Abstract
Mucosal tissues are major sites of HIV entry and initial infection. Thus, the induction of a mucosal cytotoxic T lymphocyte (CTL) response is an important feature for an effective HIV vaccine. However, little is known about approaches to induce such a protective CTL response in the mucosa. Here for the first time we show that intrarectal immunization with a synthetic, multideterminant HIV peptide plus cholera toxin adjuvant induced long-lasting, antigen-specific CTL memory in both the inductive (Peyer’s patch) and effector (lamina propria) mucosal sites, as well as in systemic sites (spleen), whereas systemic immunization induced specific CTL only in the spleen. Cholera toxin adjuvant, while enhancing the response, was not essential. The CTL recognized target cells either pulsed with HIV peptide or expressing endogenous whole envelope glycoprotein of Mr 160,000 (gp160). Exploring the requirements for CTL induction, we show that mucosal CTL responses are both interleukin 12 and interferon-γ dependent by using antibody-treated and knock-out mice. Finally, to determine whether a mucosal response is actually protective against local mucosal challenge with virus, we show that intrarectal immunization with the synthetic HIV peptide vaccine protected mice against infection via mucosal challenge with a recombinant vaccinia virus expressing HIV-1IIIB gp160. These studies provide an approach to development of an HIV vaccine that induces CTL immunity in the mucosal and systemic immune systems and protects against mucosal infection with a virus expressing HIV-1 gp160.
Keywords: mucosal immune system
Recent studies have established that HIV-1 can enter the body via mucosal surfaces even if the latter are completely intact (1–3). This strongly implies that HIV-specific immune elements at mucosal sites will be critical for the initial (and timely) control of infection in many or most individuals exposed to the virus. Studies of candidate HIV vaccines that are designed to elicit mucosal immune responses have so far focused mainly on HIV-induced B cell (IgA) and helper T cell responses (4–6) or cytotoxic T lymphocytes (CTL) in the spleen (SP) (7). There is only limited information on mucosal vaccine induction of CTL responses in mucosal tissues themselves, by using either live viruses (8) or targeted lymph node immunization (9, 10). The development of vaccines capable of inducing CTL in mucosal sites, however, is becoming more critical with the emergence of recent evidence that natural genital infection with HIV-1 leads to the development of major histocompatibility complex (MHC) class I-restricted cytotoxic T cells in the cervix (11) and that mucosal administration of live attentuated simian immunodeficiency virus (SIV)mac virus, which protects against subsequent infection with virulent virus, elicits a CTL response in gut-associated lymph nodes (12). However, little is known about approaches to induce a CTL response in the mucosa with a nonliving vaccine, or about the efficacy of such CTL in protection against mucosal challenge. In the present work we address both these issues with studies of mucosal CTL responses to a prototype synthetic HIV peptide vaccine that contains only antigenic sites that induce protective immunity or help for such protective immunity, and that exclude sites inducing potentially harmful immune responses (13–19). We show both induction of CTL in inductive and effector sites of the mucosal immune system and induction of protective immunity against mucosal challenge with virus that cannot be explained by neutralizing antibodies.
MATERIALS AND METHODS
Mice.
Female BALB/c mice were purchased from Frederick Cancer Research Center (Frederick, MD). Interferon-γ−/− (IFN-γ−/−) mice were purchased from The Jackson Laboratory and were maintained in a specific pathogen-free microisolator environment. Mice used in this study were 6–12 weeks old.
Immunization.
Mice were immunized with four doses of the synthetic HIV peptide PCLUS3–18IIIB (50 μg per mouse for each immunization) (14–18) on days 0, 7, 14, and 21 in combination with cholera toxin (CT) (10 μg per mouse) or without CT by intrarectal (IR) administration, intranasally or intragastrically. For subcutaneous immunization, incomplete Freund’s adjuvant was used.
Cell Purification.
