyl-CoA under aerobic conditions (Fig. 1A; see Table S2 in the supplemental material). Similarly, the TCA cycle activity should be repressed under oxygen limitation. Surprisingly, in our approach the transcription of most of these genes was not significantly influenced by lowering the oxygen concentration (Fig. 1A).
Among the genes whose transcription rate was decreased are also the genes that are involved in the translational machinery of S. aureus, indicating lower translational activity under conditions analyzed in the present study (see Table S2 in the supplemental material). This includes genes encoding ribosomal proteins, several tRNA synthetases, and the elongation factor G. Moreover, the transcription of genes whose gene products may function in DNA metabolism and several transport processes was also downregulated (see Table S2 in the supplemental material).
Detailed transcriptional analyses of oxygen-regulated genes.
Anaerobic regulation of transcription of selected genes was subsequently confirmed by Northern blotting experiments to validate the data obtained from DNA microarray analyses. We selected especially those genes whose transcription was induced under anaerobic conditions: the SACOL2363 and SACOL0301 genes encoding a putative lactate permease and a putative formate nitrite transporter, respectively, and the nir operon involved in nitrite respiration. Moreover, we analyzed the transcription of glycolytic genes: the pfkA-pyc operon and the gap operon (gapR, gapA1, pgk, tpi, and eno). These experiments clearly demonstrate that the transcription of all these genes was influenced by the oxygen concentration (Fig. 2). While transcripts specific for the SACOL0203 gene, the gap operon, and the pfkA operon were already detectable in aerobically growing cells, transcripts specific for the SACOL2363 and SACOL0301 genes and the nir operon were present in detectable amounts only after the shift to anaerobic conditions (Fig. 2). This correlates with our data obtained by microarray experiments (see Table S1 in the supplemental material).
FIG. 2.
Northern blot analyses of genes whose transcription was induced under anaerobic conditions. For RNA preparation, cells were grown in synthetic medium without nitrate to an OD500 of 0.5 and shifted to anaerobic conditions. RNA was isolated before (c0) and 10, 20, 30, and 60 min after shift to anaerobic conditions. To ensure that the mRNA levels of the respective genes remained unchanged during the course of the experiment in an aerobically grown culture, RNA was also prepared from a nontreated culture 60 min after the shift (c1). The RNA was separated on a denaturating RNA gel and blotted onto positively charged nylon membranes. The membranes were hybridized with digoxigenin-labeled RNA probes of the respective genes. Relevant transcripts are indicated by arrows. Schematic representations of the gene loci based on the sequence of S. aureus COL are shown.
Shift to anaerobic conditions influences the synthesis of enzymes belonging to the central carbon metabolic and fermentation pathways.
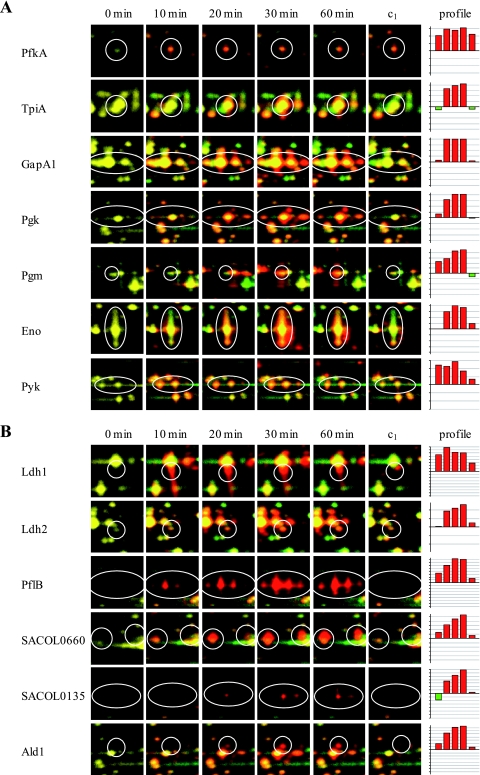
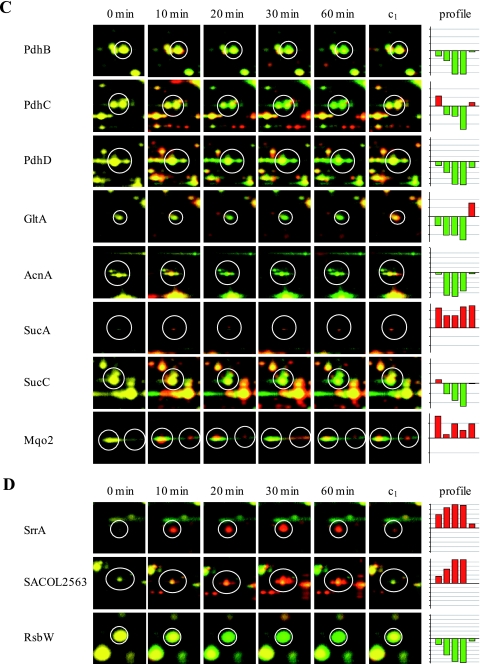
In the present approach, quantification of the synthesis of cytoplasmic proteins before and at different times after the shifting of cells to anaerobic conditions was performed. All proteins whose synthesis was significantly influenced by oxygen availability are shown in Tables S3 and S4 in the supplemental material. As a result, of the 1,073 detected protein spots, the synthesis rate of 357 protein spots was induced, whereas at the same time the synthesis rate of at least 478 protein spots seemed to be repressed. A total of 135 of these protein spots were identified, representing 112 proteins (see Tables S3 and S4 in the supplemental material). The remaining spots represent proteins that did not accumulate in detectable amounts in the cell and therefore are difficult to access for protein identification on 2D gels. As expected, among the proteins with the highest levels of induction are enzymes of the fermentation pathways such as PflB, Adh, and Ldh1 (Fig. 1C and 3B; see Table S3 in the supplemental material). Also, the synthesis of glycolytic enzymes such as GapA, Eno, Pgk, and Pyk was increased whereas at the same time the synthesis of enzymes belonging to the PDH complex and to the TCA cycle (GltA, AcnA, and SucC) was repressed (Fig. 1A and 3A and C; see Tables S3 and S4 in the supplemental material).
