Abstract
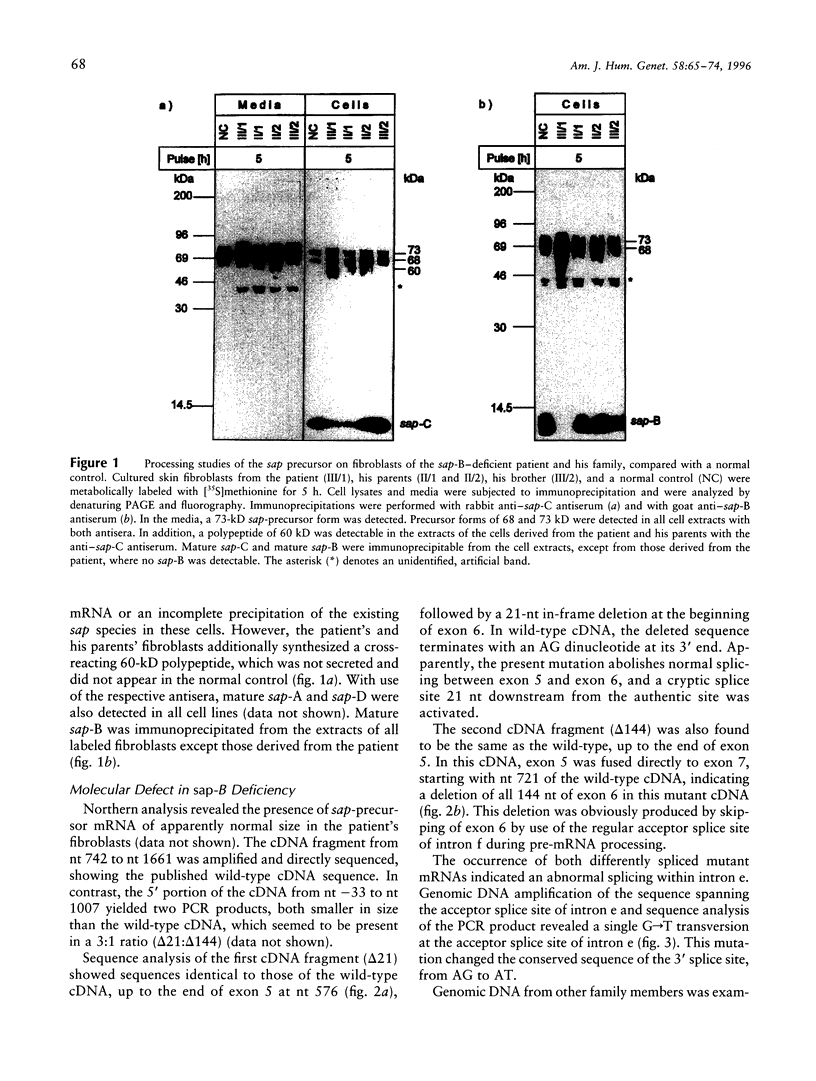
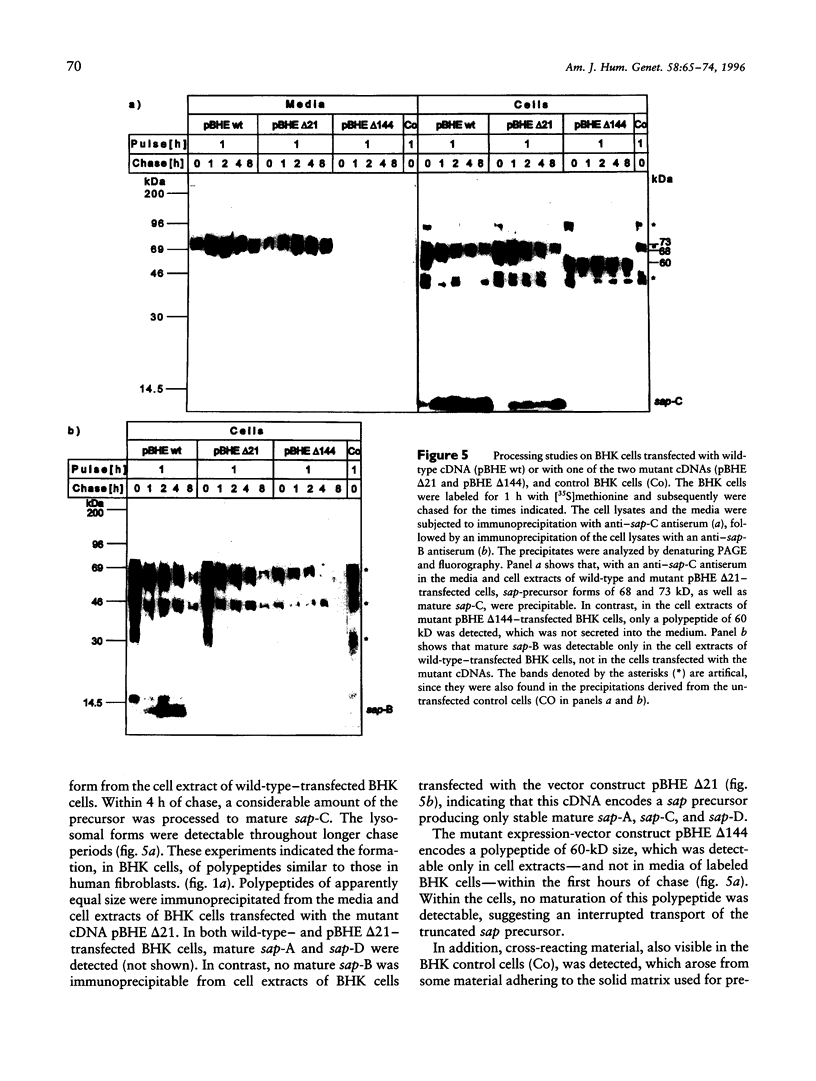
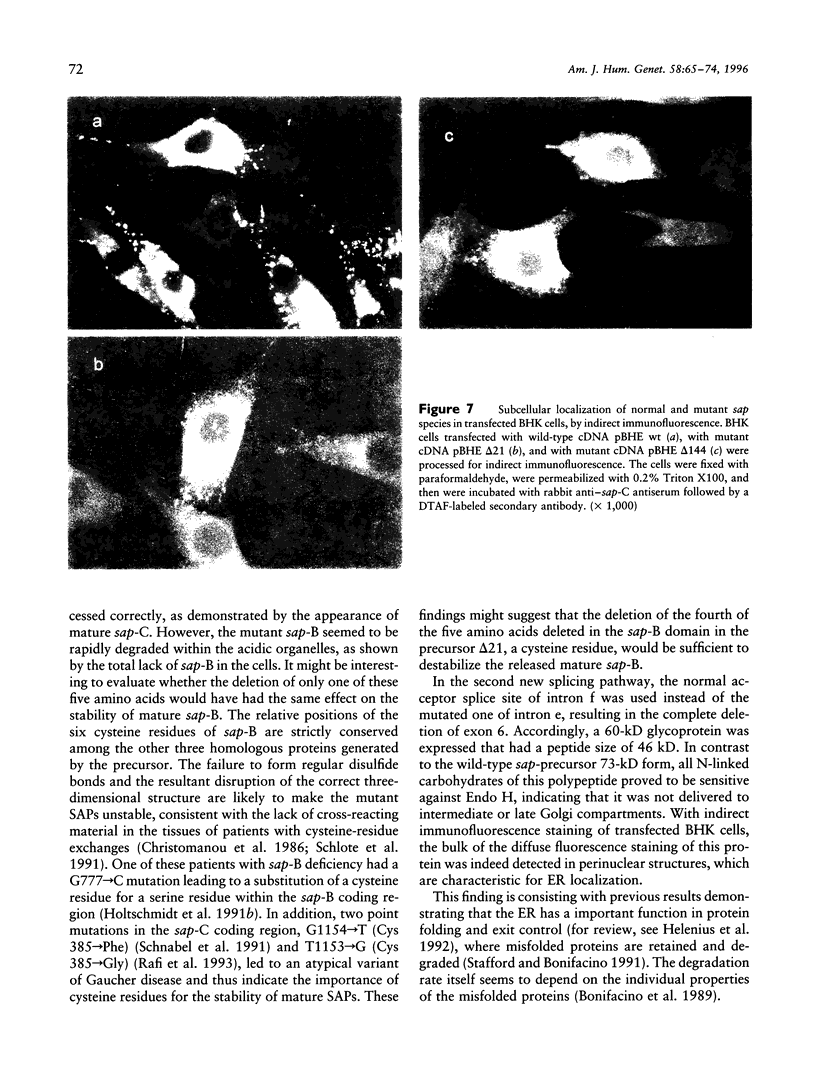
Sphingolipid activator proteins (SAPs) are small, nonenzymatic glycoproteins required for the lysosomal degradation of various sphingolipids with a short oligosaccharide chain by their exohydrolases. Four of the five known activator proteins (sap-A-sap-D), also called "saposins," are derived from a common precursor by proteolytic processing. sap-B stimulates hydrolysis of sulfatides by arylsulfatase A in vivo. Its recessively inherited deficiency results in a metabolic disorder similar to classical metachromatic leukodystrophy, which is caused by a defect of arylsulfatase A. Here we report on a patient with sap-B deficiency. Reverse-transcription-PCR studies on the patient's mRNA revealed the occurrence of two distinct mutant species: one with an in-frame deletion of the first 21 bases of exon 6, the other with a complete in-frame deletion of this exon. The patient was homozygous for the underlying mutation, which was found to be a G-->T transversion within the acceptor splice site between intron e and exon 6, abolishing normal RNA splicing. Allele-specific oligonucleotide hybridization revealed that the parents and both grandfathers of the patient were carriers of this mutation. In order to analyze the fate of the mutant precursor proteins, both abnormal cDNAs were stably expressed in baby hamster kidney cells. Pulse-chase experiments showed that the deletion of 21 bp had no effect on the transport and the maturation of the encoded precursor. All sap forms except sap-B were detectable by immunochemical methods. The cDNA bearing a complete deletion of exon 6 encoded a shortened precursor of only 60 kD, and no mature SAPs were detectable. The carbohydrate chains of this polypeptide were of the high-mannose and hybrid type, indicating no transport of the mutant precursor beyond early Golgi apparatus. An endoplasmic-reticulum localization of this polypeptide was supported by indirect immunofluorescence analysis.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Artelt P., Morelle C., Ausmeier M., Fitzek M., Hauser H. Vectors for efficient expression in mammalian fibroblastoid, myeloid and lymphoid cells via transfection or infection. Gene. 1988 Sep 7;68(2):213–219. doi: 10.1016/0378-1119(88)90023-6. [DOI] [PubMed] [Google Scholar]
