Abstract
Background
Cholangiocarcinoma (CCA) – cancer of the bile ducts – is associated with chronic infection with the liver fluke, Opisthorchis viverrini. Despite being the only eukaryote that is designated as a 'class I carcinogen' by the International Agency for Research on Cancer, little is known about its genome.
Results
Approximately 5,000 randomly selected cDNAs from the adult stage of O. viverrini were characterized and accounted for 1,932 contigs, representing ~14% of the entire transcriptome, and, presently, the largest sequence dataset for any species of liver fluke. Twenty percent of contigs were assigned GO classifications. Abundantly represented protein families included those involved in physiological functions that are essential to parasitism, such as anaerobic respiration, reproduction, detoxification, surface maintenance and feeding. GO assignments were well conserved in relation to other parasitic flukes, however, some categories were over-represented in O. viverrini, such as structural and motor proteins. An assessment of evolutionary relationships showed that O. viverrini was more similar to other parasitic (Clonorchis sinensis and Schistosoma japonicum) than to free-living (Schmidtea mediterranea) flatworms, and 105 sequences had close homologues in both parasitic species but not in S. mediterranea. A total of 164 O. viverrini contigs contained ORFs with signal sequences, many of which were platyhelminth-specific. Examples of convergent evolution between host and parasite secreted/membrane proteins were identified as were homologues of vaccine antigens from other helminths. Finally, ORFs representing secreted proteins with known roles in tumorigenesis were identified, and these might play roles in the pathogenesis of O. viverrini-induced CCA.
Conclusion
This gene discovery effort for O. viverrini should expedite molecular studies of cholangiocarcinogenesis and accelerate research focused on developing new interventions, drugs and vaccines, to control O. viverrini and related flukes.
Background
Throughout East Asia, there is a strikingly high prevalence of cholangiocarcinoma (CCA – cancer of the bile ducts) in regions where the human liver fluke is endemic. No stronger link occurs between a human malignancy and infection with a eukaryotic parasite than that between CCA and infection with the liver fluke, Opisthorchis viverrini (Digenea) [1]. Indeed, the International Agency for Research on Cancer (IARC) recognizes O. viverrini as a 'category I carcinogen' [2,3]. CCA is highly prevalent in Northeast Thailand, areas where uncooked cyprinoid fish are a dietary staple. Due to poor sanitation practices and inadequate sewerage infrastructure, O. viverrini-infected people pass the trematode's eggs in their feces into natural bodies of fresh water. Aquatic snails, which represent the first intermediate hosts of O. viverrini, ingest the eggs from which the miracidia undergo asexual reproduction before a population of the free swimming larval stage, called a cercaria, is shed from the infected snails. The cercaria then locates a cyprinoid fish, encysts in the fins, skin and musculature of the fish, and becomes a metacercaria. The metacercarial stage is infective to humans and other fish-eating mammals. Infection is acquired when people ingest raw or undercooked fish. The young adult worm escapes from the metacercarial cyst in the upper small intestine and then migrates through the ampulla of Vater into the biliary tree, where it develops to sexual maturity over four to six weeks, thus completing the life cycle. The adult worms, which are hermaphrodites, can live for many years in the liver, even decades, shedding as many as 200 eggs per day which pass out via bile into the chyme and feces [4].
In Thailand, ~6 million people are infected with O. viverrini. Despite widespread chemotherapy with the compound, praziquantel, the prevalence of O. viverrini in some endemic areasapproaches 70% (reviewed in [1]). Moreover, in Thailand, liver cancer is the most prevalent of the malignant/fatal neoplasias, and the prevalence of CCA in regions in which the parasite is endemic is unprecedented [5].
While sexual reproduction takes place in the mature adults of O. viverrini within the bile ducts, asexual reproduction in the snail leads to a massive increase in the number of infectious cercarial stages exiting and swimming off to locate then infect the fish host. The adult fluke is a diploid organism which reproduces by meiosis; self fertilization of the male and female organs occurs, but it is believed that cross-fertilization between adjacent adult worms is the normal pattern. Although the genome size of O. viverrini has not yet been reported, it is known to have six pairs of chromosomes, i.e. 2n = 12 [6], distinct from the closely related liver fluke, Clonorchis sinensis, which possesses 2n = 56 chromosomes [7].
Despite its public health importance, only a small number of O. viverrini sequences (mostly ribosomal genes) have been available in public databases prior to the present study. Characterization of the genes expressed in this organism should provide a foundation for elucidating the immunopathogenesis of CCA, particularly the molecular mechanisms by which infection with this parasite induces cancer. Indeed, secreted proteins of O. viverrini induce hyper-proliferation of cells (or hyperplasia) in vitro [8], implying that carcinogenesis may not be just a consequence of chronic inflammation, but that the parasite actively secretes gene products which initiate neoplasia.
Here, we undertake gene discovery for O. viverrini after the construction of a cDNA library and characterization of ~5,000 expressed sequence tags (ESTs) from this carcinogenic parasite. A similar dataset exists for C. sinensis [9], which, despite its widespread prevalence [10], is not recognized as a 'class I carcinogen' [3]. Therefore, we compared the available transcriptomic dataset from O. viverrini with those from C. sinensis, and from several other flatworms, both free-living and parasitic in humans.
Results and Discussion
Features of the dataset
Of 5,159 randomly selected ESTs, a total of 4,241 yielded acceptably high quality sequences. These in turn were clustered into contigs, establishing a catalogue of 1,932 non-redundant OvAEs. This apparently represents the largest dataset thus far for any of the liver flukes. Table 1 summarizes the key features of the dataset. Of note is that the identities for 1,070 (55%) of these OvAEs could not yet be established, as they did not share sequence homology (BLASTx/tBLASTx) with any other predicted or known molecules in public databases, including dbEST which contains 2,678 ESTs from the related liver fluke, C. sinensis [9]. The average insert size of these novel OvAEs was 550 nt; 47 of these 1,070 OvAEs had insert sizes of less than 150 nt. These ESTs may in fact be O. viverrini-specific or even digenetic fluke-specific genes. A similar situation currently pertains to the human blood fluke where a large percentage of known transcripts, and indeed proteins, are assumed to be Schistosoma- or indeed phylum Platyhelminthes-specific [11,12]. If the O. viverrini genome has 14,000 protein-coding genes (like the blood fluke S. mansoni) [13], and if each of the 1,932 O. viverrini contigs represented a protein coding gene, these newly discovered genes from the adult stage of O. viverrini are predicted to represent ~14% of the entire transcriptome of this liver fluke. EST sequences described herein have been deposited in dbEST under accession numbers EL618683–EL620614.
Table 1.
Features of the Opisthorchis viverrini EST catalogue.
| Feature | Number |
| Initial Sequences | 5159 |
| Usable sequencesa | 4241 |
| Contigs | 1995 (1632 singletons; 363 clusters) |
| Contigs after clean-upb | 1932 |
| Contigs identical to known proteinsc | 68 |
| Contigs similar to other proteinsd | 794 |
| Contigs with gene ontology assignments | 383 |
| Novel contigs | 1070 |
| Novel contigs with signal sequences | 75 (29 signal peptides; 46 signal anchors) |
| Average insert size | 548 bp (ESTs); 660 bp (contigs) |
| Percentage of recombinant clones | 95% |
| Number of ribosomal seqs | 1184 ESTs; 136 clusters |
aUsable sequences were determined using seqclean – sequences that were removed were either non-recombinant, of low complexity and/or quality and those of < 100 nt in length.
bclean-up refers to removal of sequences from contaminating sources; eg. Mycoplasma
cidentity determined by ≥ 95% identity over ≥ 50 amino acids.
dbased on BLASTx and tBLASTx searchers of GenBank nr and dbEST respectively.
Abundantly expressed transcripts
After manual filtering of 136 ribosomal sequences, the 10 most abundantly represented mRNAs encoded proteins with known or unknown functions, including one contig that did not have homologues in any public databases (Table 2). Abundant contigs encoded proteins involved in a range of physiological functions which are considered essential to parasitism, such as anaerobic respiration (myoglobin) [14], reproduction (vitelline precursors and egg shell proteins) and detoxification of xenobiotic compounds (glutathione-S-transferase). Other abundantly expressed OvAEs encoded proteins of likely key relevance to the host-parasite relationship, and included proteases (papain-like and legumain-like enzymes), saposin-like proteins and dynein light chains. Homologues of some of the most abundantly represented OvAEs were also highly represented in C. sinensis ESTs (cysteine proteases, myoglobin, vitelline B precursor), whereas others were uniquely over-expressed in each species. In particular, structural molecules, including tubulin and actin-binding proteins, were among the 10 most abundant clones from C. sinensis [9], but were not highly represented in the dataset from O. viverrini. An in-depth comparison of the O. viverrini and C. sinensis datasets is presented below.