FIG. 3.
Synthesis patterns of selected proteins representing different branches of cellular metabolism: glycolysis (A), fermentation (B), PDH and TCA cycle (C), and miscellaneous (D). The protein synthesis pattern under aerobic conditions (0 min; shown in green) was compared with the protein synthesis pattern at different time points after shift to fermentation conditions (10, 20, 30, and 60 min; shown in red) and with that of an aerobically grown culture 60 min after shift (c1). The autoradiograms were normalized by using total normalization. The bar graphs on the right display relative synthesis rates (logarithm to the base 2) of the individual proteins at the different time points.
In a second experiment, the global protein synthesis of cytoplasmic proteins was compared between aerobic and anaerobic conditions in the presence of 8 mM nitrate. This experiment was designed to monitor the influence of an excess amount of nitrate (5, 16) on the anaerobic protein synthesis pattern in S. aureus. In this way, we could show that the synthesis of 235 protein spots was induced under these conditions. Simultaneously, the synthesis rate of at least 518 protein spots seemed to be repressed. From these protein spots 118 could be identified representing 90 proteins (Tables S3 and S4 in the supplemental material). Most of the glycolytic and fermentation enzymes detectable on 2D gels were induced in the presence of nitrate with very similar induction ratios as found under fermentation conditions (Table S3 in the supplemental material). Only glyceraldehyde dehydrogenase and enolase seemed to be induced at slightly higher levels in the medium without nitrate. Enzymes involved in reduction of nitrate and nitrite have not yet been quantified since they were synthesized at very low rates.
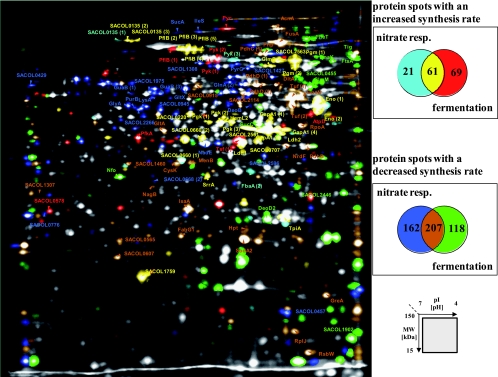
By applying the color-coding software tool provided by the Decodon GmbH (Greifswald, Germany), proteins induced or repressed by more than one stimulus can be visualized. The protein expression profiles of cells 30 min after the shift to fermentative conditions and of cells grown anaerobically in the presence of nitrate (8 mM) were simultaneously compared with the profile of aerobically grown cells (Fig. 4). Accordingly, 61 protein spots were induced both in the presence and in the absence of nitrate, whereas 69 protein spots were specifically induced under fermentative conditions, and 21 protein spots were induced only when nitrate was present. Among the proteins whose synthesis was decreased under anaerobic conditions, we found 118 protein spots whose synthesis was repressed only under fermentative conditions and 162 whose synthesis was specifically impaired in the presence of nitrate. The synthesis of 207 protein spots was repressed under both conditions (Fig. 4).
FIG. 4.
Multicolor imaging of protein synthesis patterns of S. aureus COL under anaerobic conditions in the presence and absence of nitrate. Delta 2D software was used to visualize complex protein expression patterns on the 2D image in the standard pH range of 4 to 7. The color code is shown on the right side. Proteins whose synthesis was induced under anaerobic conditions only in the presence of nitrate appear light blue, proteins whose synthesis was induced only in the absence of nitrate appear red, and proteins induced under both conditions appear yellow. In addition, proteins whose synthesis was repressed in the presence of nitrate are shown in dark blue, proteins repressed in the absence of nitrate are shown in green, and proteins repressed under both conditions are shown in brown.
Analyses of metabolites under aerobic and anaerobic conditions.
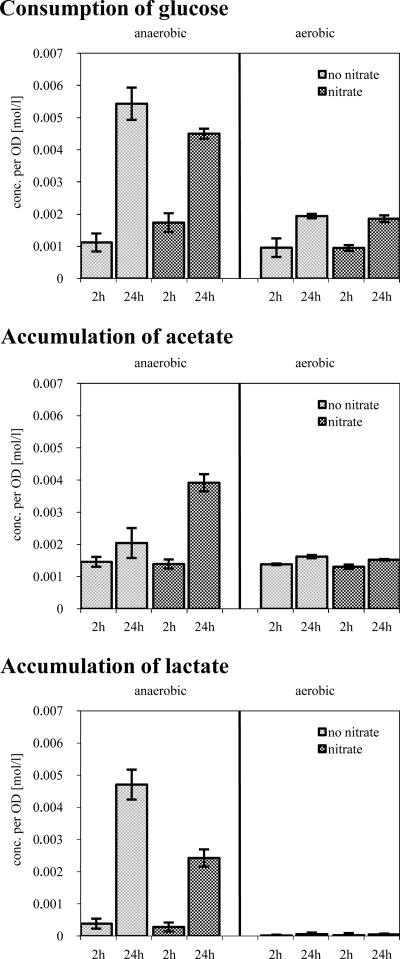
Apparently, under anaerobic conditions both in the presence and in the absence of nitrate, enzymes belonging to the glycolysis and the mixed-acid and butanediol fermentation pathways were induced in S. aureus. For this reason, the amounts of glucose, lactate, and acetate were measured in the supernatant of anaerobically (with or without nitrate) and aerobically growing cells (Fig. 5). As expected, the consumption of glucose strongly increased as soon as the oxygen concentration was diminished. The consumption was higher under fermentative conditions (Fig. 5). Simultaneous with the degradation of glucose, lactate and acetate were produced under anaerobic conditions (Fig. 5). In the absence of external electron acceptors, the reduction of metabolic intermediates by fermentation might be the only way to oxidize NADH. Expectedly, lactate production was higher under fermentative conditions (see also reference 16). Moreover, in the presence of nitrate S. aureus secreted higher amounts of acetate.