- Christomanou H., Aignesberger A., Linke R. P. Immunochemical characterization of two activator proteins stimulating enzymic sphingomyelin degradation in vitro. Absence of one of them in a human Gaucher disease variant. Biol Chem Hoppe Seyler. 1986 Sep;367(9):879–890. doi: 10.1515/bchm3.1986.367.2.879. [DOI] [PubMed] [Google Scholar]
- Fujibayashi S., Wenger D. A. Biosynthesis of the sulfatide/GM1 activator protein (SAP-1) in control and mutant cultured skin fibroblasts. Biochim Biophys Acta. 1986 Feb 28;875(3):554–562. doi: 10.1016/0005-2760(86)90077-9. [DOI] [PubMed] [Google Scholar]
- Fürst W., Machleidt W., Sandhoff K. The precursor of sulfatide activator protein is processed to three different proteins. Biol Chem Hoppe Seyler. 1988 May;369(5):317–328. doi: 10.1515/bchm3.1988.369.1.317. [DOI] [PubMed] [Google Scholar]
- Fürst W., Sandhoff K. Activator proteins and topology of lysosomal sphingolipid catabolism. Biochim Biophys Acta. 1992 Jun 5;1126(1):1–16. doi: 10.1016/0005-2760(92)90210-m. [DOI] [PubMed] [Google Scholar]
- Helenius A., Marquardt T., Braakman I. The endoplasmic reticulum as a protein-folding compartment. Trends Cell Biol. 1992 Aug;2(8):227–231. doi: 10.1016/0962-8924(92)90309-b. [DOI] [PubMed] [Google Scholar]
- Holtschmidt H., Sandhoff K., Fürst W., Kwon H. Y., Schnabel D., Suzuki K. The organization of the gene for the human cerebroside sulfate activator protein. FEBS Lett. 1991 Mar 25;280(2):267–270. doi: 10.1016/0014-5793(91)80308-p. [DOI] [PubMed] [Google Scholar]
- Holtschmidt H., Sandhoff K., Kwon H. Y., Harzer K., Nakano T., Suzuki K. Sulfatide activator protein. Alternative splicing that generates three mRNAs and a newly found mutation responsible for a clinical disease. J Biol Chem. 1991 Apr 25;266(12):7556–7560. [PubMed] [Google Scholar]