Table 2.
The 10 most abundant contigsa from the Opisthorchis viverrini EST dataset.
| Contig | ESTs/contig | Closest homologue in GenBank nr (accession no.) | %identities (no. of aa) | Score (Bits) | Closest homologue in dbEST (accession no.) | %identities (no. of aa) | Score (Bits) |
| OvAE1587 | 100 | vitelline B precursor, O. viverrini (AAL23712) | 99% (230) | 493 | C. sinensis cDNA clone CSAD-01-D02 (AT007557) | 92% (225) | 524 |
| OvAE1588 | 77 | 17 kDa myoglobin, Clonorchis sinensis (AAM18464) | 81% (149) | 244 | C. sinensis cDNA clone CSAD-29-A12 (AT009373) | 77% (188) | 344 |
| OvAE1585 | 77 | hypothetical protein, C. sinensis (AAM55183) | 84% (90) | 156 | C. sinensis cDNA clone CS30 (AT006763) | 84% (100) | 199 |
| OvAE1593 | 41 | egg protein, C. sinensis (AAN64160) | 89% (253) | 389 | C. sinensis cDNA clone CSAD-20-B05 (AT008604) | 82% (237) | 477 |
| OvAE1584 | 37 | hypothetical protein, Macaca fascicularis (BAE73006) | 67% (59) | 82 | SJA_AAF_D11.T3 SJA S. japonicum (CX857852) | 85% (94) | 183 |
| OvAE1602 | 21 | histone H1, Schistosoma japonicum (AAP06509) | 74% (70) | 112 | C.sinensis cDNA clone CSAD-25-H03 (AT009091) | 85% (177) | 302 |
| OvAE1607 | 17 | egg protein, C. sinensis (AAN64160) | 59% (252) | 288 | C. sinensis cDNA clone CSAD-01-B01 (AT007532) | 82% (252) | 493 |
| OvAE1595 | 16 | retrotransposon gag region, Monascus pilosus (ABC24965) | 31% (57)b | 33 | NA | NA | NA |
| OvAE1608 | 15 | translationally controlled tumor protein, C. sinensis (AAX84199) | 98% (169) | 306 | C. sinensis cDNA clone CSAD-24-E07(AT008979) | 92% (122) | 228 |
| OvAE1601 | 11 | glutathione-S-transferase, O. viverrini (AAL23713) | 98%(213) | 429 | C. sinensis cDNA clone CSAD-32-E05 (AT009695) | 86% (232) | 484 |
aNon-ribosomal sequences only were used in this analysis.
bSequence identity was low but diagnostic motifs of gag were detected over 57 amino acids.
Gene ontology assignments of ESTs from O. viverrini and related flukes
Three hundred and eighty three (383) of the total 1,932 OvAEs (19.8%) could be assigned GO classifications (Figure 1). The most abundant groups represented under the molecular function category were linked to binding (34.8%), catalytic activity (27.9%) and structural molecule activity (13.9%). Other sequences of interest identified in this category were inferred to relate to caspase activity (0.2%) and transporter activity (5.3%). The most abundant groups represented under the biological process category corresponded to physiological processes (41.2%), cellular processes (39.7%) and unknown biological processes (15.6%).
Figure 1.

Summary of predicted gene product function and location using gene ontology terms. Gene ontology (GO) terms for annotated Opisthorchis viverrini assembled ESTs were extracted, if present, from the GO database and sorted into the immediate subcategories for molecular function, cellular component and biological process. The GO subcategory and percentage relative to the total number of extracted terms is indicated in the legend. Although cellular and physiological processes, structural proteins and catalytic activity were strongly represented other categories of interest include the caspases and transporter activity that may represent proteins important for a parasitic lifestyle. The large number of unknowns in each of the three categories highlights the lack of knowledge regarding many of the proteins found in these parasites.
We then undertook a comparative assessment of GO assignments of sequences from O. viverrini and two other trematode parasites of humans in Asia – the liver fluke, C. sinensis (2,679 contigs) and the blood fluke, S. japonicum (107,427 contigs). In general, the percentages of ESTs allocated to each GO category among these three flukes was similar (Figure 2). However, some categories were over- or under-represented in one species. For example, contigs encoding structural proteins were ~four times more abundantly represented in the two liver flukes than in S. japonicum, whereas contigs encoding motor proteins were ~three times more abundant in O. viverrini than they were in C. sinensis or S. japonicum. Sample sizes were too small to determine whether these differences were statistically significant. Both structural and motor proteins are important components of fluke teguments [15], playing roles in surface maintenance and turnover in schistosomes [16,17] and liver flukes [18]. Therefore, these molecular differences might reflect the specialised niches and physiological requirements of each parasite. From just 1932 OvAEs, 15 different contigs had ORFs encoding components of the dynein complex of motor proteins, a category of motion- related, and surface and gut-localized EF-hand motif- containing antigens which, at least in schistosomes, represent potent allergens and targets of protective immunological responses [17,19].
Figure 2.
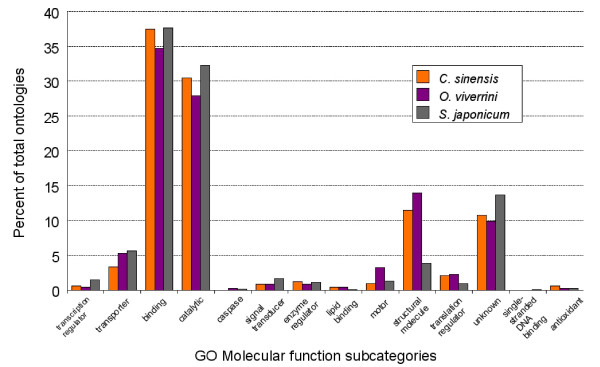
Comparison of the gene ontology molecular function terms for expressed sequence tags from Opisthorchis viverrini, Clonorchis sinensis and Schistosoma japonicum. Expressed sequence tags from C. sinensis and S. japonicum were downloaded from NCBI and subjected to the same analyses used for O. viverrini sequences. A comparison of the percentage of terms correlating to the molecular function subcategory for each organism shows a broad similarity, although in some cases, such as categories for structural or motor proteins, categories are over- or under-represented in certain species.
Evolutionary relationships between O. viverrini and other platyhelminths
To assess the evolutionary relationships between O. viverrini and other platyhelminths (both parasitic and free-living), we used SimiTri [20] to plot the relative similarities of predicted polypeptide sequences (Figure 3). OvAEs shared most sequence identity with sequences from C. sinensis (2,679 publicly available ESTs), and S. japonicum (107, 427 ESTs). OvAEs were less similar to sequences from the free-living turbellarian platyhelminth, Schmidtea mediterranea (171,472 ESTs) [21], which was not altogether surprising given the phylogenetic distance between parasitic and free-living members of the phylum Platyhelminthes [22,23]. The bulk of this phylum (including those species analysed here) represents a monophyletic group based on 18S rDNA sequences [22] and morphological characters [24], and is often referred to as the Rhabditophora [22]. Therefore, the members of the Rhabditophora are considered to be more closely related to each other than to other turbellarian clades, such as the Polycladida [22]. A total of 105 OvAEs had homologues in the ESTs from the two parasitic flukes but not in the free-living Schmidtea ([see additional file 1]; selected examples are shown in the table in Figure 3), suggesting that at least some of these are parasitism-specific genes. Thirty-three (33) of the conserved parasitic fluke genes were novel and did not have homologues of known function. Predicted proteins of known function included homologues of legumain, fatty acid binding proteins, myoglobin and potential anti-inflammatory proteins such as Ly6/UPAR domain-containing proteins. Of the parasitic fluke-specific genes, 38 encoded ORFs with N-terminal signal sequences; 14 of these OvAEs had homologues in just S. japonicum and 24 had homologues in both S. japonicum and C. sinensis.
Figure 3.