FIG. 5.
Detection of selected metabolites under aerobic and anaerobic conditions. Graphs show the consumption of glucose and accumulation of acetate and lactate in moles per OD unit under aerobic and anaerobic conditions at different time points. Cells were grown in synthetic medium in the presence or absence of nitrate to an OD500 of 0.5 and shifted to anaerobic conditions. The amount of the respective metabolites in the supernatant of S. aureus COL under these conditions was measured by using test kits from Boehringer Mannheim, and results were compared to the amount measured immediately before shifting the cells to anaerobic conditions. The amounts of lactate and acetate were set to zero at the shift.
DISCUSSION
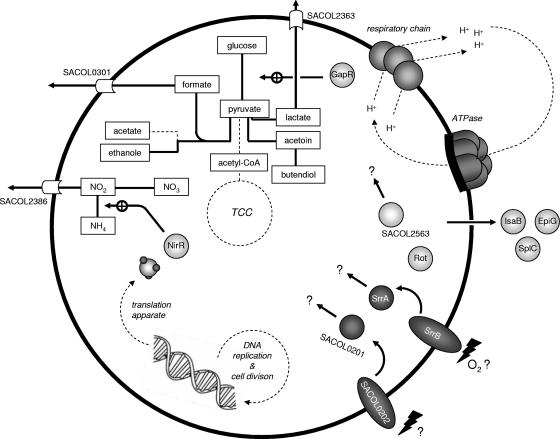
Bacteria sense environmental changes in oxygen concentration and then respond by switching their regulatory mechanisms to ensure that the most energetically favorable process is active under a given environmental condition. It was shown previously that S. aureus grows anaerobically both in the presence of nitrate by nitrate respiration and under fermentative conditions on glucose (6, 19, 68). In the present work, anaerobic gene expression in S. aureus was analyzed by using a transcriptomic as well as a proteomic approach to get deeper insights into the physiology of the cells under these conditions. The obtained results indicate that under fermentative conditions S. aureus may undergo mixed-acid (lactate, formate, and acetate) and butanediol fermentation, as shown in Fig. 1C and 6. Therefore, pyruvate could be reduced either to lactate by the activity of lactate dehydrogenase or metabolized to acetoin and 2,3-butanediol by the activity of acetolactate synthase (BudB), α-acetolactate decarboxylase (BudA1), and acetoin reductase (SACOL0111). Each conversion described above contributes to oxidation of NADH, which is a requisite under fermentation conditions. Finally, pyruvate could be converted to acetyl-CoA or acetyl-phosphate, which is further metabolized to acetate or ethanol. While the former reaction is accompanied by the synthesis of ATP, the latter involves recycling of NADH (Fig. 1C and 6). Under aerobic conditions the conversion of pyruvate to acetyl-CoA is catalyzed by the PDH complex characterized by the production of NADH. As in Escherichia coli (29), the synthesis of the PDH complex is repressed under anaerobic conditions in S. aureus (Fig. 1A 3C; see Tables S1 and S3 in the supplemental material), and residual PDH activity is possibly inhibited by NADH. The S. aureus genome codes for a protein (PflB) highly similar to pyruvate formate lyase (PFL). The expression of the respective gene was highly induced under anaerobic conditions. Accordingly, in S. aureus PDH activity might also be replaced by the activity of PFL which is favorable to cells grown under fermentation conditions, in which NADH generated during glycolysis is not reoxidized by functional respiratory chains. It is presumed that S. aureus generates acetyl-CoA and formate from pyruvate via PFL activity. Analyses of metabolites showed that significant amounts of lactate were secreted into the growth medium, indicating that S. aureus recycles NADH by reducing pyruvate. The production of ethanol, formate, and 2,3-butanediol was not clearly evident so far.
FIG. 6.
Physiological aspects of S. aureus grown under anaerobic conditions. Pathways and processes found to play a role under anaerobic conditions are shown. TCC, tricarbonic acid cycle.
Among the upregulated genes are also genes encoding proteins that might actively secrete lactate (SACOL2363) and formate (SACOL0301) into the medium. The SACOL2363 gene product shows strong similarities to LctP in B. subtilis, which is induced under anaerobic conditions as well, and codes for a lactate H+ symporter (15). Interestingly, in B. subtilis the lctP gene forms an operon with lctE encoding a lactate dehydrogenase while in S. aureus both genes are transcribed monocistronically. Moreover, the SACOL2386 gene whose gene product shows similarities to the nitrite transport protein NarK in E. coli (17, 61) was also found to be induced under anaerobic conditions.
In E. coli and other organisms, TCA cycle enzyme synthesis was shown to be affected by oxygen. Under fermentative growth conditions during which NADH cannot be reoxidized by the respiratory chain, TCA cycle activity is reduced to a minimal level needed for biosynthetic pathways. Synthesis of α-ketoglutarate dehydrogenase is repressed, and succinate dehydrogenase is replaced by fumarate reductase. Other TCA cycle enzymes such as aconitase and citrate synthase are also repressed (26, 27, 36, 50, 55, 66). In the present study only the synthesis of GltA, AcnA, and SucC was observed to be slightly repressed in S. aureus under anaerobic conditions. The expression of Icd, SucB, SdhA-C, and Mqo2 seems to be unaffected under these conditions. However, even in the presence of oxygen the expression of these enzymes was very low. This might be explained by the fact that almost all amino acids and glucose are available in the synthetic medium, and the TCA cycle is required only to a limited degree for anabolic purposes.
