Abstract
Bacterial biofilms cause serious problems, such as antibiotic resistance and medical device-related infections. To further understand bacterium-surface interactions and to develop efficient control strategies, self-assembled monolayers (SAMs) of alkanethiols presenting different functional groups on gold films were analyzed to determine their resistance to biofilm formation. Escherichia coli was labeled with green florescence protein, and its biofilm formation on SAM-modified surfaces was monitored by confocal laser scanning microscopy. The three-dimensional structures of biofilms were analyzed with the COMSTAT software to obtain information about biofilm thickness and surface coverage. SAMs presenting methyl, l-gulonamide (a sugar alcohol tethered with an amide bond), and tri(ethylene glycol) (TEG) groups were tested. Among these, the TEG-terminated SAM was the most resistant to E. coli biofilm formation; e.g., it repressed biofilm formation by E. coli DH5α by 99.5% ± 0.1% for 1 day compared to the biofilm formation on a bare gold surface. When surfaces were patterned with regions consisting of methyl-terminated SAMs surrounded by TEG-terminated SAMs, E. coli formed biofilms only on methyl-terminated patterns. Addition of TEG as a free molecule to growth medium at concentrations of 0.1 and 1.0% also inhibited biofilm formation, while TEG at concentrations up to 1.5% did not have any noticeable effects on cell growth. The results of this study suggest that the reduction in biofilm formation on surfaces modified with TEG-terminated SAMs is a result of multiple factors, including the solvent structure at the interface, the chemorepellent nature of TEG, and the inhibitory effect of TEG on cell motility.
Bacterial biofilms are sessile microbial communities formed on solid surfaces. They cause serious health problems in humans, such as dental decay and persistent lung infections in cystic fibrosis patients (9, 30). Biofilms are also responsible for medical device-related infections, including infections related to intravenous catheters, joint prostheses, cardiac pacemakers, prosthetic heart valves, peritoneal dialysis catheters, and cerebrospinal fluid shunts (12, 39). Deleterious biofilms also cause persistent biofouling and corrosion problems in water delivery systems and many industrial processes (7, 46). Once bacteria attach to a surface and form a biofilm, they are up to 1,000 times more tolerant to antimicrobials and disinfection treatment than their free-swimming counterparts (9, 17, 38). Thus, it is critical to understand biofilm formation and develop novel strategies for efficient biofilm control.
Biofilm formation is a complex process that involves multicellular behaviors, such as quorum sensing (bacterial gene regulation by sensing and responding to cell density [11, 42]) and swarming motility (coordinated movement of hyperflagellated and elongated cells [1, 21]). It is generally accepted that biofilm formation is a dynamic process including initial attachment, microcolony formation, maturation, and dispersion (12). Therefore, one promising approach is modifying the surface in order to alter cell attachment, which can hinder the subsequent steps in biofilm formation.
There are a number of different approaches that are used for surface modification (32). Conceptually, the easiest approach is to repress bacteria with bactericidal or bacteriostatic agents. This approach could be achieved either by releasing antimicrobials from the surface (e.g., cross-linking a drug-containing hydrogel to the surface) or through covalent modification. While each of these methods has certain advantages, neither is completely effective (10). In addition, there is increasing concern that the release of antibiotics may cause the spread of antimicrobial-resistant pathogens. Although it is not a problem for covalent modification, the antimicrobial activity could be masked by host proteins which quickly cover the surface of an implanted device (10). Thus, it is important to develop novel approaches for modifying the surface covalently with bioinert groups.
Self-assembled monolayers (SAMs) formed by adsorption of functional alkanethiols to a gold, silver, or copper surface are particularly well-ordered monolayers compared to other surface modifications (3, 23). Generally, SAMs formed by alkanethiols consist of three components. The first component is a strong complex of an Au-S bond connecting alkanethiol to the surface; the second component is a monolayer comprised of aliphatic chains that allow close packing of the monolayer; and the third component is a designed functional group covalently attached to the aliphatic chain (37). With the versatility of functional groups introduced by organic synthesis, the surface of SAMs can be tailored with a wide variety of chemical and physical properties while the well-ordered monolayer structure that is absent in other types of surface modifications is maintained. For instance, the functional group can be chosen so that certain properties are obtained (e.g., properties that can change the wettability of the surface so that it is hydrophilic [hydroxyl groups] or hydrophobic [methyl groups]). The class of functionally versatile surfaces presented by well-ordered monolayer structures forms a well-defined system for studying and controlling cell-surface interactions.
Recently, Luk et al. (25) found that SAMs of alkanethiols presenting mannitol groups are inert to protein adsorption and attachment of mammalian 3T3 fibroblasts. In addition, Hoffmann and Tovar (16) reported that nonspecific adsorption of proteins on oxidic surfaces is significantly reduced by mixed SAMs presenting methoxy-tri(ethylene glycol) and alkyl-terminated silanes. Thus, SAMs presenting different functional groups on gold-coated surfaces are promising model surfaces for mechanistic studies of cell-surface interactions and the development of novel biofilm control strategies. Another advantage of SAMs is that two different surface chemistries can be presented on a single surface in patterns at the micrometer scale using a technique known as microcontact printing or stamping (20). The patterned surfaces have well-defined regions with different chemical and physical properties (23).
While SAMs have great potential for controlling cell-surface interactions, their applications in biofilm control have not been well investigated. Wiencek and Fletcher (43) studied the attachment of Pseudomonas sp. strain MI-1A cells on SAMs with different hydrophobicities (terminated with a methyl group or a hydroxyl group or a mixture of methyl and hydroxyl groups). Ista et al. (18) reported that SAMs presenting hexa(ethylene glycol) resist the attachment of Staphylococcus epidermidis and Deleya marina based on cell counting. To control biofilm formation using SAMs, it is necessary to compare the effects of different functional groups, to analyze the biofilm structures on SAMs, and to explore the mechanism by which biofilm formation is reduced. In this study, Escherichia coli was used as a model system to study biofilm control by SAMs presenting three different functional groups, methyl, l-gulonamide, and tri(ethylene glycol) (TEG). We focused on E. coli strains as their genotypes are well known and isogenic mutants are available (2, 6, 36). Thus, they are an ideal system for a fundamental study and for comparison with our previous E. coli biofilm results (33, 35). The E. coli cells were labeled with green fluorescent protein (GFP) to study biofilm formation with confocal laser scanning microscopy (CLSM), which allows visualization of the three-dimensional structure of a biofilm nondestructively. The surface coverage and maximum thickness were calculated using the COMSTAT software (15). The biofilm control by TEG-terminated SAMs was explored by studying the effects of TEG on surface hydrophobicity and the growth, chemotaxis, and motility of E. coli.