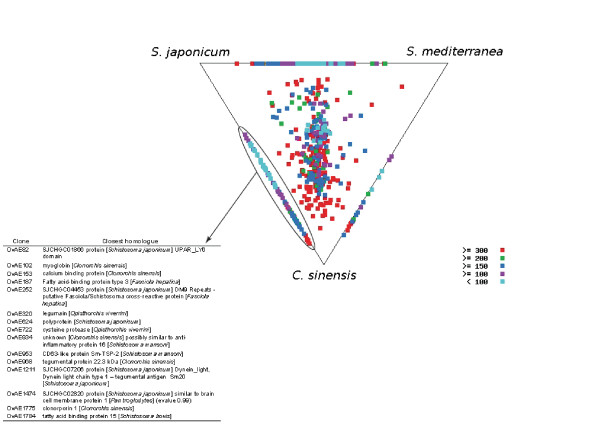
Evolutionary relationships between Opisthorchis viverrini and related platyhelminths based on similarities of protein coding genes using SimiTri. Similarity of O. viverrini ORFs (1,932 ESTs) to those from the liver fluke Clonorchis sinensis (2,679 ESTs), the blood fluke Schistosoma japonicum (107, 427 ESTs) and the free-living turbellarian Schmidtea mediterranea (171,472 ESTs). SimiTri [20] was used to plot 1,932 O. viverrini contigs against related species database entries (A). Each spot represents a unique contig and its sequence similarity to each of the three selected databases as determined by tBLASTx scores. Sequences showing similarity to only one database are not shown. Sequences showing sequence similarity to only two databases appear on the lines joining the two databases. Spots are coloured by their highest tBLASTx score to each of the databases. O. viverrini sequences with homologues in the parasitic flukes only (not in Schmidtea) are highlighted in the dotted region and the identities of selected examples are shown in the table (B). The entire list (105) of these putative parasite-specific proteins is shown in Table S1.
Secreted and membrane proteins
We conducted an analysis of ORFs containing an N-terminal signal peptide or signal anchor. A total of 164 OvAEs contained ORFs with signal sequences. The dataset was divided into three categories – sequences that were (i) novel; (ii) platyhelminth-specific; (iii) conserved across multiple phyla. Novel sequences constituted 55.4% of the total, but only 5.2% of them encoded proteins with a signal sequence. Conserved sequences constituted 36.3% of the total, and 10.7% of these encoded proteins with signal sequences. Finally, the sequences inferred to be platyhelminth-specific accounted for 8.3% of the total dataset, but 20.6% of these encoded proteins with a signal sequence (Figure 4). It should be noted, however, that not all of the OvAEs contained full-length nt sequences, and therefore the true percentage of sequences with signal peptides cannot be definitively inferred in the absence of full genome coverage.
Figure 4.
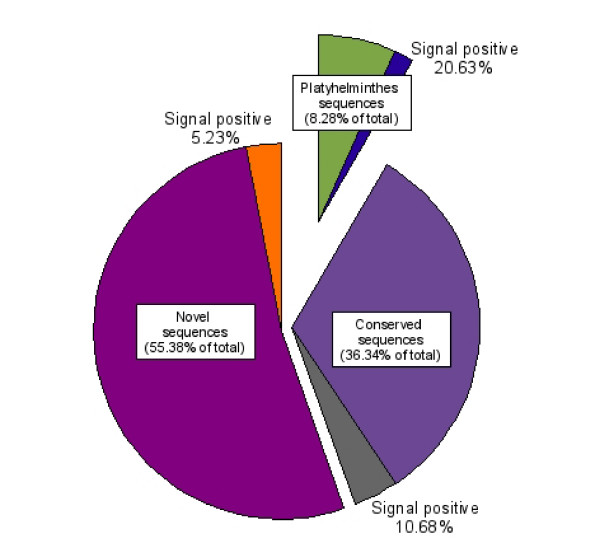
Distribution of Opisthorchis viverrini assembled ESTs (OvAEs) that contain predicted signal peptides or signal anchors. OvAEs that had BLAST hits greater than 1 × 10-5 were sorted into conserved (those matching entries for species other than platyhelminths), phylum Platyhelminthes-specific (only matching platyhelminth entries) and novel (no significant homology to any database entry). The sequences in each category were then analysed for the presence of a signal sequence using SignalP. The relative percentages of each category are indicated along with the sub-category of signal sequence positive contigs.
Sequences encoding novel secreted and/or membrane proteins (without orthologues/paralogues in other organisms or phyla) may be of particular interest for the development of vaccines and drugs, because the absence of host homologues enhances the prospect for therapeutic margins of safety. OvAEs encoding secreted/membrane proteins involved in many aspects of parasitism were identified (Table 3), and some of these are discussed in the following section. Two of the OvAEs presented in Table 3, which are inferred to encode transforming growth factor β receptor (see section "Host-parasite cross-talk) and calumenin, were more similar in sequence to vertebrate proteins from both the non-redundant and dbEST databases than they were to platyhelminth sequences, suggesting that they have evolved independently to bind host ligands. These results are reminiscent of reports of the schistosome transcriptome where, for example, receptors for mammalian hormones, including insulin, fibroblast growth factor and cytokines, have been hypothesized to bind host molecules (reviewed in [25]).
Table 3.
Selected Opsithorchis viverrini contigs that encode families of secreted/membrane proteins that potentially interact with or are exposed to host tissues. Genera of the closest homologues from BLAST × (nr) searches are shown. Where the closest homologue was from a vertebrate (bold font), a tBLASTx search against dbEST was conducted.
| Predicted function | Examples/comments and genera of closest orthologues/paralogues | Contigs | %identities |
| TGF-β receptor | bone morphogenic protein receptor type I (Sus nr/Macaca est) | OvAE22 | 44 |
| Seven transmembrane receptor | DC-STAMP (Strongylocentrotus); laminin receptor (Bos nr/Clonorchis est) | OvAE92, OvAE1722 | 51 |
| Tetraspanin | stabilize cell membranes – expressed in the tegument of schistosomes (Schistosoma) | OvAE953 | 34 |
| C1 family papain-like cysteine protease | cathepsin L (Paragonimus), cathepsin B (Fasciola; Clonorchis) | OvAE1795, OvAE813, OvAE1171, OvAE532, OvAE1070, OvAE1613, OvAE1711, OvAE615, OvAE398 | > 80 |
| C13 family asparaginyl endopeptidase | legumain (O. viverrini) | OvAE1624, OvAE1824 | 94 |
| S1 family serine protease | HtrA-like (Macaca nr/Schistosoma est) and kallikrein-like (Schistosoma) peptidases | OvAE622, OvAE1918 | 47–53 |
| A1 family aspartic protease | cathepsin D-like; digestive enzyme in helminths (Clonorchis) | OvAE1300 | 80 |
| M41 family metalloprotease | mitochondrial membrane proteinase (Schistosoma) | OvAE65 | 91 |
| Granulin | mitogen associated with cancer (Bos nr/Clonorchis est) | OvAE1732 | 45 |
| Aquaporin | water channel protein (Schistosoma) | OvAE6 | 48 |
| Tyrosinase | critical for S. mansoni egg shell production (Schistosoma) | OvAE1900, OvAE1854 | 63 |
| Phospholipase A2 | similar to vertebrate venom proteins; (Heloderma nr/Clonorchis est) | OvAE1644 | 55 |
| Thioredoxin peroxidase | immunomodulatory in fasciolosis (Schistosoma) | OvAE54 | 74 |
| EF-hand secreted Ca2+-binding protein | calumenin (Rattus nr/Xenopus est) | OvAE61 | 47 |
| Saposin-like protein | pore forming; similar to fluke cytolysins (Clonorchis) | OvAE1692 | 64 |
| Pathogenesis related protein | similar to helminth venom allergen homologues (Schistosoma) | OvAE534, OvAE1862 | 38 |
| Glutathione-S-transferase | detoxification of heme and free radicals (Clonorchis) | OvAE1057, OvAE1892, OvAE1601, OvAE1729 | 86 |
| Synaptobrevin | neurotransmission/vesicular docking – vesicle associated (Schistosoma) | OvAE1001 | 73 |
| Innexin | integral membrane protein forming gap junctions (Schistosoma) | OvAE631 | 78 |
| Fibroblast growth factor (FGF) receptor substrate 2 | host FGF is essential for growth of schistosomes (Schistosoma) | OvAE1563 | 32 |
| Ly6c | Immune cell differentiation antigen | OvAE82 | 26 |
Proteases
As with other parasitic helminth transcriptomes [11-13,25,26], proteins with catalytic activity were abundantly represented in the O. viverrini dataset (27.9% of contigs that were assigned GO molecular functions). Many of these enzymes encoded endo- and exo-proteases belonging to established families (MEROPS classification), but which have not yet been described from liver flukes (Table 3). Of particular interest were members of the S1A serine protease family with sequence similarity to kallikrein and chymotrypsin, and, therefore, potentially involved in feeding or tissue migration [27]. Other proteases included homologues of enzymes that digest hemoglobin in blood-feeding helminths, including cathepsin D-like aspartic and cathepsin B-like cysteine proteases [28-30] as well as an asparaginyl endopeptidase, which is known to activate the gastrodermal cathepsin B enzyme, and probably other gut proteases in S. mansoni [31]. We also identified O. viverrini homologues of the cell death enzyme, caspase-2, and the neutral cysteine protease from the tegument of schistosomes, calpain.
