Abstract
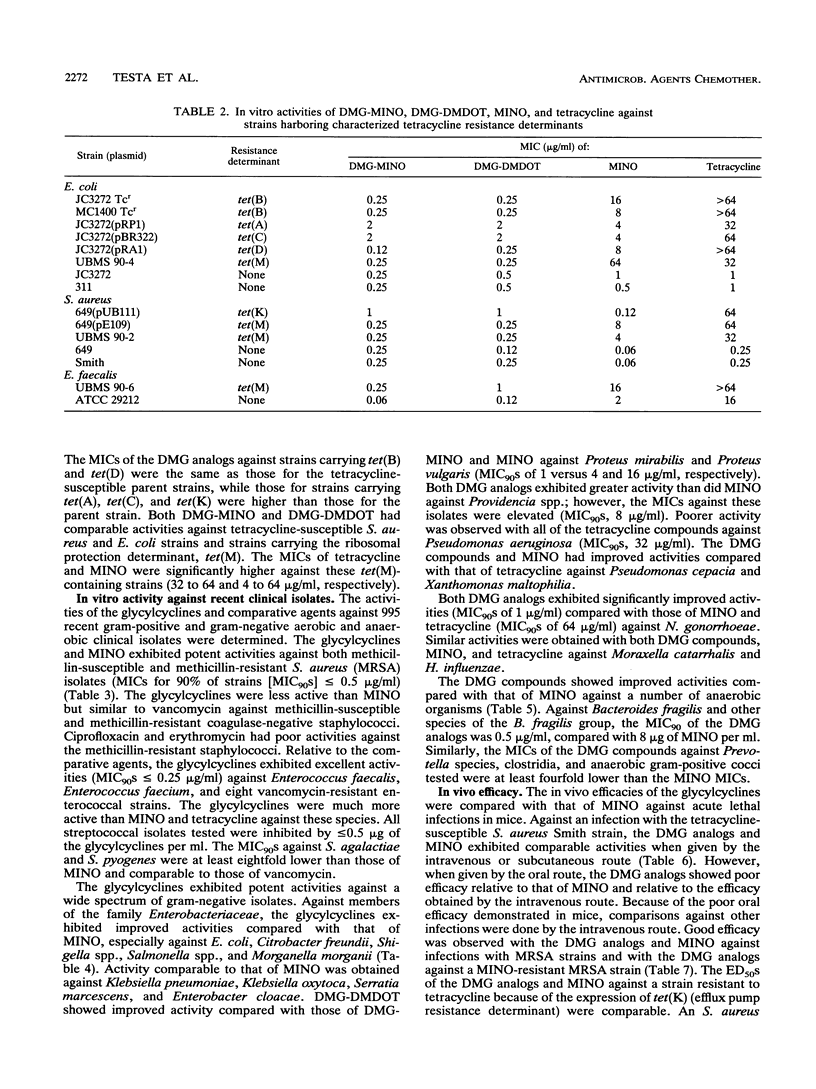
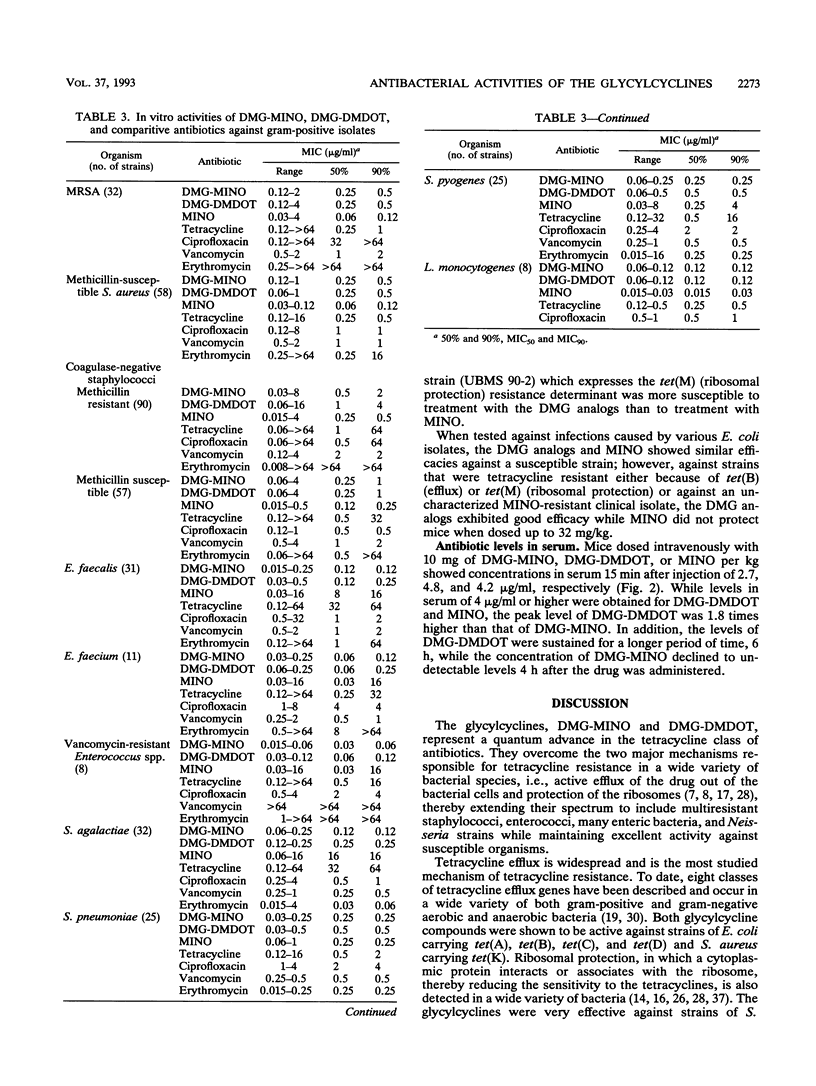
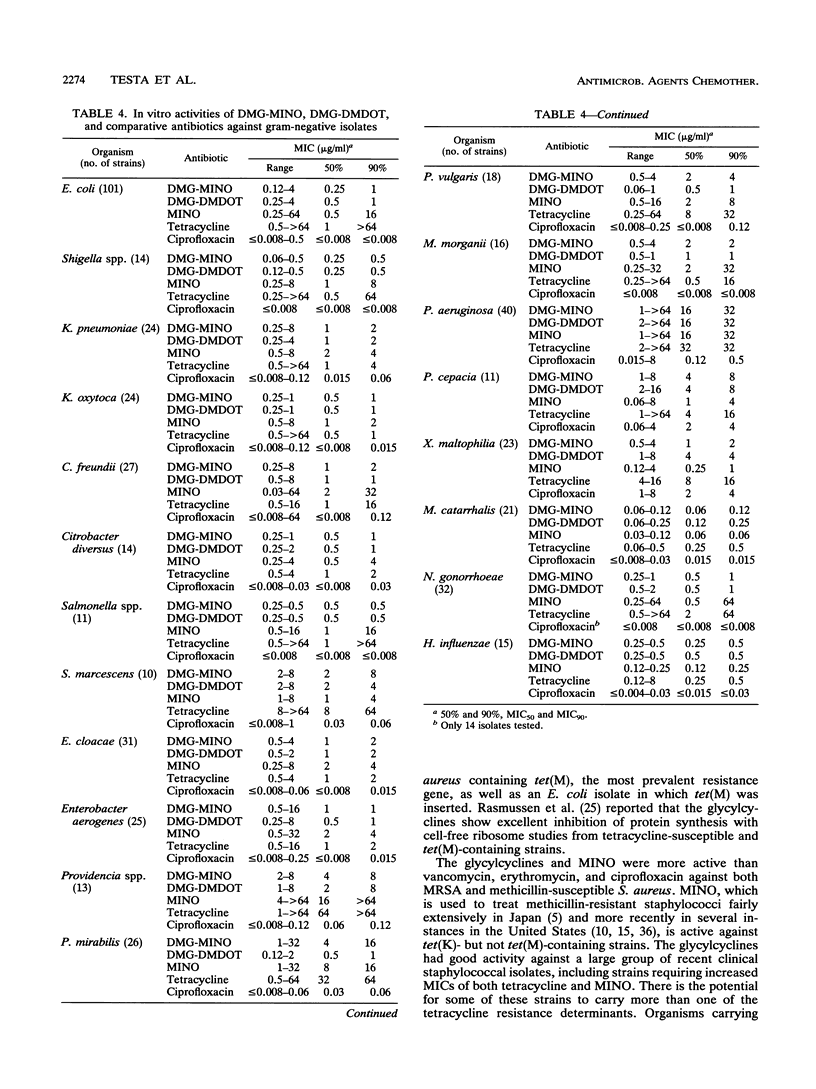
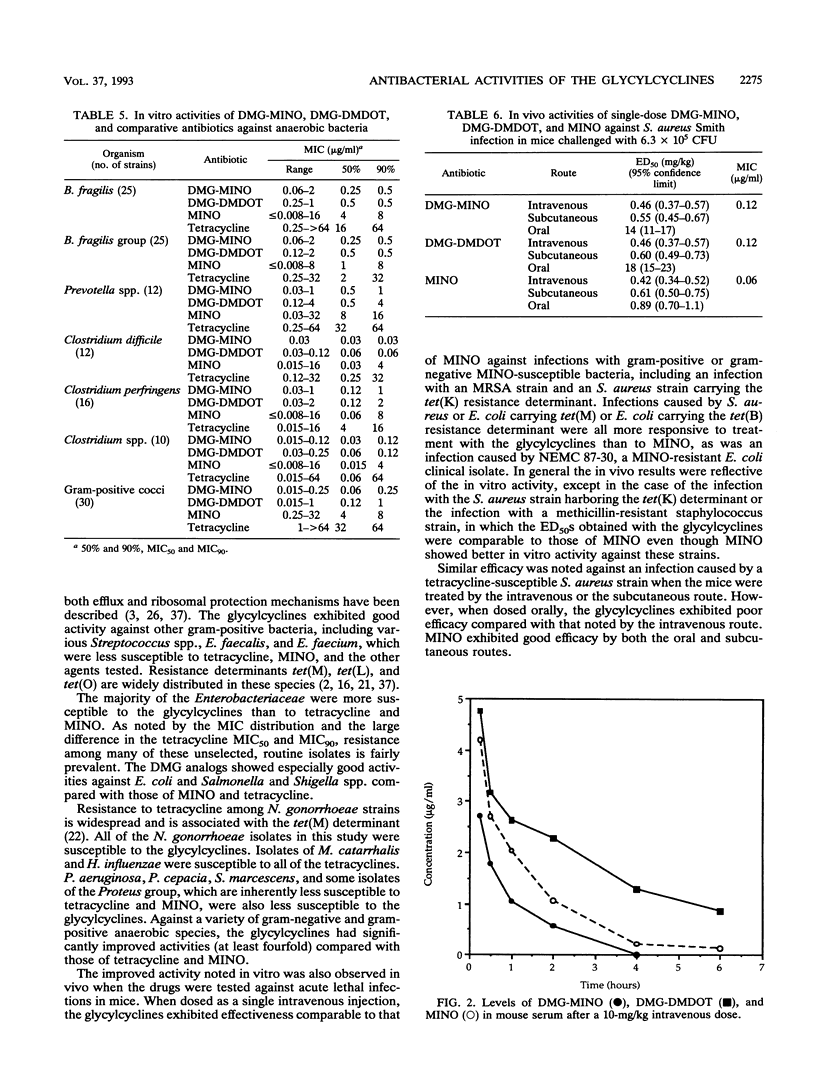
N,N-Dimethylglycylamido (DMG) derivatives of minocycline and 6-demethyl-6-deoxytetracycline are new semisynthetic tetracyclines referred to as the glycylcyclines. The in vitro activities of the glycylcyclines were evaluated in comparison with those of minocycline and tetracycline against strains carrying characterized tetracycline resistance determinants and against 995 recent clinical isolates obtained from geographically distinct medical centers in North America. The glycylcyclines were active against tetracycline-resistant strains carrying efflux [tet(A), tet(B), tet(C), and tet(D) in Escherichia coli and tet(K) in Staphylococcus aureus] and ribosomal protection [tet(M) in S. aureus, Enterococcus faecalis, and E. coli)] resistance determinants. Potent activity (MIC for 90% of strains, < or = 0.5 microgram/ml) was obtained with the glycylcyclines against methicillin-susceptible and methicillin-resistant S. aureus, E. faecalis, Enterococcus faecium, and various streptococcal species. The glycylcyclines exhibited good activity against a wide diversity of gram-negative aerobic and anaerobic bacteria, most of which were less susceptible to minocycline and tetracycline. The activities of the glycylcyclines against most organisms tested were comparable to each other. The in vivo efficacies of the glycylcyclines against acute lethal infections in mice when dosed intravenously were reflective of their in vitro activities. The glycylcyclines had efficacies comparable to that of minocycline