Five weeks to 6 months after the first dose, antigen-specific T cells were isolated from Peyer’s patch (PP), lamina propria (LP), and SP. The PP were carefully excised from the intestinal wall and dissociated into single cells by use of the collagenase type VIII, 300 units/ml (Sigma) as described (20). Our data showed that most PP CD3+ T cells isolated from normal mice were CD4+, while CD3+CD8+ T cells were less frequent. Further, collagenase did not alter expression of CD3, CD4, or CD8 on splenic T cells treated with this enzyme. LP lymphocyte isolation was performed as described (20). The large and small intestines were dissected from individual mice and the mesenteric and connective tissues carefully removed. Fecal material was flushed from the lumen with unsupplemented medium (RPMI 1640 medium). After the PP were identified and removed from the intestinal wall, the intestines were opened longitudinally, cut into short segments, and washed extensively in RPMI 1640 medium containing 2% fetal bovine serum. To remove the epithelial cell layer, tissues were placed into 100 ml of 1 mM EDTA and incubated twice (first for 40 min and then for 20 min) at 37°C with stirring. After the EDTA treatment, tissues were washed in complete RPMI 1640 medium for 10 min at room temperature and then placed into 50 ml of RPMI 1640 medium containing 10% FCS and incubated for 15 min at 37°C with stirring. The tissues and medium were transferred to a 50 ml tube and shaken vigorously for 15 sec, and then the medium containing epithelial cells was removed. This mechanical removal of cells was repeated twice more, by using fresh medium each time, to completely remove the epithelial cell layer. Histologic examination revealed that the structure of the villi and LP were preserved. To isolate LP lymphocytes, tissues were cut into small pieces and incubated in RPMI 1640 medium containing collagenase type VIII, 300 units/ml (Sigma) for 50 min at 37°C with stirring. Supernatants containing cells were collected, washed and then resuspended in complete RPMI 1640 medium. This collagenase dissociation procedure was repeated two times and the isolated cells pooled and washed again. Cells were passed through a cotton-glass wool column to remove dead cells and tissue debris and then layered onto a discontinuous gradient containing 75% and 40% Percoll (Pharmacia). After centrifugation (4°C, 600 × g, 20 min), the interface layer between the 75% and 40% Percoll was carefully removed and washed with incomplete medium. This procedure provided >90% viable lymphocytes with a cell yield of 1.5–2 × 106 lymphocytes per mouse. SP were aseptically removed and single cell suspensions prepared by gently teasing them through sterile screens. The erythrocytes were lysed in Tris-buffered ammonium chloride and the remaining cells washed extensively in RPMI 1640 medium containing 2% fetal bovine serum.
CTL Assay.
Immune cells from SP, PP, and LP were cultured at 5 × 106/ml in 24-well culture plates in complete T cell medium: RPMI 1640 medium containing 10% fetal bovine serum, 2 mM L-glutamine, penicillin (100 units/ml), streptomycin (100 μg/ml), and 5 × 10−5 M 2-mercaptoethanol. After 3 days we added 10% Con A supernatant-containing medium as a source of interleukin 2 (IL-2). LP lymphocytes were studied after 7 days stimulation with 1 μM P18IIIB-I10 peptide together with 4 × 106 of 3,300-rad irradiated syngeneic SP cells. SP and PP cells were stimulated in vitro similarly for two 7-day culture periods before assay. Cytolytic activity of CTL lines was measured by a 4-hr assay with 51Cr labeled targets. Two different cell lines were used as target cells: (i) 15–12 cells (21) (BALB/c 3T3 fibroblasts transfected with HIV-1IIIB envelope glycoprotein of Mr 160,000 (gp160) and endogenously expressing HIV gp160; these were compared with 18 Neo BALB/c 3T3 fibroblasts transfected with NeoR alone as a control. (ii) P815 targets were tested in the presence or absence of I10 peptide (1 μM). For testing the peptide specificity of CTL, 51Cr-labeled P815 targets were pulsed for 2 hr with peptide at the beginning of the assay. The percent specific 51Cr release was calculated as 100 × (experimental release − spontaneous release)/(maximum release − spontaneous release). Maximum release was determined from supernatants of cells that were lysed by addition of 5% Triton-X 100. Spontaneous release was determined from target cells incubated without added effector cells.
Study of the Virus Titer in the Ovary.
On day 35, mice were challenged intrarectally with 2.5 × 107 plaque-forming units (pfu) of vaccinia virus expressing gp160IIIB (vPE16). Six days after the challenge with recombinant vaccinia virus expressing HIV-gp160, the mice were killed and ovaries were removed, homogenized, sonicated, and assayed for vPE16 titer by plating serial 10-fold dilutions on a plate of BSC-2 indicator cells, staining with crystal violet and counting plaques at each dilution. The minimal detectable level of virus was 100 pfu.
RESULTS
Peptide constructs composed of multideterminant T helper (Th) peptides from the envelope glycoprotein of HIV were colinearly synthesized with peptide 18 (P18IIIB) (RIQRGPGRAFVTIGK) of the V3 loop of HIV-1 gp160, corresponding to the principal neutralizing determinant of the HIV1-IIIB strain and a CTL epitope in several MHC types (16, 21, 22). The minimal CTL epitope was mapped to the 10-mer peptide I10 (RGPGRAFVTI) (22–24). The segments containing clusters of overlapping Th epitopes were designated cluster peptides PCLUS3 and PCLUS6.1 (25). We have previously found these vaccine constructs to elicit high titers of neutralizing antibodies, CTL, and Th cells in mice of several MHC types (14–16). A similar vaccine in which the P18 CTL epitope portion is replaced with the homologous segment of the MN strain of HIV is currently in phase I clinical trials for systemic immunization (26).