Multiple membrane-spanning proteins
Predicted proteins with multiple membrane spanning domains were identified. Tetraspanins, an abundantly represented family of four-transmembrane proteins in the tegument of schistosomes [32,33], were identified from O. viverrini (Figure 5). These proteins are thought to stabilize the cell membrane by forming a network of interactions, called the tetraspanin web, with other membrane-bound and -associated proteins, particularly on the surface of cells of the immune system [34]. A homologue of the six-transmembrane domain family of water channel proteins, aquaporin [35], was identified. Seven transmembrane proteins are common drug targets [36], and at least three distinct members of this family were identified, including receptors for dystroglycan and lamin b, and a protein with homology to a the DC-STAMP receptor from the surface of dendritic cells (Table 3).
Figure 5.

An Opisthorchis viverrini homologue of Sm-TSP-2, a vaccine antigen expressed in the tegument of Schistosoma mansoni. Multiple sequence alignment comparing the ORF of OvAE953 with Sm-TSP-2 from S. mansoni (GenBank AF521091) and human CD63 (NM_001780). Both Sm-TSP-2 and CD63 sequences shown here are truncated at the C-terminus (fourth transmembrane domain and C-terminal tail are not shown) for comparative purposes because OvAE953 is a partial sequence. Black boxes denote identical residues shared by two or more of the sequences. Grey boxes denote conservative substitutions. Dashed lines denote the predicted transmembrane (TM) domains of Sm-TSP-2; the solid line represents the extracellular (EC) loop 2 region of Sm-TSP-2 [33].
Host-parasite "cross talk"
Parasitic helminths receive host-derived signals for growth and reproduction via surface receptors for host ligands, [37-39]. Convergent evolution of extracellular parasite proteins to promote their interactions with host tissues is well documented [40,41], and we identified O. viverrini ORFs encoding membrane and secreted proteins, some of which were clearly more similar to vertebrate than to invertebrate homologues (Table 3). Transforming growth factor-beta (TGF-β) regulates cell growth and differentiation and is acquired on the cell surface by specific TGF-β receptors [42]. An ORF encoding a member of the TGF-β receptor type Ib family was identified in O. viverrini. The ORF included a 28 amino acid insertion absent from other type I TGF-β receptors, except for TR1 from the hydatid tapeworms of the genus Echinococcus (also members of the phylum Platyhelminthes) [43]. However, these two insertions did not share sequence identity (Figure 6A). Unlike many of the ESTs identified for which the closest homologues were from parasitic trematodes, the O. viverrini TGF-β receptor type I was divergent from SmRK-I of S. mansoni [44] and instead grouped more closely with proteins from Echinococcus multilocularis and from parasitic and free-living nematodes (Figure 6B). In pairwise sequence comparisons, however, the O. viverrini partial ORF was more similar to pig and macaque sequences (44% identity over 181 amino acids) than it was to Echinococcus TR1 (40% over 180 residues) or SmRK1 (40% over 182 residues). SmRK-I is known to bind to human TGF-β [40], suggesting that the O. viverrini receptor might also bind host growth factors for maturation and reproduction. Another OvAE encoding a protein which is potentially involved in the acquisition of host signals (and subsequent signaling) for growth and development was a fibroblast growth factor (FGF) receptor substrate 2. Parasitic flatworms induce fibrosis (and FGF) [45], and the parasites might acquire and utilize the host FGF that they induce for development and reproduction. Indeed, schistosomes are dependent upon FGF and transferrin for growth and maturation in vitro [46]. Of the sequences presented in Table 3, another OvAE which shared greatest identity with vertebrate homologues, encoded for calumenin, an EF-hand calcium binding protein localized to the secretory pathway. Calumenin is an inhibitor of the gamma-carboxylation system [47] and is expressed in thrombin-activated thrombocytes. It has a modulating effect on the organization of the actin cytoskeleton and may be involved in the pathophysiology of thrombosis or in wound healing [48]. The predicted calumenin of O. viverrini was most similar to rat and frog orthologues/paralogues, suggesting that it might interact with actin on the surface of host cells which are damaged during parasite feeding and migration.
Figure 6.

A TGF-β receptor type I from Opisthorchis viverrini. Multiple sequence alignment of the ORFs of OvAE22 with homologues from Schistosoma mansoni (SmRK-I – GenBank AF031557), the hydatid tapeworm Echinococcus multilocularis (TR1 – AJ841786) and human (TGF-β receptor type I – L11695) (A). The overlined region denotes the putative serine-threonine kinase active site in SmRK-I [44]. Residues highlighted in red font in OvAE22 are putative sites of serine/threonine phosphorylation. Both SmRK-I and human TGF-β receptor type I sequences shown here are truncated at the N-terminus and SmRK-I is truncated at the C-terminus for comparative purposes with the partial sequence from O. viverrini. Black boxes denote identical residues shared by two or more of the sequences. Grey boxes denote conservative substitutions. Neighbour joining phylogenetic tree showing the relationship between the ORF of OvAE22 and other members of the TGF-β receptor type I family (B). Numbers on branches denote bootstrap values from 100 samplings. The nominated outgroup was the type 2 receptor, SmRK-2. GenBank accession numbers not already provided above are as follows: pig bone morphogenic protein (BMP) receptor type I (AY065994); dog hookworm Ancylostoma caninum S/T kinase (AY053388); Caenorhabditis briggsae CBG02627 (CAAC01000012); filarial nematode Brugia pahangi trk-1 (AF013991); S. mansoni SmRK-2 (AY550912).
Molecules associated with cancer?
O. viverrini is the major cause of CCA in South-East Asia [1]. The molecular mechanisms underlying induction of O. viverrini-induced CCA are thought to be multi-factorial (reviewed in [49]), but recent evidence suggests that O. viverrini secretes mitogenic proteins into host tissues [1,8]. OvAEs encoding secreted proteins with prospective mitogenic activity were identified in the EST dataset. Of note, first, progranulin (pgrn) is a pluripotent secreted growth factor that mediates cell cycle progression, cell motility [50] and wound repair [51]. We identified an OvAE (OvAE1732) that shared sequence identity with pgrn (data not shown). Of particular importance is that pgrn has been implicated in regulating the proliferation of tumour cells, and its expression is up-regulated in more aggressive cancers (reviewed in [50]). The kallikrein-like serine proteases are another family of enzymes whose over-expression has been linked to cancer. The expression of some kallikreins in prostate cells leads to changes indicative of an epithelial to mesenchymal transition, an important process in cancer progression [52]. An OvAE (OvAE1918) with sequence identity to kallikrein-like secreted proteases is present in the new O. viverrini gene catalogue. Phospholipase A2 (PLA-2) regulates the provision of arachidonic acid to both cyclooxygenase- and lipoxygenase-derived eicosanoids (reviewed by [53]), and the upregulation of cyclooxygenase-2 is thought to be an important feature of cholangiocarcinogenesis in both humans and experimental rodent models [49,54,55]. We identified an OvAE (OvAE1644) that encodes a secreted PLA-2 which shared greatest sequence identity with PLA-2 from venom of Heloderma (Gila monster) and an EST from C. sinensis (Table 3). Parasites utilize secreted serine proteases [56] and PLA-2s [57] to invade host tissues, and homologues of these proteins (and granulin) are potentially secreted by O. viverrini into host tissues where they might promote cell proliferation, mutagenesis and ultimately carcinogensis. Ongoing studies in our laboratories are now focused on the physiological roles of these putative carcinogens in the host-parasite relationship and in cholangiocarcinogenesis induced by O. viverrini infection.
Potential vaccines
Digenean flukes develop through a series of morphologically and developmentally discrete stages within their mammalian hosts, and each stage can be expected to display a characteristic transcritpome, confounding efforts to develop new control measures. Adult parasitic flukes are bound by an outer epithelial tegument, a structure that is widely regarded as the most vulnerable target for vaccines and drugs [32]. Homologues and orthologues of vaccine antigens identified in the tegument (and other structures from larval stages) of other flatworms were identified in the O. viverrini dataset (Table 4). Of particular note were the membrane spanning proteins, including an orthologue of the protective tetraspanin from S. mansoni, Sm-TSP-2 [32,33] (Figure 5) and the 22.6 kDa family of antigens from the schistosome tegument [58]. Homologues of gut proteases used by blood-feeding helminths to digest their blood-meal were identified from O. viverrini, including cathepsin D-like aspartic proteases [59,60], 11 distinct papain-like cysteine proteases [61-63] and the neutral protease, calpain, which associates with the inner tegument of schistosomes [64]. Other potential immunogens include lipid-binding proteins which are efficacious vaccines in the rabbit model against the western liver fluke, Fasciola hepatica, including saposin-like proteins [65] and fatty acid-binding proteins [66,67].
Table 4.