MATERIALS AND METHODS
Bacterial strains and growth media.
The following three E. coli strains were used in this study: DH5α [supE44 ΔlacU169 (φ80lacZΔM15) hsdR17 recA1 endA1 gyrA96 thi-1 relA1], RP437 [wild-type strain for chemotaxis; 7hr(Am)-1 leuB6 his-4 metF(Am)159 eda-50 rpsL13t65thi-1 ara-14 lacY1 mtl-1 xyl-5 tonA31 tsx-78], and RP5700 [(tsr)Δ7028 chemotaxis mutant of RP437] (2, 6, 36). The strains were labeled with GFP by transformation of plasmid pGLO (Bio-Rad Laboratories, Inc., Hercules, CA) to study biofilm formation on SAM-modified surfaces with CLSM. E. coli strains were grown in Luria-Bertani (LB) medium (36) containing 10 g/liter tryptone, 5 g/liter yeast extract, and 10 g/liter sodium chloride. Ampicillin was added at a concentration of 100 μg/ml to maintain the plasmid. To induce the expression of GFP, arabinose was included in the media at a final concentration of 6 g/liter.
For the chemotaxis assay, wild-type strain RP437 (2) and its isogenic Δtsr mutant strain RP5700 (6) were grown at 37°C with shaking (200 rpm) in tryptone broth (28) containing 10 g/liter tryptone, 5 g/liter sodium chloride, and 1.12 g/liter sodium lactate. Each overnight culture was washed with and resuspended in motility medium (pH 7.0) (28), which contained 2.7 g/liter KH2PO4, 3.5 g/liter K2HPO4, 0.044 g/liter potassium EDTA, and 1.12 g/liter sodium lactate.
Preparation of SAM-modified gold surfaces.
Preparation of SAMs on gold has been described in detail previously (25). We compared biofilm formations on SAMs presenting TEG, l-gulonamide, and methyl groups (Fig. 1). A bare gold surface was used as the negative control. To corroborate the results for the biofilm control, two microcontact-printed SAM patterns were also prepared on gold surfaces: an HS(CH2)14CH3 (hydrophobic) pattern with HS(CH)11(OCH2CH2)3OH (TEG, hydrophilic) as the background and an HS(CH2)14CH3 (hydrophobic) pattern with HS(CH)11-l-gulonamide (hydrophilic) as the background.
FIG. 1.
(A) Structures of the alkanethiols used in this work. (B) Schematic representation of SAM terminated with l-gulonamide-terminated-undecanethiol (B1), (1-mercatpundec-11-yl)tri(ethylene glycol) (B2), and pentadecanethiol (PDT) (B3).
Fabrication of gold films on glass substrates. (i) Cleaning of the substrates.
Fisher's Finest microscope slides were cleaned with a Piranha solution (26) (warning: Piranha solution should be handled with extreme caution; in some circumstances, probably when it has been mixed with significant quantities of an oxidizable organic material, it has detonated unexpectedly). Slides were soaked in a Piranha solution (3 parts 35% [wt/wt] H2O2 in water and 7 parts concentrated H2SO4) at 75°C for 45 min. The slides were then thoroughly rinsed with deionized water (18.2 MΩ·cm), followed by ethanol and then methanol. The cleaned slides were dried with a stream of nitrogen and were stored in an oven at 100°C overnight prior to metal deposition by electron beam evaporation.
(ii) Gold deposition on the slides.
Semitransparent gold films were deposited onto glass slides with an electron beam evaporator (Thermionics, Santa Clara, CA). The gold was deposited on plain microscope glass slides at a height of 43.2 cm from the gold source. The slides were positioned such that the angle of gold incidence was 45° from the normal of the glass slides (26). A thin film of titanium (18 Å) was initially deposited to enhance the adhesion of the gold. After the system was cooled, 370 Å of gold was deposited onto the titanium-coated slides. The rate of deposition was 0.2 Å/s for both gold and titanium. The pressure was maintained at no higher than 5 × 10−7 torr during deposition.
Hydrophobicity of the modified surfaces.
Hydrophobicity is an important factor for understanding cell-surface interactions. To characterize this property, 3 μl of deionized water was dropped on different SAM-modified surfaces, as well as bare gold surfaces. The diameter of each droplet was measured as an indicator of hydrophobicity.
Microcontact printing of methyl-terminated SAMs surrounded by TEG- or l-gulonamide-terminated SAMs.
The gold slides were cut into 7.6- by 1.25-cm pieces, washed with absolute ethanol (200 proof), and dried with a stream of nitrogen. Microcontact printing was conducted using a slight modification of a previously reported procedure (4). A solution of 1 mM pentadecanethiol in 200-proof ethanol was used for microcontact printing with polydimethylsiloxane stamps. The polydimethylsiloxane stamps dabbed with pentadecanethiol were placed in contact with the gradient gold slides for 15 s. The pentadecanethiol-printed slides were then washed with ethanol, dried with nitrogen, and soaked in 1 mM solutions of the TEG- or l-gulonamide-terminated alkanethiols in ethanol for 10 h. The slides were then taken out of the solutions, washed with absolute ethanol (200 proof), and dried with nitrogen.
Growth measurement.