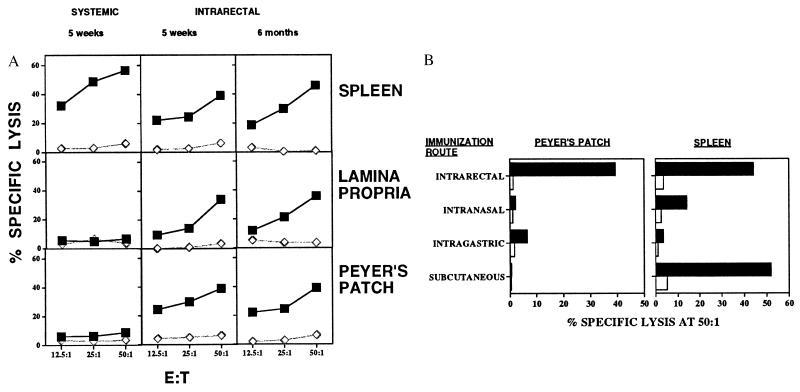
To determine whether these cluster peptide HIV vaccine constructs would elicit mucosal CTL, we immunized BALB/c mice intrarectally with peptide in the presence or absence of CT as a mucosal adjuvant on days 0, 7, 14, and 21. Mice were studied either 2 weeks later (day 35) or at 6 months for memory CTL in the SP, PP, or LP (tissues isolated as described in ref. 20). We found that IR synthetic HIV peptide vaccine PCLUS3-P18IIIB with CT (Fig. 1A) can induce P18IIIB-I10 specific CTL memory in PP and LP of the intestine as well as the SP. In contrast, systemic administration (subcutaneously) of peptide vaccine produced HIV peptide specific CTL only in the SP. Thus, responses were asymmetric. The systemic and mucosal CTL memory after mucosal administration with synthetic HIV peptide vaccine in combination with CT was long-lasting. We found significant PP, LP, and SP CTL memory 6 months after IR immunization with PCLUS 3–18IIIB (Fig. 1A).
Figure 1.
(A) IR HIV peptide immunization induces both mucosal and systemic long-lasting immunity. In contrast, systemic immunization induces only systemic CTL. Closed squares show killing of P18IIIB-I10-pulsed targets and open diamonds show killing on unpulsed targets. (B) Induction of the mucosal and systemic CTL responses by different routes of immunization with synthetic peptide HIV vaccine. Killing of peptide-pulsed targets (▪) is compared with killing of unpulsed targets (□) at an effector-to-target ratio of 50:1. Similar results were obtained at ratios of 25 and 12.5:1. In both A and B, SEM of triplicate cultures were all<5% of the mean.
P18IIIB CTL epitope peptide alone without a Th epitope in combination with CT was not able to induce either a mucosal or systemic CTL response (data not shown). Further, the IR route of immunization with HIV vaccine peptide was optimal for induction of the mucosal CTL responses in the gut: neither intranasal nor intragastric routes of immunizations were as effective for inducing CTL in the PP, LP (not shown), or even SP (Fig. 1B).
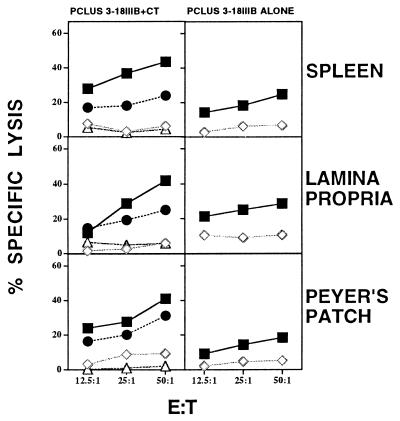
CT causes unacceptable diarrhea and other complications in humans, and thus it cannot be used as an adjuvant for a human vaccine as it can be in mice. Therefore, we asked whether the peptide vaccine would induce CTL after IR administration without any adjuvant. We found that IR administration of HIV peptide vaccine alone, without mucosal adjuvant, can induce significant CTL memory in both inductive (PP) and effector (LP) sites of mucosal immune systems, although not at quite as high a level as was obtained when CT was included (Fig. 2).