Opisthorchis viverrini ESTs with sequence identity to mRNAs encoding proteins efficacious as vaccines against other flatworm and nematode parasites.
| Predicted protein family | Vaccine antigen and helminth targeted | References |
| Aspartic protease | APR-1 for hookworm; cathepsin D for Schistosoma japonicum | [59, 60] |
| Glutathione-S-transferase | Ac-GST-1 for hookworm; bilvax for schistosomes | [79, 80] |
| Cysteine protease | TSBP for Haemonchus contortus; AcCP1 for hookworm; Cathepsins L1 and L2 for Fasciola hepatica | [61–63] |
| Tetraspanin | TSP-1, TSP-2 and Sm23 for Schistosoma mansoni | [33, 81] |
| Pathogenesis related protein | ASP-2 for hookworm; ASP-1 for Onchocerca volvulus | [82, 83] |
| calpain | Smp80 for schistosomes | [64] |
| Fatty acid binding protein | Sm14 for S. mansoni and F. hepatica | [66, 67] |
| Saposin-like protein | FhSAP-2 for F. hepatica | [65] |
| 14-3-3 | Sm14-3-3 for schistosomes | [84] |
| 22.6 (unknown function) | Sm22.6 for S. mansoni | [58] |
Conclusion
This report provides the first description of gene discovery for the liver fluke O. viverrini. Infection with O. viverrini is an important tropical health issue, but even more important and enigmatic is that chronic O. viverrini infection leads to the development of CCA. Indeed, there is no stronger link between a human parasite and cancer than that between O. viverrini and CCA [68]. The new gene catalogue for O. viverrini represents the largest EST dataset in the public domain for any species of liver fluke, and provides a platform for explorations into the molecular basis of host-helminth parasite interactions. We [1] and others [8] are interested in the molecules secreted into host tissues by O. viverrini that induce hyper-proliferation of biliary cells which can subsequently undergo malignant transformation. Given the number of O. viverrini ESTs sequenced herein, it is possible that mRNAs corresponding to these parasite mitogens are already present in the current dataset. Proteomic analysis of proteins secreted by adult O. viverrini maintained in vitro also is underway in our laboratories, and linking peptide sequences to corresponding mRNAs can be expected to be facilitated by this gene discovery program [12]. Finally, this gene discovery information for O. viverrini should expedite molecular studies of cholangiocarcinogenesis and accelerate research focused on developing new interventions, drugs and vaccines, to control O. viverrini and related flukes.
Methods
Parasite material
Adult O. viverrini were collected from experimentally infected hamsters (Mesocricetus auratas) maintained at the animal facility of the Khon Kaen University Faculty of Medicine. Protocols approved by the Khon Kaen University Animal Ethics Committee were used for all animal research conducted in this study. Briefly, metacercariae of O. viverrini were collected from naturally infected cyprinoid fish by pepsin digestion. Metacercariae (100 per hamster) were administered intragastrically to hamsters. Hamsters were euthanazed 6 weeks after inoculation, and adult worms were flushed with saline from the bile ducts [69]. Worms were washed extensively with sterile phosphate-buffered saline (pH 7.2), after which they were snap frozen and stored in liquid nitrogen or employed immediately as the source of fluke RNA.
Construction and mass excision of cDNA library
Total RNA from adult O. viverrini was extracted using Trizol (Invitrogen), following the manufacturer's instructions. Ten μg of O. viverrini total RNA was used as a template for the synthesis of double-stranded cDNA using the SMART cDNA kit (BD Bioscience), after which the cDNA modified with adapters was cloned into the Sfi I site of the pTriplEx2 plasmid (BD Bioscience) and packaged into λ arms. The titer and percentage of recombinant phages in the library were determined using the protocols recommended by the manufacturer. Escherichia coli strain BM25.8 cells were transduced with recombinant phage, from which the excision of the pTriplEx phagemid library was accomplished.
Clone selection, sequencing and annotation
Five thousand clones were randomly selected from the phagemid library and grown overnight in Luria Bertani (LB) broth supplemented with ampicillin to a final concentration of 25 μg/ml. Overnight cultures were shipped at 4°C in LB broth/ampicillin to the University of Melbourne (Department of Veterinary Science). The sequencing was performed by AgGenomics Inc., Australia, using a 3730xl DNA analyzer (Applied Biosystems). The TempliPhi™ DNA Sequencing Template Amplification system (GE Healthcare) was used to sequence each clone using the 5'λ TriplEx2 sequencing primer.
Bioinformatic analyses
Edited sequences were condensed into contigs or singletons using TGICL [70] with the default parameters of 40 bp overlap, a minimum of 95% identity and a 30 bp maximum mismatched overhang. Sequences of less than 100 nt were discarded. Sequences were named using the same convention as that used for the human blood fluke, Schistosoma mansoni [13]; OvAE for O. viverrini Assembled EST. Sequences were compared with those available in the NCBI non-redundant protein and nucleotide databases using BLASTx and BLASTn. searches, respectively in October 2006. The dbEST database was queried using BLASTn and tBLASTx searches. BLAST alignments with an E-value of ≤ 1.0 × 10-5 were reported. OvAEs were functionally categorized by querying a local copy of the Gene Ontology (GO) database [71] (downloaded November, 2006) with an E-value cutoff of 1.0 × 10-5. All ESTs from C. sinensis [9] and Schistosoma japonicum [11,12] were downloaded from NCBI [72], and the same methodology was used to derive ontology classifications for the C. sinensis ESTs. ORF predictions were performed using GENSCAN [73] using the HumanIso parameter set. Signal sequence prediction was accomplished using SignalP 3.0 [74], incorporating both hidden Markov models and neural networks. Positive signal sequence predictions from either method and positive signal anchor predictions using Markov models were reported. Predictions of transmembrane domains were conducted using TMPred [75]. All multiple sequence alignments were carried out using ClustalW. Clan and family assignments of proteolytic enzymes were analyzed via the MEROPS protease database [76]. Putative phosphorylation sites were predicted using the NetPhos 2.0 server [77].
Phylogenetic trees
Multiple sequence alignments were assembled using ClustalW. Only regions which completely overlapped with partial ORFs of O. viverrini ESTs were used for tree construction. Alignments were imported into PAUP version 4.0 beta [78] to construct trees using the neighbour joining and maximum parsimony methods. Robustness was assessed by bootstrap analysis using 100 replicates. Clades with more than 50% support were denoted with bootstrap values on the branches.
Cross-taxon similarity analysis
OvAEs were compared with all entries for other organisms in the NCBI dbEST database using tBLASTx. The highest BLAST scores (above a cut-off value of 50) were used to generate SimiTri plots [20] using software developed in-house (J. Mulvenna, unpublished).
Authors' contributions
TL, PP and JM generated and analyzed the data and contributed to drafting of the ms. BS provided parasite material, helped conceive the study and edited the drafted ms. MS and MJS provided technical assistance and edited the drafted ms. RBG facilitated interactions with the sequencing unit and edited the drafted ms. PB helped conceive the study, supervised the research and helped draft the ms. AL helped conceive the study, supervised the research, and took the lead on drafting the ms. All authors read and approved the final ms.
Supplementary Material
Opisthorchis viverrini sequences that had homologues in the parasitic flukes, Clonorchis sinensis and Schistosoma japonicum, but not in the free-living platyhelminth, Schmidtea mediterranea.
Acknowledgments
Acknowledgements
We are grateful for grant support from the Sandler Foundation, NIH-NIAID (award number AI065871), Khon Kaen University Research Fund, Thailand-Tropical Diseases Research Program and the Australian Research Council (LP0667795 and DP0665230). JM was supported by a Peter Doherty Training Fellowship from the National Health and Medical Research Council of Australia (NHMRC). AL was supported by an R. Douglas Wright Career Development Award from NHMRC. We thank Ian Smith, Jacqui Batley, Nonglack Kewgrai, Bronwyn Campbell and David Blair for advice or support.
Contributor Information
Thewarach Laha, Email: thewa_la@kku.ac.th.
Porntip Pinlaor, Email: porawa@kku.ac.th.
Jason Mulvenna, Email: jason.mulvenna@qimr.edu.au.
Banchob Sripa, Email: banchob@kku.ac.th.
Manop Sripa, Email: thewa_la@kku.ac.th.
Michael J Smout, Email: michael.smout@qimr.edu.au.
Robin B Gasser, Email: robinbg@unimelb.edu.au.
Paul J Brindley, Email: paul.brindley@tulane.edu.
Alex Loukas, Email: Alex.Loukas@qimr.edu.au.