To investigate the effects of TEG on E. coli growth, DH5α was grown in LB medium overnight at 37°C. Then it was used to inoculate LB medium in a 96-well plate to an optical density at 600 nm (OD600) of 0.05. The 96-well plate was incubated at 37°C with shaking (200 rpm), and the growth was monitored by measuring the OD600 with a microplate reader (FLx800; Bio-Tek Instruments, Inc., Winooski, VT). The growth rates were calculated based on the OD600 in the exponential phase.
Biofilm formation.
E. coli cells were used to form biofilms on SAM-modified surfaces. An overnight E. coli culture grown in LB medium with 100 μg/ml ampicillin was used to inoculate biofilm cultures in the same medium supplemented with 6 g/liter arabinose to an OD600 of 0.05 as measured with a Genesis 5 spectrophotometer (Spectronic Instruments, Rochester, NY). The SAM-modified gold surfaces and bare gold surfaces were sterilized by soaking them in 70% ethanol for 15 min and then dried with a vacuum at room temperature. Biofilms were cultured in plastic petri dishes (100 by 15 mm) at 37°C without shaking. To minimize the variation in sample preparation, different SAM-modified surfaces were incubated in the same petri dish containing 20 ml medium. The experiment was repeated four times, and consistent results were obtained.
CLSM and image analysis.
To analyze the E. coli biofilms using CLSM, each surface was washed gently by dipping it vertically in 0.85% NaCl buffer three times (fresh buffer was used for each dipping procedure). Then the surface was put upside down on a microscope cover slide and analyzed with a 5-pascal inverted confocal LSM (Carl Zeiss, Inc., Berlin, Germany). GFP was visualized by excitation with an argon laser at 488 nm. Emission of fluorescence was detected with an LP 505-nm emission filter. A series of images were obtained for each position at 1-μm intervals in the z section for a three-dimensional view of the biofilm (from the substratum to the top of the biofilm). At least five spots were examined for each sample. The surface coverage, maximum thickness, biomass, and roughness were calculated using the COMSTAT software (15). Since the biofilm formation on certain surfaces (e.g., TEG-terminated SAMs) was strongly reduced, the roughness data were not useful. The biomass data were consistent with the data for surface coverage; therefore, only the surface coverage and thickness were used to compare the biofilms.
Biofilm formation with TEG added as a free molecule in liquid medium.
To study if the biofilm inhibition by TEG is surface specific, TEG was also added as a free molecule (final concentration, 0, 0.1, or 1.0% [wt/vol]) to LB medium supplemented with 100 μg/ml ampicillin and 6 g/liter arabinose. Biofilms were cultured on bare gold surfaces (in a plastic petri dish) at 37°C without shaking. The experiment was performed in duplicate, and consistent results were obtained.
Chemical-in-plug method for a negative chemotaxis assay.
A negative chemotaxis assay was performed by following the procedure described previously (28), with slight modifications. Overnight cultures of RP437 and RP5700 in tryptone broth were harvested by centrifugation at 4,000 × g with a Marathon 21000 centrifuge (Fisher Scientific, Pittsburgh, PA) for 10 min at room temperature and washed once with motility medium. The cells were concentrated, and about 109 cells were mixed with 20 ml of motility medium containing 0.3% agar at 50°C. The agar mixture was immediately poured into a petri dish. After the dish stood for 5 min at room temperature, plugs of hard agar (2.0%) containing TEG at different concentrations (0, 1.5 × 10−3, 0.015, 0.15, 1.5, and 15% [wt/vol]) were put into the cell-agar mixture. The petri dish was incubated for 60 min at 37°C, and images were taken to evaluate the chemotaxis.
Motility assay.
To determine the best agar concentration for the motility assay, overnight cultures of RP437, RP5700, and DH5α in LB medium were used to inoculate soft LB agar plates with agar concentrations of 0.2, 0.4, 0.6, 0.8, 1.0, and 1.2% using toothpicks. The swarming colonies (with an agar concentration higher than 0.3%) were measured every 2 h, and the swarming rates (mm/h) were calculated. The agar concentration that gave the highest swarming rate (0.4%) was used in experiments the effects of TEG (0, 0.15%, and 1.5%).
RESULTS
E. coli biofilm formation on SAM-modified surfaces.
To test the effects of surface modification with functional groups, three SAM-modified gold surfaces (Fig. 1) and a bare gold surface were used to form E. coli DH5α biofilms. The structural parameters of biofilms were calculated using the COMSTAT software, and the relative amounts of biofilms (based on surface coverage) are shown in Fig. 2.
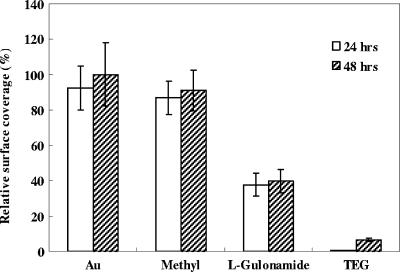
FIG. 2.
Relative surface coverage of biofilms on bare gold and SAM-modified surfaces presenting methyl, l-gulonamide, and TEG groups. The coverage of a 48-h biofilm on a bare gold surface was defined as 100%.
The results suggest that SAM-modified surfaces presenting TEG are strongly resistant to E. coli biofilm formation and that l-gulonamide groups are only moderately resistant compared to bare gold surfaces. SAMs presenting TEG reduced biofilm formation by 99.5% ± 0.1% (P < 0.01, as determined by a t test) for 24 h after inoculation and by 93.1% ± 0.8% (P < 0.01) for 48 h after inoculation (based on surface coverage). In comparison, the SAMs presenting l-gulonamide reduced biofilm formation by 59.2% ± 6.8% (P < 0.01) for 24 h and by 56.9% ± 6.9% (P < 0.01) for 48 h. SAMs presenting methyl groups did not show significant resistance to biofilm formation (Fig. 2). A similar trend was also observed for biofilm thickness (based on the average maximum thickness of four biological replicates for each SAM with five positions on each sample). For example, the thicknesses of the 24-h biofilms were 13.0 ± 1.4 μm for bare gold surfaces, 12.0 ± 1.4 μm for methyl-terminated SAMs, 5.7 ± 0.6 μm for l-gulonamide-terminated SAMs, and 2.3 ± 0.4 μm for TEG-terminated SAMs, while the thicknesses of the 48-h biofilms were 16.0 ± 2.0 μm for bare gold surfaces, 14.0 ± 1.8 μm for methyl-terminated SAMs, 9.8 ± 1.2 μm for l-gulonamide-terminated SAMs, and 6.2 ± 0.7 μm for TEG-terminated SAMs.