- Klein A., Henseler M., Klein C., Suzuki K., Harzer K., Sandhoff K. Sphingolipid activator protein D (sap-D) stimulates the lysosomal degradation of ceramide in vivo. Biochem Biophys Res Commun. 1994 May 16;200(3):1440–1448. doi: 10.1006/bbrc.1994.1612. [DOI] [PubMed] [Google Scholar]
- Kretz K. A., Carson G. S., Morimoto S., Kishimoto Y., Fluharty A. L., O'Brien J. S. Characterization of a mutation in a family with saposin B deficiency: a glycosylation site defect. Proc Natl Acad Sci U S A. 1990 Apr;87(7):2541–2544. doi: 10.1073/pnas.87.7.2541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kudoh T., Wenger D. A. Diagnosis of metachromatic leukodystrophy, Krabbe disease, and Farber disease after uptake of fatty acid-labeled cerebroside sulfate into cultured skin fibroblasts. J Clin Invest. 1982 Jul;70(1):89–97. doi: 10.1172/JCI110607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morimoto S., Martin B. M., Yamamoto Y., Kretz K. A., O'Brien J. S., Kishimoto Y. Saposin A: second cerebrosidase activator protein. Proc Natl Acad Sci U S A. 1989 May;86(9):3389–3393. doi: 10.1073/pnas.86.9.3389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien J. S., Kretz K. A., Dewji N., Wenger D. A., Esch F., Fluharty A. L. Coding of two sphingolipid activator proteins (SAP-1 and SAP-2) by same genetic locus. Science. 1988 Aug 26;241(4869):1098–1101. doi: 10.1126/science.2842863. [DOI] [PubMed] [Google Scholar]
- Rafi M. A., Zhang X. L., DeGala G., Wenger D. A. Detection of a point mutation in sphingolipid activator protein-1 mRNA in patients with a variant form of metachromatic leukodystrophy. Biochem Biophys Res Commun. 1990 Jan 30;166(2):1017–1023. doi: 10.1016/0006-291x(90)90912-7. [DOI] [PubMed] [Google Scholar]
- Rafi M. A., de Gala G., Zhang X. L., Wenger D. A. Mutational analysis in a patient with a variant form of Gaucher disease caused by SAP-2 deficiency. Somat Cell Mol Genet. 1993 Jan;19(1):1–7. doi: 10.1007/BF01233949. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlote W., Harzer K., Christomanou H., Paton B. C., Kustermann-Kuhn B., Schmid B., Seeger J., Beudt U., Schuster I., Langenbeck U. Sphingolipid activator protein 1 deficiency in metachromatic leucodystrophy with normal arylsulphatase A activity. A clinical, morphological, biochemical, and immunological study. Eur J Pediatr. 1991 Jun;150(8):584–591. doi: 10.1007/BF02072213. [DOI] [PubMed] [Google Scholar]