Figure 2.
CT adjuvant is not essential for IR peptide vaccine induction of CTL. Mucosal peptide-induced CTL lyse targets endogenously expressing HIV gp160 envelope protein. Two different cell lines were used as a target cells: (i) 15–12 cells (BALB/c 3T3 fibroblasts transfected with HIV-1IIIB gp160 and endogenously expressing HIV gp160) (•, Left). These were compared with 18 Neo BALB/c 3T3 fibroblasts transfected with NeoR alone as a control (▵, Left). (ii) P815 targets were tested in the presence (▪) or absence (◊) of I10 peptide (1 μM). SEM of triplicate cultures were all <5% of the mean.
Because the P18lllB-I10 peptide is being recognized in the above studies on peptide-pulsed P815 target cells that do not express MHC class II molecules, the data obtained imply that PP, LP, and SP CTL recognize exogenously-acquired peptide in the context of MHC class I molecules. To determine whether the CTL induced would also recognize endogenously-acquired peptide, as would be the case for HIV-1 gp160 expressed in an infected cell, we tested whether the CTL induced by PCLUS3–18IIIB in mucosal sites would kill cell line 15–12 targets (BALB/c 3T3 fibroblasts, which express only MHC class I, transfected with HIV-1IIIB gp160 (21)). As shown in Fig. 2, we found that the killing of such target cells was nearly as great as the killing of the peptide pulsed P815 target cells, indicating that the CTL do in fact recognize and lyse target cells with endogenously acquired specific antigen. Parenthetically, by adding anti-CD8 mAb to cultures, we also established that the CTL were CD8+ T cells (data not shown).
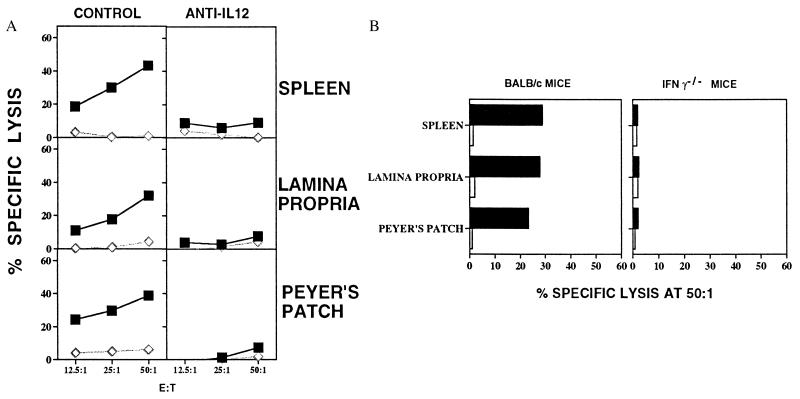
To determine the cytokine requirements for the induction of mucosal CTL, we first treated mice i.p. one day before and after each immunization with 0.5 mg monoclonal anti-IL-12 antibody (clone C17.8, a kind gift of G. Trinchieri, Wistar Institute, Philadelphia). Such treatment led to an almost total inhibition of the generation of CTL memory at both mucosal and splenic sites (Fig. 3A), implying that CTL induction was IL-12 dependent. Because of the interdependence of IL-12 and IFN-γ (17, 27, 28), we also tested the dependence of CTL induction on IFN-γ, by using knock-out mice. BALB/c IFN-γ knock-out (IFN-γ−/−) mice (29–32) were immunized intrarectally with synthetic HIV peptide vaccine (Fig. 3B). In contrast to the control wild type mice, the IFN-γ−/− mice produced no detectable memory CTL in the SP, PP, or LP. Thus, the mucosal CTL response is dependent on IFN-γ as well as on IL-12, and thus probably on an intact Th1 pathway. However, at least two explanations remain possible. On the one hand, IL-12 is necessary for the induction of Th1 helper cells that make IFN-γ, and the induction of CTL may be dependent on this form of help. On the other hand, IFN-γ induces the expression of the β2 subunit of the IL-12 receptor (33), and so may be necessary for the response to IL-12. Therefore, these two cytokines are interdependent, and further work will be necessary to determine which of these cytokines is most directly involved in mucosal CTL induction.
Figure 3.