Since a TEG-terminated SAM presents multiple water-soluble ethylene glycol units and hydroxyl groups on the surface (Fig. 1), we compared the hydrophobicities of the three surfaces studied. It was found that SAMs presenting methyl groups were more hydrophobic than TEG- and l-gulonamide-terminated SAMs based on a comparison of the droplet diameters. TEG- and l-gulonamide-terminated SAMs were found to have similar hydrophobicities, which is consistent with the previous report based on contact angles of the droplet curvatures on these two surfaces (advancing contact angle, ∼37° [18, 29]).
Biofilm formation on SAM-modified surfaces with patterns.
To corroborate the results described above, SAM-modified surfaces with different patterns were used to culture E. coli DH5α biofilms; the patterns included microcontact-printed HS(CH2)14CH3 patterns with an HS(CH2)11(OCH2CH2)3OH background and microcontact-printed HS(CH2)14CH3 patterns with an HS(CH2)11-l-gulonamide background. Consistent with the biofilm inhibition on SAM-modified surfaces presenting TEG groups, there was no noticeable biofilm growth with the HS(CH2)11(OCH2CH2)3OH background. However, the alkanethiol HS(CH2)14CH3 was not resistant to biofilm formation. Thus, biofilms were formed only in the areas modified with HS(CH2)14CH3 patterns (125- by 125-μm squares), as expected, and the surface coverage and thickness were consistent with those in nonpattern experiments (see Fig. S1 in the supplemental material). Patterned biofilms were also observed for SAM-modified surfaces with the HS(CH2)11-l-gulonamide background. However, the shape was less regular than the shape on the surfaces with the TEG background (see Fig. S1 in the supplemental material), consistent with the finding that SAMs presenting l-gulonamide were less resistant to biofilm formation than SAMs presenting TEG, as shown by the nonpattern experiments described above.
Biofilm formation with TEG added as a free molecule in liquid medium.
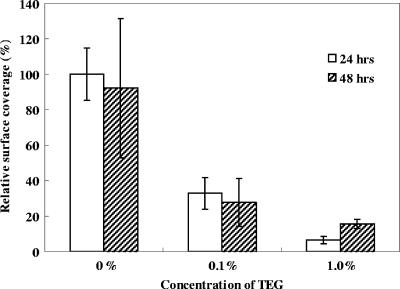
To determine if the biofilm control by TEG is surface specific, TEG was also added as a free molecule in LB medium at final concentrations of 0, 0.1, and 1.0% (wt/vol). Biofilms were grown on bare gold surfaces in batch cultures and observed with CLSM at 24 and 48 h after inoculation. The relative surface coverage on the substrate is shown in Fig. 3. Addition of TEG greatly reduced the biofilm formation. For instance, 1% TEG in the cell culture inhibited biofilm formation by 93.6% ± 2.2% (P < 0.01) and 84.4% ± 2.6% (P < 0.05) at 24 and 48 h after inoculation, respectively (Fig. 3), whereas 0.1% TEG inhibited the biofilm formation by 67.1% ± 9.0% (P < 0.01) and 72.1% ± 14.4% (P < 0.05) for 24 and 48 h, respectively. Compared to the effect on surface coverage, the effect of TEG on biofilm thickness was less apparent; e.g., the biofilm thicknesses at 24 h after inoculation were 11.2 ± 1.6 μm, 10.8 ± 0.8 μm, and 7.0 ± 0.7 μm for cultures containing 0, 0.1, and 1.0% TEG, respectively.
FIG. 3.
Relative surface coverage of biofilms on a bare gold surface in the presence of 0, 0.1, and 1.0% TEG. The coverage of a 24-h biofilm without TEG was defined as 100%.
TEG has no effect on E. coli growth.
We noted that the TEG concentrations used in the noncoating experiment were relatively high. To investigate if the biofilm inhibition was due to effects on cell growth, growth curves of DH5α in LB medium supplemented with 0, 0.01, 0.1, 1, and 1.5% TEG were measured. TEG did not have any noticeable effects on general cell growth since the specific growth rates were about 0.75 h−1 for all the samples (growth curves are shown in Fig. S2 in the supplemental material). Oligo(ethylene glycol) and poly(ethylene glycol) are in general nontoxic and have been used for drug delivery and other medical purposes (13). Specifically, TEG has been used for drug discovery and development (40). These findings suggest that TEG inhibited biofilm formation by interrupting bacterium-surface interactions.
Long-term resistance by SAMs on gold surfaces.
To corroborate the results described above and to study the biofilm resistance of SAMs with extended exposure to cells, a long-term experiment was performed involving SAM-modified surfaces with the microcontact-printed HS(CH2)14CH3 pattern and the HS(CH2)11-(OCH2CH2)3OH background. The medium was replaced with fresh medium every day, and the surfaces were analyzed every 2 days using CLSM. Within 7 days, the DH5α cells spread and proliferated to occupy the nonresistant region (pattern), but they did not invade the resistant region (background) (see Fig. S3 in the supplemental material). This result indicates that the TEG-terminated SAMs were resistant to biofilm formation for at least 7 days under the experimental conditions used in this study.
Negative chemotaxis to TEG.