References
- Sripa B, Kaewkes S, Sithithaworn P, Mairiang E, Laha T, Smout M, Pairojkul C, Bhudhisawasdi V, Tesana S, Thinkamrop B, Bethony JM, Loukas A, Brindley PJ. Liver fluke induces cholangiocarcinoma. PLoS Med. 2007;in press doi: 10.1371/journal.pmed.0040201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- IARC Infection with liver flukes (Opisthorchis viverrini, Opisthorchis felineus and Clonorchis sinensis) IARC Monogr Eval Carcinog Risks Hum. 1994;61:121–175. [PMC free article] [PubMed] [Google Scholar]
- IARC monogrpahs on the evaluation of carcinogenic risks to humans
- Sithithaworn P, Tesana S, Pipitgool V, Kaewkes S, Pairojkul C, Sripa B, Paupairoj A, Thaiklar K. Relationship between faecal egg count and worm burden of Opisthorchis viverrini in human autopsy cases. Parasitology. 1991;102 Pt 2:277–281. doi: 10.1017/s0031182000062594. [DOI] [PubMed] [Google Scholar]
- Srivatanakul P. Epidemiology of Liver Cancer in Thailand. Asian Pac J Cancer Prev. 2001;2:117–121. [PubMed] [Google Scholar]
- Komalamisra C. Chromosomes and C-banding of Opisthorchis viverrini. Southeast Asian J Trop Med Public Health. 1999;30:576–579. [PubMed] [Google Scholar]
- Park GM, Im K, Huh S, Yong TS. Chromosomes of the liver fluke, Clonorchis sinensis. Korean J Parasitol. 2000;38:201–206. doi: 10.3347/kjp.2000.38.3.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thuwajit C, Thuwajit P, Kaewkes S, Sripa B, Uchida K, Miwa M, Wongkham S. Increased cell proliferation of mouse fibroblast NIH-3T3 in vitro induced by excretory/secretory product(s) from Opisthorchis viverrini. Parasitology. 2004;129:455–464. doi: 10.1017/S0031182004005815. [DOI] [PubMed] [Google Scholar]
- Cho PY, Lee MJ, Kim TI, Kang SY, Hong SJ. Expressed sequence tag analysis of adult Clonorchis sinensis, the Chinese liver fluke. Parasitol Res. 2006;99:602–608. doi: 10.1007/s00436-006-0204-1. [DOI] [PubMed] [Google Scholar]
- Lun ZR, Gasser RB, Lai DH, Li AX, Zhu XQ, Yu XB, Fang YY. Clonorchiasis: a key foodborne zoonosis in China. Lancet Infect Dis. 2005;5:31–41. doi: 10.1016/S1473-3099(04)01252-6. [DOI] [PubMed] [Google Scholar]
- Hu W, Yan Q, Shen DK, Liu F, Zhu ZD, Song HD, Xu XR, Wang ZJ, Rong YP, Zeng LC, Wu J, Zhang X, Wang JJ, Xu XN, Wang SY, Fu G, Zhang XL, Wang ZQ, Brindley PJ, McManus DP, Xue CL, Feng Z, Chen Z, Han ZG. Evolutionary and biomedical implications of a Schistosoma japonicum complementary DNA resource. Nat Genet. 2003;35:139–147. doi: 10.1038/ng1236. [DOI] [PubMed] [Google Scholar]
- Liu F, Lu J, Hu W, Wang SY, Cui SJ, Chi M, Yan Q, Wang XR, Song HD, Xu XN, Wang JJ, Zhang XL, Zhang X, Wang ZQ, Xue CL, Brindley PJ, McManus DP, Yang PY, Feng Z, Chen Z, Han ZG. New perspectives on host-parasite interplay by comparative transcriptomic and proteomic analyses of Schistosoma japonicum. PLoS Pathog. 2006;2:e29. doi: 10.1371/journal.ppat.0020029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verjovski-Almeida S, DeMarco R, Martins EA, Guimaraes PE, Ojopi EP, Paquola AC, Piazza JP, Nishiyama MY, Jr., Kitajima JP, Adamson RE, Ashton PD, Bonaldo MF, Coulson PS, Dillon GP, Farias LP, Gregorio SP, Ho PL, Leite RA, Malaquias LC, Marques RC, Miyasato PA, Nascimento AL, Ohlweiler FP, Reis EM, Ribeiro MA, Sa RG, Stukart GC, Soares MB, Gargioni C, Kawano T, Rodrigues V, Madeira AM, Wilson RA, Menck CF, Setubal JC, Leite LC, Dias-Neto E. Transcriptome analysis of the acoelomate human parasite Schistosoma mansoni. Nat Genet. 2003;35:148–157. doi: 10.1038/ng1237. [DOI] [PubMed] [Google Scholar]
- Sim S, Park GM, Yong TS. Cloning and characterization of Clonorchis sinensis myoglobin using immune sera against excretory-secretory antigens. Parasitol Res. 2003;91:338–343. doi: 10.1007/s00436-003-0869-7. [DOI] [PubMed] [Google Scholar]
- Jones MK, Gobert GN, Zhang L, Sunderland P, McManus DP. The cytoskeleton and motor proteins of human schistosomes and their roles in surface maintenance and host-parasite interactions. Bioessays. 2004;26:752–765. doi: 10.1002/bies.20058. [DOI] [PubMed] [Google Scholar]
- Matsumoto Y, Perry G, Levine RJ, Blanton R, Mahmoud AA, Aikawa M. Paramyosin and actin in schistosomal teguments. Nature. 1988;333:76–78. doi: 10.1038/333076a0. [DOI] [PubMed] [Google Scholar]
- Zhang LH, McManus DP, Sunderland P, Lu XM, Ye JJ, Loukas A, Jones MK. The cellular distribution and stage-specific expression of two dynein light chains from the human blood fluke Schistosoma japonicum. Int J Biochem Cell Biol. 2005;37:1511–1524. doi: 10.1016/j.biocel.2005.01.015. [DOI] [PubMed] [Google Scholar]
- Tansatit T, Sahaphong S, Riengrojpitak S, Viyanant V, Sobhon P. Immunolocalization of cytoskeletal components in the tegument of the 3-week-old juvenile and adult Fasciola gigantica. Vet Parasitol. 2006;135:269–278. doi: 10.1016/j.vetpar.2005.10.018. [DOI] [PubMed] [Google Scholar]
- Fitzsimmons CM, McBeath R, Joseph S, Jones FM, Walter K, Hoffmann KF, Kariuki HC, Mwatha JK, Kimani G, Kabatereine NB, Vennervald BJ, Ouma JH, Dunne DW. Factors affecting human IgE and IgG responses to allergen-like Schistosoma mansoni antigens: Molecular structure and patterns of in vivo exposure. Int Arch Allergy Immunol. 2007;142:40–50. doi: 10.1159/000095997. [DOI] [PubMed] [Google Scholar]
- Parkinson J, Blaxter M. SimiTri--visualizing similarity relationships for groups of sequences. Bioinformatics. 2003;19:390–395. doi: 10.1093/bioinformatics/btf870. [DOI] [PubMed] [Google Scholar]
- Zayas RM, Hernandez A, Habermann B, Wang Y, Stary JM, Newmark PA. The planarian Schmidtea mediterranea as a model for epigenetic germ cell specification: analysis of ESTs from the hermaphroditic strain. Proc Natl Acad Sci U S A. 2005;102:18491–18496. doi: 10.1073/pnas.0509507102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carranza S, Baguna J, Riutort M. Are the Platyhelminthes a monophyletic primitive group? An assessment using 18S rDNA sequences. Mol Biol Evol. 1997;14:485–497. doi: 10.1093/oxfordjournals.molbev.a025785. [DOI] [PubMed] [Google Scholar]
- Le TH, Blair D, McManus DP. Mitochondrial genomes of parasitic flatworms. Trends Parasitol. 2002;18:206–213. doi: 10.1016/S1471-4922(02)02252-3. [DOI] [PubMed] [Google Scholar]
- Karling TG. On the anatomy and affinities of the turbellarian orders. In: Riser NW, Morse MP, editor. Biology of the turbellaria. New York , McGraw Hill; 1974. pp. 1–16. [Google Scholar]
- Hu W, Brindley PJ, McManus DP, Feng Z, Han ZG. Schistosome transcriptomes: new insights into the parasite and schistosomiasis. Trends Mol Med. 2004;10:217–225. doi: 10.1016/j.molmed.2004.03.002. [DOI] [PubMed] [Google Scholar]
- Ranjit N, Jones MK, Stenzel DJ, Gasser RB, Loukas A. A survey of the intestinal transcriptomes of the hookworms, Necator americanus and Ancylostoma caninum, using tissues isolated by laser microdissection microscopy. Int J Parasitol. 2006;36:701–710. doi: 10.1016/j.ijpara.2006.01.015. [DOI] [PubMed] [Google Scholar]
- Paliouras M, Diamandis EP. The kallikrein world: an update on the human tissue kallikreins. Biol Chem. 2006;387:643–652. doi: 10.1515/BC.2006.083. [DOI] [PubMed] [Google Scholar]