against infections with methicillin-susceptible and methicillin-resistant S. aureus strains, a strain carrying tet(K), and a tetracycline-susceptible E. coli strain but exceeded the effectiveness of minocycline against infections with resistant isolates, including strains harboring tet(M) or tet(B). Levels of DMG-6-deoxytetracycline in serum were higher and more sustained than those of DMG-minocycline or minocycline. Our results show that the glycylcyclines have potent in vitro activities against a wide spectrum of gram-positive and gram-negative, aerobic and anaerobic bacteria, including many resistant strains. On the basis of their in vitro and in vivo activities, the glycylcyclines represent a significant advance to the tetracycline class of antibiotics and have good potential value for clinical efficacy.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bentorcha F., De Cespédès G., Horaud T. Tetracycline resistance heterogeneity in Enterococcus faecium. Antimicrob Agents Chemother. 1991 May;35(5):808–812. doi: 10.1128/aac.35.5.808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bismuth R., Zilhao R., Sakamoto H., Guesdon J. L., Courvalin P. Gene heterogeneity for tetracycline resistance in Staphylococcus spp. Antimicrob Agents Chemother. 1990 Aug;34(8):1611–1614. doi: 10.1128/aac.34.8.1611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burdett V. Streptococcal tetracycline resistance mediated at the level of protein synthesis. J Bacteriol. 1986 Feb;165(2):564–569. doi: 10.1128/jb.165.2.564-569.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chimura T. [A therapy for methicillin-resistant Staphylococcus aureus (MRSA) infections in obstetrics and gynecology]. Jpn J Antibiot. 1991 Aug;44(8):854–859. [PubMed] [Google Scholar]