TEG inhibited biofilm formation without noticeable effects on growth, suggesting that TEG controlled biofilm formation by interrupting bacterium-surface interactions. Ethylene glycol has been reported to be a chemorepellent of E. coli. It causes a decrease in the methylation level of methyl-accepting chemotaxis proteins (MCPs) and an increase in tumbling, which enables the cells to swim away from it (28). We hypothesized that the TEG in our experiments may have similar effects on chemotaxis. To test this hypothesis, the method reported by Oosawa and Imae (28) was used to study the chemotaxis of E. coli in the presence of different concentrations of TEG (0, 1.5 × 10−3, 0.015, 0.15, 1.5, and 15%). Both wild-type strain RP437 and the Δtsr mutant RP5700 were tested.
On the RP437 chemotaxis plates, a clear zone around each plug containing TEG was formed within 60 min after addition of the hard agar plug (see Fig. S4 in the supplemental material). This suggests that the cells were moving away from the plugs containing TEG, similar to the results for ethylene glycol reported by Oosawa and Imae (28). No clear zone was seen for RP5700, except for the preparations with 15% (1 M) TEG (the clear zone was much smaller than that of RP437 [data not shown]). Thus, RP5700 demonstrated reduced but not fully abolished chemotaxis. E. coli has different MCPs (e.g., Tsr, Tar, and Trg) for sensing different chemicals (28). The results described above suggest that Tsr plays an important role in the response to TEG, but there might be redundancy in TEG sensing by other MCPs since RP5700 also exhibited negative chemotaxis in response to 15% TEG.
TEG controls the motility of E. coli.
Interestingly, the biofilm cells on bare gold surfaces were found to be elongated (12.1 ± 2.7 μm), while the few cells on TEG-terminated SAMs were 3.1 ± 0.8 μm long (see Fig. S5 in the supplemental material). Bacteria are well known for a highly organized multicellular behavior called swarming, by which the elongated cells move quickly across the surface (1). Swarming has been reported for E. coli (14), and swarming cells have been observed in biofilms (21). Thus, the elongated cells found on bare gold surfaces in this study could have been swarming cells, and this led to the hypothesis that the swarming motility might be affected by TEG.
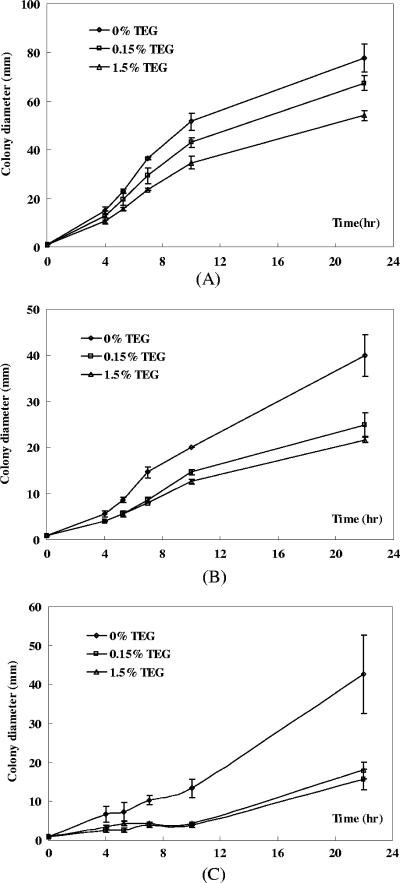
A motility assay was performed to evaluate the effects of TEG on E. coli swarming. LB medium with different concentrations of agar were first tested to optimize the experimental conditions for the swarming of DH5α. The best swarming motility was observed with 0.4% agar, and these conditions were used for the following experiments. Of the three strains tested, RP437 exhibited the highest motility, with an average swarming rate of 5.2 ± 0.4 mm/h (based on the swarming in the first 10 h after inoculation), followed by RP5700 (2.0 ± 0.1 mm/h) and DH5α (1.3 ± 0.2 mm/h). Addition of TEG (0.15 and 1.5%) resulted in clear inhibition of motility for all the three strains (Fig. 4). For instance, the average swarming rates for RP437 with different concentrations of TEG were 5.2 ± 0.4 mm/h (no TEG), 4.3 ± 0.2 mm/h (0.15% TEG), and 3.4 ± 0.3 mm/h (1.5% TEG).
FIG. 4.
Swarming of E. coli in the presence of 0, 0.15, and 1.5% TEG. (A) RP437. (B) RP5700. (C) DH5α.
Biofilm formation by strains with different motilities.
Motility has been shown to be important for biofilm formation (31, 44). Since TEG inhibited biofilm formation by DH5α and the motilities of DH5α, RP437, and RP5700, it was interesting to study the effects of TEG on the biofilm formation by the more motile strains, strains RP437 and RP5700. These two strains were labeled with GFP (pGLO) and tested for biofilm formation on a SAM-modified surface with an HS(CH2)11(OCH2CH2)3OH background and microcontact-printed HS(CH2)14CH3 patterns. Consistent with the results for DH5α, the TEG-terminated SAMs were also resistant to biofilm formation by RP437, as biofilms were formed only in expected patterns (Fig. 5). In addition, RP437 was found to form more biofilm than DH5α, consistent with its higher motility compared to the other two strains. Interestingly, RP5700 did not form an apparent biofilm. This suggests that tsr plays an important role in biofilm formation (see Discussion).
FIG. 5.
Representative image for RP437 biofilms formed on a SAM-modified surface with the HS(CH2)11(OCH2CH2)3OH background and microcontact-printed HS(CH2)14CH3 patterns. The image is a Z section of a 20-μm-thick biofilm obtained 10 μm from the substratum.
DISCUSSION
In this study, we obtained evidence that SAMs presenting certain functional groups are resistant to E. coli biofilm formation, and the TEG group was found to be the most resistant; e.g., it repressed DH5α biofilm formation by 99.5% ± 0.1% for 24 h after inoculation. These results were corroborated by experiments using surfaces with regions consisting of nonresistant SAMs surrounded by resistant SAMs. TEG added as a free molecule in growth medium also inhibited biofilm formation and motility but had no effect on general cell growth, suggesting that TEG inhibited biofilm formation by interrupting cell-surface interactions.