(A) IR peptide induction of CTL is IL-12 dependent. One day before and one day after immunization with peptide mice were treated i.p. with anti-IL12 antibody (0.5 mg per injection; 4 mg per mouse total dose (Right) or were untreated (Left). For testing the peptide specificity of CTL, 51Cr-labeled P815 targets were pulsed with peptide at the beginning of the assay (▪) or without peptide (◊). (B) IR peptide induction of CTL is IFN-γ dependent. Wild-type BALB/c mice (Left) or IFN-γ−/− (Right) were immunized as in A, and tested at 5 weeks. Killing of peptide-pulsed targets (▪) is compared with killing of unpulsed targets (░⃞) at an effector-to-target ratio of 50:1. Similar results were obtained at ratios of 25 and 12.5:1. (A and B) SEM of triplicate cultures were all <5% of the mean.
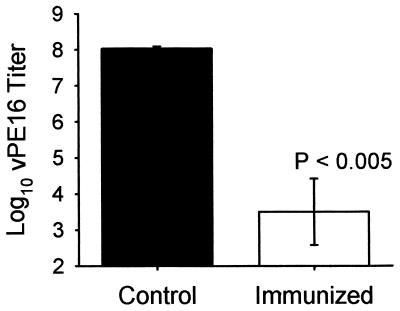
Finally, we determined the ability of the mucosal immune responses induced by the cluster HIV peptide vaccine to protect against virus challenge via a mucosal route. We challenged the IR immunized mice, on day 35 after the start of immunization, by IR infusion with vaccinia virus expressing HIV-1IIIB gp160 (vPE16). Six days after the challenge, mice were sacrificed and tissues were removed and assayed for vPE16 titer. Although very low levels of vaccinia were sometimes detected in the colon and the SP on day 2 or 6 after IR challenge in preliminary studies (data not shown), most of the virus was consistently found in the ovaries, because this particular vaccinia virus has been found to replicate best in the ovaries (34). IR immunization with the synthetic HIV peptide vaccine protected mice against an IR challenge with this gp160-recombinant vaccinia virus, as shown by a 4.5-log reduction in viral pfu in ovaries of immunized mice compared with those in unimmunized animals (P < 0.005) (Fig. 4).
Figure 4.
IR peptide immunization protects against IR challenge with HIV-gp160 expressing recombinant vaccinia virus. The left bar (▪) shows virus as the log of the number of pfus in ovaries of unimmunized mice and the right bar (□) shows virus titer in the ovaries of immunized mice. Bars = SEM of five mice per group. The difference is significant at P < 0.005 by Student’s t test.
DISCUSSION
HIV prevention by vaccine administration is based on the potential ability of the vaccine-induced immune response to limit the initial proliferative burst of the virus and to eradicate virus or virus-infected cells prior to viral dissemination. The HIV peptide vaccine studied here begins to fulfill these objectives. (i) IR peptide vaccine administration results in substantial CTL memory cells in both mucosal and systemic sites. CTL responses have been correlated with initial limitation of natural viral infection (35–37) and could thus be effective in preventing infection if in place at the time of viral entry. (ii) In that the CTL responses elicited were mediated by CD8+ T cells, the protection against HIV infection afforded by such responses could be multifaceted and involve, not only cytolysis of infected cells, but also elaboration of antigen-nonspecific factors (38, 39) and/or chemokines (40–44) that have been shown to limit HIV infection. (iii) The response was shown to actually limit infection by a recombinant vaccinia virus expressing gp160. The IR peptide immunization also induced V3-loop specific IgG and IgA antibody in fecal washes and specific IgG in the serum (data not shown). However, interestingly, because the gp160 is expressed by the vPE16 vaccinia virus only after it infects a cell, but not as part of the virion, the protection effected by the vaccine must be due to killing of virally infected cells expressing gp160 epitopes and cannot be due to neutralization of virus by antibodies. Thus, although other antibody-mediated mechanisms cannot be excluded, such as lysis of infected cells by complement or by antibody-dependent cellular cytotoxicity, it is likely that the mechanism of HIV peptide vaccine efficacy was via the induction of the CTL detected rather than via the induction of neutralizing antibodies.
Previous studies have shown that a synthetic HIV peptide very similar to that utilized here and administered intranasally to mice (also with CT) elicted a serum and vaginal anti-HIV antibody response, as well as a helper T cell response characterized by proliferation and delayed type hypersensitivity (5). In addition, an HIV peptide containing only the CTL epitope given intranasally evoked CTL responses in the SP, but CTL in mucosal tissues were not examined (7). In complementary studies of CTL induction in relation to SIV, it was shown that protection against SIV (or SHIV) infection in macaques could be achieved with rectal administration of attenuated SIV (12) or by “targeted lymph node” immunization of iliac nodes with a virus-like particle (10) or subunit SIV envelope/core vaccine (9) and that in both instances the immune response was characterized, at least in part, by the appearance of virus-specific CTL. Likewise, a recombinant live adenovirus induced mucosal CTL (8). The present studies showing the induction by a peptide vaccine of a protective immune response associated with mucosal memory CTL, however, is the first evidence that protective mucosal CTL can be elicited with a nonliving soluble antigen vaccine delivered directly to the mucosa itself. The ability of the peptide vaccine used here to induce this response may be due to the fact that the peptide contains epitopes in the same molecule that address both helper and effector cells. Thus, the relevant antigen-presenting cells interact with responding T cells in the same manner as would presenting cells processing real virus. The ability to elicit T cell immunity with a designed nonliving antigen is important because it has been shown that whole HIV can potentially induce antibodies that actually enhance viral entry into cells (45–47).