- Chopra I., Bennett P. M., Lacey R. W. A variety of Staphylococcal plasmids present as multiple copies. J Gen Microbiol. 1973 Dec;79(2):343–345. doi: 10.1099/00221287-79-2-343. [DOI] [PubMed] [Google Scholar]
- Chopra I., Hawkey P. M., Hinton M. Tetracyclines, molecular and clinical aspects. J Antimicrob Chemother. 1992 Mar;29(3):245–277. doi: 10.1093/jac/29.3.245. [DOI] [PubMed] [Google Scholar]
- Chopra I., Shales S., Ball P. Tetracycline resistance determinants from groups A to D vary in their ability to confer decreased accumulation of tetracycline derivatives by Escherichia coli. J Gen Microbiol. 1982 Apr;128(4):689–692. doi: 10.1099/00221287-128-4-689. [DOI] [PubMed] [Google Scholar]
- Darouiche R., Wright C., Hamill R., Koza M., Lewis D., Markowski J. Eradication of colonization by methicillin-resistant Staphylococcus aureus by using oral minocycline-rifampin and topical mupirocin. Antimicrob Agents Chemother. 1991 Aug;35(8):1612–1615. doi: 10.1128/aac.35.8.1612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finland M. Twenty-fifth anniversary of the discovery of Aureomycin: the place of the tetracyclines in antimicrobial therapy. Clin Pharmacol Ther. 1974 Jan;15(1):3–8. doi: 10.1002/cpt19741513. [DOI] [PubMed] [Google Scholar]
- Goldman R. A., Hasan T., Hall C. C., Strycharz W. A., Cooperman B. S. Photoincorporation of tetracycline into Escherichia coli ribosomes. Identification of the major proteins photolabeled by native tetracycline and tetracycline photoproducts and implications for the inhibitory action of tetracycline on protein synthesis. Biochemistry. 1983 Jan 18;22(2):359–368. doi: 10.1021/bi00271a020. [DOI] [PubMed] [Google Scholar]