The results of this study indicate that different functional groups have different effects on cell attachment. Specifically, the resistance to biofilm formation was found to increase in the order methyl < l-gulonamide < TEG. Previously, Tidwell et al. (41) found that the growth of bovine aortic endothelial cells on SAMs increases in the following order: hydroxyl (—CH2OH) < methyl ester (—COOCH3) < methyl (—CH3) ≪ carboxyl (—COOH). In addition, Lee et al. (22) found that the strength of adhesion of K100 erythroleukemia cells to functional groups has the following trend: methyl (—CH3) < carboxyl (—COOH) ≈ epoxide ≪ amine (—NH2). Therefore, altering the adhesion strength could be a mechanism for controlling cell attachment, and it would be helpful to test our system with other bacterial species.
Since hydrophobic surface structures of bacteria (e.g., fimbriae) have been shown to mediate cell attachment (27, 31, 33, 45) and TEG is hydrophilic, it's possible that a TEG-terminated SAM created a hydrophilic surface which increased the energy barrier for cell attachment. For the early stage of biofilm formation, we studied the attachment of RP437 cells on bare gold surfaces and on TEG-terminated SAMs at 2 and 6 h after inoculation. It was found that cells attached to bare gold quickly, with 0.54% ± 0.17% and 2.65% ± 0.92% of the surface covered at 2 and 6 h after inoculation, respectively. However, no noticeable attachment was seen on TEG-terminated SAMs (see Fig. S6 in the supplemental material), suggesting that the initial attachment was inhibited. This is consistent with the data obtained at 24 and 48 h after inoculation. Our data also suggest that the reduction in biofilm formation on SAMs is caused not only by the increase in hydrophilicity since the TEG-terminated SAMs were more resistant to biofilm formation than the l-gulonamide-terminated SAMs (Fig. 2), while these SAMs have similar hydrophobicities and both present highly water-soluble functional groups (hydroxyl groups). In an earlier report, D. marina was found to attach more to SAMs presenting a hexa(ethylene glycol) group than to SAMs presenting a COOH group, although COOH is more hydrophilic than hexa(ethylene glycol) (18). This finding and our results indicate that the hydrophobicity is not the determining factor in biofilm formation. Our motility assay and chemotaxis results indicate that TEG may affect biofilm formation at the molecular level.
It has been reported that motility is critical for initial attachment during E. coli biofilm formation (31). Wood et al. (44) reported that motility also influences the maturation of biofilms of E. coli since strains with higher motility (MG1655 > ATCC 25404 > BW25113 > JM109 > DH5α) can form thicker biofilms with greater surface coverage than strains with lower motility. Consistently, we also found that RP437 (the most motile strain in this study) formed more biofilm than RP5700 and DH5α formed. Interestingly, RP5700 formed much less biofilm than RP437 and DH5α formed [no apparent biofilm was seen with the HS(CH2)14CH3 pattern (see Fig. S7 in the supplemental material)], even though the motility of RP5700 was slightly higher than that of DH5α. Burkart et al. (5) showed that either Tsr or Tar alone can support E. coli swarming, which is consistent with the finding that RP5700 has some motility. RP5700 lacks the serine chemoreceptor Tsr compared to RP437. Although the mutation in tsr did not completely abolish motility, the role that tsr plays in biofilm formation may be critical. Previously, a brominated furanone from a marine alga, Delisea pulchra, was found to inhibit E. coli biofilm formation, and DNA microarray results suggest that furanone represses the motility genes, including tsr (34).
In the experiment in which TEG was added as a free molecule in liquid medium, although the surface coverage of biofilm was significantly reduced, SAMs did not have effects on the maximum thickness of biofilm to the same extent. It is generally accepted that biofilm formation includes attachment, microcolony formation, maturation, and dispersion (12). Our results suggest that TEG may have stronger inhibition on cell attachment than on biofilm maturation. However, the biofilms in this study were relatively thin (10 to 20 μm). Further study with thicker biofilms (e.g., biofilms in flow cells [S. Hou and D. Ren, unpublished data]) should help test this hypothesis.
Although surfaces modified with certain SAMs are resistant to biofilm formation in vitro, the in vivo applications may have additional challenges. It is well documented that medical devices are quickly covered by host proteins once they are implanted in the human body (8, 10); therefore, the surfaces must resist both bacterial attachment and protein absorption in vivo. In an earlier study, TEG-terminated SAMs were found to resist protein absorption (25). Thus, this surface modification strategy is promising for biofilm control, and further tests of biofilm formation in the presence of proteins should be helpful for evaluating the application of these SAMs in vivo.
To conclude, this work demonstrated that bioinert SAMs of TEG-terminated alkanethiols on gold films that resist protein adsorption and mammalian cell adhesion also resist bacterial biofilm formation. Our results further indicate that surface hydrophobicity is not the determining factor in biofilm formation, as l-gulonamide-terminated SAMs have hydrophobicity similar to that of TEG-terminated SAMs but exhibit significantly lower resistance to biofilm formation than TEG-terminated SAMs. The results of chemotaxis and motility experiments suggest that TEG-terminated SAMs may also repel cells and have an inhibitory effect on cell motility, which is known to be important for biofilm formation (31). Thus, the reduction in biofilm formation on TEG-terminated SAMs is a result of multiple factors, including the solvent structure at the interface (19, 24), the chemorepellent nature of TEG, and the inhibitory effects of TEG on cell motility. With the ability to resist bacterial adhesion and biofilm formation, SAMs are potential materials for development of antibiofilm coatings for medical applications.
Supplementary Material
Acknowledgments
This work was funded by the Syracuse Center of Excellence under EPA grant X-83232501-0.
We thank Melissa Pepling at Syracuse University for helping with CLSM, as well as Arne Heydorn at the Technical University of Denmark for providing the COMSTAT software. We are also grateful to John S. Parkinson (University of Utah) for providing E. coli strains RP437 and RP5700.
Footnotes
Published ahead of print on 4 May 2007.
Supplemental material for this article may be found at http://aem.asm.org/.