The CTL response elicited by the peptide vaccine in these studies was both IL-12 and IFN-γ dependent because it was abrogated by administration of anti-IL-12 or in mice deficient in IFN-γ production. Because IL-12 contributes to the induction of Th1 type helper T cells that make IFN-γ, these results are consistent with previous studies showing that CTL induction is facilitated by Th1 cells, although a mechanism involving IFN-γ induction of IL-12 receptors (33) remains a possibility. The fact that the CTL response obtained was enhanced by CT, an adjuvant thought to favor Th2 responses (which suppress IL-12 production) is somewhat paradoxical and suggests that, in this context at least, CT has a more complex activity. In any case, the effects obtained with anti-IL-12 strongly suggest that a mature mucosal vaccine should encompass the concomitant exogenous administration of IL-12 or substances that stimulate endogenous IL-12 production.
Thus, this study makes the novel observation that a synthetic multideterminant HIV peptide vaccine can induce an antigen-specific protective CTL response in the mucosal immune system. Although an ultimate HIV vaccine probably must incorporate more epitopes and may be made as a recombinant construct rather than a synthetic peptide, the results and approaches of this study can potentially lead to the development of a new type of protective HIV vaccine in humans.
Acknowledgments
We thank William Biddison and Jonathan Yewdell for critical reading of the manuscript and helpful suggestions.
ABBREVIATIONS
- CTL
cytotoxic T lymphocytes
- IR
intrarectal
- IL-2 and IL-12
interleukin 2 and 12
- IFN-γ
interferon-γ
- SP
spleen
- CT
cholera toxin
- PP
Peyer’s patch
- LP
lamina propria
- Th
T helper
- pfu
plaque-forming unit
- SIV
simian immunodeficiency virus
- MHC
major histocompatibility complex
Note Added in Proof
Recent results that support the conclusions of the paper are: (i) that the protection is specific in that no protection was observed against a control vaccinia virus expressing an unrelated protein antigen; (ii) that mucosal immunization is significantly more effective than s.c. immunization in protecting against a mucosal challenge; and (iii) that the protection could be prevented by treating the mice with anti-CD8 antibodies, confirming that the protection was dependent on CD8-positive cells. This result confirms the ability of CD8-positive CTL to protect against mucosal challenge with virus. These results will be reported more fully elsewhere.
References
- 1.Neutra M R, Pringault E, Kraehenbuhl J-P. Annu Rev Immunol. 1996;14:275–300. doi: 10.1146/annurev.immunol.14.1.275. [DOI] [PubMed] [Google Scholar]
- 2.Miller C J, Alexander N J, Sutjipto S, Lackner A A, Gettie A, Hendrickx A G, Lowenstine L J, Jennings M, Marx P A. J Virol. 1989;63:4277–4284. doi: 10.1128/jvi.63.10.4277-4284.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bomsel M. Nat Med. 1997;3:42–47. doi: 10.1038/nm0197-42. [DOI] [PubMed] [Google Scholar]
- 4.Lehner T, Bergmeier L A, Panagiotidi C, Tao L, Brookes R, Klavinskis L S, Walker P, Walker J, Ward R G, et al. Science. 1992;258:1365–1369. doi: 10.1126/science.1360702. [DOI] [PubMed] [Google Scholar]
- 5.Staats H F, Nichols W G, Palker T J. J Immunol. 1996;157:462–472. [PubMed] [Google Scholar]