For both S. aureus and B. subtilis a growth-stimulating effect of pyruvate under fermentative conditions was observed (19, 46, 58). In B. subtilis a mixture of 20 amino acids can substitute for pyruvate in a medium supporting fermentative growth. In the present approach bacteria were grown under fermentative conditions in the presence of glucose and a mixture of 16 amino acids. As in B. subtilis (46), addition of pyruvate did not affect growth (data not shown). To date, the stimulating effect of pyruvate on both B. subtilis and S. aureus is not always clear. However, in S. aureus high concentrations of pyruvate could be crucial for the activity of PFL, which is important for the allocation of acetyl-CoA under these conditions both for anabolic and energetic processes. In the case of amino acids, the effect could be provoked in particular by those amino acids that could be metabolized to pyruvate, e.g., alanine, glycine, and serine. One of the genes with the highest level of induction under anaerobic conditions codes for an alanine dehydrogenase (Ald1) which is able to catalyze the formation of pyruvate from alanine. Very similar results were also obtained for other bacteria such as mycobacteria (59, 64, 67). However, the degradation of alanine under anaerobic conditions has to be determined by measuring the amount of alanine.
The ability of S. aureus to adapt to extreme changes in external oxygen concentrations implies the existence of one or more systems that regulate anaerobic gene expression. To date, the mechanism of anaerobic gene expression in S. aureus has not been fully characterized. Two regulatory systems were shown to be involved in anaerobic gene expression in S. aureus: the two-component systems SrrAB (SrhSR) and NreBC (35, 71, 74). The NreBC system measures oxygen concentration directly by an iron-sulfur cluster of the Fnr-type which is unstable in the presence of oxygen and which was shown to control dissimilatory nitrate/nitrite reduction systems (35). Initial studies to characterize the SrrAB regulon using a proteomic approach revealed that the two-component system positively regulates lactate dehydrogenase and alcohol dehydrogenase and seems to be a negative regulator of some TCA cycle enzymes (71). The signal that activates the SrrAB system under anaerobic conditions is so far unclear. In previous studies (38, 62) it has been shown that the expression of fermentation enzymes such as lactate dehydrogenase, PFL, and alcohol dehydrogenase was also highly induced under aerobic conditions when the electron transport chain was interrupted. These results clearly indicated that the oxygen concentration per se might not be crucial for the regulation of genes involved in fermentation processes. Therefore, it could be speculated that the reduced state of component(s) of the respiratory chain, the membrane potential, and/or the increased level of NADH might be a signal for anaerobic gene regulation in S. aureus. In order to get some new insights into the regulatory mechanism of anaerobic gene expression, we analyzed the upstream regions of genes regulated by oxygen concentration. Surprisingly, we found an inverted repeat in front of some of them, and the consensus sequence shows strong similarities to Fnr binding sites in B. subtilis and to possible Rex binding sites in B. subtilis and Streptomyces (see Table S5 in the supplemental material) (4, 28, 57).
Remarkably, among the genes whose transcription was induced under oxygen-restricting conditions, we found three genes that might encode regulatory proteins of yet unknown function: SACOL0201, SACOL2360, and SACOL2658. The SACOL0201 gene comprises an operon with two additional genes, SACOL0202 and SACOL0203. The latter genes encode a sensor kinase or a response regulator, respectively, typical of two-component systems, whereas the gene product of the first gene shows high similarities to the periplasmic iron binding proteins of an ABC transporter present, for instance, in Brachyspira hyodysenteriae (18). The operon is highly conserved in S. aureus and S. epidermidis and not found in other staphylococci such as Staphylococcus saprophyticus and Staphylococcus haemolyticus, whose genome sequences are available (40, 70) (www.tigr.org).
In the presence of nitrate or nitrite, ATP could be generated by oxidative phosphorylation under anaerobic conditions. In terms of energetics, nitrate respiration is a more favorable pathway for NADH recycling than fermentation. In E. coli, nitrate represses the synthesis of enzymes belonging to less energetic anaerobic respiratory and fermentation pathways by the two-component systems NarXL and/or NarQP, whereas genes involved in nitrate respiration are activated only in the presence of nitrate (12, 32, 34, 41, 52). In S. aureus, the influence of nitrate on anaerobic gene regulation seems to be different from that in E. coli. The transcription of genes related to nitrate respiration were found to be induced also in the absence of nitrate. These results correlate with previous studies demonstrating the expression of the nitrate/nitrite reduction system also in the absence of nitrate (21). Moreover, the data presented here show that the synthesis of fermentation enzymes was induced under anaerobic respiration conditions in S. aureus at a rate similar to that observed under fermentative conditions, which is in contrast to findings in E. coli and B. subtilis (15, 44, 76). Accordingly, there seems to be no regulating mechanism at the gene expression level in S. aureus ensuring that nitrate is preferred to endogenously generated electron acceptors. However, analysis of fermentation end products indicates that lactate is mainly produced in cells grown under fermentative conditions, whereas in the presence of nitrate acetate was synthesized in higher amounts. Acetate production is the energetically more efficient fermentative pathway. Usually, 1 mol of ATP per mol of acetate could be produced. Higher amounts of lactate under fermentative conditions indicate the importance of NADH reoxidation by lactate dehydrogenase. Considering the fact that the respective enzymes are synthesized in equal amounts both in the absence and presence of nitrate, lactate dehydrogenase should be more active under fermentative conditions, possibly due to an increased NADH/NAD ratio. At the same time the formation of acetate might be repressed.
Among the anaerobically induced proteins are also proteins belonging to the Clp machinery: the proteolytic component ClpP and the chaperone ClpL (Fig. 3D; see Table S1 in the supplemental material). ClpL belongs to the Clp ATPases and was shown to be regulated by the alternative sigma factor σB (24, 53). Interestingly, the transcription of the sigB operon and of other sigB-dependent genes was repressed under these conditions (Fig. 3D; see Table S1 in the supplemental material). Nevertheless, in a SigB-deficient background, ClpL induction under anaerobic conditions was almost completely abolished (unpublished data). The mechanism by which the transcription of clpL is induced under anaerobic conditions and the function of the protein are currently under investigation. For ClpP the role of the proteolytic component in anaerobic gene regulation was, as already discussed, possibly mediated by controlling the activity of one or more regulatory protein(s) under anaerobic conditions (45).