- Brindley PJ, Kalinna BH, Wong JY, Bogitsh BJ, King LT, Smyth DJ, Verity CK, Abbenante G, Brinkworth RI, Fairlie DP, Smythe ML, Milburn PJ, Bielefeldt-Ohmann H, Zheng Y, McManus DP. Proteolysis of human hemoglobin by schistosome cathepsin D. Mol Biochem Parasitol. 2001;112:103–112. doi: 10.1016/S0166-6851(00)00351-0. [DOI] [PubMed] [Google Scholar]
- Delcroix M, Sajid M, Caffrey CR, Lim KC, Dvorak J, Hsieh I, Bahgat M, Dissous C, McKerrow JH. A multienzyme network functions in intestinal protein digestion by a platyhelminth parasite. J Biol Chem. 2006;281:39316–39329. doi: 10.1074/jbc.M607128200. [DOI] [PubMed] [Google Scholar]
- Williamson AL, Lecchi P, Turk BE, Choe Y, Hotez PJ, McKerrow JH, Cantley LC, Sajid M, Craik CS, Loukas A. A multi-enzyme cascade of hemoglobin proteolysis in the intestine of blood-feeding hookworms. J Biol Chem. 2004;279:35950–35957. doi: 10.1074/jbc.M405842200. [DOI] [PubMed] [Google Scholar]
- Sajid M, McKerrow JH, Hansell E, Mathieu MA, Lucas KD, Hsieh I, Greenbaum D, Bogyo M, Salter JP, Lim KC, Franklin C, Kim JH, Caffrey CR. Functional expression and characterization of Schistosoma mansoni cathepsin B and its trans-activation by an endogenous asparaginyl endopeptidase. Mol Biochem Parasitol. 2003;131:65–75. doi: 10.1016/S0166-6851(03)00194-4. [DOI] [PubMed] [Google Scholar]
- Loukas A, Tran MH, Pearson MS. Schistosome membrane proteins as vaccines. Int J Parasitol. 2007;37:257–263. doi: 10.1016/j.ijpara.2006.12.001. [DOI] [PubMed] [Google Scholar]
- Tran MH, Pearson MS, Bethony JM, Smyth DJ, Jones MK, Duke M, Don TA, McManus DP, Correa-Oliveira R, Loukas A. Tetraspanins on the surface of Schistosoma mansoni are protective antigens against schistosomiasis. Nat Med. 2006;12:835–840. doi: 10.1038/nm1430. [DOI] [PubMed] [Google Scholar]
- Levy S, Shoham T. The tetraspanin web modulates immune-signalling complexes. Nat Rev Immunol. 2005;5:136–148. doi: 10.1038/nri1548. [DOI] [PubMed] [Google Scholar]
- Agre P. The aquaporin water channels. Proc Am Thorac Soc. 2006;3:5–13. doi: 10.1513/pats.200510-109JH. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simmons DL. What makes a good anti-inflammatory drug target? Drug Discov Today. 2006;11:210–219. doi: 10.1016/S1359-6446(05)03721-9. [DOI] [PubMed] [Google Scholar]
- Amiri P, Locksley RM, Parslow TG, Sadick M, Rector E, Ritter D, McKerrow JH. Tumour necrosis factor alpha restores granulomas and induces parasite egg-laying in schistosome-infected SCID mice. Nature. 1992;356:604–607. doi: 10.1038/356604a0. [DOI] [PubMed] [Google Scholar]
- Blank RB, Lamb EW, Tocheva AS, Crow ET, Lim KC, McKerrow JH, Davies SJ. The common gamma chain cytokines interleukin (IL)-2 and IL-7 indirectly modulate blood fluke development via effects on CD4+ T cells. J Infect Dis. 2006;194:1609–1616. doi: 10.1086/508896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies SJ, Grogan JL, Blank RB, Lim KC, Locksley RM, McKerrow JH. Modulation of blood fluke development in the liver by hepatic CD4+ lymphocytes. Science. 2001;294:1358–1361. doi: 10.1126/science.1064462. [DOI] [PubMed] [Google Scholar]
- Beall MJ, Pearce EJ. Human transforming growth factor-beta activates a receptor serine/threonine kinase from the intravascular parasite Schistosoma mansoni. J Biol Chem. 2001;276:31613–31619. doi: 10.1074/jbc.M104685200. [DOI] [PubMed] [Google Scholar]
- Osman A, Niles EG, Verjovski-Almeida S, LoVerde PT. Schistosoma mansoni TGF-beta receptor II: role in host ligand-induced regulation of a schistosome target gene. PLoS Pathog. 2006;2:e54. doi: 10.1371/journal.ppat.0020054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keah HH, Hearn MT. A molecular recognition paradigm: promiscuity associated with the ligand-receptor interactions of the activin members of the TGF-beta superfamily. J Mol Recognit. 2005;18:385–403. doi: 10.1002/jmr.715. [DOI] [PubMed] [Google Scholar]
- Zavala-Gongora R, Kroner A, Bernthaler P, Knaus P, Brehm K. A member of the transforming growth factor-beta receptor family from Echinococcus multilocularis is activated by human bone morphogenetic protein 2. Mol Biochem Parasitol. 2006;146:265–271. doi: 10.1016/j.molbiopara.2005.12.011. [DOI] [PubMed] [Google Scholar]
- Davies SJ, Shoemaker CB, Pearce EJ. A divergent member of the transforming growth factor beta receptor family from Schistosoma mansoni is expressed on the parasite surface membrane. J Biol Chem. 1998;273:11234–11240. doi: 10.1074/jbc.273.18.11234. [DOI] [PubMed] [Google Scholar]
- Wynn TA. Fibrotic disease and the T(H)1/T(H)2 paradigm. Nat Rev Immunol. 2004;4:583–594. doi: 10.1038/nri1412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clemens LE, Basch PF. Schistosoma mansoni: effect of transferrin and growth factors on development of schistosomula in vitro. J Parasitol. 1989;75:417–421. doi: 10.2307/3282599. [DOI] [PubMed] [Google Scholar]
- Wajih N, Sane DC, Hutson SM, Wallin R. The inhibitory effect of calumenin on the vitamin K-dependent gamma-carboxylation system. Characterization of the system in normal and warfarin-resistant rats. J Biol Chem. 2004;279:25276–25283. doi: 10.1074/jbc.M401645200. [DOI] [PubMed] [Google Scholar]
- Ostergaard M, Hansen GA, Vorum H, Honore B. Proteomic profiling of fibroblasts reveals a modulating effect of extracellular calumenin on the organization of the actin cytoskeleton. Proteomics. 2006;6:3509–3519. doi: 10.1002/pmic.200500686. [DOI] [PubMed] [Google Scholar]
- Sirica AE. Cholangiocarcinoma: molecular targeting strategies for chemoprevention and therapy. Hepatology. 2005;41:5–15. doi: 10.1002/hep.20537. [DOI] [PubMed] [Google Scholar]
- He Z, Bateman A. Progranulin (granulin-epithelin precursor, PC-cell-derived growth factor, acrogranin) mediates tissue repair and tumorigenesis. J Mol Med. 2003;81:600–612. doi: 10.1007/s00109-003-0474-3. [DOI] [PubMed] [Google Scholar]
- He Z, Ong CH, Halper J, Bateman A. Progranulin is a mediator of the wound response. Nat Med. 2003;9:225–229. doi: 10.1038/nm816. [DOI] [PubMed] [Google Scholar]
- Whitbread AK, Veveris-Lowe TL, Lawrence MG, Nicol DL, Clements JA. The role of kallikrein-related peptidases in prostate cancer: potential involvement in an epithelial to mesenchymal transition. Biol Chem. 2006;387:707–714. doi: 10.1515/BC.2006.089. [DOI] [PubMed] [Google Scholar]
- Dong Q, Patel M, Scott KF, Graham GG, Russell PJ, Sved P. Oncogenic action of phospholipase A2 in prostate cancer. Cancer Lett. 2006;240:9–16. doi: 10.1016/j.canlet.2005.08.012. [DOI] [PubMed] [Google Scholar]
- Endo K, Yoon BI, Pairojkul C, Demetris AJ, Sirica AE. ERBB-2 overexpression and cyclooxygenase-2 up-regulation in human cholangiocarcinoma and risk conditions. Hepatology. 2002;36:439–450. doi: 10.1053/jhep.2002.34435. [DOI] [PubMed] [Google Scholar]
- Sirica AE, Lai GH, Endo K, Zhang Z, Yoon BI. Cyclooxygenase-2 and ERBB-2 in cholangiocarcinoma: potential therapeutic targets. Semin Liver Dis. 2002;22:303–313. doi: 10.1055/s-2002-34507. [DOI] [PubMed] [Google Scholar]
- Salter JP, Choe Y, Albrecht H, Franklin C, Lim KC, Craik CS, McKerrow JH. Cercarial elastase is encoded by a functionally conserved gene family across multiple species of schistosomes. J Biol Chem. 2002;277:24618–24624. doi: 10.1074/jbc.M202364200. [DOI] [PubMed] [Google Scholar]