- 6.Kozlowski P A, Cu-Uvin S, Neutra M R, Flanigan T P. Infect Immun. 1997;65:1387–1394. doi: 10.1128/iai.65.4.1387-1394.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Porgador A, Staats H F, Faiola B, Gilboa E, Palker T J. J Immunol. 1997;158:834–841. [PubMed] [Google Scholar]
- 8.Gallichan W S, Rosenthal K L. J Exp Med. 1996;184:1879–1890. doi: 10.1084/jem.184.5.1879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lehner T, Wang Y, Cranage M, Bergmeier L A, Mitchell E, Tao L, Hall G, Dennis M, Cook N, Brookes R, et al. Nat Med. 1996;2:767–775. doi: 10.1038/nm0796-767. [DOI] [PubMed] [Google Scholar]
- 10.Klavinskis L S, Bergmeier L A, Gao L, Mitchell E, Ward R G, Layton G, Brookes R, Meyers N J, Lehner T. J Immunol. 1996;157:2521–2527. [PubMed] [Google Scholar]
- 11.Musey L, Hu Y, Eckert L, Christensen M, Karchmer T, McElrath M J. J Exp Med. 1997;185:293–303. doi: 10.1084/jem.185.2.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cranage M P, Whatmore A M, Sharpe S A, Cook N, Polyanskaya N, Leech S, Smith J D, Rud E W, Dennis M J, Hall G A. Virology. 1997;229:143–154. doi: 10.1006/viro.1996.8419. [DOI] [PubMed] [Google Scholar]
- 13.Takahashi H, Nakagawa Y, Pendleton C D, Houghten R A, Yokomuro K, Germain R N, Berzofsky J A. Science. 1992;255:333–336. doi: 10.1126/science.1372448. [DOI] [PubMed] [Google Scholar]
- 14.Ahlers J D, Pendleton C D, Dunlop N, Minassian A, Nara P L, Berzofsky J A. J Immunol. 1993;150:5647–5665. [PubMed] [Google Scholar]
- 15.Shirai M, Pendleton C D, Ahlers J, Takeshita T, Newman M, Berzofsky J A. J Immunol. 1994;152:549–556. [PubMed] [Google Scholar]
- 16.Ahlers J D, Dunlop N, Pendleton C D, Newman M, Nara P L, Berzofsky J A. AIDS Res Hum Retroviruses. 1996;12:259–272. doi: 10.1089/aid.1996.12.259. [DOI] [PubMed] [Google Scholar]
- 17.Ahlers J D, Dunlop N, Alling D W, Nara P L, Berzofsky J A. J Immunol. 1997;158:3947–3958. [PubMed] [Google Scholar]
- 18.Ahlers J D, Takeshita T, Pendleton C D, Berzofsky J A. Proc Natl Acad Sci USA. 1997;94:10856–10861. doi: 10.1073/pnas.94.20.10856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Berzofsky, J. A. & Berkower, I. J. (1995) AIDS9 (Suppl. A), S143–S157. [PubMed]
- 20.Mega J, Bruce M G, Beagley K W, McGhee J R, Taguchi T, Pitts A M, McGhee M L, Bucy R P, Eldridge J H, Mestecky J, et al. Int Immunol. 1991;3:793–805. doi: 10.1093/intimm/3.8.793. [DOI] [PubMed] [Google Scholar]
- 21.Takahashi H, Cohen J, Hosmalin A, Cease K B, Houghten R, Cornette J, DeLisi C, Moss B, Germain R N, Berzofsky J A. Proc Natl Acad Sci USA. 1988;85:3105–3109. doi: 10.1073/pnas.85.9.3105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Shirai M, Pendleton C D, Berzofsky J A. J Immunol. 1992;148:1657–1667. [PubMed] [Google Scholar]
- 23.Kozlowski S, Corr M, Takeshita T, Boyd L F, Pendleton C D, Germain R N, Berzofsky J A, Margulies D H. J Exp Med. 1992;175:1417–1422. doi: 10.1084/jem.175.6.1417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Takeshita T, Takahashi H, Kozlowski S, Ahlers J D, Pendleton C D, Moore R L, Nakagawa Y, Yokomuro K, Fox B S, Margulies D H, Berzofsky J A. J Immunol. 1995;154:1973–1986. [PubMed] [Google Scholar]
- 25.Berzofsky J A, Pendleton C D, Clerici M, Ahlers J, Lucey D R, Putney S D, Shearer G M. J Clin Invest. 1991;88:876–884. doi: 10.1172/JCI115389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Humphrey, R. W., Pinto, L. A., Fowke, K., Merced-Galindez, F., Ahlers, J. D., Little, R., Dunlop, N., Pendleton, C. D., Kohler, D., Nara, P., et al. (1998) J. Hum. Virol, in press.