REFERENCES
- 1.Allison, C., and C. Hughes. 1991. Bacterial swarming: an example of prokaryotic differentiation and multicellular behaviour. Sci. Prog. 75:403-422. [PubMed] [Google Scholar]
- 2.Ames, P., and J. S. Parkinson. 1988. Transmembrane signaling by bacterial chemoreceptors: E. coli transducers with locked signal output. Cell 55:817-826. [DOI] [PubMed] [Google Scholar]
- 3.Bain, C. D., E. B. Troughton, Y.-T. Tao, J. Evall, G. M. Whitesides, and R. G. Nuzzo. 1989. Formation of monolayer films by the spontaneous assembly of organic thiols from solution onto gold. J. Am. Chem. Soc. 111:321-335. [Google Scholar]
- 4.Brock, A., E. Chang, C.-C. Ho, P. LeDuc, X. Jiang, G. M. Whitesides, and D. E. Ingber. 2003. Geometric determinants of directional cell motility revealed using microcontact printing. Langmuir 19:1611-1617. [DOI] [PubMed] [Google Scholar]
- 5.Burkart, M., A. Toguchi, and R. M. Harshey. 1998. The chemotaxis system, but not chemotaxis, is essential for swarming motility in Escherichia coli. Proc. Natl. Acad. Sci. USA 95:2568-2573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Callahan, A. M., B. L. Frazier, and J. S. Parkinson. 1987. Chemotaxis in Escherichia coli: construction and properties of λ tsr transducing phage. J. Bacteriol. 169:1246-1253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Characklis, W. G. 1981. Fouling biofilm development: a process analysis. Biotechnol. Bioeng. 23:1923-1960. [DOI] [PubMed] [Google Scholar]
- 8.Chatzinikolaou, I., and I. I. Raad. 2000. Intravascular catheter-related infections: a preventable challenge in the critically ill. Semin. Respir. Infect. 15:264-271. [DOI] [PubMed] [Google Scholar]
- 9.Costerton, J. W., P. S. Stewart, and E. P. Greenberg. 1999. Bacterial biofilms: a common cause of persistent infections. Science 284:1318-1322. [DOI] [PubMed] [Google Scholar]
- 10.Danese, P. N. 2002. Antibiofilm approaches: prevention of catheter colonization. Chem. Biol. 9:873-880. [DOI] [PubMed] [Google Scholar]
- 11.Davies, D. G., M. R. Parsek, J. P. Pearson, B. H. Iglewski, J. W. Costerton, and E. P. Greenberg. 1998. The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science 280:295-298. [DOI] [PubMed] [Google Scholar]
- 12.Hall-Stoodley, L., J. W. Costerton, and P. Stoodley. 2004. Bacterial biofilms: from the natural environment to infectious diseases. Nat. Rev. Microbiol. 2:95-108. [DOI] [PubMed] [Google Scholar]
- 13.Harris, J. M., and S. Zalipsky. 1997. Poly(ethylene glycol): chemistry and biological applications. American Chemical Society, Washington, DC.
- 14.Harshey, R. M., and T. Matsuyama. 1994. Dimorphic transition in Escherichia coli and Salmonella typhimurium: surface-induced differentiation into hyperflagellate swarmer cells. Proc. Natl. Acad. Sci. USA 91:8631-8635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Heydorn, A., A. T. Nielsen, M. Hentzer, C. Sternberg, M. Givskov, B. K. Ersbøll, and S. Molin. 2000. Quantification of biofilm structures by the novel computer program COMSTAT. Microbiology 146:2395-2407. [DOI] [PubMed] [Google Scholar]
- 16.Hoffmann, C., and G. E. M. Tovar. 2006. Mixed self-assembled monolayers (SAMs) consisting of methoxy-tri(ethylene glycol)-terminated and alkyl-terminated dimethylchlorosilanes control the non-specific adsorption of proteins at oxidic surfaces. J. Colloid Interface Sci. 295:427-435. [DOI] [PubMed] [Google Scholar]
- 17.Hoyle, B. D., and J. W. Costerton. 1991. Bacterial resistance to antibiotics: the role of biofilms. Prog. Drug Res. 37:91-105. [DOI] [PubMed] [Google Scholar]
- 18.Ista, L. K., H. Fan, O. Baca, and G. P. Lopez. 1996. Attachment of bacteria to model solid surfaces: oligo(ethylene glycol) surfaces inhibit bacterial attachment. FEMS Microbiol. Lett. 142:59-63. [DOI] [PubMed] [Google Scholar]
- 19.Kane, R. S., P. Deschatelets, and G. M. Whitesides. 2003. Kosmotropes form the basis of protein-resistant surfaces. Langmuir 19:2388-2391. [Google Scholar]
- 20.Kumar, A., H. A. Biebuyck, and G. M. Whitesides. 1994. Patterning self-assembled monolayers: applications in materials science. Langmuir 10:1498-1511. [Google Scholar]
- 21.Lawrence, J. R., D. R. Korber, B. D. Hoyle, J. W. Costerton, and D. E. Caldwell. 1991. Optical sectioning of microbial biofilms. J. Bacteriol. 173:6558-6567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee, M. H., D. A. Brass, R. Morris, R. J. Composto, and P. Ducheyne. 2005. The effect of non-specific interactions on cellular adhesion using model surfaces. Biomaterials 26:1721-1730. [DOI] [PubMed] [Google Scholar]
- 23.Love, C. J., L. A. Estroff, J. K. Kriebel, R. G. Nuzzo, and G. M. Whitesides. 2005. Self-assembled monolayers of thiolates on metals as a form of nanotechnology. Chem. Rev. 105:1103-1169. [DOI] [PubMed] [Google Scholar]
- 24.Luk, Y.-Y. 2004. Biosurfaces: the significance of water structure at interfaces, p. 389-403. In J. A. Schwarz, C. Contescu, and K. Putyera (ed.), Encyclopedia of nanoscience and nanotechnology. Marcel Dekker, Inc., New York, NY.