- Lawlor M. T., Sullivan M. C., Levitz R. E., Quintiliani R., Nightingale C. Treatment of prosthetic valve endocarditis due to methicillin-resistant Staphylococcus aureus with minocycline. J Infect Dis. 1990 Apr;161(4):812–814. doi: 10.1093/infdis/161.4.812. [DOI] [PubMed] [Google Scholar]
- LeBlanc D. J., Lee L. N., Titmas B. M., Smith C. J., Tenover F. C. Nucleotide sequence analysis of tetracycline resistance gene tetO from Streptococcus mutans DL5. J Bacteriol. 1988 Aug;170(8):3618–3626. doi: 10.1128/jb.170.8.3618-3626.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy S. B. Evolution and spread of tetracycline resistance determinants. J Antimicrob Chemother. 1989 Jul;24(1):1–3. doi: 10.1093/jac/24.1.1. [DOI] [PubMed] [Google Scholar]
- Levy S. B., McMurry L. M., Burdett V., Courvalin P., Hillen W., Roberts M. C., Taylor D. E. Nomenclature for tetracycline resistance determinants. Antimicrob Agents Chemother. 1989 Aug;33(8):1373–1374. doi: 10.1128/aac.33.8.1373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMurry L. M., Park B. H., Burdett V., Levy S. B. Energy-dependent efflux mediated by class L (tetL) tetracycline resistance determinant from streptococci. Antimicrob Agents Chemother. 1987 Oct;31(10):1648–1650. doi: 10.1128/aac.31.10.1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morse S. A., Johnson S. R., Biddle J. W., Roberts M. C. High-level tetracycline resistance in Neisseria gonorrhoeae is result of acquisition of streptococcal tetM determinant. Antimicrob Agents Chemother. 