- Saffer LD, Schwartzman JD. A soluble phospholipase of Toxoplasma gondii associated with host cell penetration. J Protozool. 1991;38:454–460. doi: 10.1111/j.1550-7408.1991.tb04816.x. [DOI] [PubMed] [Google Scholar]
- Pacifico LG, Fonseca CT, Chiari L, Oliveira SC. Immunization with Schistosoma mansoni 22.6 kDa antigen induces partial protection against experimental infection in a recombinant protein form but not as DNA vaccine. Immunobiology. 2006;211:97–104. doi: 10.1016/j.imbio.2005.06.004. [DOI] [PubMed] [Google Scholar]
- Loukas A, Bethony JM, Mendez S, Fujiwara RT, Goud GN, Ranjit N, Zhan B, Jones K, Bottazzi ME, Hotez PJ. Vaccination with recombinant aspartic hemoglobinase reduces parasite load and blood loss after hookworm infection in dogs. PLoS Med. 2005;2:e295. doi: 10.1371/journal.pmed.0020295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verity CK, McManus DP, Brindley PJ. Vaccine efficacy of recombinant cathepsin D aspartic protease from Schistosoma japonicum. Parasite Immunol. 2001;23:153–162. doi: 10.1046/j.1365-3024.2001.00369.x. [DOI] [PubMed] [Google Scholar]
- Redmond DL, Knox DP. Further protection studies using recombinant forms of Haemonchus contortus cysteine proteinases. Parasite Immunol. 2006;28:213–219. doi: 10.1111/j.1365-3024.2006.00823.x. [DOI] [PubMed] [Google Scholar]
- Loukas A, Bethony JM, Williamson AL, Goud GN, Mendez S, Zhan B, Hawdon JM, Elena Bottazzi M, Brindley PJ, Hotez PJ. Vaccination of dogs with a recombinant cysteine protease from the intestine of canine hookworms diminishes the fecundity and growth of worms. J Infect Dis. 2004;189:1952–1961. doi: 10.1086/386346. [DOI] [PubMed] [Google Scholar]
- Dalton JP, McGonigle S, Rolph TP, Andrews SJ. Induction of protective immunity in cattle against infection with Fasciola hepatica by vaccination with cathepsin L proteinases and with hemoglobin. Infect Immun. 1996;64:5066–5074. doi: 10.1128/iai.64.12.5066-5074.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hota-Mitchell S, Siddiqui AA, Dekaban GA, Smith J, Tognon C, Podesta RB. Protection against Schistosoma mansoni infection with a recombinant baculovirus-expressed subunit of calpain. Vaccine. 1997;15:1631–1640. doi: 10.1016/S0264-410X(97)00081-9. [DOI] [PubMed] [Google Scholar]
- Espino AM, Hillyer GV. A novel Fasciola hepatica saposinlike recombinant protein with immunoprophylactic potential. J Parasitol. 2004;90:876–879. doi: 10.1645/GE-215R. [DOI] [PubMed] [Google Scholar]
- Almeida MS, Torloni H, Lee-Ho P, Vilar MM, Thaumaturgo N, Simpson AJ, Tendler M. Vaccination against Fasciola hepatica infection using a Schistosoma mansoni defined recombinant antigen, Sm14. Parasite Immunol. 2003;25:135–137. doi: 10.1046/j.1365-3024.2003.00619.x. [DOI] [PubMed] [Google Scholar]
- Tendler M, Brito CA, Vilar MM, Serra-Freire N, Diogo CM, Almeida MS, Delbem AC, Da Silva JF, Savino W, Garratt RC, Katz N, Simpson AS. A Schistosoma mansoni fatty acid-binding protein, Sm14, is the potential basis of a dual-purpose anti-helminth vaccine. Proc Natl Acad Sci U S A. 1996;93:269–273. doi: 10.1073/pnas.93.1.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parkin DM. The global health burden of infection-associated cancers in the year 2002. Int J Cancer. 2006;118:3030–3044. doi: 10.1002/ijc.21731. [DOI] [PubMed] [Google Scholar]
- Sithithaworn P, Pipitgool V, Srisawangwong T, Elkins DB, Haswell-Elkins MR. Seasonal variation of Opisthorchis viverrini infection in cyprinoid fish in north-east Thailand: implications for parasite control and food safety. Bull World Health Organ. 1997;75:125–131. [PMC free article] [PubMed] [Google Scholar]
- Pertea G, Huang X, Liang F, Antonescu V, Sultana R, Karamycheva S, Lee Y, White J, Cheung F, Parvizi B, Tsai J, Quackenbush J. TIGR Gene Indices clustering tools (TGICL): a software system for fast clustering of large EST datasets. Bioinformatics. 2003;19:651–652. doi: 10.1093/bioinformatics/btg034. [DOI] [PubMed] [Google Scholar]
- Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, Kasarskis A, Lewis S, Matese JC, Richardson JE, Ringwald M, Rubin GM, Sherlock G. Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat Genet. 2000;25:25–29. doi: 10.1038/75556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- National Center for Biotechnology Information
- Burge C, Karlin S. Prediction of complete gene structures in human genomic DNA. J Mol Biol. 1997;268:78–94. doi: 10.1006/jmbi.1997.0951. [DOI] [PubMed] [Google Scholar]
- Bendtsen JD, Nielsen H, von Heijne G, Brunak S. Improved prediction of signal peptides: SignalP 3.0. J Mol Biol. 2004;340:783–795. doi: 10.1016/j.jmb.2004.05.028. [DOI] [PubMed] [Google Scholar]
- Hofmann K, Stoffel W. TMbase - A database of membrane spanning proteins segments. Biol Chem Hoppe-Seyler. 1993;374:166. [Google Scholar]
- MEROPS the peptidase database
- NetPhos 2.0 server
- Swofford DL. PAUP*. Phylogenetic Analysis Using Parsimony (*and other methods). Version 4. Sunderland, MA , Sinauer Associates; 1998. [Google Scholar]
- Capron A, Riveau G, Capron M, Trottein F. Schistosomes: the road from host-parasite interactions to vaccines in clinical trials. Trends Parasitol. 2005;21:143–149. doi: 10.1016/j.pt.2005.01.003. [DOI] [PubMed] [Google Scholar]
- Zhan B, Liu S, Perally S, Xue J, Fujiwara R, Brophy P, Xiao S, Liu Y, Feng J, Williamson A, Wang Y, Bueno LL, Mendez S, Goud G, Bethony JM, Hawdon JM, Loukas A, Jones K, Hotez PJ. Biochemical characterization and vaccine potential of a heme-binding glutathione transferase from the adult hookworm Ancylostoma caninum. Infect Immun. 2005;73:6903–6911. doi: 10.1128/IAI.73.10.6903-6911.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Da'dara AA, Skelly PJ, Wang MM, Harn DA. Immunization with plasmid DNA encoding the integral membrane protein, Sm23, elicits a protective immune response against schistosome infection in mice. Vaccine. 2001;20:359–369. doi: 10.1016/S0264-410X(01)00374-7. [DOI] [PubMed] [Google Scholar]
- Bethony J, Loukas A, Smout M, Brooker S, Mendez S, Plieskatt J, Goud G, Bottazzi ME, Zhan B, Wang Y, Williamson A, Lustigman S, Correa-Oliveira R, Xiao S, Hotez PJ. Antibodies against a secreted protein from hookworm larvae reduce the intensity of hookworm infection in humans and vaccinated laboratory animals. Faseb J. 2005;19:1743–1745. doi: 10.1096/fj.05-3936fje. [DOI] [PubMed] [Google Scholar]
- MacDonald AJ, Tawe W, Leon O, Cao L, Liu J, Oksov Y, Abraham D, Lustigman S. Ov-ASP-1, the Onchocerca volvulus homologue of the activation associated secreted protein family is immunostimulatory and can induce protective anti-larval immunity. Parasite Immunol. 2004;26:53–62. doi: 10.1111/j.0141-9838.2004.00685.x. [DOI] [PubMed] [Google Scholar]
- Schechtman D, Tarrab-Hazdai R, Arnon R. The 14-3-3 protein as a vaccine candidate against schistosomiasis. Parasite Immunol. 2001;23:213–217. doi: 10.1046/j.1365-3024.2001.00378.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Opisthorchis viverrini sequences that had homologues in the parasitic flukes, Clonorchis sinensis and Schistosoma japonicum, but not in the free-living platyhelminth, Schmidtea mediterranea.