- 27.McKnight A J, Zimmer G J, Fogelman I, Wolf S F, Abbas A K. J Immunol. 1994;152:2171–2179. [PubMed] [Google Scholar]
- 28.Bliss J, Van Cleave V, Murray K, Wiencis A, Ketchum M, Maylor R, Haire T, Resmini C, Abbas A K, Wolf S F. J Immunol. 1996;156:887–894. [PubMed] [Google Scholar]
- 29.Dalton D K, Pitts-Meek S, Keshav S, Figari I S, Bradley A, Stewart T A. Science. 1993;259:1739–1742. doi: 10.1126/science.8456300. [DOI] [PubMed] [Google Scholar]
- 30.Huang S, Hendriks W, Althage A, Hemmi S, Bluethmann H, Kamijo R, Vilcek J, Zinkernagel R M, Aguet M. Science. 1993;259:1742–1745. doi: 10.1126/science.8456301. [DOI] [PubMed] [Google Scholar]
- 31.van den Broek M F, Müller U, Huang S, Aguet M, Zinkernagel R M. J Virol. 1995;69:4792–4796. doi: 10.1128/jvi.69.8.4792-4796.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tishon A, Lewicki H, Rall G, von Herrath M, Oldstone M B A. Virology. 1995;212:244–250. doi: 10.1006/viro.1995.1477. [DOI] [PubMed] [Google Scholar]
- 33.Szabo S J, Dighe A S, Gubler U, Murphy K M. J Exp Med. 1997;185:817–824. doi: 10.1084/jem.185.5.817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Alexander-Miller M A, Leggatt G R, Berzofsky J A. Proc Natl Acad Sci USA. 1996;93:4102–4107. doi: 10.1073/pnas.93.9.4102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Koup R A, Ho D D. Nature (London) 1994;370:416. doi: 10.1038/370416a0. [DOI] [PubMed] [Google Scholar]
- 36.Koup R A, Safrit J T, Cao Y, Andrews C A, McLeod G, Borkowsky W, Farthing C, Ho D D. J Virol. 1994;68:4650–4655. doi: 10.1128/jvi.68.7.4650-4655.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Reimann K A, Tenner-Racz K, Racz P, Montefiori D C, Yasutomi Y, Lin W, Ransil B J, Letvin N L. J Virol. 1994;68:2362–2370. doi: 10.1128/jvi.68.4.2362-2370.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Walker C M, Moody D J, Stites D P, Levy J A. Science. 1986;234:1563–1566. doi: 10.1126/science.2431484. [DOI] [PubMed] [Google Scholar]
- 39.Levy J A. Microbiol Rev. 1993;57:183–289. doi: 10.1128/mr.57.1.183-289.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cocchi F, DeVico A L, Garzino-Demo A, Arya S K, Gallo R C, Lusso P. Science. 1995;270:1811–1815. doi: 10.1126/science.270.5243.1811. [DOI] [PubMed] [Google Scholar]
- 41.Feng Y, Broder C C, Kennedy P E, Berger E A. Science. 1996;272:872–877. [Google Scholar]
- 42.Alkhatib G, Combadiere C, Broder C C, Feng Y, Kennedy P E, Murphy P M, Berger E A. Science. 1996;272:1955–1958. doi: 10.1126/science.272.5270.1955. [DOI] [PubMed] [Google Scholar]
- 43.Deng H, Liu R, Ellmeier W, Choe S, Unutmaz D, Burkhart M, Di Marzio P, Marmon S, Sutton R, Hill C M, et al. Nature (London) 1996;381:661–666. doi: 10.1038/381661a0. [DOI] [PubMed] [Google Scholar]
- 44.Dragic T, Litwin V, Allaway G P, Martin S R, Huang Y, Nagashima K A, Cayanan C, Maddon P J, Koup R A, Moore J P, et al. Nature (London) 1996;381:667–673. doi: 10.1038/381667a0. [DOI] [PubMed] [Google Scholar]
- 45.Robinson W E, Jr, Montefiori D C, Mitchell W M, Prince A M, Alter H J, Dreesman G R, Eichberg J W. Proc Natl Acad Sci USA. 1989;86:4710–4714. doi: 10.1073/pnas.86.12.4710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Robinson W E, Jr, Montefiori D C, Mitchell W M. Lancet. 1988;1988:790–794. doi: 10.1016/s0140-6736(88)91657-1. [DOI] [PubMed] [Google Scholar]
- 47.Robinson W E, Jr, Kawamura T, Gorny M K, Lake D, Xu J-Y, Matsumoto Y, Sugano T, Masuho Y, Mitchell W M, Hersh E, et al. Proc Natl Acad Sci USA. 1990;87:3185–3189. doi: 10.1073/pnas.87.8.3185. [DOI] [PMC free article] [PubMed] [Google Scholar]