The role of oxygen in virulence gene expression has been discussed for many pathogenic bacteria. Several recent in vitro studies for S. aureus show that oxygen concentration affects the production of agr, tst, and spa (56, 74, 75). Here we demonstrate that the transcription of the genes pls, hly, splC and splD, epiG, and isaB and of the SACOL0470, SACOL2004, and SACOL2006 genes was increased under anaerobic conditions, whereas at the same time isaA transcription seemed to be repressed. The transcription of these genes could be either directly regulated by SrrA or mediated by other regulatory proteins, in particular, those whose transcription was affected by oxygen concentration as well (e.g., rot and sarZ). The level of RNAIII was not increased in our experiments, possibly due to very low optical densities (data not shown). Regarding hly (hla) the observed increase of transcription was in contrast to results already published by Ohlsen et al. (51), who found a drastically reduced activity of the hla promoter under anaerobic conditions in strain Wood 46-3. In addition, the stimulating effect of oxygen on ica gene expression observed in three different S. aureus strains (SA113, 3AI, and Wood 46) by Cramton et al. (14) was not confirmed in the present approach. However, strain- and/or medium-specific effects could not be excluded.
Oxygen concentration seems to fluctuate within the human host (54), and the influence of oxygen availability on physiology and virulence factor production might thus be of some importance for understanding the pathogenicity of S. aureus in different tissues. While in abscesses bacteria live under very anaerobic conditions, the cardiac endothelial tissues might be characterized by high oxygen concentrations. In the present approach we could identify genes of S. aureus whose expression is highly stimulated under low-oxygen concentrations and might therefore be used as indicator genes for anaerobic conditions. By using a gfp reporter gene assay, expression of these genes in different animal models could be monitored. This might help us to understand the physiology of S. aureus under in vivo conditions and could provide some ideas about the environmental signals that could specifically influence virulence factor synthesis in different tissues.
Supplementary Material
Acknowledgments
We are indebted to Richard A. Proctor for lengthy, fruitful discussions, for sharing unpublished results, and for critical reading of the manuscript. Moreover, we thank Volkmar Liebscher, Christin Weinberg, and Dirk Höper for fruitful discussions on DNA microarray data analyses. Birgit Voigt, Haike Henkel, and Dirk Albrecht are acknowledged for support in protein digestion and identification and Susanne Freund for metabolite analyses. We are grateful to Thomas Meier and Anita Harang for excellent technical assistance. Furthermore, we thank Decodon GmbH (Greifswald, Germany) for providing Delta2D software.
This work was supported by grants of the BMBF (031U107A/-207A and 031U213B), the DFG (GK212/3-00 and SFB/TR34), the Land MV, and the Fonds der Chemischen Industrie to M.H. and S.E.
Footnotes
Published ahead of print on 23 March 2007.
Supplemental material for this article may be found at http://jb.asm.org/.
REFERENCES
- 1.Baruah, A., B. Lindsey, Y. Zhu, and M. M. Nakano. 2004. Mutational analysis of the signal-sensing domain of ResE histidine kinase from Bacillus subtilis. J. Bacteriol. 186:1694-1704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bernhardt, J., K. Büttner, C. Scharf, and M. Hecker. 1999. Dual channel imaging of two-dimensional electropherograms in Bacillus subtilis. Electrophoresis 20:2225-2240. [DOI] [PubMed] [Google Scholar]
- 3.Blum, H., H. Beier, and H. J. Gross. 1987. Improved silver staining of plant proteins, RNA and DNA in polyacrylamide gels. Electrophoresis 8:93-99. [Google Scholar]
- 4.Brekasis, D., and M. S. Paget. 2003. A novel sensor of NADH/NAD+ redox poise in Streptomyces coelicolor A3(2). EMBO J. 22:4856-4865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Burke, K. A., A. E. Brown, and J. Lascelles. 1981. Membrane and cytoplasmic nitrate reductase of Staphylococcus aureus and application of crossed immunoelectrophoresis. J. Bacteriol. 148:724-727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Burke, K. A., and J. Lascelles. 1975. Nitrate reductase system in Staphylococcus aureus wild type and mutants. J. Bacteriol. 123:308-316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Büttner, K., J. Bernhardt, C. Scharf, R. Schmid, U. Mäder, C. Eymann, H. Antelmann, A. Völker, U. Völker, and M. Hecker. 2001. A comprehensive two-dimensional map of cytosolic proteins of Bacillus subtilis. Electrophoresis 22:2908-2935. [DOI] [PubMed] [Google Scholar]
- 8.Candiano, G., M. Bruschi, L. Musante, L. Santucci, G. M. Ghiggeri, B. Carnemolla, P. Orecchia, L. Zardi, and P. G. Righetti. 2004. Blue silver: a very sensitive colloidal Coomassie G-250 staining for proteome analysis. Electrophoresis 25:1327-1333. [DOI] [PubMed] [Google Scholar]
- 9.Chan, P. F., and S. J. Foster. 1998. The role of environmental factors in the regulation of virulence-determinant expression in Staphylococcus aureus 8325-4. Microbiology 144:2469-2479. [DOI] [PubMed] [Google Scholar]
- 10.Clements, M. O., S. P. Watson, R. K. Poole, and S. J. Foster. 1999. CtaA of Staphylococcus aureus is required for starvation survival, recovery, and cytochrome biosynthesis. J. Bacteriol. 181:501-507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Coleman, G., I. T. Garbutt, and U. Demnitz. 1983. Ability of a Staphylococcus aureus isolate from a chronic osteomyelitic lesion to survive in the absence of air. Eur. J. Clin. Microbiol. 2:595-597. [DOI] [PubMed] [Google Scholar]