1986 Nov;30(5):664–670. doi: 10.1128/aac.30.5.664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts M. C., Hillier S. L. Genetic basis of tetracycline resistance in urogenital bacteria. Antimicrob Agents Chemother. 1990 Feb;34(2):261–264. doi: 10.1128/aac.34.2.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salyers A. A., Speer B. S., Shoemaker N. B. New perspectives in tetracycline resistance. Mol Microbiol. 1990 Jan;4(1):151–156. doi: 10.1111/j.1365-2958.1990.tb02025.x. [DOI] [PubMed] [Google Scholar]
- Shales S. W., Chopra I., Ball P. R. Evidence for more than one mechanism of plasmid-determined tetracycline resistance in Escherichia coli. J Gen Microbiol. 1980 Nov;121(1):221–229. doi: 10.1099/00221287-121-1-221. [DOI] [PubMed] [Google Scholar]
- Speer B. S., Shoemaker N. B., Salyers A. A. Bacterial resistance to tetracycline: mechanisms, transfer, and clinical significance. Clin Microbiol Rev. 1992 Oct;5(4):387–399. doi: 10.1128/cmr.5.4.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valcavi U., Brandt A., Corsi G. B., Minoja F., Pascucci G. Chemical modifications in the tetracycline series. J Antibiot (Tokyo) 1981 Jan;34(1):34–39. doi: 10.7164/antibiotics.34.34. [DOI] [PubMed] [Google Scholar]
- Yuk J. H., Dignani M. C., Harris R. L., Bradshaw M. W., Williams T. W., Jr Minocycline as an alternative antistaphylococcal agent. Rev Infect Dis. 1991 Sep-Oct;13(5):1023–1024. doi: 10.1093/clinids/13.5.1023. [DOI] [PubMed] [Google Scholar]
- Zilhao R., Papadopoulou B., Courvalin P. Occurrence of the Campylobacter resistance gene tetO in Enterococcus and Streptococcus spp. Antimicrob Agents Chemother. 1988 Dec;32(12):1793–1796. doi: 10.1128/aac.32.12.1793. [DOI] [PMC free article] [PubMed] [Google Scholar]