- 25.Luk, Y.-Y., M. Kato, and M. Mrksich. 2000. Self-assembled monolayers of alkanethiolates presenting mannitol groups are inert to protein adsorption and cell attachment. Langmuir 16:9604-9608. [Google Scholar]
- 26.Luk, Y.-Y., K.-L. Yang, K. Cadwell, and N. L. Abbott. 2004. Deciphering the interactions between liquid crystals and chemically functionalized surfaces: role of hydrogen bonding on orientations of liquid crystals. Surface Sci. 570:43-56. [Google Scholar]
- 27.Öhman, L., K.-E. Magnusson, and O. Stendahl. 1982. The mannose-specific lectin activity of Escherichia coli type I fimbriae assayed by agglutination of glycolipid-containing liposomes, erythrocytes, and yeast cells and hydrophobic interaction chromatography. FEMS Microbiol. Lett. 14:149-153. [Google Scholar]
- 28.Oosawa, K., and Y. Imae. 1983. Glycerol and ethylene glycol: members of a new class of repellents of Escherichia coli chemotaxis. J. Bacteriol. 154:104-112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pale-Grosdemange, C., E. S. Simon, K. L. Prime, and G. M. Whitesides. 1991. Formation of self-assembled monolayers by chemisorption of derivatives of oligo(ethylene glycol) of structure HS(CH2)11(OCH2CH2)mOH on gold. J. Am. Chem. Soc. 113:12-20. [Google Scholar]
- 30.Potera, C. 1999. Forging a link between biofilms and disease. Science 283:1837-1839. [DOI] [PubMed] [Google Scholar]
- 31.Pratt, L. A., and R. Kolter. 1998. Genetic analysis of Escherichia coli biofilm formation: roles of flagella, motility, chemotaxis and type I pili. Mol. Microbiol. 30:285-293. [DOI] [PubMed] [Google Scholar]
- 32.Ratner, B. D. 1995. Surface modification of polymers: chemical, biological and surface analytical challenges. Biosens. Bioelectron. 10:797-804. [DOI] [PubMed] [Google Scholar]
- 33.Ren, D., L. A. Bedzyk, S. M. Thomas, R. W. Ye, and T. K. Wood. 2004. Gene expression in Escherichia coli biofilms. Appl. Microbiol. Biotechnol. 64:515-524. [DOI] [PubMed] [Google Scholar]
- 34.Ren, D., L. A. Bedzyk, R. W. Ye, S. M. Thomas, and T. K. Wood. 2004. Differential gene expression shows natural brominated furanones interfere with the autoinducer-2 bacterial signaling system of Escherichia coli. Biotechnol. Bioeng. 88:630-642. [DOI] [PubMed] [Google Scholar]
- 35.Ren, D., J. J. Sims, and T. K. Wood. 2001. Inhibition of biofilm formation and swarming of Escherichia coli by (5Z)-4-bromo-5-(bromomethylene)-3-butyl-2(5H)-furanone. Environ. Microbiol. 3:731-736. [DOI] [PubMed] [Google Scholar]
- 36.Sambrook, J., E. F. Fritsch, and T. Maniatis. 1989. Molecular cloning: a laboratory manual, 2nd ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
- 37.Sellers, H., A. Ulman, Y. Shnidman, and J. E. Eilers. 1993. Structure and binding of alkanethiolates on gold and silver surfaces: implications for self-assembled monolayers. J. Am. Chem. Soc. 115:9389-9401. [Google Scholar]
- 38.Smith, A. W. 2005. Biofilms and antibiotic therapy: is there a role for combating bacterial resistance by the use of novel drug delivery systems? Adv. Drug Deliv. Rev. 57:1539-1550. [DOI] [PubMed] [Google Scholar]
- 39.Stewart, P. S., and J. W. Costerton. 2001. Antibiotic resistance of bacteria in biofilms. Lancet 358:135-138. [DOI] [PubMed] [Google Scholar]
- 40.Tejada, F. R., P. I. Nagy, M. Xu, C. Wu, T. Katz, J. Dorsey, M. Rieman, E. Lawlor, M. Warrier, and W. S. Messer, Jr. 2006. Design and synthesis of novel derivatives of the muscarinic agonist tetra(ethylene glycol)(3-methoxy-1,2,5-thiadiazol-4-yl) [3-(1-methyl-1,2,5,6-tetrahydropyrid-3-yl)-1,2,5-thiadiazol-4-yl] ether (CDD-0304): effects of structural modifications on the binding and activity at muscarinic receptor subtypes and chimeras. J. Med. Chem. 49:7518-7531. [DOI] [PubMed] [Google Scholar]
- 41.Tidwell, C. D., S. I. Ertel, B. D. Ratner, B. J. Tarasevich, S. Atre, and D. L. Allara. 1997. Endothelial cell growth and protein adsorption on terminally functionalized, self-assembled monolayers of alkanethiolates on gold. Langmuir 13:3404-3413. [Google Scholar]
- 42.Waters, C. M., and B. L. Bassler. 2005. Quorum sensing: cell-to-cell communication in bacteria. Annu. Rev. Cell Dev. Biol. 21:319-346. [DOI] [PubMed] [Google Scholar]
- 43.Wiencek, K. M., and M. Fletcher. 1995. Bacterial adhesion to hydroxyl- and methyl-terminated alkanethiol self-assembled monolayers. J. Bacteriol. 177:1959-1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wood, T. K., A. F. G. Barrios, M. Herzberg, and J. Lee. 2006. Motility influences biofilm architecture in Escherichia coli. Appl. Microbiol. Biotechnol. 72:361-367. [DOI] [PubMed] [Google Scholar]
- 45.Zita, A., and M. Hermansson. 1997. Effects of bacterial cell surface structures and hydrophobicity on attachment to activated sludge flocs. Appl. Environ. Microbiol. 63:1168-1170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zottola, E. A., and K. C. Sasahara. 1994. Microbial biofilms in the food processing industry—should they be a concern? Int. J. Food Microbiol. 23:125-148. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.