- 12.Constantinidou, C., J. L. Hobman, L. Griffiths, M. D. Patel, C. W. Penn, J. A. Cole, and T. W. Overton. 2006. A reassessment of the FNR regulon and transcriptomic analysis of the effects of nitrate, nitrite, NarXL, and NarQP as Escherichia coli K12 adapts from aerobic to anaerobic growth. J. Biol. Chem. 281:4802-4815. [DOI] [PubMed] [Google Scholar]
- 13.Cramton, S. E., C. Gerke, N. F. Schnell, W. W. Nichols, and F. Götz. 1999. The intercellular adhesion (ica) locus is present in Staphylococcus aureus and is required for biofilm formation. Infect. Immun. 67:5427-5433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cramton, S. E., M. Ulrich, F. Götz, and G. Döring. 2001. Anaerobic conditions induce expression of polysaccharide intercellular adhesin in Staphylococcus aureus and Staphylococcus epidermidis. Infect. Immun. 69:4079-4085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cruz Ramos, H., T. Hoffmann, M. Marino, H. Nedjari, E. Presecan-Siedel, O. Dreesen, P. Glaser, and D. Jahn. 2000. Fermentative metabolism of Bacillus subtilis: physiology and regulation of gene expression. J. Bacteriol. 182:3072-3080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dassy, B., and J. M. Fournier. 1996. Respiratory activity is essential for post-exponential-phase production of type 5 capsular polysaccharide by Staphylococcus aureus. Infect. Immun. 64:2408-2414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.DeMoss, J. A., and P. Y. Hsu. 1991. NarK enhances nitrate uptake and nitrite excretion in Escherichia coli. J. Bacteriol. 173:3303-3310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dugourd, D., C. Martin, C. R. Rioux, M. Jacques, and J. Harel. 1999. Characterization of a periplasmic ATP-binding cassette iron import system of Brachyspira (Serpulina) hyodysenteriae. J. Bacteriol. 181:6948-6957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Evans, J. B. 1975. Uracil and pyruvate requirements of anaerobic growth of staphylococci. J. Clin. Microbiol. 2:14-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Eymann, C., A. Dreisbach, D. Albrecht, J. Bernhardt, D. Becher, S. Gentner, L. T. Tam, K. Büttner, G. Buurman, C. Scharf, S. Venz, U. Völker, and M. Hecker. 2004. A comprehensive proteome map of growing Bacillus subtilis cells. Proteomics 4:2849-2876. [DOI] [PubMed] [Google Scholar]
- 21.Fedtke, I., A. Kamps, B. Krismer, and F. Götz. 2002. The nitrate reductase and nitrite reductase operons and the narT gene of Staphylococcus carnosus are positively controlled by the novel two-component system NreBC. J. Bacteriol. 184:6624-6634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Garrard, W., and J. Lascelles. 1968. Regulation of Staphylococcus aureus lactate dehydrogenase. J. Bacteriol. 95:152-156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gertz, S., S. Engelmann, R. Schmid, K. Ohlsen, J. Hacker, and M. Hecker. 1999. Regulation of sigmaB-dependent transcription of sigB and asp23 in two different Staphylococcus aureus strains. Mol. Gen. Genet. 261:558-566. [DOI] [PubMed] [Google Scholar]
- 24.Gertz, S., S. Engelmann, R. Schmid, A. K. Ziebandt, K. Tischer, C. Scharf, J. Hacker, and M. Hecker. 2000. Characterization of the σB regulon in Staphylococcus aureus. J. Bacteriol. 182:6983-6991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Glaser, P., A. Danchin, F. Kunst, P. Zuber, and M. M. Nakano. 1995. Identification and isolation of a gene required for nitrate assimilation and anaerobic growth of Bacillus subtilis. J. Bacteriol. 177:1112-1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gray, C. T., J. W. Wimpenny, and M. R. Mossman. 1966. Regulation of metabolism in facultative bacteria. II. Effects of aerobiosis, anaerobiosis and nutrition on the formation of Krebs cycle enzymes in Escherichia coli. Biochim. Biophys. Acta 117:33-41. [DOI] [PubMed] [Google Scholar]
- 27.Gunsalus, R. P., and S. J. Park. 1994. Aerobic-anaerobic gene regulation in Escherichia coli: control by the ArcAB and Fnr regulons. Res. Microbiol. 145:437-450. [DOI] [PubMed] [Google Scholar]
- 28.Gyan, S., Y. Shiohira, I. Sato, M. Takeuchi, and T. Sato. 2006. Regulatory loop between redox sensing of the NADH/NAD(+) ratio by Rex (YdiH) and oxidation of NADH by NADH dehydrogenase Ndh in Bacillus subtilis. J. Bacteriol. 188:7062-7071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hansen, H. G., and U. Henning. 1966. Regulation of pyruvate dehydrogenase activity in Escherichia coli K12. Biochim. Biophys. Acta 122:355-358. [DOI] [PubMed] [Google Scholar]
- 30.Hoffmann, T., N. Frankenberg, M. Marino, and D. Jahn. 1998. Ammonification in Bacillus subtilis utilizing dissimilatory nitrite reductase is dependent on resDE. J. Bacteriol. 180:186-189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hoffmann, T., B. Troup, A. Szabo, C. Hungerer, and D. Jahn. 1995. The anaerobic life of Bacillus subtilis: cloning of the genes encoding the respiratory nitrate reductase system. FEMS Microbiol. Lett. 131:219-225. [DOI] [PubMed] [Google Scholar]
- 32.Iuchi, S., and E. C. Lin. 1987. The narL gene product activates the nitrate reductase operon and represses the fumarate reductase and trimethylamine N-oxide reductase operons in Escherichia coli. Proc. Natl. Acad. Sci. USA 84:3901-3905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jorgensen, E. D., R. K. Durbin, S. S. Risman, and W. T. McAllister. 1991. Specific contacts between the bacteriophage T3, T7, and SP6 RNA polymerases and their promoters. J. Biol. Chem. 266:645-651. [PubMed] [Google Scholar]