- Schnabel D., Schröder M., Fürst W., Klein A., Hurwitz R., Zenk T., Weber J., Harzer K., Paton B. C., Poulos A. Simultaneous deficiency of sphingolipid activator proteins 1 and 2 is caused by a mutation in the initiation codon of their common gene. J Biol Chem. 1992 Feb 15;267(5):3312–3315. [PubMed] [Google Scholar]
- Schnabel D., Schröder M., Sandhoff K. Mutation in the sphingolipid activator protein 2 in a patient with a variant of Gaucher disease. FEBS Lett. 1991 Jun 17;284(1):57–59. doi: 10.1016/0014-5793(91)80760-z. [DOI] [PubMed] [Google Scholar]
- Stafford F. J., Bonifacino J. S. A permeabilized cell system identifies the endoplasmic reticulum as a site of protein degradation. J Cell Biol. 1991 Dec;115(5):1225–1236. doi: 10.1083/jcb.115.5.1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tayama M., Soeda S., Kishimoto Y., Martin B. M., Callahan J. W., Hiraiwa M., O'Brien J. S. Effect of saposins on acid sphingomyelinase. Biochem J. 1993 Mar 1;290(Pt 2):401–404. doi: 10.1042/bj2900401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vara J. A., Portela A., Ortín J., Jiménez A. Expression in mammalian cells of a gene from Streptomyces alboniger conferring puromycin resistance. Nucleic Acids Res. 1986 Jun 11;14(11):4617–4624. doi: 10.1093/nar/14.11.4617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wirth M., Bode J., Zettlmeissl G., Hauser H. Isolation of overproducing recombinant mammalian cell lines by a fast and simple selection procedure. Gene. 1988 Dec 20;73(2):419–426. doi: 10.1016/0378-1119(88)90506-9. [DOI] [PubMed] [Google Scholar]
- Zhang X. L., Rafi M. A., DeGala G., Wenger D. A. Insertion in the mRNA of a metachromatic leukodystrophy patient with sphingolipid activator protein-1 deficiency. Proc Natl Acad Sci U S A. 1990 Feb;87(4):1426–1430. doi: 10.1073/pnas.87.4.1426. [DOI] [PMC free article] [PubMed] [Google Scholar]