- 34.Kaiser, M., and G. Sawers. 1995. Nitrate repression of the Escherichia coli pfl operon is mediated by the dual sensors NarQ and NarX and the dual regulators NarL and NarP. J. Bacteriol. 177:3647-3655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kamps, A., S. Achebach, I. Fedtke, G. Unden, and F. Götz. 2004. Staphylococcal NreB: an O2-sensing histidine protein kinase with an O2-labile iron-sulphur cluster of the FNR type. Mol. Microbiol. 52:713-723. [DOI] [PubMed] [Google Scholar]
- 36.Kang, Y., K. D. Weber, Y. Qiu, P. J. Kiley, and F. R. Blattner. 2005. Genome-wide expression analysis indicates that FNR of Escherichia coli K-12 regulates a large number of genes of unknown function. J. Bacteriol. 187:1135-1160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kass, E. H., M. I. Kendrick, Y. C. Tsai, and J. Parsonnet. 1987. Interaction of magnesium ion, oxygen tension, and temperature in the production of toxic-shock-syndrome toxin-1 by Staphylococcus aureus. J. Infect. Dis. 155:812-815. [DOI] [PubMed] [Google Scholar]
- 38.Kohler, C., C. von Eiff, G. Peters, R. A. Proctor, M. Hecker, and S. Engelmann. 2003. Physiological characterization of a heme-deficient mutant of Staphylococcus aureus by a proteomic approach. J. Bacteriol. 185:6928-6937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kohler, C., S. Wolff, D. Albrecht, S. Fuchs, D. Becher, K. Büttner, S. Engelmann, and M. Hecker. 2005. Proteome analyses of Staphylococcus aureus in growing and non-growing cells: a physiological approach. Int. J. Med. Microbiol. 295:547-565. [DOI] [PubMed] [Google Scholar]
- 40.Kuroda, M., A. Yamashita, H. Hirakawa, M. Kumano, K. Morikawa, M. Higashide, A. Maruyama, Y. Inose, K. Matoba, H. Toh, S. Kuhara, M. Hattori, and T. Ohta. 2005. Whole genome sequence of Staphylococcus saprophyticus reveals the pathogenesis of uncomplicated urinary tract infection. Proc. Natl. Acad. Sci. USA 13:500-504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Li, J., S. Kustu, and V. Stewart. 1994. In vitro interaction of nitrate-responsive regulatory protein NarL with DNA target sequences in the fdnG, narG, narK and frdA operon control regions of Escherichia coli K-12. J. Mol. Biol. 241:150-165. [DOI] [PubMed] [Google Scholar]
- 42.Maeda, H., S. Matsu-ura, Y. Yamauchi, and H. Ohmori. 2001. Resazurin as an electron acceptor in glucose oxidase-catalyzed oxidation of glucose. Chem. Pharm. Bull. 49:622-625. [DOI] [PubMed] [Google Scholar]
- 43.Majumdar, D., Y. J. Avissar, and J. H. Wyche. 1991. Simultaneous and rapid isolation of bacterial and eukaryotic DNA and RNA: a new approach for isolating DNA. BioTechniques 11:94-101. [PubMed] [Google Scholar]
- 44.Marino, M., T. Hoffmann, R. Schmid, H. Mobitz, and D. Jahn. 2000. Changes in protein synthesis during the adaptation of Bacillus subtilis to anaerobic growth conditions. Microbiology 146:97-105. [DOI] [PubMed] [Google Scholar]
- 45.Michel, A., F. Agerer, C. R. Hauck, M. Herrmann, J. Ullrich, J. Hacker, and K. Ohlsen. 2006. Global regulatory impact of ClpP protease of Staphylococcus aureus on regulons involved in virulence, oxidative stress response, autolysis, and DNA repair. J. Bacteriol. 188:5783-5796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nakano, M. M., Y. P. Dailly, P. Zuber, and D. P. Clark. 1997. Characterization of anaerobic fermentative growth of Bacillus subtilis: identification of fermentation end products and genes required for growth. J. Bacteriol. 179:6749-6755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Nakano, M. M., and F. M. Hulett. 1997. Adaptation of Bacillus subtilis to oxygen limitation. FEMS Microbiol. Lett. 157:1-7. [DOI] [PubMed] [Google Scholar]
- 48.Nakano, M. M., Y. Zhu, M. Lacelle, X. Zhang, and F. M. Hulett. 2000. Interaction of ResD with regulatory regions of anaerobically induced genes in Bacillus subtilis. Mol. Microbiol. 37:1198-1207. [DOI] [PubMed] [Google Scholar]
- 49.Nakano, M. M., P. Zuber, P. Glaser, A. Danchin, and F. M. Hulett. 1996. Two-component regulatory proteins ResD-ResE are required for transcriptional activation of fnr upon oxygen limitation in Bacillus subtilis. J. Bacteriol. 178:3796-3802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nakano, M. M., P. Zuber, and A. L. Sonenshein. 1998. Anaerobic regulation of Bacillus subtilis Krebs cycle genes. J. Bacteriol. 180:3304-3311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ohlsen, K., K. P. Koller, and J. Hacker. 1997. Analysis of expression of the alpha-toxin gene (hla) of Staphylococcus aureus by using a chromosomally encoded hla::lacZ gene fusion. Infect. Immun. 65:3606-3614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Overton, T. W., L. Griffiths, M. D. Patel, J. L. Hobman, C. W. Penn, J. A. Cole, and C. Constantinidou. 2006. Microarray analysis of gene regulation by oxygen, nitrate, nitrite, FNR, NarL and NarP during anaerobic growth of Escherichia coli: new insights into microbial physiology. Biochem. Soc. Trans. 34:104-107. [DOI] [PubMed] [Google Scholar]
- 53.Pané-Farré, J., B. Jonas, K. Förstner, S. Engelmann, and M. Hecker. 2006. The σB regulon in Staphylococcus aureus and its regulation. Int. J. Med. Microbiol. 296:237-258. [DOI] [PubMed] [Google Scholar]
- 54.Park, M. K., R. A. Myers, and L. Marzella. 1992. Oxygen tensions and infections: modulation of microbial growth, activity of antimicrobial agents, and immunologic responses. Clin. Infect. Dis. 14:720-740. [DOI] [PubMed] [Google Scholar]
- 55.Park, S. J., J. McCabe, J. Turna, and R. P. Gunsalus. 1994. Regulation of the citrate synthase (gltA) gene of Escherichia coli in response to anaerobiosis and carbon supply: role of the arcA gene product. J. Bacteriol. 176:5086-5092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Pragman, A. A., J. M. Yarwood, T. J. Tripp, and P. M. Schlievert. 2004. Characterization of virulence factor regulation by SrrAB, a two-component system in Staphylococcus aureus. J. Bacteriol. 186:2430-2438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Reents, H., R. Munch, T. Dammeyer, D. Jahn, and E. Härtig. 2006. The Fnr regulon of Bacillus subtilis. J. Bacteriol. 188:1103-1112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Richardson, G. M. 1936. The nutrition of Staphylococcus aureus. Necessity of uracil in anaerobic growth. Biochem. J. 30:2184-2190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Rosenkrands, I., R. A. Slayden, J. Crawford, C. Aagaard, C. E. Barry III, and P. Andersen. 2002. Hypoxic response of Mycobacterium tuberculosis studied by metabolic labeling and proteome analysis of cellular and extracellular proteins. J. Bacteriol. 184:3485-3491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ross, R. A., and A. B. Onderdonk. 2000. Production of toxic shock syndrome toxin 1 by Staphylococcus aureus requires both oxygen and carbon dioxide. Infect. Immun. 68:5205-5209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Rowe, J. J., T. Ubbink-Kok, D. Molenaar, W. N. Konings, and A. J. Driessen. 1994. NarK is a nitrite-extrusion system involved in anaerobic nitrate respiration by Escherichia coli. Mol. Microbiol. 12:579-586. [DOI] [PubMed] [Google Scholar]
- 62.Seggewiss, J., K. Becker, O. Kotte, M. Eisenacher, M. R. Yazdi, A. Fischer, P. McNamara, N. Al Laham, R. Proctor, G. Peters, M. Heinemann, and C. von Eiff. 2006. Reporter metabolite analysis of transcriptional profiles of a Staphylococcus aureus strain with normal phenotype and its isogenic hemB mutant displaying the small-colony-variant phenotype. J. Bacteriol. 188:7765-7777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Shafer, W. M., and J. J. Iandolo. 1979. Genetics of staphylococcal enterotoxin B in methicillin-resistant isolates of Staphylococcus aureus. Infect. Immun. 25:902-911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sherman, D. R., M. Voskuil, D. Schnappinger, R. Liao, M. I. Harrell, and G. K. Schoolnik. 2001. Regulation of the Mycobacterium tuberculosis hypoxic response gene encoding α-crystallin. Proc. Natl. Acad. Sci. USA 98:7534-7539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Song, Y., and B. E. Logan. 2004. Effect of O2 exposure on perchlorate reduction by Dechlorosoma sp. KJ. Water Res. 38:1626-1632. [DOI] [PubMed] [Google Scholar]
- 66.Spiro, S., and J. R. Guest. 1991. Adaptive responses to oxygen limitation in Escherichia coli. Trends Biochem. Sci. 16:310-314. [DOI] [PubMed] [Google Scholar]
- 67.Starck, J., G. Kallenius, B. I. Marklund, D. I. Andersson, and T. Akerlund. 2004. Comparative proteome analysis of Mycobacterium tuberculosis grown under aerobic and anaerobic conditions. Microbiology 150:3821-3829. [DOI] [PubMed] [Google Scholar]
- 68.Strasters, K. C., and K. C. Winkler. 1963. Carbohydrate metabolism of Staphylococcus aureus. J. Gen. Microbiol. 33:213-229. [DOI] [PubMed] [Google Scholar]
- 69.Sun, G., E. Sharkova, R. Chesnut, S. Birkey, M. F. Duggan, A. Sorokin, P. Pujic, S. D. Ehrlich, and F. M. Hulett. 1996. Regulators of aerobic and anaerobic respiration in Bacillus subtilis. J. Bacteriol. 178:1374-1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Takeuchi, F., S. Watanabe, T. Baba, H. Yuzawa, T. Ito, Y. Morimoto, M. Kuroda, L. Cui, M. Takahashi, A. Ankai, S. Baba, S. Fukui, J. C. Lee, and K. Hiramatsu. 2005. Whole-genome sequencing of Staphylococcus haemolyticus uncovers the extreme plasticity of its genome and the evolution of human-colonizing staphylococcal species. J. Bacteriol. 187:7292-7308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Throup, J. P., F. Zappacosta, R. D. Lunsford, R. S. Annan, S. A. Carr, J. T. Lonsdale, A. P. Bryant, D. McDevitt, M. Rosenberg, and M. K. Burnham. 2001. The srhSR gene pair from Staphylococcus aureus: genomic and proteomic approaches to the identification and characterization of gene function. Biochemistry 40:10392-10401. [DOI] [PubMed] [Google Scholar]
- 72.Reference deleted.
- 73.Wetzstein, M., U. Völker, J. Dedio, S. Löbau, U. Zuber, M. Schiesswohl, C. Herget, M. Hecker, and W. Schumann. 1992. Cloning, sequencing, and molecular analysis of the dnaK locus from Bacillus subtilis. J. Bacteriol. 174:3300-3310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Yarwood, J. M., J. K. McCormick, and P. M. Schlievert. 2001. Identification of a novel two-component regulatory system that acts in global regulation of virulence factors of Staphylococcus aureus. J. Bacteriol. 183:1113-1123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Yarwood, J. M., and P. M. Schlievert. 2000. Oxygen and carbon dioxide regulation of toxic shock syndrome toxin 1 production by Staphylococcus aureus MN8. J. Clin. Microbiol. 38:1797-1803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ye, R. W., W. Tao, L. Bedzyk, T. Young, M. Chen, and L. Li. 2000. Global gene expression profiles of Bacillus subtilis grown under anaerobic conditions. J. Bacteriol. 182:4458-4465. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.